Get your patient on Actemra (Tocilizumab)
Actemra prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Actemra patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
For RA, pJIA and sJIA, ACTEMRA may be used alone or in combination with methotrexate: and in RA, other non-biologic DMARDs may be used. (2 )
General Administration and Dosing Information (2.1 )
- RA, GCA, SSc-ILD, PJIA and SJIA – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm 3 , platelet count below 100,000 per mm 3 , or ALT or AST above 1.5 times the upper limit of normal (ULN) (5.3 , 5.4 ) .
- COVID-19 – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 1000 per mm 3 , platelet count below 50,000 mm 3 , or ALT or AST above 10 times ULN (5.3 , 5.4 ) .
- In RA, CRS or COVID-19 patients, ACTEMRA doses exceeding 800 mg per infusion are not recommended. (2.2 , 2.7 , 12.3 )
- In GCA patients, ACTEMRA doses exceeding 600 mg per infusion are not recommended. (2.3 , 12.3 )
Rheumatoid Arthritis (2.2 )
Recommended Adult Intravenous Dosage:
When used in combination with non-biologic DMARDs or as monotherapy the recommended starting dose is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response.
Recommended Adult Subcutaneous Dosage:
| Patients less than 100 kg weight | 162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response |
| Patients at or above 100 kg weight | 162 mg administered subcutaneously every week |
Giant Cell Arteritis (2.3 )
Recommended Adult Intravenous Dosage:
The recommended dose is 6 mg per kg every 4 weeks in combination with a tapering course of glucocorticoids. ACTEMRA can be used alone following discontinuation of glucocorticoids.
Recommended Adult Subcutaneous Dosage:
The recommended dose is 162 mg given once every week as a subcutaneous injection, in combination with a tapering course of glucocorticoids.
A dose of 162 mg given once every other week as a subcutaneous injection, in combination with a tapering course of glucocorticoids, may be prescribed based on clinical considerations.
ACTEMRA can be used alone following discontinuation of glucocorticoids.
Systemic Sclerosis-Associated Interstitial Lung Disease (SSc-ILD) (2.4 )
Recommended Adult Subcutaneous Dosage:
The recommended dose of ACTEMRA for adult patients with SSc-ILD is 162 mg given once every week as a subcutaneous injection.
Polyarticular Juvenile Idiopathic Arthritis (2.5 )
| Recommended Intravenous PJIA Dosage Every 4 Weeks | |
|---|---|
| Patients less than 30 kg weight | 10 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Recommended Subcutaneous PJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg once every three weeks |
| Patients at or above 30 kg weight | 162 mg once every two weeks |
Systemic Juvenile Idiopathic Arthritis (2.6 )
| Recommended Intravenous SJIA Dosage Every 2 Weeks | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Recommended Subcutaneous SJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg every two weeks |
| Patients at or above 30 kg weight | 162 mg every week |
Cytokine Release Syndrome (2.7 )
| Recommended Intravenous CRS Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Alone or in combination with corticosteroids. | |
Coronavirus Disease 2019 (2.8 )
| Recommended Intravenous COVID-19 Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Administered by a 60-minute intravenous infusion | |
Administration of Intravenous formulation (2.9 )
- For patients with RA, GCA, COVID-19, CRS, PJIA, and SJIA patients at or above 30 kg, dilute to 100 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
- For PJIA, SJIA, CRS and COVID-19 patients less than 30 kg, dilute to 50 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
- Administer as a single intravenous drip infusion over 1 hour; do not administer as bolus or push.
Administration of Subcutaneous formulation (2.10 )
- Follow the Instructions for Use for prefilled syringe and prefilled ACTPen ® autoinjector
Dose Modifications (2.11 )
- Recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia.
General Considerations for Administration
Not Recommended for Concomitant Use with Biological DMARDs
ACTEMRA has not been studied in combination with biological DMARDs such as TNF antagonists, IL-1R antagonists, anti-CD20 monoclonal antibodies and selective co-stimulation modulators because of the possibility of increased immunosuppression and increased risk of infection. Avoid using ACTEMRA with biological DMARDs.
Baseline Laboratory Evaluation Prior to Treatment
Obtain and assess baseline complete blood count (CBC) and liver function tests prior to treatment.
- RA, GCA, SSc-ILD, PJIA and SJIA – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm 3 , platelet count below 100,000 per mm 3 , or ALT or AST above 1.5 times the upper limit of normal (ULN) [see Warnings and Precautions (5.3 , 5.4) ] .
- CRS – Patients with severe or life-threatening CRS frequently have cytopenias or elevated ALT or AST due to the lymphodepleting chemotherapy or the CRS. The decision to administer ACTEMRA should take into account the potential benefit of treating the CRS versus the risks of short-term treatment with ACTEMRA.
- COVID-19 – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 1000 per mm 3 , platelet count below 50,000 mm 3 , or ALT or AST above 10 times ULN [see Warnings and Precautions (5.3 , 5.4) ] .
Recommended Dosage for Rheumatoid Arthritis
ACTEMRA may be used as monotherapy or concomitantly with methotrexate or other non-biologic DMARDs as an intravenous infusion or as a subcutaneous injection.
Recommended Intravenous Dosage Regimen:
The recommended dosage of ACTEMRA for adult patients given as a 60-minute single intravenous drip infusion is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response.
- Reduction of dose from 8 mg per kg to 4 mg per kg is recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) , Warnings and Precautions (5.3 , 5.4) , and Adverse Reactions (6.1) ] .
- Doses exceeding 800 mg per infusion are not recommended in RA patients [see Clinical Pharmacology (12.3) ] .
Recommended Subcutaneous Dosage Regimen:
| Patients less than 100 kg weight | 162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response |
| Patients at or above 100 kg weight | 162 mg administered subcutaneously every week |
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration administer the first subcutaneous dose instead of the next scheduled intravenous dose.
Interruption of dose or reduction in frequency of administration of subcutaneous dose from every week to every other week dosing is recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) , Warnings and Precautions (5.3 , 5.4) , and Adverse Reactions (6.2) ].
Recommended Dosage for Giant Cell Arteritis
Recommended Intravenous Dosage Regimen:
The recommended dosage of ACTEMRA for adult patients given as a 60-minute single intravenous drip infusion is 6 mg per kg every 4 weeks in combination with tapering course of glucocorticoids.
ACTEMRA can be used alone following discontinuation of glucocorticoids.
- Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
- Doses exceeding 600 mg per infusion are not recommended in GCA patients [see Clinical Pharmacology (12.3) ].
Recommended Subcutaneous Dosage Regimen:
The recommended dose of ACTEMRA for adult patients with GCA is 162 mg given once every week as a subcutaneous injection in combination with a tapering course of glucocorticoids.
A dose of 162 mg given once every other week as a subcutaneous injection in combination with a tapering course of glucocorticoids may be prescribed based on clinical considerations.
ACTEMRA can be used alone following discontinuation of glucocorticoids.
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration, administer the first subcutaneous dose instead of the next scheduled intravenous dose.
Interruption of dose or reduction in frequency of administration of subcutaneous dose from every week to every other week dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
Recommended Dosage for Systemic Sclerosis-Associated Interstitial Lung Disease
The recommended dose of ACTEMRA for adult patients with SSc-ILD is 162 mg given once every week as a subcutaneous injection.
- Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
- Subcutaneous administration with the prefilled ACTPen ® autoinjector has not been studied in SSc-ILD.
- Intravenous administration is not approved for SSc-ILD.
Recommended Dosage for Polyarticular Juvenile Idiopathic Arthritis
ACTEMRA may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change dose based solely on a single visit body weight measurement, as weight may fluctuate.
Recommended Intravenous Dosage Regimen:
The recommended dosage of ACTEMRA for PJIA patients given once every 4 weeks as a 60-minute single intravenous drip infusion is:
| Recommended Intravenous PJIA Dosage Every 4 Weeks | |
|---|---|
| Patients less than 30 kg weight | 10 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
Recommended Subcutaneous Dosage Regimen:
| Recommended Subcutaneous PJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg once every 3 weeks |
| Patients at or above 30 kg weight | 162 mg once every 2 weeks |
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration, administer the first subcutaneous dose instead of the next scheduled intravenous dose.
Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11 ] .
Recommended Dosage for Systemic Juvenile Idiopathic Arthritis
ACTEMRA may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change a dose based solely on a single visit body weight measurement, as weight may fluctuate.
Recommended Intravenous Dosage Regimen:
The recommended dose of ACTEMRA for SJIA patients given once every 2 weeks as a 60-minute single intravenous drip infusion is:
| Recommended Intravenous SJIA Dosage Every 2 Weeks | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
Recommended Subcutaneous Dosage Regimen:
| Recommended Subcutaneous SJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg once every two weeks |
| Patients at or above 30 kg weight | 162 mg once every week |
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration, administer the first subcutaneous dose when the next scheduled intravenous dose is due.
Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
Recommended Dosage for Cytokine Release Syndrome (CRS)
Use only the intravenous route for treatment of CRS. The recommended dose of ACTEMRA for treatment of CRS given as a 60-minute intravenous infusion is:
| Recommended Intravenous CRS Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Alone or in combination with corticosteroids | |
- If no clinical improvement in the signs and symptoms of CRS occurs after the first dose, up to 3 additional doses of ACTEMRA may be administered. The interval between consecutive doses should be at least 8 hours.
- Doses exceeding 800 mg per infusion are not recommended in CRS patients.
- Subcutaneous administration is not approved for CRS.
Recommended Dosage for Coronavirus Disease 2019 (COVID-19)
Administer ACTEMRA by intravenous infusion only.
The recommended dosage of ACTEMRA for treatment of patients with COVID-19 given as a single 60-minute intravenous infusion is:
| Recommended Intravenous COVID-19 Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
If clinical signs or symptoms worsen or do not improve after the first dose, one additional infusion of ACTEMRA may be administered at least 8 hours after the initial infusion.
- Doses exceeding 800 mg per infusion are not recommended in patients with COVID-19.
- Subcutaneous administration is not approved for COVID-19.
Preparation and Administration Instructions for Intravenous Infusion
ACTEMRA for intravenous infusion should be diluted by a healthcare professional using aseptic technique as follows:
- Use a sterile needle and syringe to prepare ACTEMRA.
- Patients less than 30 kg : use a 50 mL infusion bag or bottle of 0.9% or 0.45% Sodium Chloride Injection, USP, and then follow steps 1 and 2 below.
- Patients at or above 30 kg weight : use a 100 mL infusion bag or bottle, and then follow steps 1 and 2 below.
- Step 1. Withdraw a volume of 0.9% or 0.45% Sodium Chloride Injection, USP, equal to the volume of the ACTEMRA injection required for the patient's dose from the infusion bag or bottle [see Dosage and Administration (2.2 , 2.5 , 2.6 , 2.7) ] .
| For Intravenous Use: Volume of ACTEMRA Injection per kg of Body Weight | ||
|---|---|---|
| Dosage | Indication | Volume of ACTEMRA injection per kg of body weight |
| 4 mg/kg | Adult RA | 0.2 mL/kg |
| 6 mg/kg | Adult GCA | 0.3 mL/kg |
| 8 mg/kg | Adult RA SJIA, PJIA, CRS and COVID-19 (greater than or equal to 30 kg of body weight) | 0.4 mL/kg |
| 10 mg/kg | PJIA (less than 30 kg of body weight) | 0.5 mL/kg |
| 12 mg/kg | SJIA, CRS and COVID-19 (less than 30 kg of body weight) | 0.6 mL/kg |
- Step 2. Withdraw the amount of ACTEMRA for intravenous infusion from the vial(s) and add slowly into the 0.9% or 0.45% Sodium Chloride Injection, USP infusion bag or bottle. To mix the solution, gently invert the bag to avoid foaming.
- The fully diluted ACTEMRA solutions for infusion using 0.9% Sodium Chloride Injection, USP may be stored at 36°F to 46°F (2°C to 8°C) or room temperature for up to 24 hours and should be protected from light.
- The fully diluted ACTEMRA solutions for infusion using 0.45% Sodium Chloride Injection, USP may be stored at 36°F to 46°F (2°C to 8°C) for up to 24 hours or room temperature for up to 4 hours and should be protected from light.
- ACTEMRA solutions do not contain preservatives; therefore, unused product remaining in the vials should not be used.
- Allow the fully diluted ACTEMRA solution to reach room temperature prior to infusion.
- The infusion should be administered over 60 minutes, and must be administered with an infusion set. Do not administer as an intravenous push or bolus.
- ACTEMRA should not be infused concomitantly in the same intravenous line with other drugs. No physical or biochemical compatibility studies have been conducted to evaluate the co-administration of ACTEMRA with other drugs.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If particulates and discolorations are noted, the product should not be used.
- Fully diluted ACTEMRA solutions are compatible with polypropylene, polyethylene and polyvinyl chloride infusion bags and polypropylene, polyethylene and glass infusion bottles.
Preparation and Administration Instructions for Subcutaneous Injection
- ACTEMRA for subcutaneous injection is not intended for intravenous drip infusion.
- Assess suitability of patient for subcutaneous home use and instruct patients to inform a healthcare professional before administering the next dose if they experience any symptoms of allergic reaction. Patients should seek immediate medical attention if they develop symptoms of serious allergic reactions. ACTEMRA subcutaneous injection is intended for use under the guidance of a healthcare practitioner. After proper training in subcutaneous injection technique, a patient may self-inject ACTEMRA or the patient's caregiver may administer ACTEMRA if a healthcare practitioner determines that it is appropriate. PJIA and SJIA patients may self-inject with the ACTEMRA prefilled syringe or ACTPen ® autoinjector, or the patient's caregiver may administer ACTEMRA if both the healthcare practitioner and the parent/legal guardian determines it is appropriate [see Use in Specific Populations (8.4) ] . Patients, or patient caregivers, should be instructed to follow the directions provided in the Instructions for Use (IFU) for additional details on medication administration.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use ACTEMRA prefilled syringes (PFS) or prefilled ACTPen ® autoinjectors exhibiting particulate matter, cloudiness, or discoloration. ACTEMRA for subcutaneous administration should be clear and colorless to pale yellow. Do not use if any part of the PFS or ACTPen ® autoinjector appears to be damaged.
- Patients using ACTEMRA for subcutaneous administration should be instructed to inject the full amount in the syringe (0.9 mL) or full amount in the ACTPen ® autoinjector (0.9 mL), which provides 162 mg of ACTEMRA, according to the directions provided in the IFU.
- Injection sites should be rotated with each injection and should never be given into moles, scars, or areas where the skin is tender, bruised, red, hard, or not intact.
Dosage Modifications due to Serious Infections or Laboratory Abnormalities
Serious Infections
Hold ACTEMRA treatment if a patient develops a serious infection until the infection is controlled.
Laboratory Abnormalities
Rheumatoid Arthritis, Giant Cell Arteritis and Systemic Sclerosis-Associated Interstitial Lung Disease
| Liver Enzyme Abnormalities [see Warnings and Precautions (5.3 , 5.4) ] | ||
|---|---|---|
| Lab Value | Recommendation for RA and SSc-ILD | Recommendation for GCA |
| Greater than 1 to 3× ULN | Dose modify concomitant DMARDs if appropriate For persistent increases in this range:
| Dose modify immunomodulatory agents if appropriate For persistent increases in this range:
|
| Greater than 3 to 5× ULN (confirmed by repeat testing) | Hold ACTEMRA dosing until less than 3× ULN and follow recommendations above for greater than 1 to 3× ULN For persistent increases greater than 3× ULN, discontinue ACTEMRA | Hold ACTEMRA dosing until less than 3x ULN and follow recommendations above for greater than 1 to 3x ULN For persistent increases greater than 3x ULN, discontinue ACTEMRA |
| Greater than 5× ULN | Discontinue ACTEMRA | Discontinue ACTEMRA |
| Low Absolute Neutrophil Count (ANC) [see Warnings and Precautions (5.4) ] | ||
|---|---|---|
| Lab Value (cells per mm 3 ) | Recommendation for RA and SSc-ILD | Recommendation for GCA |
| ANC greater than 1000 | Maintain dose | Maintain dose |
| ANC 500 to 1000 | Hold ACTEMRA dosing When ANC greater than 1000 cells per mm 3 :
| Hold ACTEMRA dosing When ANC greater than 1000 cells per mm 3 :
|
| ANC less than 500 | Discontinue ACTEMRA | Discontinue ACTEMRA |
| Low Platelet Count [see Warnings and Precautions (5.4) ] | ||
|---|---|---|
| Lab Value (cells per mm 3 ) | Recommendation for RA and SSc-ILD | Recommendation for GCA |
| 50,000 to 100,000 | Hold ACTEMRA dosing When platelet count is greater than 100,000 cells per mm 3 :
| Hold ACTEMRA dosing When platelet count is greater than 100,000 cells per mm 3 :
|
| Less than 50,000 | Discontinue ACTEMRA | Discontinue ACTEMRA |
Polyarticular and Systemic Juvenile Idiopathic Arthritis
Dose reduction of ACTEMRA has not been studied in the PJIA and SJIA populations. Dose interruptions of ACTEMRA are recommended for liver enzyme abnormalities, low neutrophil counts, and low platelet counts in patients with PJIA and SJIA at levels similar to what is outlined above for patients with RA and GCA. If appropriate, dose modify or stop concomitant methotrexate and/or other medications and hold ACTEMRA dosing until the clinical situation has been evaluated. In PJIA and SJIA the decision to discontinue ACTEMRA for a laboratory abnormality should be based upon the medical assessment of the individual patient.

Actemra prescribing information
WARNING: RISK OF SERIOUS INFECTIONS
Patients treated with ACTEMRA are at increased risk for developing serious infections that may lead to hospitalization or death [see Warnings and Precautions (5.1) , Adverse Reactions (6.1) ] . Most patients who developed these infections were taking concomitant immunosuppressants such as methotrexate or corticosteroids.
If a serious infection develops, interrupt ACTEMRA until the infection is controlled.
Reported infections include:
- Active tuberculosis, which may present with pulmonary or extrapulmonary disease. Patients, except those with COVID-19, should be tested for latent tuberculosis before ACTEMRA use and during therapy. Treatment for latent infection should be initiated prior to ACTEMRA use.
- Invasive fungal infections, including candidiasis, aspergillosis, and pneumocystis. Patients with invasive fungal infections may present with disseminated, rather than localized, disease.
- Bacterial, viral and other infections due to opportunistic pathogens.
The risks and benefits of treatment with ACTEMRA should be carefully considered prior to initiating therapy in patients with chronic or recurrent infection.
Patients should be closely monitored for the development of signs and symptoms of infection during and after treatment with ACTEMRA, including the possible development of tuberculosis in patients who tested negative for latent tuberculosis infection prior to initiating therapy [see Warnings and Precautions (5.1) ] .
INDICATIONS AND USAGE
ACTEMRA ® (tocilizumab) is an interleukin-6 (IL-6) receptor antagonist indicated for treatment of:
Rheumatoid Arthritis (RA) (1.1 )
- Adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs).
Giant Cell Arteritis (GCA) (1.2 )
- Adult patients with giant cell arteritis.
Systemic Sclerosis-Associated Interstitial Lung Disease (SSc-ILD) (1.3 )
- Slowing the rate of decline in pulmonary function in adult patients with systemic sclerosis-associated interstitial lung disease (SSc-ILD)
Polyarticular Juvenile Idiopathic Arthritis (PJIA) (1.4 )
- Patients 2 years of age and older with active polyarticular juvenile idiopathic arthritis.
