Verticalm Prescribing Information
Based on a review of this drug by the National Academy of Sciences - National Research Council and/or other information, FDA has classified the indications as follows:
Effective: Management of nausea and vomiting, and dizziness associated with motion sickness.
Final classification of the less than effective indications requires further investigation.
Motion Sickness
The initial dose of 25 to 50 mg of meclizine hydrochloride should be taken one hour prior to embarkation for protection against motion sickness. Thereafter, the dose may be repeated every 24 hours for the duration of the journey.
Meclizine hydrochloride is contraindicated in individuals who have shown a previous hypersensitivity to it.
Anaphylactoid reaction, drowsiness, dry mouth, headache, fatigue, vomiting and, on rare occasions, blurred vision have been reported.
Chemically, meclizine hydrochloride is 1-(p-chloro-α-phenylbenzyl)-4-(m-methylbenzyl) piperazine dihydrochloride monohydrate.
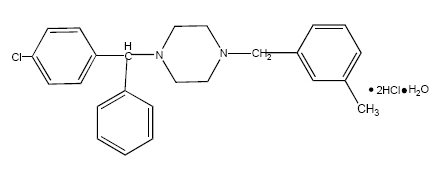
C25H27CIN2·2HCl·H20 M.W. 481.89
Meclizine hydrochloride tablets, USP are available in two different strengths, 12.5 mg and 25 mg. Inactive ingredients: microcrystalline cellulose, lactose monohydrate, croscarmellose sodium and magnesium stearate. The 12.5 mg tablet also contains FD&C Blue #1 Aluminum Lake.
Meclizine hydrochloride is an antihistamine that shows marked protective activity against nebulized histamine and lethal doses of intravenously injected histamine in guinea pigs. It has a marked effect in blocking the vasodepressor response to histamine, but only a slight blocking action against acetylcholine. Its activity is relatively weak in inhibiting the spasmogenic action of histamine on isolated guinea pig ileum.
Pharmacokinetics
The available pharmacokinetic information for meclizine following oral administration has been summarized from published literature.
Absorption
Meclizine is absorbed after oral administration with maximum plasma concentrations reaching at a median Tmax value of 3 hours post-dose (range: 1.5 to 6 hours) for the tablet dosage form.
Distribution
Drug distribution characteristics for meclizine in humans are unknown.
Metabolism
The metabolic fate of meclizine in humans is unknown. In an in vitro metabolic study using human hepatic microsome and recombinant CYP enzyme, CYP 2D6 was found to be the dominant enzyme for metabolism of meclizine.
The genetic polymorphism of CYP2D6 that results in extensive-, poor-, intermediate- and ultrarapid metabolizer phenotypes could contribute to large inter-individual variability in meclizine exposure.
Elimination
Meclizine has a plasma elimination half-life of about 5-6 hours in humans.