Get your patient on Calcipotriene - Calcipotriene ointment (Calcipotriene)
Calcipotriene - Calcipotriene ointment prescribing information
INDICATIONS AND USAGE
Calcipotriene Ointment, 0.005%, is indicated for the treatment of plaque psoriasis in adults. The safety and effectiveness of topical calcipotriene in dermatoses other than psoriasis have not been established.
DOSAGE AND ADMINISTRATION
Apply a thin layer of Calcipotriene Ointment once or twice daily and rub in gently and completely.
CONTRAINDICATIONS
Calcipotriene Ointment is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation. It should not be used by patients with demonstrated hypercalcemia or evidence of vitamin D toxicity. Calcipotriene Ointment should not be used on the face.
ADVERSE REACTIONS
Clinical Trials Experience
In controlled clinical trials, the most frequent adverse reactions reported for Calcipotriene Ointment were burning, itching and skin irritation, which occurred in approximately 10-15% of patients.
Erythema, dry skin, peeling, rash, dermatitis, worsening of psoriasis including development of facial/scalp psoriasis were reported in 1 to 10% of patients. Other experiences reported in less than 1% of patients included skin atrophy, hyperpigmentation, hypercalcemia, and folliculitis. Once daily dosing has not been shown to be superior in safety to twice daily dosing.
Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions associated with the use of Calcipotriene Ointment have been identified post-approval: contact dermatitis, including allergic contact dermatitis.
DESCRIPTION
Calcipotriene Ointment, 0.005% contains the compound calcipotriene, a synthetic vitamin D 3 derivative, for topical dermatological use.
Chemically, calcipotriene is (5Z,7E,22E,24S)-24-cyclopropyl-9,10-secochola-5,7,10(19),22-tetraene-1α,3β,24-triol-, with the empirical formula C 27 H 40 O 3 , a molecular weight of 412.6, and the following structural formula:
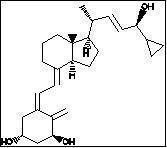
Calcipotriene is a white or off-white crystalline substance. Calcipotriene Ointment contains calcipotriene 50 μg/g in an ointment base of dibasic sodium phosphate, edetate disodium, mineral oil, petrolatum, propylene glycol, tocopherol, steareth-2 and water.
CLINICAL PHARMACOLOGY
In humans, the natural supply of vitamin D depends mainly on exposure to the ultraviolet rays of the sun for conversion of 7-dehydrocholesterol to vitamin D 3 (cholecalciferol) in the skin. Calcipotriene is a synthetic analog of vitamin D 3 .
Clinical studies with radiolabelled Calcipotriene Ointment indicate that approximately 6% (± 3%, SD) of the applied dose of calcipotriene is absorbed systemically when the ointment is applied topically to psoriasis plaques, or 5% (± 2.6%, SD) when applied to normal skin, and much of the absorbed active is converted to inactive metabolites within 24 hours of application.
Vitamin D and its metabolites are transported in the blood, bound to specific plasma proteins. The active form of the vitamin, 1,25-dihydroxy vitamin D 3 (calcitriol), is known to be recycled via the liver and excreted in the bile. Calcipotriene metabolism following systemic uptake is rapid, and occurs via a similar pathway to the natural hormone. The primary metabolites are much less potent than the parent compound.
There is evidence that maternal 1,25-dihydroxy vitamin D 3 (calcitriol) may enter the fetal circulation, but it is not known whether it is excreted in human milk. The systemic disposition of calcipotriene is expected to be similar to that of the naturally occurring vitamin.
CLINICAL STUDIES
Adequate and well-controlled trials of patients treated with Calcipotriene Ointment have demonstrated improvement usually beginning after 2 weeks of therapy. This improvement continued in patients using Calcipotriene Ointment once daily and twice daily. After 8 weeks of once daily Calcipotriene Ointment, 56.7% of patients showed at least marked improvements (6.4% showed complete clearing). After 8 weeks of twice daily Calcipotriene Ointment, 70.0% of patients showed at least marked improvement (11.3% showed complete clearing).
Subtracting percentages of patients using placebo (vehicle only) from percentages of patients using Calcipotriene Ointment who had at least marked improvements after 8 weeks yields 39.9% for once daily and 49.6% for twice daily. This adjustment for placebo effect indicates that what might appear to be differences between once and twice daily use may reflect differences in the studies independent from the frequency of dosing. Although there was a numerical difference in comparison across studies, twice daily dosing has not been shown to be superior in efficacy to once daily dosing.
Over 400 patients have been treated in open label clinical studies of Calcipotriene Ointment for periods of up to one year. In half of these studies, patients who previously had not responded well to Calcipotriene Ointment were excluded. The adverse events in these extended studies included skin irritation in approximately 25% of patients and worsening of psoriasis in approximately 10% of patients. In one of these open label studies, half of the patients no longer required Calcipotriene Ointment by 16 weeks of treatment, because of satisfactory therapeutic results.
HOW SUPPLIED
Calcipotriene Ointment, 0.005% is available in:
60 gram aluminum tubes NDC 66993-878-61
120 gram aluminum tubes NDC 66993-878-78