Systemic Juvenile Idiopathic Arthritis (SJIA) (1.5 )
- Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
Cytokine Release Syndrome (CRS) (1.6 )
- Adults and pediatric patients 2 years of age and older with chimeric antigen receptor (CAR) T cell-induced severe or life-threatening cytokine release syndrome.
Coronavirus Disease 2019 (COVID-19) (1.7 )
- Hospitalized adult and pediatric patients aged 2 years and older with coronavirus disease 2019 (COVID-19) who are receiving systemic corticosteroids and require supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).
Rheumatoid Arthritis (RA)
ACTEMRA ® (tocilizumab) is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more Disease-Modifying Anti-Rheumatic Drugs (DMARDs).
Giant Cell Arteritis (GCA)
ACTEMRA ® (tocilizumab) is indicated for the treatment of giant cell arteritis (GCA) in adult patients.
Systemic Sclerosis-Associated Interstitial Lung Disease (SSc-ILD)
ACTEMRA ® (tocilizumab) is indicated for slowing the rate of decline in pulmonary function in adult patients with systemic sclerosis-associated interstitial lung disease.
Polyarticular Juvenile Idiopathic Arthritis (PJIA)
ACTEMRA ® (tocilizumab) is indicated for the treatment of active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older.
Systemic Juvenile Idiopathic Arthritis (SJIA)
ACTEMRA ® (tocilizumab) is indicated for the treatment of active systemic juvenile idiopathic arthritis in patients 2 years of age and older.
Cytokine Release Syndrome (CRS)
ACTEMRA ® (tocilizumab) is indicated for the treatment of chimeric antigen receptor (CAR) T cell-induced severe or life-threatening cytokine release syndrome in adults and pediatric patients 2 years of age and older.
Coronavirus Disease 2019 (COVID-19)
ACTEMRA® (tocilizumab) is indicated for the treatment of coronavirus disease 2019 (COVID-19) in hospitalized adult and pediatric patients aged 2 years and older who are receiving systemic corticosteroids and require supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).
DOSAGE AND ADMINISTRATION
For RA, pJIA and sJIA, ACTEMRA may be used alone or in combination with methotrexate: and in RA, other non-biologic DMARDs may be used. (2 )
General Administration and Dosing Information (2.1 )
- RA, GCA, SSc-ILD, PJIA and SJIA – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm 3 , platelet count below 100,000 per mm 3 , or ALT or AST above 1.5 times the upper limit of normal (ULN) (5.3 , 5.4 ) .
- COVID-19 – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 1000 per mm 3 , platelet count below 50,000 mm 3 , or ALT or AST above 10 times ULN (5.3 , 5.4 ) .
- In RA, CRS or COVID-19 patients, ACTEMRA doses exceeding 800 mg per infusion are not recommended. (2.2 , 2.7 , 12.3 )
- In GCA patients, ACTEMRA doses exceeding 600 mg per infusion are not recommended. (2.3 , 12.3 )
Rheumatoid Arthritis (2.2 )
Recommended Adult Intravenous Dosage:
When used in combination with non-biologic DMARDs or as monotherapy the recommended starting dose is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response.
Recommended Adult Subcutaneous Dosage:
| Patients less than 100 kg weight | 162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response |
| Patients at or above 100 kg weight | 162 mg administered subcutaneously every week |
Giant Cell Arteritis (2.3 )
Recommended Adult Intravenous Dosage:
The recommended dose is 6 mg per kg every 4 weeks in combination with a tapering course of glucocorticoids. ACTEMRA can be used alone following discontinuation of glucocorticoids.
Recommended Adult Subcutaneous Dosage:
The recommended dose is 162 mg given once every week as a subcutaneous injection, in combination with a tapering course of glucocorticoids.
A dose of 162 mg given once every other week as a subcutaneous injection, in combination with a tapering course of glucocorticoids, may be prescribed based on clinical considerations.
ACTEMRA can be used alone following discontinuation of glucocorticoids.
Systemic Sclerosis-Associated Interstitial Lung Disease (SSc-ILD) (2.4 )
Recommended Adult Subcutaneous Dosage:
The recommended dose of ACTEMRA for adult patients with SSc-ILD is 162 mg given once every week as a subcutaneous injection.
Polyarticular Juvenile Idiopathic Arthritis (2.5 )
| Recommended Intravenous PJIA Dosage Every 4 Weeks | |
|---|---|
| Patients less than 30 kg weight | 10 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Recommended Subcutaneous PJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg once every three weeks |
| Patients at or above 30 kg weight | 162 mg once every two weeks |
Systemic Juvenile Idiopathic Arthritis (2.6 )
| Recommended Intravenous SJIA Dosage Every 2 Weeks | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Recommended Subcutaneous SJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg every two weeks |
| Patients at or above 30 kg weight | 162 mg every week |
Cytokine Release Syndrome (2.7 )
| Recommended Intravenous CRS Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Alone or in combination with corticosteroids. | |
Coronavirus Disease 2019 (2.8 )
| Recommended Intravenous COVID-19 Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Administered by a 60-minute intravenous infusion | |
Administration of Intravenous formulation (2.9 )
- For patients with RA, GCA, COVID-19, CRS, PJIA, and SJIA patients at or above 30 kg, dilute to 100 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
- For PJIA, SJIA, CRS and COVID-19 patients less than 30 kg, dilute to 50 mL in 0.9% or 0.45% Sodium Chloride Injection, USP for intravenous infusion using aseptic technique.
- Administer as a single intravenous drip infusion over 1 hour; do not administer as bolus or push.
Administration of Subcutaneous formulation (2.10 )
- Follow the Instructions for Use for prefilled syringe and prefilled ACTPen ® autoinjector
Dose Modifications (2.11 )
- Recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia.
General Considerations for Administration
Not Recommended for Concomitant Use with Biological DMARDs
ACTEMRA has not been studied in combination with biological DMARDs such as TNF antagonists, IL-1R antagonists, anti-CD20 monoclonal antibodies and selective co-stimulation modulators because of the possibility of increased immunosuppression and increased risk of infection. Avoid using ACTEMRA with biological DMARDs.
Baseline Laboratory Evaluation Prior to Treatment
Obtain and assess baseline complete blood count (CBC) and liver function tests prior to treatment.
- RA, GCA, SSc-ILD, PJIA and SJIA – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 2000 per mm 3 , platelet count below 100,000 per mm 3 , or ALT or AST above 1.5 times the upper limit of normal (ULN) [see Warnings and Precautions (5.3 , 5.4) ] .
- CRS – Patients with severe or life-threatening CRS frequently have cytopenias or elevated ALT or AST due to the lymphodepleting chemotherapy or the CRS. The decision to administer ACTEMRA should take into account the potential benefit of treating the CRS versus the risks of short-term treatment with ACTEMRA.
- COVID-19 – It is recommended that ACTEMRA not be initiated in patients with an absolute neutrophil count (ANC) below 1000 per mm 3 , platelet count below 50,000 mm 3 , or ALT or AST above 10 times ULN [see Warnings and Precautions (5.3 , 5.4) ] .
Recommended Dosage for Rheumatoid Arthritis
ACTEMRA may be used as monotherapy or concomitantly with methotrexate or other non-biologic DMARDs as an intravenous infusion or as a subcutaneous injection.
Recommended Intravenous Dosage Regimen:
The recommended dosage of ACTEMRA for adult patients given as a 60-minute single intravenous drip infusion is 4 mg per kg every 4 weeks followed by an increase to 8 mg per kg every 4 weeks based on clinical response.
- Reduction of dose from 8 mg per kg to 4 mg per kg is recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) , Warnings and Precautions (5.3 , 5.4) , and Adverse Reactions (6.1) ] .
- Doses exceeding 800 mg per infusion are not recommended in RA patients [see Clinical Pharmacology (12.3) ] .
Recommended Subcutaneous Dosage Regimen:
| Patients less than 100 kg weight | 162 mg administered subcutaneously every other week, followed by an increase to every week based on clinical response |
| Patients at or above 100 kg weight | 162 mg administered subcutaneously every week |
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration administer the first subcutaneous dose instead of the next scheduled intravenous dose.
Interruption of dose or reduction in frequency of administration of subcutaneous dose from every week to every other week dosing is recommended for management of certain dose-related laboratory changes including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) , Warnings and Precautions (5.3 , 5.4) , and Adverse Reactions (6.2) ].
Recommended Dosage for Giant Cell Arteritis
Recommended Intravenous Dosage Regimen:
The recommended dosage of ACTEMRA for adult patients given as a 60-minute single intravenous drip infusion is 6 mg per kg every 4 weeks in combination with tapering course of glucocorticoids.
ACTEMRA can be used alone following discontinuation of glucocorticoids.
- Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
- Doses exceeding 600 mg per infusion are not recommended in GCA patients [see Clinical Pharmacology (12.3) ].
Recommended Subcutaneous Dosage Regimen:
The recommended dose of ACTEMRA for adult patients with GCA is 162 mg given once every week as a subcutaneous injection in combination with a tapering course of glucocorticoids.
A dose of 162 mg given once every other week as a subcutaneous injection in combination with a tapering course of glucocorticoids may be prescribed based on clinical considerations.
ACTEMRA can be used alone following discontinuation of glucocorticoids.
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration, administer the first subcutaneous dose instead of the next scheduled intravenous dose.
Interruption of dose or reduction in frequency of administration of subcutaneous dose from every week to every other week dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
Recommended Dosage for Systemic Sclerosis-Associated Interstitial Lung Disease
The recommended dose of ACTEMRA for adult patients with SSc-ILD is 162 mg given once every week as a subcutaneous injection.
- Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
- Subcutaneous administration with the prefilled ACTPen ® autoinjector has not been studied in SSc-ILD.
- Intravenous administration is not approved for SSc-ILD.
Recommended Dosage for Polyarticular Juvenile Idiopathic Arthritis
ACTEMRA may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change dose based solely on a single visit body weight measurement, as weight may fluctuate.
Recommended Intravenous Dosage Regimen:
The recommended dosage of ACTEMRA for PJIA patients given once every 4 weeks as a 60-minute single intravenous drip infusion is:
| Recommended Intravenous PJIA Dosage Every 4 Weeks | |
|---|---|
| Patients less than 30 kg weight | 10 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
Recommended Subcutaneous Dosage Regimen:
| Recommended Subcutaneous PJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg once every 3 weeks |
| Patients at or above 30 kg weight | 162 mg once every 2 weeks |
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration, administer the first subcutaneous dose instead of the next scheduled intravenous dose.
Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11 ] .
Recommended Dosage for Systemic Juvenile Idiopathic Arthritis
ACTEMRA may be used as an intravenous infusion or as a subcutaneous injection alone or in combination with methotrexate. Do not change a dose based solely on a single visit body weight measurement, as weight may fluctuate.
Recommended Intravenous Dosage Regimen:
The recommended dose of ACTEMRA for SJIA patients given once every 2 weeks as a 60-minute single intravenous drip infusion is:
| Recommended Intravenous SJIA Dosage Every 2 Weeks | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
Recommended Subcutaneous Dosage Regimen:
| Recommended Subcutaneous SJIA Dosage | |
|---|---|
| Patients less than 30 kg weight | 162 mg once every two weeks |
| Patients at or above 30 kg weight | 162 mg once every week |
When transitioning from ACTEMRA intravenous therapy to subcutaneous administration, administer the first subcutaneous dose when the next scheduled intravenous dose is due.
Interruption of dosing may be needed for management of dose-related laboratory abnormalities including elevated liver enzymes, neutropenia, and thrombocytopenia [see Dosage and Administration (2.11) ] .
Recommended Dosage for Cytokine Release Syndrome (CRS)
Use only the intravenous route for treatment of CRS. The recommended dose of ACTEMRA for treatment of CRS given as a 60-minute intravenous infusion is:
| Recommended Intravenous CRS Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
| Alone or in combination with corticosteroids | |
- If no clinical improvement in the signs and symptoms of CRS occurs after the first dose, up to 3 additional doses of ACTEMRA may be administered. The interval between consecutive doses should be at least 8 hours.
- Doses exceeding 800 mg per infusion are not recommended in CRS patients.
- Subcutaneous administration is not approved for CRS.
Recommended Dosage for Coronavirus Disease 2019 (COVID-19)
Administer ACTEMRA by intravenous infusion only.
The recommended dosage of ACTEMRA for treatment of patients with COVID-19 given as a single 60-minute intravenous infusion is:
| Recommended Intravenous COVID-19 Dosage | |
|---|---|
| Patients less than 30 kg weight | 12 mg per kg |
| Patients at or above 30 kg weight | 8 mg per kg |
If clinical signs or symptoms worsen or do not improve after the first dose, one additional infusion of ACTEMRA may be administered at least 8 hours after the initial infusion.
- Doses exceeding 800 mg per infusion are not recommended in patients with COVID-19.
- Subcutaneous administration is not approved for COVID-19.
Preparation and Administration Instructions for Intravenous Infusion
ACTEMRA for intravenous infusion should be diluted by a healthcare professional using aseptic technique as follows:
- Use a sterile needle and syringe to prepare ACTEMRA.
- Patients less than 30 kg : use a 50 mL infusion bag or bottle of 0.9% or 0.45% Sodium Chloride Injection, USP, and then follow steps 1 and 2 below.
- Patients at or above 30 kg weight : use a 100 mL infusion bag or bottle, and then follow steps 1 and 2 below.
- Step 1. Withdraw a volume of 0.9% or 0.45% Sodium Chloride Injection, USP, equal to the volume of the ACTEMRA injection required for the patient's dose from the infusion bag or bottle [see Dosage and Administration (2.2 , 2.5 , 2.6 , 2.7) ] .
| For Intravenous Use: Volume of ACTEMRA Injection per kg of Body Weight | ||
|---|---|---|
| Dosage | Indication | Volume of ACTEMRA injection per kg of body weight |
| 4 mg/kg | Adult RA | 0.2 mL/kg |
| 6 mg/kg | Adult GCA | 0.3 mL/kg |
| 8 mg/kg | Adult RA SJIA, PJIA, CRS and COVID-19 (greater than or equal to 30 kg of body weight) | 0.4 mL/kg |
| 10 mg/kg | PJIA (less than 30 kg of body weight) | 0.5 mL/kg |
| 12 mg/kg | SJIA, CRS and COVID-19 (less than 30 kg of body weight) | 0.6 mL/kg |
- Step 2. Withdraw the amount of ACTEMRA for intravenous infusion from the vial(s) and add slowly into the 0.9% or 0.45% Sodium Chloride Injection, USP infusion bag or bottle. To mix the solution, gently invert the bag to avoid foaming.
- The fully diluted ACTEMRA solutions for infusion using 0.9% Sodium Chloride Injection, USP may be stored at 36°F to 46°F (2°C to 8°C) or room temperature for up to 24 hours and should be protected from light.
- The fully diluted ACTEMRA solutions for infusion using 0.45% Sodium Chloride Injection, USP may be stored at 36°F to 46°F (2°C to 8°C) for up to 24 hours or room temperature for up to 4 hours and should be protected from light.
- ACTEMRA solutions do not contain preservatives; therefore, unused product remaining in the vials should not be used.
- Allow the fully diluted ACTEMRA solution to reach room temperature prior to infusion.
- The infusion should be administered over 60 minutes, and must be administered with an infusion set. Do not administer as an intravenous push or bolus.
- ACTEMRA should not be infused concomitantly in the same intravenous line with other drugs. No physical or biochemical compatibility studies have been conducted to evaluate the co-administration of ACTEMRA with other drugs.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. If particulates and discolorations are noted, the product should not be used.
- Fully diluted ACTEMRA solutions are compatible with polypropylene, polyethylene and polyvinyl chloride infusion bags and polypropylene, polyethylene and glass infusion bottles.
Preparation and Administration Instructions for Subcutaneous Injection
- ACTEMRA for subcutaneous injection is not intended for intravenous drip infusion.
- Assess suitability of patient for subcutaneous home use and instruct patients to inform a healthcare professional before administering the next dose if they experience any symptoms of allergic reaction. Patients should seek immediate medical attention if they develop symptoms of serious allergic reactions. ACTEMRA subcutaneous injection is intended for use under the guidance of a healthcare practitioner. After proper training in subcutaneous injection technique, a patient may self-inject ACTEMRA or the patient's caregiver may administer ACTEMRA if a healthcare practitioner determines that it is appropriate. PJIA and SJIA patients may self-inject with the ACTEMRA prefilled syringe or ACTPen ® autoinjector, or the patient's caregiver may administer ACTEMRA if both the healthcare practitioner and the parent/legal guardian determines it is appropriate [see Use in Specific Populations (8.4) ] . Patients, or patient caregivers, should be instructed to follow the directions provided in the Instructions for Use (IFU) for additional details on medication administration.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use ACTEMRA prefilled syringes (PFS) or prefilled ACTPen ® autoinjectors exhibiting particulate matter, cloudiness, or discoloration. ACTEMRA for subcutaneous administration should be clear and colorless to pale yellow. Do not use if any part of the PFS or ACTPen ® autoinjector appears to be damaged.
- Patients using ACTEMRA for subcutaneous administration should be instructed to inject the full amount in the syringe (0.9 mL) or full amount in the ACTPen ® autoinjector (0.9 mL), which provides 162 mg of ACTEMRA, according to the directions provided in the IFU.
- Injection sites should be rotated with each injection and should never be given into moles, scars, or areas where the skin is tender, bruised, red, hard, or not intact.
Dosage Modifications due to Serious Infections or Laboratory Abnormalities
Serious Infections
Hold ACTEMRA treatment if a patient develops a serious infection until the infection is controlled.
Laboratory Abnormalities
Rheumatoid Arthritis, Giant Cell Arteritis and Systemic Sclerosis-Associated Interstitial Lung Disease
| Liver Enzyme Abnormalities [see Warnings and Precautions (5.3 , 5.4) ] | ||
|---|---|---|
| Lab Value | Recommendation for RA and SSc-ILD | Recommendation for GCA |
| Greater than 1 to 3× ULN | Dose modify concomitant DMARDs if appropriate For persistent increases in this range:
| Dose modify immunomodulatory agents if appropriate For persistent increases in this range:
|
| Greater than 3 to 5× ULN (confirmed by repeat testing) | Hold ACTEMRA dosing until less than 3× ULN and follow recommendations above for greater than 1 to 3× ULN For persistent increases greater than 3× ULN, discontinue ACTEMRA | Hold ACTEMRA dosing until less than 3x ULN and follow recommendations above for greater than 1 to 3x ULN For persistent increases greater than 3x ULN, discontinue ACTEMRA |
| Greater than 5× ULN | Discontinue ACTEMRA | Discontinue ACTEMRA |
| Low Absolute Neutrophil Count (ANC) [see Warnings and Precautions (5.4) ] | ||
|---|---|---|
| Lab Value (cells per mm 3 ) | Recommendation for RA and SSc-ILD | Recommendation for GCA |
| ANC greater than 1000 | Maintain dose | Maintain dose |
| ANC 500 to 1000 | Hold ACTEMRA dosing When ANC greater than 1000 cells per mm 3 :
| Hold ACTEMRA dosing When ANC greater than 1000 cells per mm 3 :
|
| ANC less than 500 | Discontinue ACTEMRA | Discontinue ACTEMRA |
| Low Platelet Count [see Warnings and Precautions (5.4) ] | ||
|---|---|---|
| Lab Value (cells per mm 3 ) | Recommendation for RA and SSc-ILD | Recommendation for GCA |
| 50,000 to 100,000 | Hold ACTEMRA dosing When platelet count is greater than 100,000 cells per mm 3 :
| Hold ACTEMRA dosing When platelet count is greater than 100,000 cells per mm 3 :
|
| Less than 50,000 | Discontinue ACTEMRA | Discontinue ACTEMRA |
Polyarticular and Systemic Juvenile Idiopathic Arthritis
Dose reduction of ACTEMRA has not been studied in the PJIA and SJIA populations. Dose interruptions of ACTEMRA are recommended for liver enzyme abnormalities, low neutrophil counts, and low platelet counts in patients with PJIA and SJIA at levels similar to what is outlined above for patients with RA and GCA. If appropriate, dose modify or stop concomitant methotrexate and/or other medications and hold ACTEMRA dosing until the clinical situation has been evaluated. In PJIA and SJIA the decision to discontinue ACTEMRA for a laboratory abnormality should be based upon the medical assessment of the individual patient.
DOSAGE FORMS AND STRENGTHS
Intravenous Infusion
Injection: 80 mg/4 mL (20 mg/mL), 200 mg/10 mL (20 mg/mL), 400 mg/20 mL (20 mg/mL) in single-dose vials for further dilution prior to intravenous infusion (3 )
Subcutaneous Injection
Injection: 162 mg/0.9 mL in a single-dose prefilled syringe or single-dose prefilled ACTPen ® autoinjector (3 )
Intravenous Infusion
Injection: 80 mg/4 mL, 200 mg/10 mL, 400 mg/20 mL as a clear, colorless to pale yellow solution in 20 mg/mL single-dose vials for further dilution prior to intravenous infusion.
Subcutaneous Injection
Injection: 162 mg/0.9 mL clear, colorless to slightly yellowish solution in a single-dose prefilled syringe or single-dose prefilled ACTPen ® autoinjector.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
The available data with ACTEMRA from a pregnancy exposure registry, retrospective cohort study, pharmacovigilance, and published literature are insufficient to draw conclusions about a drug-associated risk of major birth defects, miscarriage, or other adverse maternal or fetal outcomes. These studies had methodological limitations, including small sample size of tocilizumab exposed groups, missing exposure and outcomes information, and lack of adjustment for confounders. Monoclonal antibodies, such as tocilizumab, are actively transported across the placenta during the third trimester of pregnancy and may affect immune response in the in utero exposed infant [see Clinical Considerations ]. In animal reproduction studies, intravenous administration of tocilizumab to Cynomolgus monkeys during organogenesis caused abortion/embryo-fetal death at doses 1.25 times and higher than the maximum recommended human dose by the intravenous route of 8 mg per kg every 2 to 4 weeks. The literature in animals suggests that inhibition of IL-6 signaling may interfere with cervical ripening and dilatation and myometrial contractile activity leading to potential delays of parturition [see Data ] . Based on the animal data, there may be a potential risk to the fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Fetal/Neonatal adverse reactions
Monoclonal antibodies are increasingly transported across the placenta as pregnancy progresses, with the largest amount transferred during the third trimester. Risks and benefits should be considered prior to administering live or live-attenuated vaccines to infants exposed to ACTEMRA in utero [see Warnings and Precautions 5.9) ] .
Disease-associated Maternal Risk
Published data suggest that the risk of adverse pregnancy outcomes in women with rheumatoid arthritis is associated with increased disease activity. Adverse pregnancy outcomes include preterm delivery (before 37 weeks of gestation), low birth weight (less than 2500 g) infants, and small for gestational age at birth.
Data
Animal Data
An embryo-fetal developmental toxicity study was performed in which pregnant Cynomolgus monkeys were treated intravenously with tocilizumab at daily doses of 2, 10, or 50 mg/ kg during organogenesis from gestation day (GD) 20-50. Although there was no evidence for a teratogenic/dysmorphogenic effect at any dose, tocilizumab produced an increase in the incidence of abortion/embryo-fetal death at doses 1.25 times and higher the MRHD by the intravenous route at maternal intravenous doses of 10 and 50 mg/ kg. Testing of a murine analogue of tocilizumab in mice did not yield any evidence of harm to offspring during the pre- and postnatal development phase when dosed at 50 mg/kg intravenously with treatment every three days from implantation (GD 6) until post-partum day 21 (weaning). There was no evidence for any functional impairment of the development and behavior, learning ability, immune competence and fertility of the offspring.
Parturition is associated with significant increases of IL-6 in the cervix and myometrium. The literature suggests that inhibition of IL-6 signaling may interfere with cervical ripening and dilatation and myometrial contractile activity leading to potential delays of parturition. For mice deficient in IL-6 (ll6 -/- null mice), parturition was delayed relative to wild-type (ll6 +/+ ) mice. Administration of recombinant IL-6 to ll6 -/- null mice restored the normal timing of delivery.
Lactation
Risk Summary
No information is available on the presence of tocilizumab in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. Maternal immunoglobulin G (IgG) is present in human milk. If tocilizumab is transferred into human milk, the effects of local exposure in the gastrointestinal tract and potential limited systemic exposure in the infant to tocilizumab are unknown. The lack of clinical data during lactation precludes clear determination of the risk of ACTEMRA to an infant during lactation; therefore the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for ACTEMRA and the potential adverse effects on the breastfed child from tocilizumab or from the underlying maternal condition.
Pediatric Use
ACTEMRA by intravenous use is indicated for the treatment of pediatric patients with:
- Active systemic juvenile idiopathic arthritis in patients 2 years of age and older
- Active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older
- Severe or life-threatening CAR T cell-induced cytokine release syndrome (CRS) in patients 2 years of age and older.
- Hospitalized Coronavirus disease 2019 (COVID-19) patients 2 years of age and older who are receiving systemic corticosteroids and require supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation (ECMO).
ACTEMRA by subcutaneous use is indicated for the treatment of pediatric patients with:
- Active polyarticular juvenile idiopathic arthritis in patients 2 years of age and older
- Active systemic juvenile idiopathic arthritis in patients 2 years of age and older
The safety and effectiveness of ACTEMRA in pediatric patients with conditions other than PJIA, SJIA, CRS or COVID-19 have not been established. The safety and effectiveness in pediatric patients below the age of 2 have not been established in PJIA, SJIA, CRS or COVID-19.
Systemic Juvenile Idiopathic Arthritis – Intravenous Use
A multicenter, open-label, single arm study to evaluate the PK, safety and exploratory PD and efficacy of ACTEMRA over 12-weeks in SJIA patients (N=11) under 2 years of age was conducted. Patients received intravenous ACTEMRA 12 mg/kg every two weeks. Concurrent use of stable background treatment with corticosteroids, MTX, and/or non-steroidal anti-inflammatory drugs was permitted. Patients who completed the 12-week period could continue to the optional extension period (a total of 52-weeks or until the age of 2 years, whichever was longer).
The primary PK endpoints (C max , C trough and AUC 2weeks ) of ACTEMRA at steady-state in this study were within the ranges of these parameters observed in patients with SJIA aged 2 to 17 years.
The safety and immunogenicity of ACTEMRA for patients with SJIA under 2 years of age was assessed descriptively. SAEs, AEs leading to discontinuation, and infectious AEs were reported by 27.3%, 36.4%, and 81.8% of patients. Six patients (54.5%) experienced hypersensitivity reactions, defined as all adverse events occurring during or within 24 hours after an infusion considered related to ACTEMRA. Three of these patients experienced serious hypersensitivity reactions and were withdrawn from the study. Three patients with hypersensitivity reactions (two with serious hypersensitivity reactions) developed treatment induced anti-tocilizumab antibodies after the event. There were no cases of MAS based on the protocol-specified criteria, but 2 cases of suspected MAS based on Ravelli criteria Ravelli A, Minoia F, Davì S on behalf of the Paediatric Rheumatology International Trials Organisation, the Childhood Arthritis and Rheumatology Research Alliance, the Pediatric Rheumatology Collaborative Study Group, and the Histiocyte Society , et al. 2016 Classification Criteria for Macrophage Activation Syndrome Complicating Systemic Juvenile Idiopathic Arthritis. Annals of the Rheumatic Diseases 2016;75:481-489. .
Cytokine Release Syndrome – Intravenous Use
In the retrospective analysis of pooled outcome data for patients treated with ACTEMRA for CAR T cell-induced CRS, 25 patients were children (2 years up to 12 years of age), and 17 patients were adolescents (12 years up to 18 years of age). There were no differences between the pediatric patients and the adults for safety or efficacy.
COVID-19 – Intravenous Use
The safety and effectiveness of ACTEMRA have been established for the treatment of pediatric patients aged 2 years and older with COVID-19. Use of ACTEMRA in these age groups is supported by evidence from adequate and well-controlled studies of ACTEMRA in adults with COVID-19 [See Adverse Reactions (6.11) and Clinical Studies (14.11) ] , predicted pharmacokinetic data modeling and simulation for pediatric patients aged 2 years and older with COVID-19 [See Clinical Pharmacology (12.3) ] , safety data from a randomized, double-blind, placebo-controlled study of ACTEMRA in pediatric patients aged 2 years and older with systemic juvenile idiopathic arthritis, and additional uncontrolled pharmacovigilance data in pediatric patients aged 2 years and older with COVID-19 who received ACTEMRA under Emergency Use Authorization.
The safety and effectiveness of ACTEMRA has not been established in pediatric COVID-19 patients younger than 2 years of age.
Geriatric Use
Of the 2644 patients who received ACTEMRA in Studies I to V [see Clinical Studies (14) ] , a total of 435 rheumatoid arthritis patients were 65 years of age and older, including 50 patients 75 years and older. Of the 1069 patients who received ACTEMRA-SC in studies SC-I and SC-II there were 295 patients 65 years of age and older, including 41 patients 75 years and older. The frequency of serious infection among ACTEMRA treated subjects 65 years of age and older was higher than those under the age of 65. As there is a higher incidence of infections in the elderly population in general, caution should be used when treating the elderly.
Clinical studies that included ACTEMRA for CRS did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients.
In the EMPACTA, COVACTA, and REMDACTA studies, of the 974 COVID-19 patients in the ACTEMRA arm, 375 (39%) were 65 years of age or older. No overall differences in safety or effectiveness of ACTEMRA were observed between patients 65 years of age and older and those under the age of 65 years of age in these studies [see Adverse Reactions (6.1) and Clinical Studies (14.11) ] .
In the RECOVERY study, of the 2022 COVID-19 patients in the ACTEMRA arm, 930 (46%) were 65 years of age or older. No overall differences in effectiveness of ACTEMRA were observed between patients 65 years of age and older and those under the age 65 years of age in this study [see Clinical Studies (14.11) ] .
Hepatic Impairment
The safety and efficacy of ACTEMRA have not been studied in patients with hepatic impairment, including patients with positive HBV and HCV serology [see Warnings and Precautions (5.8) ] .
Renal Impairment
No dose adjustment is required in patients with mild or moderate renal impairment. ACTEMRA has not been studied in patients with severe renal impairment [see Clinical Pharmacology (12.3) ] .
CONTRAINDICATIONS
ACTEMRA is contraindicated in patients with known hypersensitivity to ACTEMRA [see Warnings and Precautions (5.6) ].
WARNINGS AND PRECAUTIONS
- Serious Infections – do not administer ACTEMRA during an active infection, including localized infections. If a serious infection develops, interrupt ACTEMRA until the infection is controlled. (5.1 )
- Gastrointestinal (GI) perforation—use with caution in patients who may be at increased risk. (5.2 )
- Hepatotoxicity- Monitor patients for signs and symptoms of hepatic injury. Modify or discontinue ACTEMRA if abnormal liver tests persist or worsen or if clinical signs and symptoms of liver disease develop. (2.10 , 5.3 )
- Laboratory monitoring—recommended due to potential consequences of treatment-related changes in neutrophils, platelets, lipids, and liver function tests. (2.10 , 5.4 )
- Hypersensitivity reactions, including anaphylaxis and death, and serious cutaneous reactions including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) – discontinue ACTEMRA, treat promptly, and monitor until reaction resolves. (5.6 )
- Live vaccines—Avoid use with ACTEMRA. (5.9 , 7.3 )
Serious Infections
Serious and sometimes fatal infections due to bacterial, mycobacterial, invasive fungal, viral, protozoal, or other opportunistic pathogens have been reported in patients receiving immunosuppressive agents including ACTEMRA. The most common serious infections included pneumonia, urinary tract infection, cellulitis, herpes zoster, gastroenteritis, diverticulitis, sepsis and bacterial arthritis [see Adverse Reactions (6.1) ] . Among opportunistic infections, tuberculosis, cryptococcus, aspergillosis, candidiasis, and pneumocystosis were reported with ACTEMRA. Other serious infections, not reported in clinical studies, may also occur (e.g., histoplasmosis, coccidioidomycosis, listeriosis). Patients have presented with disseminated rather than localized disease, and were often taking concomitant immunosuppressants such as methotrexate or corticosteroids which in addition to rheumatoid arthritis may predispose them to infections.
Do not administer ACTEMRA in patients with an active infection, including localized infections. The risks and benefits of treatment should be considered prior to initiating ACTEMRA in patients:
- with chronic or recurrent infection;
- who have been exposed to tuberculosis;
- with a history of serious or an opportunistic infection;
- who have resided or traveled in areas of endemic tuberculosis or endemic mycoses; or
- with underlying conditions that may predispose them to infection.
Closely monitor patients for the development of signs and symptoms of infection during and after treatment with ACTEMRA, as signs and symptoms of acute inflammation may be lessened due to suppression of the acute phase reactants [see Dosage and Administration (2.8) , Adverse Reactions (6.1) , and Patient Counseling Information (17) ] .
Hold ACTEMRA if a patient develops a serious infection, an opportunistic infection, or sepsis. A patient who develops a new infection during treatment with ACTEMRA should undergo a prompt and complete diagnostic workup appropriate for an immunocompromised patient, initiate appropriate antimicrobial therapy, and closely monitor the patient.
COVID-19
In patients with COVID-19, monitor for signs and symptoms of new infections during and after treatment with ACTEMRA. There is limited information regarding the use of ACTEMRA in patients with COVID-19 and concomitant active serious infections. The risks and benefits of treatment with ACTEMRA in COVID-19 patients with other concurrent infections should be considered.
Tuberculosis
Evaluate patients for tuberculosis risk factors and test for latent infection prior to initiating ACTEMRA. In patients with COVID-19, testing for latent infection is not necessary prior to initiating treatment with ACTEMRA.
Consider anti-tuberculosis therapy prior to initiation of ACTEMRA in patients with a past history of latent or active tuberculosis in whom an adequate course of treatment cannot be confirmed, and for patients with a negative test for latent tuberculosis but having risk factors for tuberculosis infection. Consultation with a physician with expertise in the treatment of tuberculosis is recommended to aid in the decision whether initiating anti-tuberculosis therapy is appropriate for an individual patient.
Closely monitor patients for the development of signs and symptoms of tuberculosis including patients who tested negative for latent tuberculosis infection prior to initiating therapy.
The incidence of tuberculosis in worldwide clinical development programs is 0.1%. Patients with latent tuberculosis should be treated with standard antimycobacterial therapy before initiating ACTEMRA.
Viral Reactivation
Viral reactivation has been reported with immunosuppressive biologic therapies and cases of herpes zoster exacerbation were observed in clinical studies with ACTEMRA. No cases of Hepatitis B reactivation were observed in the trials; however patients who screened positive for hepatitis were excluded.
Gastrointestinal Perforations
Events of gastrointestinal perforation have been reported in clinical trials, primarily as complications of diverticulitis in patients treated with ACTEMRA. Use ACTEMRA with caution in patients who may be at increased risk for gastrointestinal perforation. Promptly evaluate patients presenting with fever, new onset abdominal symptoms, and a change in bowel habits for early identification of gastrointestinal perforation [see Adverse Reactions (6.1) ] .
Hepatotoxicity
Serious cases of hepatic injury have been observed in patients taking intravenous or subcutaneous ACTEMRA. Some of these cases have resulted in liver transplant or death. Time to onset for cases ranged from months to years after treatment initiation with tocilizumab. While most cases presented with marked elevations of transaminases (> 5 times ULN), some cases presented with signs or symptoms of liver dysfunction and only mildly elevated transaminases.
During randomized controlled studies, treatment with ACTEMRA was associated with a higher incidence of transaminase elevations [see Adverse Reactions (6.1 , 6.2 , 6.6 , 6.8) ]. Increased frequency and magnitude of these elevations was observed when potentially hepatotoxic drugs (e.g., MTX) were used in combination with ACTEMRA.
For RA, GCA and SSc-ILD patients, obtain a liver test panel (serum alanine aminotransferase [ALT], aspartate aminotransferase [AST], alkaline phosphatase, and total bilirubin) before initiating ACTEMRA, every 4 to 8 weeks after start of therapy for the first 6 months of treatment and every 3 months thereafter. It is not recommended to initiate ACTEMRA treatment in RA, GCA or SSc-ILD patients with elevated transaminases ALT or AST greater than 1.5× ULN. In patients who develop elevated ALT or AST greater than 5× ULN, discontinue ACTEMRA. For recommended modifications based upon increase in transaminases see Dosage and Administration (2.11) .
Patients hospitalized with COVID-19 may have elevated ALT or AST levels. Multi-organ failure with involvement of the liver is recognized as a complication of severe COVID-19. The decision to administer ACTEMRA should balance the potential benefit of treating COVID-19 against the potential risks of acute treatment with ACTEMRA. It is not recommended to initiate ACTEMRA treatment in COVID-19 patients with elevated ALT or AST above 10 × ULN. Monitor ALT and AST during treatment.
Measure liver tests promptly in patients who report symptoms that may indicate liver injury, such as fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice. In this clinical context, if the patient is found to have abnormal liver tests (e.g., ALT greater than three times the upper limit of the reference range, serum total bilirubin greater than two times the upper limit of the reference range), ACTEMRA treatment should be interrupted and investigation done to establish the probable cause. ACTEMRA should only be restarted in patients with another explanation for the liver test abnormalities after normalization of the liver tests.
A similar pattern of liver enzyme elevation is noted with ACTEMRA treatment in the PJIA and SJIA populations. Monitor liver test panel at the time of the second administration and thereafter every 4 to 8 weeks for PJIA and every 2 to 4 weeks for SJIA.
Changes in Laboratory Parameters
Patients with Rheumatoid Arthritis, Giant Cell Arteritis, Systemic Sclerosis-Associated Interstitial Lung Disease and Coronavirus Disease 2019
Neutropenia
Treatment with ACTEMRA was associated with a higher incidence of neutropenia. Infections have been uncommonly reported in association with treatment-related neutropenia in long-term extension studies and postmarketing clinical experience.
- It is not recommended to initiate ACTEMRA treatment in RA, GCA and SSc-ILD patients with a low neutrophil count, i.e., absolute neutrophil count (ANC) less than 2000 per mm 3 . In patients who develop an absolute neutrophil count less than 500 per mm 3 treatment is not recommended.
- Monitor neutrophils 4 to 8 weeks after start of therapy and every 3 months thereafter [see Clinical Pharmacology (12.2) ] . For recommended modifications based on ANC results see Dosage and Administration (2.11) .
- It is not recommended to initiate ACTEMRA treatment in COVID-19 patients with an ANC less than 1000 per mm 3 . Neutrophils should be monitored.
Thrombocytopenia
Treatment with ACTEMRA was associated with a reduction in platelet counts. Treatment-related reduction in platelets was not associated with serious bleeding events in clinical trials [see Adverse Reactions (6.1 , 6.2) ] .
- It is not recommended to initiate ACTEMRA treatment in RA, GCA and SSc-ILD patients with a platelet count below 100,000 per mm 3 . In patients who develop a platelet count less than 50,000 per mm 3 treatment is not recommended.
- Monitor platelets 4 to 8 weeks after start of therapy and every 3 months thereafter. For recommended modifications based on platelet counts see Dosage and Administration (2.11) .
- In COVID-19 patients with a platelet count less than 50,000 per mm 3 , treatment is not recommended. Platelets should be monitored.
Elevated Liver Enzymes
Refer to 5.3 Hepatotoxicity. For recommended modifications [see Dosage and Administration (2.11) ]
Lipid Abnormalities
Treatment with ACTEMRA was associated with increases in lipid parameters such as total cholesterol, triglycerides, LDL cholesterol, and/or HDL cholesterol [see Adverse Reactions (6.1 , 6.2) ] .
- Assess lipid parameters approximately 4 to 8 weeks following initiation of ACTEMRA therapy.
- Subsequently, manage patients according to clinical guidelines [e.g., National Cholesterol Educational Program (NCEP)] for the management of hyperlipidemia.
Patients with Polyarticular and Systemic Juvenile Idiopathic Arthritis
A similar pattern of liver enzyme elevation, low neutrophil count, low platelet count and lipid elevations is noted with ACTEMRA treatment in the PJIA and SJIA populations. Monitor neutrophils, platelets, ALT and AST at the time of the second administration and thereafter every 4 to 8 weeks for PJIA and every 2 to 4 weeks for SJIA. Monitor lipids as above for approved adult indications [see Dosage and Administration (2.11) ] .
Immunosuppression
The impact of treatment with ACTEMRA on the development of malignancies is not known but malignancies were observed in clinical studies [see Adverse Reactions (6.1) ] . ACTEMRA is an immunosuppressant, and treatment with immunosuppressants may result in an increased risk of malignancies.
Hypersensitivity Reactions, Including Anaphylaxis
Hypersensitivity reactions, including anaphylaxis, have been reported in association with ACTEMRA and anaphylactic events with a fatal outcome have been reported with intravenous infusion of ACTEMRA. Anaphylaxis and other hypersensitivity reactions that required treatment discontinuation were reported in 0.1% (3 out of 2644) of patients in the 6-month controlled trials of intravenous ACTEMRA, 0.2% (8 out of 4009) of patients in the intravenous all-exposure RA population, 0.7% (8 out of 1068) in the subcutaneous 6-month controlled RA trials, and in 0.7% (10 out of 1465) of patients in the subcutaneous all-exposure population. In the SJIA controlled trial with intravenous ACTEMRA, 1 out of 112 patients (0.9%) experienced hypersensitivity reactions that required treatment discontinuation. In the PJIA controlled trial with intravenous ACTEMRA, 0 out of 188 patients (0%) in the ACTEMRA all-exposure population experienced hypersensitivity reactions that required treatment discontinuation. Reactions that required treatment discontinuation included generalized erythema, rash, and urticaria. Injection site reactions were categorized separately [s ee Adverse Reactions (6) ] .
In the postmarketing setting, events of hypersensitivity reactions, including anaphylaxis and death have occurred in patients treated with a range of doses of intravenous ACTEMRA, with or without concomitant therapies. Events have occurred in patients who received premedication. Hypersensitivity, including anaphylaxis events, have occurred both with and without previous hypersensitivity reactions and as early as the first infusion of ACTEMRA [see Adverse Reactions (6.12) ] . In addition, serious cutaneous reactions, including Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), have been reported in patients with autoinflammatory conditions treated with ACTEMRA.
ACTEMRA for intravenous use should only be infused by a healthcare professional with appropriate medical support to manage anaphylaxis. For ACTEMRA subcutaneous injection, advise patients to seek immediate medical attention if they experience any symptoms of a hypersensitivity reaction. If a hypersensitivity reaction occurs, immediately discontinue ACTEMRA, treat promptly and monitor until signs and symptoms resolve.
Demyelinating Disorders
The impact of treatment with ACTEMRA on demyelinating disorders is not known, but multiple sclerosis and chronic inflammatory demyelinating polyneuropathy were reported rarely in RA clinical studies. Monitor patients for signs and symptoms potentially indicative of demyelinating disorders. Prescribers should exercise caution in considering the use of ACTEMRA in patients with preexisting or recent onset demyelinating disorders.
Active Hepatic Disease and Hepatic Impairment
Treatment with ACTEMRA is not recommended in patients with active hepatic disease or hepatic impairment [see Adverse Reactions (6.1) , Use in Specific Populations (8.6) ] .
Vaccinations
Avoid use of live vaccines concurrently with ACTEMRA as clinical safety has not been established. No data are available on the secondary transmission of infection from persons receiving live vaccines to patients receiving ACTEMRA.
No data are available on the effectiveness of vaccination in patients receiving ACTEMRA. Because IL-6 inhibition may interfere with the normal immune response to new antigens, it is recommended that all patients, particularly pediatric or elderly patients, if possible, be brought up to date with all immunizations in agreement with current immunization guidelines prior to initiating ACTEMRA therapy. The interval between live vaccinations and initiation of ACTEMRA therapy should be in accordance with current vaccination guidelines regarding immunosuppressive agents.
ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in labeling:
- Serious Infections [see Warnings and Precautions (5.1) ]
- Gastrointestinal Perforations [see Warnings and Precautions (5.2) ]
- Laboratory Parameters [see Warnings and Precautions (5.4) ]
- Immunosuppression [see Warnings and Precautions (5.5) ]
- Hypersensitivity Reactions, Including Anaphylaxis [see Warnings and Precautions (5.6) ]
- Demyelinating Disorders [see Warnings and Precautions (5.7) ]
- Active Hepatic Disease and Hepatic Impairment [see Warnings and Precautions (5.8) ]
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not predict the rates observed in a broader patient population in clinical practice.
Clinical Trials Experience in Rheumatoid Arthritis Patients Treated with Intravenous ACTEMRA (ACTEMRA-IV)
The ACTEMRA-IV data in rheumatoid arthritis (RA) includes 5 double-blind, controlled, multicenter studies. In these studies, patients received doses of ACTEMRA-IV 8 mg per kg monotherapy (288 patients), ACTEMRA-IV 8 mg per kg in combination with DMARDs (including methotrexate) (1582 patients), or ACTEMRA-IV 4 mg per kg in combination with methotrexate (774 patients).
The all exposure population includes all patients in registration studies who received at least one dose of ACTEMRA-IV. Of the 4009 patients in this population, 3577 received treatment for at least 6 months, 3309 for at least one year; 2954 received treatment for at least 2 years and 2189 for 3 years.
All patients in these studies had moderately to severely active rheumatoid arthritis. The study population had a mean age of 52 years, 82% were female and 74% were Caucasian.
The most common serious adverse reactions were serious infections [see Warnings and Precautions (5.1) ] . The most commonly reported adverse reactions in controlled studies up to 24 weeks (occurring in at least 5% of patients treated with ACTEMRA-IV monotherapy or in combination with DMARDs) were upper respiratory tract infections, nasopharyngitis, headache, hypertension and increased ALT.
The proportion of patients who discontinued treatment due to any adverse reactions during the double-blind, placebo-controlled studies was 5% for patients taking ACTEMRA-IV and 3% for placebo-treated patients. The most common adverse reactions that required discontinuation of ACTEMRA-IV were increased hepatic transaminase values (per protocol requirement) and serious infections.
Overall Infections
In the 24 week, controlled clinical studies, the rate of infections in the ACTEMRA-IV monotherapy group was 119 events per 100 patient-years and was similar in the methotrexate monotherapy group. The rate of infections in the 4 mg per kg and 8 mg per kg ACTEMRA-IV plus DMARD group was 133 and 127 events per 100 patient-years, respectively, compared to 112 events per 100 patient-years in the placebo plus DMARD group. The most commonly reported infections (5% to 8% of patients) were upper respiratory tract infections and nasopharyngitis.
The overall rate of infections with ACTEMRA-IV in the all exposure population remained consistent with rates in the controlled periods of the studies.
Serious Infections
In the 24 week, controlled clinical studies, the rate of serious infections in the ACTEMRA-IV monotherapy group was 3.6 per 100 patient-years compared to 1.5 per 100 patient-years in the methotrexate group. The rate of serious infections in the 4 mg per kg and 8 mg per kg ACTEMRA-IV plus DMARD group was 4.4 and 5.3 events per 100 patient-years, respectively, compared to 3.9 events per 100 patient-years in the placebo plus DMARD group.
In the all-exposure population, the overall rate of serious infections remained consistent with rates in the controlled periods of the studies. The most common serious infections included pneumonia, urinary tract infection, cellulitis, herpes zoster, gastroenteritis, diverticulitis, sepsis and bacterial arthritis. Cases of opportunistic infections have been reported [see Warnings and Precautions (5.1) ] .
In the cardiovascular outcomes Study WA25204, the rate of serious infections in the ACTEMRA 8 mg/kg IV every 4 weeks group, with or without DMARD, was 4.5 per 100 patient-years, and the rate in the etanercept 50 mg weekly SC group, with or without DMARD, was 3.2 per 100 patient-years [see Clinical Studies (14.1) ] .
Gastrointestinal Perforations
During the 24 week, controlled clinical trials, the overall rate of gastrointestinal perforation was 0.26 events per 100 patient-years with ACTEMRA-IV therapy.
In the all-exposure population, the overall rate of gastrointestinal perforation remained consistent with rates in the controlled periods of the studies. Reports of gastrointestinal perforation were primarily reported as complications of diverticulitis including generalized purulent peritonitis, lower GI perforation, fistula and abscess. Most patients who developed gastrointestinal perforations were taking concomitant nonsteroidal anti-inflammatory medications (NSAIDs), corticosteroids, or methotrexate [see Warnings and Precautions (5.2) ] . The relative contribution of these concomitant medications versus ACTEMRA-IV to the development of GI perforations is not known.
Infusion Reactions
In the 24 week, controlled clinical studies, adverse events associated with the infusion (occurring during or within 24 hours of the start of infusion) were reported in 8% and 7% of patients in the 4 mg per kg and 8 mg per kg ACTEMRA-IV plus DMARD group, respectively, compared to 5% of patients in the placebo plus DMARD group. The most frequently reported event on the 4 mg per kg and 8 mg per kg dose during the infusion was hypertension (1% for both doses), while the most frequently reported event occurring within 24 hours of finishing an infusion were headache (1% for both doses) and skin reactions (1% for both doses), including rash, pruritus and urticaria. These events were not treatment limiting.
Anaphylaxis
Hypersensitivity reactions requiring treatment discontinuation, including anaphylaxis, associated with ACTEMRA-IV were reported in 0.1% (3 out of 2644) in the 24 week, controlled trials and in 0.2% (8 out of 4009) in the all-exposure population. These reactions were generally observed during the second to fourth infusion of ACTEMRA-IV. Appropriate medical treatment should be available for immediate use in the event of a serious hypersensitivity reaction [see Warnings and Precautions (5.6) ] .
Laboratory Abnormalities
Neutropenia
In the 24 week, controlled clinical studies, decreases in neutrophil counts below 1000 per mm 3 occurred in 1.8% and 3.4% of patients in the 4 mg per kg and 8 mg per kg ACTEMRA-IV plus DMARD group, respectively, compared to 0.1% of patients in the placebo plus DMARD group. Approximately half of the instances of ANC below 1000 per mm 3 occurred within 8 weeks of starting therapy. Decreases in neutrophil counts below 500 per mm 3 occurred in 0.4% and 0.3% of patients in the 4 mg per kg and 8 mg per kg ACTEMRA-IV plus DMARD, respectively, compared to 0.1% of patients in the placebo plus DMARD group. There was no clear relationship between decreases in neutrophils below 1000 per mm 3 and the occurrence of serious infections.
In the all-exposure population, the pattern and incidence of decreases in neutrophil counts remained consistent with what was seen in the 24 week controlled clinical studies [see Warnings and Precautions (5.4) ] .
Thrombocytopenia
In the 24 week, controlled clinical studies, decreases in platelet counts below 100,000 per mm 3 occurred in 1.3% and 1.7% of patients on 4 mg per kg and 8 mg per kg ACTEMRA-IV plus DMARD, respectively, compared to 0.5% of patients on placebo plus DMARD, without associated bleeding events.
In the all-exposure population, the pattern and incidence of decreases in platelet counts remained consistent with what was seen in the 24 week controlled clinical studies [see Warnings and Precautions (5.4) ] .
Elevated Liver Enzymes
Liver enzyme abnormalities are summarized in Table 1 . In patients experiencing liver enzyme elevation, modification of treatment regimen, such as reduction in the dose of concomitant DMARD, interruption of ACTEMRA-IV, or reduction in ACTEMRA-IV dose, resulted in decrease or normalization of liver enzymes [see Dosage and Administration (2.11) ] . These elevations were not associated with clinically relevant increases in direct bilirubin, nor were they associated with clinical evidence of hepatitis or hepatic insufficiency [see Warnings and Precautions (5.3 , 5.4) ] .
| ACTEMRA 8 mg per kg MONOTHERAPY | Methotrexate | ACTEMRA 4 mg per kg + DMARDs | ACTEMRA 8 mg per kg + DMARDs | Placebo + DMARDs | |
|---|---|---|---|---|---|
| N = 288 (%) | N = 284 (%) | N = 774 (%) | N = 1582 (%) | N = 1170 (%) | |
| ULN = Upper Limit of Normal | |||||
| AST (U/L) | |||||
| > ULN to 3× ULN | 22 | 26 | 34 | 41 | 17 |
| > 3× ULN to 5× ULN | 0.3 | 2 | 1 | 2 | 0.3 |
| > 5× ULN | 0.7 | 0.4 | 0.1 | 0.2 | < 0.1 |
| ALT (U/L) | |||||
| > ULN to 3× ULN | 36 | 33 | 45 | 48 | 23 |
| > 3× ULN to 5× ULN | 1 | 4 | 5 | 5 | 1 |
| > 5× ULN | 0.7 | 1 | 1.3 | 1.5 | 0.3 |
In the all-exposure population, the elevations in ALT and AST remained consistent with what was seen in the 24 week, controlled clinical trials.
In Study WA25204, of the 1538 patients with moderate to severe RA [see Clinical Studies (14.1) ] and treated with tocilizumab, elevations in ALT or AST >3 × ULN occurred in 5.3% and 2.2% patients, respectively. One serious event of drug induced hepatitis with hyperbilirubinemia was reported in association with tocilizumab.
Lipids
Elevations in lipid parameters (total cholesterol, LDL, HDL, triglycerides) were first assessed at 6 weeks following initiation of ACTEMRA-IV in the controlled 24 week clinical trials. Increases were observed at this time point and remained stable thereafter. Increases in triglycerides to levels above 500 mg per dL were rarely observed. Changes in other lipid parameters from baseline to week 24 were evaluated and are summarized below:
- Mean LDL increased by 13 mg per dL in the ACTEMRA 4 mg per kg+DMARD arm, 20 mg per dL in the ACTEMRA 8 mg per kg+DMARD, and 25 mg per dL in ACTEMRA 8 mg per kg monotherapy.
- Mean HDL increased by 3 mg per dL in the ACTEMRA 4 mg per kg+DMARD arm, 5 mg per dL in the ACTEMRA 8 mg per kg+DMARD, and 4 mg per dL in ACTEMRA 8 mg per kg monotherapy.
- Mean LDL/HDL ratio increased by an average of 0.14 in the ACTEMRA 4 mg per kg+DMARD arm, 0.15 in the ACTEMRA 8 mg per kg+DMARD, and 0.26 in ACTEMRA 8 mg per kg monotherapy.
- ApoB/ApoA1 ratios were essentially unchanged in ACTEMRA-treated patients.
Elevated lipids responded to lipid lowering agents.
In the all-exposure population, the elevations in lipid parameters remained consistent with what was seen in the 24 week, controlled clinical trials.
Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to tocilizumab in the studies described below with the incidence of antibodies in other studies or to other products may be misleading.
In the 24 week, controlled clinical studies, a total of 2876 patients have been tested for anti-tocilizumab antibodies. Forty-six patients (2%) developed positive anti-tocilizumab antibodies, of whom 5 had an associated, medically significant, hypersensitivity reaction leading to withdrawal. Thirty patients (1%) developed neutralizing antibodies.
Malignancies
During the 24 week, controlled period of the studies, 15 malignancies were diagnosed in patients receiving ACTEMRA-IV, compared to 8 malignancies in patients in the control groups. Exposure-adjusted incidence was similar in the ACTEMRA-IV groups (1.32 events per 100 patient-years) and in the placebo plus DMARD group (1.37 events per 100 patient-years).
In the all-exposure population, the rate of malignancies remained consistent with the rate observed in the 24 week, controlled period [see Warnings and Precautions (5.5) ] .
Other Adverse Reactions
Adverse reactions occurring in 2% or more of patients on 4 or 8 mg per kg ACTEMRA-IV plus DMARD and at least 1% greater than that observed in patients on placebo plus DMARD are summarized in Table 2 .
| 24 Week Phase 3 Controlled Study Population | |||||
|---|---|---|---|---|---|
| ACTEMRA 8 mg per kg MONOTHERAPY | Methotrexate | ACTEMRA 4 mg per kg + DMARDs | ACTEMRA 8 mg per kg + DMARDs | Placebo + DMARDs | |
| Preferred Term | N = 288 (%) | N = 284 (%) | N = 774 (%) | N = 1582 (%) | N = 1170 (%) |
| Upper Respiratory Tract Infection | 7 | 5 | 6 | 8 | 6 |
| Nasopharyngitis | 7 | 6 | 4 | 6 | 4 |
| Headache | 7 | 2 | 6 | 5 | 3 |
| Hypertension | 6 | 2 | 4 | 4 | 3 |
| ALT increased | 6 | 4 | 3 | 3 | 1 |
| Dizziness | 3 | 1 | 2 | 3 | 2 |
| Bronchitis | 3 | 2 | 4 | 3 | 3 |
| Rash | 2 | 1 | 4 | 3 | 1 |
| Mouth Ulceration | 2 | 2 | 1 | 2 | 1 |
| Abdominal Pain Upper | 2 | 2 | 3 | 3 | 2 |
| Gastritis | 1 | 2 | 1 | 2 | 1 |
| Transaminase increased | 1 | 5 | 2 | 2 | 1 |
Other infrequent and medically relevant adverse reactions occurring at an incidence less than 2% in rheumatoid arthritis patients treated with ACTEMRA-IV in controlled trials were:
Infections and Infestations: oral herpes simplex
Gastrointestinal disorders: stomatitis, gastric ulcer
Investigations: weight increased, total bilirubin increased
Blood and lymphatic system disorders: leukopenia
General disorders and administration site conditions: edema peripheral
Respiratory, thoracic, and mediastinal disorders : dyspnea, cough
Eye disorders: conjunctivitis
Renal disorders: nephrolithiasis
Endocrine disorders: hypothyroidism
Clinical Trials Experience in Rheumatoid Arthritis Patients Treated with Subcutaneous ACTEMRA (ACTEMRA-SC)
The ACTEMRA-SC data in rheumatoid arthritis (RA) includes 2 double-blind, controlled, multicenter studies. Study SC-I was a non-inferiority study that compared the efficacy and safety of tocilizumab 162 mg administered every week subcutaneously and 8 mg/kg intravenously every four weeks in 1262 adult subjects with rheumatoid arthritis. Study SC-II was a placebo controlled superiority study that evaluated the safety and efficacy of tocilizumab 162 mg administered every other week subcutaneously or placebo in 656 patients. All patients in both studies received background non-biologic DMARDs.
The safety observed for ACTEMRA-SC administered subcutaneously was consistent with the known safety profile of intravenous ACTEMRA, with the exception of injection site reactions (ISRs), which were more common with ACTEMRA-SC compared with placebo SC injections (IV arm).
Injection Site Reactions
In the 6-month control period, in SC-I, the frequency of ISRs was 10.1% (64/631) and 2.4% (15/631) for the weekly ACTEMRA-SC and placebo SC (IV-arm) groups, respectively. In SC-II, the frequency of ISRs was 7.1% (31/437) and 4.1% (9/218) for the every other week ACTEMRA-SC and placebo groups, respectively. These ISRs (including erythema, pruritus, pain and hematoma) were mild to moderate in severity. The majority resolved without any treatment and none necessitated drug discontinuation.
Immunogenicity
In the 6-month control period in SC-I, 0.8% (5/625) in the ACTEMRA-SC arm and 0.8% (5/627) in the IV arm developed anti-tocilizumab antibodies; of these, all developed neutralizing antibodies. In SC-II, 1.6% (7/434) in the ACTEMRA-SC arm compared with 1.4 % (3/217) in the placebo arm developed anti- tocilizumab antibodies; of these, 1.4% (6/434) in the ACTEMRA-SC arm and 0.5% (1/217) in the placebo arm also developed neutralizing antibodies.
A total of 1454 (>99%) patients who received ACTEMRA-SC in the all exposure group have been tested for anti-tocilizumab antibodies. Thirteen patients (0.9%) developed anti-tocilizumab antibodies, and, of these, 12 patients (0.8%) developed neutralizing antibodies.
The rate is consistent with previous intravenous experience. No correlation of antibody development to adverse events or loss of clinical response was observed.
Laboratory Abnormalities
Neutropenia
During routine laboratory monitoring in the 6-month controlled clinical trials, a decrease in neutrophil count below 1 × 10 9 /L occurred in 2.9% and 3.7% of patients receiving ACTEMRA-SC weekly and every other week, respectively.
There was no clear relationship between decreases in neutrophils below 1 × 10 9 /L and the occurrence of serious infections.
Thrombocytopenia
During routine laboratory monitoring in the ACTEMRA-SC 6-month controlled clinical trials, none of the patients had a decrease in platelet count to ≤50,000/mm 3 .
Elevated Liver Enzymes
During routine laboratory monitoring in the 6-month controlled clinical trials, elevation in ALT or AST ≥3 × ULN occurred in 6.5% and 1.4% of patients, respectively, receiving ACTEMRA-SC weekly and 3.4% and 0.7% receiving ACTEMRA-SC every other week.
Lipid Parameters Elevations
During routine laboratory monitoring in the ACTEMRA-SC 6-month clinical trials, 19% of patients dosed weekly and 19.6% of patients dosed every other week and 10.2% of patients on placebo experienced sustained elevations in total cholesterol > 6.2 mmol/l (240 mg/dL), with 9%, 10.4% and 5.1% experiencing a sustained increase in LDL to 4.1 mmol/l (160 mg/dL) receiving ACTEMRA-SC weekly, every other week and placebo, respectively.
Clinical Trials Experience in Giant Cell Arteritis Patients Treated with Subcutaneous ACTEMRA (ACTEMRA-SC)
The safety of subcutaneous ACTEMRA (tocilizumab) has been studied in one Phase III study (WA28119) with 251 GCA patients. The total patient years duration in the ACTEMRA-SC GCA all exposure population was 138.5 patient years during the 12-month double blind, placebo-controlled phase of the study. The overall safety profile observed in the ACTEMRA-SC treatment groups was generally consistent with the known safety profile of ACTEMRA. There was an overall higher incidence of infections in GCA patients relative to RA patients. The rate of infection/serious infection events was 200.2/9.7 events per 100 patient years in the ACTEMRA-SC weekly group and 160.2/4.4 events per 100 patient years in the ACTEMRA-SC every other week group as compared to 156.0/4.2 events per 100 patient years in the placebo + 26 week prednisone taper and 210.2/12.5 events per 100 patient years in the placebo + 52 week taper groups.
Clinical Trials Experience in Giant Cell Arteritis Patients Treated With Intravenous ACTEMRA (ACTEMRA-IV)
The safety of ACTEMRA-IV was studied in an open label PK-PD and safety study in 24 patients with GCA who were in remission on ACTEMRA-IV at time of enrollment. Patients received ACTEMRA 7 mg/kg every 4 weeks for 20 weeks, followed by 6 mg/kg every 4 weeks for 20 weeks. The total patient years exposure to treatment was 17.5 years. The overall safety profile observed for ACTEMRA administered intravenously in GCA patients was consistent with the known safety profile of ACTEMRA.
Clinical Trials Experience in Systemic Sclerosis-Associated Interstitial Lung Disease Patients Treated with Subcutaneous ACTEMRA (ACTEMRA-SC)
The safety of subcutaneous ACTEMRA was evaluated in two double-blind, placebo-controlled, multicenter studies (WA29767 and WA27788). In the Phase 3 Study WA29767, 212 patients with SSc were randomized to tocilizumab 162 mg administered every week subcutaneously or placebo for 48 weeks, followed by open-label tocilizumab 162 mg administered subcutaneously every week for another 48 weeks. In the Phase 2/3 Study WA27788, 87 patients were randomized to tocilizumab 162 mg administered every week subcutaneously or placebo for 48 weeks, followed by open-label tocilizumab 162 mg administered subcutaneously every week for another 48 weeks.
The safety profile for ACTEMRA through week 48 in WA29767 was comparable for SSc-ILD and SSc patients overall, and in both studies was consistent with the known safety profile of ACTEMRA.
Immunogenicity
In the two clinical studies, WA29767 and WA27788, the incidence of treatment-induced anti-TCZ antibodies at week 96 was low (3 out of 169 patients, 1.8%). These anti-drug antibodies were of neutralizing potential, and none of the patients experienced hypersensitivity reactions.
Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Intravenous ACTEMRA (ACTEMRA-IV)
The safety of ACTEMRA-IV was studied in 188 pediatric patients 2 to 17 years of age with PJIA who had an inadequate clinical response or were intolerant to methotrexate. The total patient exposure in the ACTEMRA-IV all exposure population (defined as patients who received at least one dose of ACTEMRA-IV) was 184.4 patient years. At baseline, approximately half of the patients were taking oral corticosteroids and almost 80% were taking methotrexate. In general, the types of adverse drug reactions in patients with PJIA were consistent with those seen in RA and SJIA patients [see Adverse Reactions (6.1 and 6.8) ] .
Infections
The rate of infections in the ACTEMRA-IV all exposure population was 163.7 per 100 patient years. The most common events observed were nasopharyngitis and upper respiratory tract infections. The rate of serious infections was numerically higher in patients weighing less than 30 kg treated with 10 mg/kg tocilizumab (12.2 per 100 patient years) compared to patients weighing at or above 30 kg, treated with 8 mg/kg tocilizumab (4.0 per 100 patient years). The incidence of infections leading to dose interruptions was also numerically higher in patients weighing less than 30 kg treated with 10 mg/kg tocilizumab (21%) compared to patients weighing at or above 30 kg, treated with 8 mg/kg tocilizumab (8%).
Infusion Reactions
In PJIA patients, infusion-related reactions are defined as all events occurring during or within 24 hours of an infusion. In the ACTEMRA-IV all exposure population, 11 patients (6%) experienced an event during the infusion, and 38 patients (20.2%) experienced an event within 24 hours of an infusion. The most common events occurring during infusion were headache, nausea and hypotension, and occurring within 24 hours of infusion were dizziness and hypotension. In general, the adverse drug reactions observed during or within 24 hours of an infusion were similar in nature to those seen in RA and SJIA patients [see Adverse Reactions (6.1 and 6.8) ] .
No clinically significant hypersensitivity reactions associated with tocilizumab and requiring treatment discontinuation were reported.
Immunogenicity
One patient, in the 10 mg/kg less than 30 kg group, developed positive anti-tocilizumab antibodies without developing a hypersensitivity reaction and subsequently withdrew from the study.
Laboratory Abnormalities
Neutropenia
During routine laboratory monitoring in the ACTEMRA-IV all exposure population, a decrease in neutrophil counts below 1 × 10 9 per L occurred in 3.7% of patients.
There was no clear relationship between decreases in neutrophils below 1 × 10 9 per L and the occurrence of serious infections.
Thrombocytopenia
During routine laboratory monitoring in the ACTEMRA-IV all exposure population, 1% of patients had a decrease in platelet count at or less than 50,000 per mm 3 without associated bleeding events.
Elevated Liver Enzymes
During routine laboratory monitoring in the ACTEMRA-IV all exposure population, elevation in ALT or AST at or greater than 3 × ULN occurred in 4% and less than 1% of patients, respectively.
Lipids
During routine laboratory monitoring in the tocilizumab all exposure population, elevation in total cholesterol greater than 1.5-2 × ULN occurred in one patient (0.5%) and elevation in LDL greater than 1.5-2 × ULN occurred in one patient (0.5%).
Clinical Trials Experience in Polyarticular Juvenile Idiopathic Arthritis Patients Treated With Subcutaneous ACTEMRA (ACTEMRA-SC)
The safety of ACTEMRA-SC was studied in 52 pediatric patients 1 to 17 years of age with PJIA who had an inadequate clinical response or were intolerant to methotrexate. The total patient exposure in the PJIA ACTEMRA-SC population (defined as patients who received at least one dose of ACTEMRA-SC and accounting for treatment discontinuation) was 49.5 patient years. In general, the safety observed for ACTEMRA administered subcutaneously was consistent with the known safety profile of intravenous ACTEMRA, with the exception of injection site reactions (ISRs), and neutropenia.
Injection Site Reactions
During the 1-year study, a frequency of 28.8% (15/52) ISRs was observed in ACTEMRA-SC treated PJIA patients. These ISRs occurred in a greater proportion of patients at or above 30 kg (44.0%) compared with patients below 30 kg (14.8%). All ISRs were mild in severity and none of the ISRs required patient withdrawal from treatment or dose interruption. A higher frequency of ISRs was observed in ACTEMRA-SC treated PJIA patients compared to what was seen in adult RA or GCA patients [see Adverse Reactions (6.2 and 6.3) ] .
Immunogenicity
Three patients, 1 patient below 30 kg and 2 patients at or above 30 kg, developed positive anti-tocilizumab antibodies with neutralizing potential without developing a serious or clinically significant hypersensitivity reaction. One patient subsequently withdrew from the study.
Neutropenia
During routine laboratory monitoring in the ACTEMRA-SC all exposure population, a decrease in neutrophil counts below 1 × 10 9 per L occurred in 15.4% of patients, and was more frequently observed in the patients less than 30 kg (25.9%) compared to patients at or above 30 kg (4.0%). There was no clear relationship between decreases in neutrophils below 1 × 10 9 per L and the occurrence of serious infections.
Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated with Intravenous ACTEMRA (ACTEMRA-IV)
The data described below reflect exposure to ACTEMRA-IV in one randomized, double-blind, placebo-controlled trial of 112 pediatric patients with SJIA 2 to 17 years of age who had an inadequate clinical response to nonsteroidal anti-inflammatory drugs (NSAIDs) or corticosteroids due to toxicity or lack of efficacy. At baseline, approximately half of the patients were taking 0.3 mg/kg/day corticosteroids or more, and almost 70% were taking methotrexate. The trial included a 12 week controlled phase followed by an open-label extension. In the 12 week double-blind, controlled portion of the clinical study 75 patients received treatment with ACTEMRA-IV (8 or 12 mg per kg based upon body weight). After 12 weeks or at the time of escape, due to disease worsening, patients were treated with ACTEMRA-IV in the open-label extension phase.
The most common adverse events (at least 5%) seen in ACTEMRA-IV treated patients in the 12 week controlled portion of the study were: upper respiratory tract infection, headache, nasopharyngitis and diarrhea.
Infections
In the 12 week controlled phase, the rate of all infections in the ACTEMRA-IV group was 345 per 100 patient-years and 287 per 100 patient-years in the placebo group. In the open label extension over an average duration of 73 weeks of treatment, the overall rate of infections was 304 per 100 patient-years.
In the 12 week controlled phase, the rate of serious infections in the ACTEMRA-IV group was 11.5 per 100 patient years. In the open label extension over an average duration of 73 weeks of treatment, the overall rate of serious infections was 11.4 per 100 patient years. The most commonly reported serious infections included pneumonia, gastroenteritis, varicella, and otitis media.
Macrophage Activation Syndrome
In the 12 week controlled study, no patient in any treatment group experienced macrophage activation syndrome (MAS) while on assigned treatment; 3 per 112 (3%) developed MAS during open-label treatment with ACTEMRA-IV. One patient in the placebo group escaped to ACTEMRA-IV 12 mg per kg at Week 2 due to severe disease activity, and ultimately developed MAS at Day 70. Two additional patients developed MAS during the long-term extension. All 3 patients had ACTEMRA-IV dose interrupted (2 patients) or discontinued (1 patient) for the MAS event, received treatment, and the MAS resolved without sequelae. Based on a limited number of cases, the incidence of MAS does not appear to be elevated in the ACTEMRA-IV SJIA clinical development experience; however no definitive conclusions can be made.
Infusion Reactions
Patients were not premedicated, however most patients were on concomitant corticosteroids as part of their background treatment for SJIA. Infusion related reactions were defined as all events occurring during or within 24 hours after an infusion. In the 12 week controlled phase, 4% of ACTEMRA-IV and 0% of placebo treated patients experienced events occurring during infusion. One event (angioedema) was considered serious and life-threatening, and the patient was discontinued from study treatment.
Within 24 hours after infusion, 16% of patients in the ACTEMRA-IV treatment group and 5% of patients in the placebo group experienced an event. In the ACTEMRA-IV group the events included rash, urticaria, diarrhea, epigastric discomfort, arthralgia and headache. One of these events, urticaria, was considered serious.
Anaphylaxis
Anaphylaxis was reported in 1 out of 112 patients (less than 1%) treated with ACTEMRA-IV during the controlled and open label extension study [see Warnings and Precautions (5.6) ] .
Immunogenicity
All 112 patients were tested for anti-tocilizumab antibodies at baseline. Two patients developed positive anti-tocilizumab antibodies: one of these patients experienced serious adverse events of urticaria and angioedema consistent with an anaphylactic reaction which led to withdrawal; the other patient developed macrophage activation syndrome while on escape therapy and was discontinued from the study.
Laboratory Abnormalities
Neutropenia
During routine monitoring in the 12 week controlled phase, a decrease in neutrophil below 1 × 10 9 per L occurred in 7% of patients in the ACTEMRA-IV group, and in no patients in the placebo group. In the open label extension over an average duration of 73 weeks of treatment, a decreased neutrophil count occurred in 17% of the ACTEMRA-IV group. There was no clear relationship between decrease in neutrophils below 1 × 10 9 per L and the occurrence of serious infections.
Thrombocytopenia
During routine monitoring in the 12 week controlled phase, 1% of patients in the ACTEMRA-IV group and 3% in the placebo group had a decrease in platelet count to no more than 100,000 per mm 3 .
In the open label extension over an average duration of 73 weeks of treatment, decreased platelet count occurred in 4% of patients in the ACTEMRA-IV group, with no associated bleeding.
Elevated Liver Enzymes
During routine laboratory monitoring in the 12 week controlled phase, elevation in ALT or AST at or above 3× ULN occurred in 5% and 3% of patients, respectively in the ACTEMRA-IV group and in 0% of placebo patients.
In the open label extension over an average duration of 73 weeks of treatment, the elevation in ALT or AST at or above 3× ULN occurred in 13% and 5% of ACTEMRA-IV treated patients, respectively.
Lipids
During routine laboratory monitoring in the 12 week controlled phase, elevation in total cholesterol greater than 1.5× ULN – 2× ULN occurred in 1.5% of the ACTEMRA-IV group and in 0% of placebo patients. Elevation in LDL greater than 1.5× ULN – 2× ULN occurred in 1.9% of patients in the ACTEMRA-IV group and 0% of the placebo group.
In the open label extension study over an average duration of 73 weeks of treatment, the pattern and incidence of elevations in lipid parameters remained consistent with the 12 week controlled study data.
Clinical Trials Experience in Systemic Juvenile Idiopathic Arthritis Patients Treated with Subcutaneous ACTEMRA (ACTEMRA-SC)
The safety profile of ACTEMRA-SC was studied in 51 pediatric patients 1 to 17 years of age with SJIA who had an inadequate clinical response to NSAIDs and corticosteroids. In general, the safety observed for ACTEMRA administered subcutaneously was consistent with the known safety profile of intravenous ACTEMRA, with the exception of ISRs where a higher frequency was observed in ACTEMRA-SC treated SJIA patients compared to PJIA patients and adult RA or GCA patients [see Adverse Reactions (6.2 , 6.3 and 6.7) ] .
Injection Site Reactions (ISRs)
A total of 41.2% (21/51) SJIA patients experienced ISRs to ACTEMRA-SC. The most common ISRs were erythema, pruritus, pain, and swelling at the injection site. The majority of ISRs reported were Grade 1 events and all ISRs reported were non-serious and none required patient withdrawal from treatment or dose interruption.
Immunogenicity
Forty-six of the 51 (90.2%) patients who were tested for anti-tocilizumab antibodies at baseline had at least one post-baseline screening assay result. No patient developed positive anti-tocilizumab antibodies post-baseline.
Clinical Trials Experience in Patients with Cytokine Release Syndrome Treated with Intravenous ACTEMRA (ACTEMRA-IV)
In a retrospective analysis of pooled outcome data from multiple clinical trials 45 patients were treated with tocilizumab 8 mg/kg (12 mg/kg for patients less than 30 kg) with or without additional high-dose corticosteroids for severe or life-threatening CAR T-cell-induced CRS. A median of 1 dose of tocilizumab (range, 1-4 doses) was administered. No adverse reactions related to tocilizumab were reported [see Clinical Studies (14.10) ] .
Clinical Trials Experience in COVID-19 Patients Treated with Intravenous ACTEMRA (ACTEMRA-IV)
The safety of ACTEMRA in hospitalized adult COVID-19 patients was evaluated in a pooled safety population that includes patients enrolled in EMPACTA, COVACTA, AND REMDACTA. The analysis of adverse reactions included a total of 974 patients exposed to ACTEMRA. Patients received a single, 60-minute infusion of intravenous ACTEMRA 8 mg/kg (maximum dose of 800 mg). If clinical signs or symptoms worsened or did not improve, one additional dose of ACTEMRA 8 mg/kg could be administered between 8- 24 hours after the initial dose.
Adverse reactions summarized in Table 3 occurred in at least 3% of ACTEMRA-treated patients and more commonly than in patients on placebo in the pooled safety population.
| Adverse Reaction | ACTEMRA 8 mg per kg | Placebo |
|---|---|---|
| N = 974 (%) | N = 483 (%) | |
| Hepatic Transaminases increased | 10% | 8% |
| Constipation | 9 % | 8% |
| Urinary tract infection | 5% | 4% |
| Hypertension | 4% | 1% |
| Hypokalaemia | 4% | 3% |
| Anxiety | 4% | 2% |
| Diarrhea | 4% | 2% |
| Insomnia | 4% | 3% |
| Nausea | 3% | 2% |
In the pooled safety population, the rates of infection/serious infection events were 30%/19% in patients receiving ACTEMRA versus 32%/23% receiving placebo.
Laboratory Abnormalities
In the pooled adult safety population of EMPACTA, COVACTA, and REMDACTA, neutrophil counts <1000 cells/mcl occurred in 3.4% of patients who received ACTEMRA and 0.5% of patients who received placebo. Platelet counts <50,000 cells/mcl occurred in 3.2% of patients who received ACTEMRA and 1.5% of patients who received placebo. ALT or AST at or above 5× ULN occurred in 11.7% of patients who received ACTEMRA and 9.9% of patients who received placebo.
Postmarketing Experience
The following adverse reactions have been identified during post-approval use of ACTEMRA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hypersensitivity Reactions: Fatal anaphylaxis, Stevens-Johnson Syndrome, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) [see Warnings and Precautions (5.6) ]
- Pancreatitis
- Drug-induced liver injury, Hepatitis, Hepatic failure, Jaundice [see Warnings and Precautions (5.3) ]
- Hypofibrinogenemia
DRUG INTERACTIONS
Concomitant Drugs for Treatment of Adult Indications
In RA patients, population pharmacokinetic analyses did not detect any effect of methotrexate (MTX), non-steroidal anti-inflammatory drugs or corticosteroids on tocilizumab clearance. Concomitant administration of a single intravenous dose of 10 mg/kg ACTEMRA with 10-25 mg MTX once weekly had no clinically significant effect on MTX exposure. ACTEMRA has not been studied in combination with biological DMARDs such as TNF antagonists [see Dosage and Administration (2.2) ] .
In GCA patients, no effect of concomitant corticosteroid on tocilizumab exposure was observed.
Interactions with CYP450 Substrates
Cytochrome P450s in the liver are down-regulated by infection and inflammation stimuli including cytokines such as IL-6. Inhibition of IL-6 signaling in RA patients treated with tocilizumab may restore CYP450 activities to higher levels than those in the absence of tocilizumab leading to increased metabolism of drugs that are CYP450 substrates. In vitro studies showed that tocilizumab has the potential to affect expression of multiple CYP enzymes including CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP2D6 and CYP3A4. Its effect on CYP2C8 or transporters is unknown. In vivo studies with omeprazole, metabolized by CYP2C19 and CYP3A4, and simvastatin, metabolized by CYP3A4, showed up to a 28% and 57% decrease in exposure one week following a single dose of ACTEMRA, respectively. The effect of tocilizumab on CYP enzymes may be clinically relevant for CYP450 substrates with narrow therapeutic index, where the dose is individually adjusted. Upon initiation or discontinuation of ACTEMRA, in patients being treated with these types of medicinal products, perform therapeutic monitoring of effect (e.g., warfarin) or drug concentration (e.g., cyclosporine or theophylline) and the individual dose of the medicinal product adjusted as needed. Exercise caution when coadministering ACTEMRA with CYP3A4 substrate drugs where decrease in effectiveness is undesirable, e.g., oral contraceptives, lovastatin, atorvastatin, etc. The effect of tocilizumab on CYP450 enzyme activity may persist for several weeks after stopping therapy [see Clinical Pharmacology (12.3) ] .
Live Vaccines
Avoid use of live vaccines concurrently with ACTEMRA [see Warnings and Precautions (5.9) ] .
DESCRIPTION
Tocilizumab is a recombinant humanized anti-human interleukin 6 (IL-6) receptor monoclonal antibody of the immunoglobulin IgG1κ (gamma 1, kappa) subclass with a typical H 2 L 2 polypeptide structure. Each light chain and heavy chain consists of 214 and 448 amino acids, respectively. The four polypeptide chains are linked intra- and inter-molecularly by disulfide bonds. ACTEMRA has a molecular weight of approximately 148 kDa. The antibody is produced in mammalian (Chinese hamster ovary) cells.
Intravenous Infusion
ACTEMRA (tocilizumab) injection is a sterile, clear, colorless to pale yellow, preservative-free solution for further dilution prior to intravenous infusion with a pH of approximately 6.5. Each single-dose vial, formulated with a disodium phosphate dodecahydrate/sodium dihydrogen phosphate dihydrate buffered solution, is available at a concentration of 20 mg/mL containing 80 mg/4 mL, 200 mg/10 mL, or 400 mg/20 mL of ACTEMRA. Each mL of solution contains polysorbate 80 (0.5 mg), sucrose (50 mg), and Water for Injection, USP.
Subcutaneous Injection
ACTEMRA (tocilizumab) injection is a sterile, clear, colorless to slightly yellowish, preservative-free, histidine buffered solution for subcutaneous use with a pH of approximately 6.0.
It is supplied in a ready-to-use, single-dose 0.9 mL prefilled syringe (PFS) with a needle safety device or a ready-to-use, single-dose 0.9 mL autoinjector that delivers 162 mg tocilizumab, L-arginine hydrochloride (19 mg), L-histidine (1.52 mg), L-histidine hydrochloride monohydrate (1.74 mg), L-methionine (4.03 mg), polysorbate 80 (0.18 mg), and Water for Injection, USP.
CLINICAL PHARMACOLOGY
Mechanism of Action
Tocilizumab binds to both soluble and membrane-bound IL-6 receptors (sIL-6R and mIL-6R), and has been shown to inhibit IL-6-mediated signaling through these receptors. IL-6 is a pleiotropic pro-inflammatory cytokine produced by a variety of cell types including T- and B-cells, lymphocytes, monocytes and fibroblasts. IL-6 has been shown to be involved in diverse physiological processes such as T-cell activation, induction of immunoglobulin secretion, initiation of hepatic acute phase protein synthesis, and stimulation of hematopoietic precursor cell proliferation and differentiation. IL-6 is also produced by synovial and endothelial cells leading to local production of IL-6 in joints affected by inflammatory processes such as rheumatoid arthritis.
Pharmacodynamics
In clinical studies in RA patients with the 4 mg per kg and 8 mg per kg intravenous doses or the 162 mg weekly and every other weekly subcutaneous doses of ACTEMRA, decreases in levels of C-reactive protein (CRP) to within normal ranges were seen as early as week 2. Changes in pharmacodynamic parameters were observed (i.e., decreases in rheumatoid factor, erythrocyte sedimentation rate (ESR), serum amyloid A, fibrinogen and increases in hemoglobin) with doses, however the greatest improvements were observed with 8 mg per kg ACTEMRA. Pharmacodynamic changes were also observed to occur after ACTEMRA administration in GCA, SSc-ILD, PJIA, and SJIA patients (decreases in CRP, ESR, and increases in hemoglobin). The relationship between these pharmacodynamic findings and clinical efficacy is not known.
In healthy subjects administered ACTEMRA in doses from 2 to 28 mg per kg intravenously and 81 to 162 mg subcutaneously, absolute neutrophil counts decreased to the nadir 3 to 5 days following ACTEMRA administration. Thereafter, neutrophils recovered towards baseline in a dose dependent manner. Rheumatoid arthritis and GCA patients demonstrated a similar pattern of absolute neutrophil counts following ACTEMRA administration [see Warnings and Precautions (5.4) ] .
Pharmacokinetics
PK of tocilizumab is characterized by nonlinear elimination which is a combination of linear clearance and Michaelis-Menten elimination. The nonlinear part of tocilizumab elimination leads to an increase in exposure that is more than dose-proportional. The pharmacokinetic parameters of tocilizumab do not change with time. Due to the dependence of total clearance on tocilizumab serum concentrations, the half-life of tocilizumab is also concentration-dependent and varies depending on the serum concentration level. Population pharmacokinetic analyses in any patient population tested so far indicate no relationship between apparent clearance and the presence of anti-drug antibodies.
Rheumatoid Arthritis - Intravenous and Subcutaneous Administration
The pharmacokinetics in healthy subjects and RA patients suggest that PK is similar between the two populations.
The population PK model was developed from an analysis dataset composed of an IV dataset of 1793 patients from Study I, Study III, Study IV, and Study V, and from an IV and SC dataset of 1759 patients from Studies SC-I and SC-II. C mean is included in place of AUC tau , since for dosing regimens with different inter-dose intervals, the mean concentration over the dosing period characterizes the comparative exposure better than AUC tau.
At high serum concentrations, when total clearance of tocilizumab is dominated by linear clearance, a terminal half-life of approximately 21.5 days was derived from the population parameter estimates.
For doses of 4 mg/kg tocilizumab given every 4 weeks intravenously, the estimated median (range) C max , C trough , and C mean of tocilizumab at steady state were 86.1 (44.8–202) mcg/mL, 0.1 (0.0–14.6) mcg/mL, and 18.0 (8.9–50.7) mcg/mL, respectively. For doses of 8 mg/kg tocilizumab given every 4 weeks intravenously, the estimated median (range) C max , C trough, and C mean of tocilizumab were 176 (75.4–557) mcg/mL, 13.4 (0.1–154) mcg/mL, and 54.0 (17–260) mcg/mL, respectively. C max increased dose-proportionally between doses of 4 and 8 mg/kg IV every 4 weeks, while a greater than dose-proportional increase was observed in C mean and C trough . At steady-state, C mean and C trough were 3.0 and 134 fold higher at 8 mg/kg as compared to 4 mg/kg, respectively.
The accumulation ratios for AUC and C max after multiple doses of 4 and 8 mg/kg IV Q4W are low, while the accumulation ratios for C trough are higher (2.62 and 2.47, respectively). For C max , greater than 90% of the steady-state value was reached after the 1st IV infusion. For AUC tau and C mean , 90% of the steady-state value was reached after the 1st and 3rd infusion for 4 mg/kg and 8 mg/kg IV, while for C trough , approximately 90% of the steady-state value was reached after the 4th IV infusion after both doses.
For doses of 162 mg given every other week subcutaneously, the estimated median (range) steady-state C max , C trough , and C mean of tocilizumab were 12.1 (0.4–49.3) mcg/mL, 4.1 (0.0–34.2) mcg/mL, and 9.2 (0.2–43.6) mcg/mL, respectively.
For doses of 162 mg given every week subcutaneously, the estimated median (range) steady-state C max , C trough , and C mean of tocilizumab were 49.8 (3–150) mcg/mL, 42.9 (1.3–144) mcg/mL, and 47.3 (2.4–147) mcg/mL, respectively. Exposures after the 162 mg SC QW regimen were greater by 5.1 (C mean ) to 10.5 fold (C trough ) compared to the 162 mg SC Q2W regimen.
Accumulation ratios after multiple doses of either SC regimen were higher than after IV regimen with the highest ratios for C trough (6.02 and 6.30, for 162 mg SC Q2W and 162 mg SC QW, respectively). The higher accumulation for C trough was expected based on the nonlinear clearance contribution at lower concentrations. For C max , greater than 90% of the steady-state value was reached after the 5th SC and the 12th SC injection with the Q2W and QW regimens, respectively. For AUC tau and C mean , 90% of the steady-state value was reached after the 6th and 12th injections for the 162 mg SC Q2W and QW regimens, respectively. For C trough , approximately 90% of the steady-state value was reached after the 6th and 12th injections for the 162 mg SC Q2W and QW regimens, respectively.
Population PK analysis identified body weight as a significant covariate impacting the pharmacokinetics of tocilizumab. When given IV on a mg/kg basis, individuals with body weight ≥ 100 kg are predicted to have mean steady-state exposures higher than mean values for the patient population. Therefore, tocilizumab doses exceeding 800 mg per infusion are not recommended in patients with RA [see Dosage and Administration (2.2) ] . Due to the flat dosing employed for SC administration of tocilizumab, no modifications are necessary by this dosing route.
Giant Cell Arteritis – Subcutaneous and Intravenous Administration
The pharmacokinetics of tocilizumab SC in GCA patients was determined using a population pharmacokinetic analysis on a dataset composed of 149 GCA patients treated with 162 mg subcutaneously every week or with 162 mg subcutaneously every other week.
For the 162 mg every week dose, the estimated median (range) steady-state C max , C trough and C mean of tocilizumab SC were 72.1 (12.2–151) mcg/mL, 67.2 (10.7–145) mcg/mL, and 70.6 (11.7–149) mcg/mL, respectively. The accumulation ratios for C mean or AUC tau , C trough , and C max were 10.9, 9.6, and 8.9, respectively. Steady state was reached after 17 weeks. For the 162 mg every other week dose, the estimated median (range) steady-state C max , C trough , and C mean of tocilizumab were 17.2 (1.1–56.2) mcg/mL, 7.7 (0.1–37.3) mcg/mL, and 13.7 (0.5–49) mcg/mL, respectively. The accumulation ratios for C mean or AUC tau , C trough , and C max were 2.8, 5.6, and 2.3 respectively. Steady-state was reached after 14 weeks.
The pharmacokinetics of tocilizumab IV in GCA patients was characterized by a non-compartmental pharmacokinetic analysis which included 22 patients treated with 6 mg/kg intravenously every 4 weeks for 20 weeks. The median (range) C max , C trough and C mean of tocilizumab at steady state were 178 (115-320) mcg/mL, 22.7 (3.38-54.5) mcg/mL and 57.5 (32.9-110) mcg/mL, respectively. Steady state trough concentrations were within the range observed in GCA patients treated with 162 mg TCZ SC administered every week or every other week.
Based on pharmacokinetic exposure and extrapolation between RA and GCA patients, when given IV on a mg/kg basis, tocilizumab doses exceeding 600 mg per infusion are not recommended in patients with GCA [see Dosage and Administration (2.3) ] .
Systemic Sclerosis-Associated Interstitial Lung Disease – Subcutaneous Administration
The pharmacokinetics of tocilizumab in SSc-ILD patients was determined using a population pharmacokinetic analysis on a dataset composed of 66 SSc-ILD patients treated with 162 mg tocilizumab SC every week.
The estimated median (range) steady-state C max , C trough and C mean of tocilizumab were 52.5 (14.8-121) mcg/mL, 47.2 (10.8-114) mcg/mL, and 50.4 (13.4-119) mcg/mL, respectively. The accumulation ratios for C mean or AUC tau , C trough , and C max were 7.11, 6.56, and 5.89, respectively. Steady-state was reached after 13 weeks.
Polyarticular Juvenile Idiopathic Arthritis – Intravenous and Subcutaneous Administration
The pharmacokinetics of tocilizumab (TCZ) in PJIA patients was characterized by a population pharmacokinetic analysis which included 188 patients who were treated with TCZ IV or 52 patients treated with TCZ SC.
For doses of 8 mg/kg tocilizumab (patients with a body weight at or above 30 kg) given every 4 weeks intravenously, the estimated median (range) C max , C trough , and C mean of tocilizumab at steady state were 181 (114–331) mcg/mL, 3.28 (0.02–35.4) mcg/mL, and 38.6 (22.2–83.8) mcg/mL, respectively. For doses of 10 mg/kg tocilizumab (patients with a body weight less than 30 kg) given every 4 weeks intravenously, the estimated median (range) C max , C trough , and C mean of tocilizumab were 167 (125–220) mcg/mL, 0.35 (0–11.8) mcg/mL, and 30.8 (16.0–48.0) mcg/mL, respectively.
The accumulation ratios were 1.05 and 1.16 for AUC 4weeks , and 1.43 and 2.22 for C trough for 10 mg/kg (BW less than 30 kg) and 8 mg/kg (BW at or above 30 kg) intravenous doses, respectively. No accumulation for C max was observed. Following 10 mg/kg and 8 mg/kg TCZ IV every 4 weeks doses in PJIA patients (aged 2 to 17 years), steady state concentrations (trough and average) were within the range of exposures in adult RA patients following 4 mg/kg and 8 mg/kg every 4 weeks, and steady state peak concentrations in PJIA patients were comparable to those following 8 mg/kg every 4 weeks in adult RA patients.
For doses of 162 mg tocilizumab (patients with a body weight at or above 30 kg) given every 2 weeks subcutaneously, the estimated median (range) C max , C trough , and C mean of tocilizumab were 29.7 (7.56–50.3) mcg/mL, 12.7 (0.19–23.8) mcg/mL, and 23.0 (3.86–36.9) mcg/mL, respectively. For doses of 162 mg tocilizumab (patients with a body weight less than 30 kg) given every 3 weeks subcutaneously, the estimated median (range) C max , C trough , and C mean of tocilizumab were 62.4 (39.4–121) mcg/mL, 13.4 (0.21–52.3) mcg/mL, and 35.7 (17.4–91.8) mcg/mL, respectively.
The accumulation ratios were 1.46 and 2.04 for AUC 4weeks , 2.08 and 3.58 for C trough , and 1.32 and 1.72 for C max , for 162 mg given every 3 weeks (BW less than 30 kg) and 162 mg given every 2 weeks (BW at or above 30 kg) subcutaneous doses, respectively. Following subcutaneous dosing, steady state C trough was comparable for patients in the two body weight groups, while steady-state C max and C mean were higher for patients in the less than 30 kg group compared to the group at or above 30 kg. All patients treated with TCZ SC had steady-state C trough at or higher than that achieved with TCZ IV across the spectrum of body weights. The average and trough concentrations in patients after subcutaneous dosing were within the range of those achieved in adult patients with RA following the subcutaneous administration of the recommended regimens.
Systemic Juvenile Idiopathic Arthritis – Intravenous and Subcutaneous Administration
The pharmacokinetics of tocilizumab (TCZ) in SJIA patients was characterized by a population pharmacokinetic analysis which included 89 patients who were treated with TCZ IV or 51 patients treated with TCZ SC.
For doses of 8 mg/kg tocilizumab (patients with a body weight at or above 30 kg) given every 2 weeks intravenously, the estimated median (range) C max , C trough , and C mean of tocilizumab were 253 (120–404) mcg/mL, 70.7 (5.26–127) mcg/mL, and 117 (37.6–199) mcg/mL, respectively. For doses of 12 mg/kg tocilizumab (patients with a body weight less than 30 kg) given every 2 weeks intravenously, the estimated median (range) C max , C trough , and C mean of tocilizumab were 274 (149–444) mcg/mL, 65.9 (19.0–135) mcg/mL, and 124 (60–194) mcg/mL, respectively.
The accumulation ratios were 1.95 and 2.01 for AUC 4weeks , and 3.41 and 3.20 for C trough for 12 mg/kg (BW less than 30 kg) and 8 mg/kg (BW at or above 30 kg) intravenous doses, respectively. Accumulation data for C max were 1.37 and 1.42 for 12 mg/kg (BW less than 30 kg) and 8 mg/kg (BW at or above 30 kg) intravenous doses, respectively. Following every other week dosing with tocilizumab IV, steady state was reached by 8 weeks for both body weight groups. Mean estimated tocilizumab exposure parameters were similar between the two dose groups defined by body weight.
For doses of 162 mg tocilizumab (patients with a body weight at or above 30 kg) given every week subcutaneously, the estimated median (range) C max , C trough , and C mean of tocilizumab were 89.8 (26.4–190) mcg/mL, 72.4 (19.5–158) mcg/mL, and 82.4 (23.9–169) mcg/mL, respectively. For doses of 162 mg tocilizumab (patients with a body weight less than 30 kg) given every 2 weeks subcutaneously, the estimated median (range) C max , C trough , and C mean of tocilizumab were 127 (51.7–266) mcg/mL, 64.2 (16.6–136) mcg/mL, and 92.7 (38.5–199) mcg/mL, respectively.
The accumulation ratios were 2.27 and 4.28 for AUC 4weeks , 3.21 and 4.39 for C trough , and 1.88 and 3.66 for C max , for 162 mg given every 2 weeks (BW less than 30 kg) and 162 mg given every week (BW at or above 30 kg) subcutaneous doses, respectively. Following subcutaneous dosing, steady state was reached by 12 weeks for both body weight groups. All patients treated with tocilizumab SC had steady-state C max lower than that achieved with tocilizumab IV across the spectrum of body weights. Trough and mean concentrations in patients after SC dosing were similar to those achieved with tocilizumab IV across body weights.
COVID-19 -Intravenous Administration
The pharmacokinetics of tocilizumab in adult COVID-19 patients was characterized by a population pharmacokinetic analysis of a dataset composed of 380 adult patients treated with tocilizumab 8mg/kg intravenously (IV) in the COVACTA study [see Clinical Studies (14.11) ] and another clinical study.
For one dose of 8 mg/kg tocilizumab IV, the estimated median (range) C max and C day28 of tocilizumab were 151 (77.5-319) mcg/mL and 0.229 (0.00119-19.4) mcg/mL, respectively. For two doses of 8 mg/kg tocilizumab IV separated by at least 8 hours, the estimated median (range) C max and C day28 of tocilizumab was 290 (152-604) mcg/mL and 7.04 (0.00474-54.8) mcg/mL, respectively. The weight-tiered dosing used in RECOVERY study, 800 mg for patients >90 kg, 600 mg for patients >65 and ≤90 kg, 400 mg for patients >40 and ≤65 kg, and 8mg/kg for patients ≤40 kg, is comparable to 8 mg/kg dosing and is expected to have similar exposure.
Based on PK-sIL-6R modeling and simulation, the recommended dosing regimen for ACTEMRA is expected to result in comparable plasma exposures of tocilizumab in pediatric patients aged 2 years and older with COVID-19 as observed in adults with COVID-19 [see Use in Specific Populations (8.4) ] .
Absorption
Following subcutaneous dosing, the absorption half-life was around 4 days in RA and GCA patients and 3 days in SSc-ILD patients. The bioavailability for the subcutaneous formulation was 80%.
Following subcutaneous dosing in PJIA patients, the absorption half-life was around 2 days, and the bioavailability for the subcutaneous formulation in PJIA patients was 96%.
Following subcutaneous dosing in SJIA patients, the absorption half-life was around 2 days, and the bioavailability for the SC formulation in SJIA patients was 95%.
In RA patients the median values of T max were 2.8 days after the tocilizumab every week dose and 4.7 days after the tocilizumab every other week dose.
In GCA patients, the median values of T max were 3 days after the tocilizumab every week dose and 4.5 days after the tocilizumab every other week dose.
In SSc-ILD patients, the median value of T max was 2.8 days after the tocilizumab every week dose.
Distribution
Following intravenous dosing, tocilizumab undergoes biphasic elimination from the circulation. In rheumatoid arthritis patients the central volume of distribution was 3.5 L and the peripheral volume of distribution was 2.9 L, resulting in a volume of distribution at steady state of 6.4 L.
In GCA patients, the central volume of distribution was 4.09 L, the peripheral volume of distribution was 3.37 L resulting in a volume of distribution at steady state of 7.46 L.
In SSc-ILD patients, the central volume of distribution was 4.16 L, the peripheral volume of distribution was 2.58 L resulting in a volume of distribution at steady state of 6.74 L.
In pediatric patients with PJIA, the central volume of distribution was 1.98 L, the peripheral volume of distribution was 2.1 L, resulting in a volume of distribution at steady state of 4.08 L.
In pediatric patients with SJIA, the central volume of distribution was 1.87 L, the peripheral volume of distribution was 2.14 L resulting in a volume of distribution at steady state of 4.01 L.
In adult COVID-19 patients treated with one or two infusions of tocilizumab 8 mg/kg intravenously separated by 8 hours, the estimated central volume of distribution was 4.52 L, and the estimated peripheral volume of distribution was 4.23 L, resulting in a volume of distribution of 8.75 L.
Elimination
ACTEMRA is eliminated by a combination of linear clearance and nonlinear elimination. The concentration-dependent nonlinear elimination plays a major role at low tocilizumab concentrations. Once the nonlinear pathway is saturated, at higher tocilizumab concentrations, clearance is mainly determined by the linear clearance. The saturation of the nonlinear elimination leads to an increase in exposure that is more than dose-proportional. The pharmacokinetic parameters of ACTEMRA do not change with time.
Population pharmacokinetic analyses in any patient population tested so far indicate no relationship between apparent clearance and the presence of anti-drug antibodies.
The linear clearance in the population pharmacokinetic analysis was estimated to be 12.5 mL per h in RA patients, 6.7 mL per h in GCA patients, 8.8 mL per h in SSc-ILD patients, 5.8 mL per h in pediatric patients with PJIA, and 5.7 mL per h in pediatric patients with SJIA. In adult COVID-19 patients, serum concentrations were below the limit of quantification after 35 days on average following one infusion of tocilizumab 8 mg/kg intravenously. The average linear clearance in the population pharmacokinetic analysis was estimated to be 17.6 mL per hour in patients with baseline ordinal scale category 3 (OS 3, patients requiring supplemental oxygen), 22.5 mL per hour in patients with baseline OS 4 (patients requiring high-flow oxygen or non-invasive ventilation), 29 mL per hour in patients with baseline OS 5 (patients requiring mechanical ventilation), and 35.4 mL per hour in patients with baseline OS 6 (patients requiring extracorporeal membrane oxygenation (ECMO) or mechanical ventilation and additional organ support).
Due to the dependence of total clearance on ACTEMRA serum concentrations, the half-life of ACTEMRA is also concentration-dependent and varies depending on the serum concentration level.
For intravenous administration in RA patients, the concentration-dependent apparent t 1/2 is up to 11 days for 4 mg per kg and up to 13 days for 8 mg per kg every 4 weeks in patients with RA at steady-state. For subcutaneous administration in RA patients, the concentration-dependent apparent t 1/2 is up to 13 days for 162 mg every week and 5 days for 162 mg every other week in patients with RA at steady-state.
In GCA patients at steady state, the effective t 1/2 of tocilizumab varied between 18.3 and 18.9 days for 162 mg subcutaneously every week dosing regimen and between 4.2 and 7.9 days for 162 mg subcutaneously every other week dosing regimen. For intravenous administration in GCA patients, the TCZ concentration-dependent apparent t 1/2 was 13.2 days following 6 mg/kg every 4 weeks.
In SSc-ILD patients at steady state, the effective t 1/2 of tocilizumab varied between 12.1 and 13.0 days for the 162 mg subcutaneous every week dosing regimen.
The t 1/2 of tocilizumab in children with PJIA is up to 17 days for the two body weight categories (8 mg/kg for body weight at or above 30 kg or 10 mg/kg for body weight below 30 kg) during a dosing interval at steady state. For subcutaneous administration, the t 1/2 of tocilizumab in PJIA patients is up to 10 days for the two body weight categories (every other week regimen for body weight at or above 30 kg or every 3 week regimen for body weight less than 30 kg) during a dosing interval at steady state.
The t 1/2 of tocilizumab intravenous in pediatric patients with SJIA is up to 16 days for the two body weight categories (8 mg/kg for body weight at or above 30 kg and 12 mg/kg for body weight below 30 kg every other week) during a dosing interval at steady-state. Following subcutaneous administration, the effective t 1/2 of tocilizumab subcutaneous in SJIA patients is up to 14 days for both the body weight categories (162 mg every week for body weight at or above 30 kg and 162 mg every two weeks for body weight below 30 kg) during a dosing interval at steady state.
Specific Populations
Population pharmacokinetic analyses in adult rheumatoid arthritis patients and GCA patients showed that age, gender and race did not affect the pharmacokinetics of tocilizumab. Linear clearance was found to increase with body size. In RA patients, the body weight-based dose (8 mg per kg) resulted in approximately 86% higher exposure in patients who are greater than 100 kg in comparison to patients who are less than 60 kg. There was an inverse relationship between tocilizumab exposure and body weight for flat dose subcutaneous regimens.
In GCA patients treated with ACTEMRA-SC, higher exposure was observed in patients with lower body weight. For the 162 mg every week subcutaneous dosing regimen, the steady-state C mean was 51% higher in patients with body weight less than 60 kg compared to patients weighing between 60 to 100 kg. For the 162 mg every other week subcutaneous regimen, the steady-state C mean was 129% higher in patients with body weight less than 60 kg compared to patients weighing between 60 to 100 kg. There is limited data for patients above 100 kg (n=7).
In COVID-19 patients, exposure following body-weight-based intravenous dosing (8 mg per kg tocilizumab up to 100 kg body weight with a maximum dose of 800 mg) was dependent on body weight and disease severity assessed by an ordinal scale (OS). Within an OS category, compared to patients with a mean body weight of 80 kg, exposure was 20% lower in patients weighing less than 60 kg. Exposure in patients weighing more than 100 kg was in the same range as exposure in patients with a mean body weight of 80 kg. For an 80 kg patient, exposure decreases as OS category increases; for each category increase, exposure decreases by 13%.
Patients with Hepatic Impairment
No formal study of the effect of hepatic impairment on the pharmacokinetics of tocilizumab was conducted.
Patients with Renal Impairment
No formal study of the effect of renal impairment on the pharmacokinetics of tocilizumab was conducted.
Most of the RA, GCA, and SSc-ILD patients in the population pharmacokinetic analysis had normal renal function or mild renal impairment. Mild renal impairment (estimated creatinine clearance less than 80 mL per min and at or above 50 mL per min based on Cockcroft-Gault formula) did not impact the pharmacokinetics of tocilizumab.
Approximately one-third of the patients in the ACTEMRA-SC GCA clinical trial had moderate renal impairment at baseline (estimated creatinine clearance of 30-59 mL/min). No impact on tocilizumab exposure was noted in these patients.
No dose adjustment is required in patients with mild or moderate renal impairment.
Drug Interaction Studies
In vitro data suggested that IL-6 reduced mRNA expression for several CYP450 isoenzymes including CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP2D6 and CYP3A4, and this reduced expression was reversed by co-incubation with tocilizumab at clinically relevant concentrations. Accordingly, inhibition of IL-6 signaling in RA patients treated with tocilizumab may restore CYP450 activities to higher levels than those in the absence of tocilizumab leading to increased metabolism of drugs that are CYP450 substrates. Its effect on CYP2C8 or transporters (e.g., P-gp) is unknown. This is clinically relevant for CYP450 substrates with a narrow therapeutic index, where the dose is individually adjusted. Upon initiation of ACTEMRA, in patients being treated with these types of medicinal products, therapeutic monitoring of the effect (e.g., warfarin) or drug concentration (e.g., cyclosporine or theophylline) should be performed and the individual dose of the medicinal product adjusted as needed. Caution should be exercised when ACTEMRA is coadministered with drugs where decrease in effectiveness is undesirable, e.g., oral contraceptives (CYP3A4 substrates) [see Drug Interactions (7.2) ] .
Simvastatin
Simvastatin is a CYP3A4 and OATP1B1 substrate. In 12 RA patients not treated with ACTEMRA, receiving 40 mg simvastatin, exposures of simvastatin and its metabolite, simvastatin acid, was 4- to 10-fold and 2-fold higher, respectively, than the exposures observed in healthy subjects. One week following administration of a single infusion of ACTEMRA (10 mg per kg), exposure of simvastatin and simvastatin acid decreased by 57% and 39%, respectively, to exposures that were similar or slightly higher than those observed in healthy subjects. Exposures of simvastatin and simvastatin acid increased upon withdrawal of ACTEMRA in RA patients. Selection of a particular dose of simvastatin in RA patients should take into account the potentially lower exposures that may result after initiation of ACTEMRA (due to normalization of CYP3A4) or higher exposures after discontinuation of ACTEMRA.
Omeprazole
Omeprazole is a CYP2C19 and CYP3A4 substrate. In RA patients receiving 10 mg omeprazole, exposure to omeprazole was approximately 2 fold higher than that observed in healthy subjects. In RA patients receiving 10 mg omeprazole, before and one week after ACTEMRA infusion (8 mg per kg), the omeprazole AUC inf decreased by 12% for poor (N=5) and intermediate metabolizers (N=5) and by 28% for extensive metabolizers (N=8) and were slightly higher than those observed in healthy subjects.
Dextromethorphan
Dextromethorphan is a CYP2D6 and CYP3A4 substrate. In 13 RA patients receiving 30 mg dextromethorphan, exposure to dextromethorphan was comparable to that in healthy subjects. However, exposure to its metabolite, dextrorphan (a CYP3A4 substrate), was a fraction of that observed in healthy subjects. One week following administration of a single infusion of ACTEMRA (8 mg per kg), dextromethorphan exposure was decreased by approximately 5%. However, a larger decrease (29%) in dextrorphan levels was noted after ACTEMRA infusion.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to establish the carcinogenicity potential of tocilizumab. Literature indicates that the IL-6 pathway can mediate anti-tumor responses by promoting increased immune cell surveillance of the tumor microenvironment. However, available published evidence also supports that IL-6 signaling through the IL-6 receptor may be involved in pathways that lead to tumorigenesis. The malignancy risk in humans from an antibody that disrupts signaling through the IL-6 receptor, such as tocilizumab, is presently unknown.
Fertility and reproductive performance were unaffected in male and female mice that received a murine analogue of tocilizumab administered by the intravenous route at a dose of 50 mg/kg every three days.
CLINICAL STUDIES
Rheumatoid Arthritis – Intravenous Administration
The efficacy and safety of intravenously administered ACTEMRA was assessed in five randomized, double-blind, multicenter studies in patients greater than 18 years with active rheumatoid arthritis diagnosed according to American College of Rheumatology (ACR) criteria. Patients had at least 8 tender and 6 swollen joints at baseline. ACTEMRA was given intravenously every 4 weeks as monotherapy (Study I), in combination with methotrexate (MTX) (Studies II and III) or other disease-modifying anti-rheumatic drugs (DMARDs) (Study IV) in patients with an inadequate response to those drugs, or in combination with MTX in patients with an inadequate response to TNF antagonists (Study V).
Study I (NCT00109408) evaluated patients with moderate to severe active rheumatoid arthritis who had not been treated with MTX within 24 weeks prior to randomization, or who had not discontinued previous methotrexate treatment as a result of clinically important toxic effects or lack of response. In this study, 67% of patients were MTX-naïve, and over 40% of patients had rheumatoid arthritis less than 2 years. Patients received ACTEMRA 8 mg per kg monotherapy or MTX alone (dose titrated over 8 weeks from 7.5 mg to a maximum of 20 mg weekly). The primary endpoint was the proportion of ACTEMRA patients who achieved an ACR 20 response at Week 24.
Study II (NCT00106535) was a 104-week study with an optional 156-week extension phase that evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response to MTX. Patients received ACTEMRA 8 mg per kg, ACTEMRA 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). Upon completion of 52-weeks, patients received open-label treatment with ACTEMRA 8 mg per kg through 104 weeks or they had the option to continue their double-blind treatment if they maintained a greater than 70% improvement in swollen/tender joint count. Two pre-specified interim analyses at week 24 and week 52 were conducted. The primary endpoint at week 24 was the proportion of patients who achieved an ACR 20 response. At weeks 52 and 104, the primary endpoints were change from baseline in modified total Sharp-Genant score and the area under the curve (AUC) of the change from baseline in HAQ-DI score.
Study III (NCT00106548) evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response to MTX. Patients received ACTEMRA 8 mg per kg, ACTEMRA 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
Study IV (NCT00106574) evaluated patients who had an inadequate response to their existing therapy, including one or more DMARDs. Patients received ACTEMRA 8 mg per kg or placebo every four weeks, in combination with the stable DMARDs. The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
Study V (NCT00106522) evaluated patients with moderate to severe active rheumatoid arthritis who had an inadequate clinical response or were intolerant to one or more TNF antagonist therapies. The TNF antagonist therapy was discontinued prior to randomization. Patients received ACTEMRA 8 mg per kg, ACTEMRA 4 mg per kg, or placebo every four weeks, in combination with MTX (10 to 25 mg weekly). The primary endpoint was the proportion of patients who achieved an ACR 20 response at week 24.
Clinical Response
The percentages of intravenous ACTEMRA-treated patients achieving ACR 20, 50 and 70 responses are shown in Table 4 . In all intravenous studies, patients treated with 8 mg per kg ACTEMRA had higher ACR 20, ACR 50, and ACR 70 response rates versus MTX- or placebo-treated patients at week 24.
During the 24 week controlled portions of Studies I to V, patients treated with ACTEMRA at a dose of 4 mg per kg in patients with inadequate response to DMARDs or TNF antagonist therapy had lower response rates compared to patients treated with ACTEMRA 8 mg per kg.
| Percent of Patients | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Response Rate | Study I | Study II | Study III | Study IV | Study V | ||||||||
| MTX | ACTEMRA 8 mg per kg | Placebo + MTX | ACTEMRA 4 mg per kg + MTX | ACTEMRA 8 mg per kg + MTX | Placebo + MTX | ACTEMRA 4 mg per kg + MTX | ACTEMRA 8 mg per kg + MTX | Placebo + DMARDs | ACTEMRA 8 mg per kg + DMARDs | Placebo + MTX | ACTEMRA 4 mg per kg + MTX | ACTEMRA 8 mg per kg + MTX | |
| N=284 | N=286 | N=393 | N=399 | N=398 | N=204 | N=213 | N=205 | N=413 | N=803 | N=158 | N=161 | N=170 | |
| (95% CI) CI: 95% confidence interval of the weighted difference to placebo adjusted for site (and disease duration for Study I only) | ( 95% CI) | (95% CI) | ( 95% CI) | (95% CI) | (95% CI) | ( 95% CI) | (95% CI) | ||||||
| ACR 20 | |||||||||||||
| Week 24 | 53% | 70% (0.11, 0.27) | 27% | 51% (0.17, 0.29) | 56% (0.23, 0.35) | 27% | 48% (0.15, 0.32) | 59% (0.23, 0.41) | 24% | 61% (0.30, 0.40) | 10% | 30% (0.15, 0.36) | 50% (0.36, 0.56) |
| Week 52 | N/A | N/A | 25% | 47% (0.15, 0.28) | 56% (0.25, 0.38) | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| ACR 50 | |||||||||||||
| Week 24 | 34% | 44% (0.04, 0.20) | 10% | 25% (0.09, 0.20) | 32% (0.16, 0.28) | 11% | 32% (0.13, 0.29) | 44% (0.25, 0.41) | 9% | 38% (0.23, 0.33) | 4% | 17% (0.05, 0.25) | 29% (0.21, 0.41) |
| Week 52 | N/A | N/A | 10% | 29% (0.14, 0.25) | 36% (0.21, 0.32) | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| ACR 70 | |||||||||||||
| Week 24 | 15% | 28% (0.07, 0.22) | 2% | 11% (0.03, 0.13) | 13% (0.05, 0.15) | 2% | 12% (0.04, 0.18) | 22% (0.12, 0.27) | 3% | 21% (0.13, 0.21) | 1% | 5% (-0.06, 0.14) | 12% (0.03, 0.22) |
| Week 52 | N/A | N/A | 4% | 16% (0.08, 0.17) | 20% (0.12, 0.21) | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
| Major Clinical Responses Major clinical response is defined as achieving an ACR 70 response for a continuous 24 week period | |||||||||||||
| Week 52 | N/A | N/A | 1% | 4% (0.01, 0.06) | 7% (0.03, 0.09) | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A |
In study II, a greater proportion of patients treated with 4 mg per kg and 8 mg per kg ACTEMRA + MTX achieved a low level of disease activity as measured by a DAS 28-ESR less than 2.6 compared with placebo +MTX treated patients at week 52. The proportion of ACTEMRA-treated patients achieving DAS 28-ESR less than 2.6, and the number of residual active joints in these responders in Study II are shown in Table 5 .
| Study II | |||
|---|---|---|---|
| Placebo + MTX N = 393 | ACTEMRA 4 mg per kg + MTX N = 399 | ACTEMRA 8 mg per kg + MTX N = 398 | |
| •n denotes numerator of all the percentage. Denominator is the intent-to-treat population. Not all patients received DAS28 assessments at Week 52. | |||
| DAS28-ESR less than 2.6 | |||
| Proportion of responders at week 52 (n) 95% confidence interval | 3% (12) | 18% (70) 0.10, 0.19 | 32% (127) 0.24, 0.34 |
| Of responders, proportion with 0 active joints (n) | 33% (4) | 27% (19) | 21% (27) |
| Of responders, proportion with 1 active joint (n) | 8% (1) | 19% (13) | 13% (16) |
| Of responders, proportion with 2 active joints (n) | 25% (3) | 13% (9) | 20% (25) |
| Of responders, proportion with 3 or more active joints (n) | 33% (4) | 41% (29) | 47% (59) |
The results of the components of the ACR response criteria for Studies III and V are shown in Table 6 . Similar results to Study III were observed in Studies I, II and IV.
| Study III | Study V | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ACTEMRA 4 mg per kg + MTX | ACTEMRA 8 mg per kg + MTX | Placebo + MTX | ACTEMRA 4 mg per kg + MTX | ACTEMRA 8 mg per kg + MTX | Placebo + MTX | |||||||
| N=213 | N=205 | N=204 | N=161 | N=170 | N=158 | |||||||
| Component (mean) | Baseline | Week 24 Data shown is mean at week 24, difference in adjusted mean change from baseline compared with placebo + MTX at week 24 and 95% confidence interval for that difference | Baseline | Week 24 | Baseline | Week 24 | Baseline | Week 24 | Baseline | Week 24 | Baseline | Week 24 |
| Number of tender joints (0-68) | 33 | 19 -7.0 (-10.0, -4.1) | 32 | 14.5 -9.6 (-12.6, -6.7) | 33 | 25 | 31 | 21 -10.8 (-14.6, -7.1) | 32 | 17 -15.1 (-18.8, -11.4) | 30 | 30 |
| Number of swollen joints (0-66) | 20 | 10 -4.2 (-6.1, -2.3) | 19.5 | 8 -6.2 (-8.1, -4.2) | 21 | 15 | 19.5 | 13 -6.2 (-9.0, -3.5) | 19 | 11 -7.2 (-9.9, -4.5) | 19 | 18 |
| Pain Visual analog scale: 0 = best, 100 = worst | 61 | 33 -11.0 (-17.0, -5.0) | 60 | 30 -15.8 (-21.7, -9.9) | 57 | 43 | 63.5 | 43 -12.4 (-22.1, -2.1) | 65 | 33 -23.9 (-33.7, -14.1) | 64 | 48 |
| Patient global assessment | 66 | 34 -10.9 (-17.1, -4.8) | 65 | 31 -14.9 (-20.9, -8.9) | 64 | 45 | 70 | 46 -10.0 (-20.3, 0.3) | 70 | 36 -17.4 (-27.8, -7.0) | 71 | 51 |
| Physician global assessment | 64 | 26 -5.6 (-10.5, -0.8) | 64 | 23 -9.0 (-13.8, -4.2) | 64 | 32 | 66.5 | 39 -10.5 (-18.6, -2.5) | 66 | 28 -18.2 (-26.3, -10.0) | 67.5 | 43 |
| Disability index (HAQ) Health Assessment Questionnaire: 0 = best, 3 = worst; 20 questions; 8 categories: dressing and grooming, arising, eating, walking, hygiene, reach, grip, and activities | 1.64 | 1.01 -0.18 (-0.34, -0.02) | 1.55 | 0.96 -0.21 (-0.37, -0.05) | 1.55 | 1.21 | 1.67 | 1.39 -0.25 (-0.42, -0.09) | 1.75 | 1.34 -0.34 (-0.51, -0.17) | 1.70 | 1.58 |
| CRP (mg per dL) | 2.79 | 1.17 -1.30 (-2.0, -0.59) | 2.61 | 0.25 -2.156 (-2.86, -1.46) | 2.36 | 1.89 | 3.11 | 1.77 -1.34 (-2.5, -0.15) | 2.80 | 0.28 -2.52 (-3.72, -1.32) | 3.705 | 3.06 |
The percent of ACR 20 responders by visit for Study III is shown in Figure 1 . Similar response curves were observed in studies I, II, IV, and V.
| Figure 1 Percent of ACR 20 Responders by Visit for Study III (Inadequate Response to MTX) The same patients may not have responded at each timepoint. |
 |
Radiographic Response
In Study II, structural joint damage was assessed radiographically and expressed as change in total Sharp-Genant score and its components, the erosion score and joint space narrowing score. Radiographs of hands/wrists and forefeet were obtained at baseline, 24 weeks, 52 weeks, and 104 weeks and scored by readers unaware of treatments group and visit number. The results from baseline to week 52 are shown in Table 7 . ACTEMRA 4 mg per kg slowed (less than 75% inhibition compared to the control group) and ACTEMRA 8 mg per kg inhibited (at least 75% inhibition compared to the control group) the progression of structural damage compared to placebo plus MTX at week 52.
| Placebo + MTX | ACTEMRA 4 mg per kg + MTX | ACTEMRA 8 mg per kg + MTX | |
|---|---|---|---|
| N=294 | N=343 | N=353 | |
| SD = standard deviation | |||
| Week 52 Week 52 analysis employs linearly extrapolated data for patients after escape, withdrawal, or loss to follow up. | |||
| Total Sharp-Genant Score, Mean (SD) | 1.17 (3.14) | 0.33 (1.30) | 0.25 (0.98) |
| Adjusted Mean difference Difference between the adjusted means (ACTEMRA + MTX - Placebo + MTX) (95%CI) | -0.83 (-1.13, -0.52) | -0.90 (-1.20, -0.59) | |
| Erosion Score, Mean (SD) | 0.76 (2.14) | 0.20 (0.83) | 0.15 (0.77) |
| Adjusted Mean difference (95%CI) | -0.55 (-0.76, -0.34) | -0.60 (-0.80, -0.39) | |
| Joint Space Narrowing Score, Mean (SD) | 0.41 (1.71) | 0.13 (0.72) | 0.10 (0.49) |
| Adjusted Mean difference (95%CI) | -0.28 (-0.44, -0.11) | -0.30 (-0.46, -0.14) | |
The mean change from baseline to week 104 in Total Sharp-Genant Score for the ACTEMRA 4 mg per kg groups was 0.47 (SD = 1.47) and for the 8 mg per kg groups was 0.34 (SD = 1.24). By the week 104, most patients in the control (placebo + MTX) group had crossed over to active treatment, and results are therefore not included for comparison. Patients in the active groups may have crossed over to the alternate active dose group, and results are reported per original randomized dose group.
In the placebo group, 66% of patients experienced no radiographic progression (Total Sharp-Genant Score change ≤ 0) at week 52 compared to 78% and 83% in the ACTEMRA 4 mg per kg and 8 mg per kg, respectively. Following 104 weeks of treatment, 75% and 83% of patients initially randomized to ACTEMRA 4 mg per kg and 8 mg per kg, respectively, experienced no progression of structural damage compared to 66% of placebo treated patients.
Health Related Outcomes
In Study II, physical function and disability were assessed using the Health Assessment Questionnaire Disability Index (HAQ-DI). Both dosing groups of ACTEMRA demonstrated a greater improvement compared to the placebo group in the AUC of change from baseline in the HAQ-DI through week 52. The mean change from baseline to week 52 in HAQ-DI was 0.6, 0.5, and 0.4 for ACTEMRA 8 mg per kg, ACTEMRA 4 mg per kg, and placebo treatment groups, respectively. Sixty-three percent (63%) and sixty percent (60%) of patients in the ACTEMRA 8 mg per kg and ACTEMRA 4 mg per kg treatment groups, respectively, achieved a clinically relevant improvement in HAQ-DI (change from baseline of ≥ 0.3 units) at week 52 compared to 53% in the placebo treatment group.
Other Health-Related Outcomes
General health status was assessed by the Short Form Health Survey (SF-36) in Studies I – V. Patients receiving ACTEMRA demonstrated greater improvement from baseline compared to placebo in the Physical Component Summary (PCS), Mental Component Summary (MCS), and in all 8 domains of the SF-36.
Cardiovascular Outcomes
Study WA25204 (NCT01331837) was a randomized, open-label (sponsor-blinded), 2-arm parallel-group, multicenter, non-inferiority, cardiovascular (CV) outcomes trial in patients with a diagnosis of moderate to severe RA. This CV safety study was designed to exclude a moderate increase in CV risk in patients treated with ACTEMRA compared with a TNF inhibitor standard of care (etanercept).
The study included 3,080 seropositive RA patients with active disease and an inadequate response to non-biologic disease-modifying anti-rheumatic drugs, who were aged ≥50 years with at least one additional CV risk factor beyond RA. Patients were randomized 1:1 to IV ACTEMRA 8 mg/kg Q4W or SC etanercept 50 mg QW and followed for an average of 3.2 years. The primary endpoint was the comparison of the time-to-first occurrence of any component of a composite of major adverse CV events (MACE; non-fatal myocardial infarction, non-fatal stroke, or CV death), with the final intent-to-treat analysis based on a total of 161 confirmed CV events (83/1538 [5.4%] for ACTEMRA; 78/1542 [5.1%] for etanercept) reviewed by an independent and blinded adjudication committee.
Non-inferiority of ACTEMRA to etanercept for cardiovascular risk was determined by excluding >80% relative increase in the risk of MACE. The estimated hazard ratio (HR) for the risk of MACE comparing ACTEMRA to etanercept was 1.05; 95% CI (0.77, 1.43).
Rheumatoid Arthritis – Subcutaneous Administration
The efficacy and safety of subcutaneously administered ACTEMRA was assessed in two double-blind, controlled, multicenter studies in patients with active RA. One study, SC-I (NCT01194414), was a non-inferiority study that compared the efficacy and safety of ACTEMRA 162 mg administered every week subcutaneously to 8 mg per kg intravenously every four weeks. The second study, SC-II (NCT01232569), was a placebo controlled superiority study that evaluated the safety and efficacy of ACTEMRA 162 mg administered every other week subcutaneously to placebo. Both SC-I and SC-II required patients to be >18 years of age with moderate to severe active rheumatoid arthritis diagnosed according to ACR criteria who had at least 4 tender and 4 swollen joints at baseline (SC-I) or at least 8 tender and 6 swollen joints at baseline (SC-II), and an inadequate response to their existing DMARD therapy, where approximately 20% also had a history of inadequate response to at least one TNF inhibitor. All patients in both SC studies received background non-biologic DMARD(s).
In SC-I, 1262 patients were randomized 1:1 to receive ACTEMRA-SC 162 mg every week or intravenous ACTEMRA 8 mg/kg every four weeks in combination with DMARD(s). In SC-II, 656 patients were randomized 2:1 to ACTEMRA-SC 162 mg every other week or placebo, in combination with DMARD(s). The primary endpoint in both studies was the proportion of patients who achieved an ACR20 response at Week 24.
The clinical response to 24 weeks of ACTEMRA-SC therapy is shown in Table 8 . In SC-I, the primary outcome measure was ACR20 at Week 24. The pre-specified non-inferiority margin was a treatment difference of 12%. The study demonstrated non-inferiority of ACTEMRA with respect to ACR20 at Week 24; ACR50, ACR70, and DAS28 responses are also shown in Table 8 . In SC-II, a greater portion of patients treated with ACTEMRA 162 mg subcutaneously every other week achieved ACR20, ACR50, and ACR70 responses compared to placebo-treated patients (Table 8 ). Further, a greater proportion of patients treated with ACTEMRA 162 mg subcutaneously every other week achieved a low level of disease activity as measured by a DAS28-ESR less than 2.6 at Week 24 compared to those treated with placebo (Table 8 ).
| SC-I Per Protocol Population | SC-II Intent To Treat Population | |||
|---|---|---|---|---|
| TCZ SC 162 mg every week + DMARD | TCZ IV 8mg/kg + DMARD | TCZ SC 162 mg every other week + DMARD | Placebo + DMARD | |
| N=558 | N=537 | N=437 | N=219 | |
| TCZ = tocilizumab | ||||
| ACR20 | ||||
| Week 24 | 69% | 73.4% | 61% | 32% |
| Weighted difference (95% CI) | -4% (-9.2, 1.2) | 30% (22.0, 37.0) | ||
| ACR50 | ||||
| Week 24 | 47% | 49% | 40% | 12% |
| Weighted difference (95% CI) | -2% (-7.5, 4.0) | 28% (21.5, 34.4) | ||
| ACR70 | ||||
| Week 24 | 24% | 28% | 20% | 5% |
| Weighted difference (95% CI) | -4% (-9.0, 1.3) | 15% (9.8, 19.9) | ||
| Change in DAS28 [Adjusted mean] | ||||
| Week 24 | -3.5 | -3.5 | -3.1 | -1.7 |
| Adjusted mean difference (95% CI) | 0 (-0.2, 0.1) | -1.4 (-1.7; -1.1) | ||
| DAS28 < 2.6 | ||||
| Week 24 | 38.4% | 36.9% | 32.0% | 4.0% |
| Weighted difference (95% CI) | 0.9 (-5.0, 6.8) | 28.6 (22.0, 35.2) | ||
The results of the components of the ACR response criteria and the percent of ACR20 responders by visit for ACTEMRA-SC in Studies SC-I and SC-II were consistent with those observed for ACTEMRA-IV.
Radiographic Response
In study SC-II, the progression of structural joint damage was assessed radiographically and expressed as a change from baseline in the van der Heijde modified total Sharp score (mTSS). At week 24, significantly less radiographic progression was observed in patients receiving ACTEMRA-SC every other week plus DMARD(s) compared to placebo plus DMARD(s); mean change from baseline in mTSS of 0.62 vs. 1.23, respectively, with an adjusted mean difference of -0.60 (-1.1, -0.1). These results are consistent with those observed in patients treated with intravenous ACTEMRA.
Health Related Outcomes
In studies SC-I and SC-II, the mean decrease from baseline to week 24 in HAQ-DI was 0.6, 0.6, 0.4 and 0.3, and the proportion of patients who achieved a clinically relevant improvement in HAQ-DI (change from baseline of ≥ 0.3 units) was 65%, 67%, 58% and 47%, for the subcutaneous every week, intravenous 8 mg/kg, subcutaneous every other week, and placebo treatment groups, respectively.
Other Health-Related Outcomes
General health status was assessed by the SF-36 in Studies SC-I and SC-II. In Study SC-II, patients receiving ACTEMRA every other week demonstrated greater improvement from baseline compared to placebo in the PCS, MCS, and in all 8 domains of the SF-36. In Study SC-I, improvements in these scores were similar between ACTEMRA-SC every week and ACTEMRA-IV 8 mg/kg.
Giant Cell Arteritis – Subcutaneous Administration
The efficacy and safety of subcutaneously administered ACTEMRA was assessed in a single, randomized, double-blind, multicenter study in patients with active GCA. In Study WA28119 (NCT01791153), 251 screened patients with new-onset or relapsing GCA were randomized to one of four treatment arms. Two subcutaneous doses of ACTEMRA (162 mg every week and 162 mg every other week) were compared to two different placebo control groups (pre-specified prednisone-taper regimen over 26 weeks and 52 weeks) randomized 2:1:1:1. The study consisted of a 52-week blinded period, followed by a 104-week open-label extension.
All patients received background glucocorticoid (prednisone) therapy. Each of the ACTEMRA-treated groups and one of the placebo-treated groups followed a pre-specified prednisone-taper regimen with the aim to reach 0 mg by 26 weeks, while the second placebo-treated group followed a pre-specified prednisone-taper regimen with the aim to reach 0 mg by 52 weeks designed to be more in keeping with standard practice.
The primary efficacy endpoint was the proportion of patients achieving sustained remission from Week 12 through Week 52. Sustained remission was defined by a patient attaining a sustained (1) absence of GCA signs and symptoms from Week 12 through Week 52, (2) normalization of erythrocyte sedimentation rate (ESR) (to < 30 mm/hr without an elevation to ≥ 30 mm/hr attributable to GCA) from Week 12 through Week 52, (3) normalization of C-reactive protein (CRP) (to < 1 mg/dL, with an absence of successive elevations to ≥ 1mg/dL) from Week 12 through Week 52, and (4) successful adherence to the prednisone taper defined by not more than 100 mg of excess prednisone from Week 12 through Week 52. ACTEMRA 162 mg weekly and 162 mg every other week + 26 weeks prednisone taper both showed superiority in achieving sustained remission from Week 12 through Week 52 compared with placebo + 26 weeks prednisone taper (Table 9 ). Both ACTEMRA treatment arms also showed superiority compared to the placebo + 52 weeks prednisone taper (Table 9 ).
| PBO + 26 weeks prednisone taper N=50 | PBO + 52 weeks prednisone taper N=51 | TCZ 162mg SC QW + 26 weeks prednisone taper N=100 | TCZ 162 mg SC Q2W + 26 weeks prednisone taper N=49 | |
|---|---|---|---|---|
| Patients not completing the study to week 52 were classified as non-responders in the primary and key secondary analysis: PBO+26: 6 (12.0%), PBO+52: 5 (9.8%), TCZ QW: 15 (15.0%), TCZ Q2W: 9 (18.4%). CRP = C-reactive protein ESR = erythrocyte sedimentation rate PBO = placebo Q2W = every other week dose QW = every week dose TCZ = tocilizumab | ||||
| Sustained remission Sustained remission was achieved by a patient meeting all of the following components: absence of GCA signs and symptoms † , normalization of ESR ‡ , normalization of CRP § and adherence to the prednisone taper regimen ¶ . | ||||
| Responders, n (%) | 7 (14.0%) | 9 (17.6%) | 56 (56.0%) | 26 (53.1%) |
| Unadjusted difference in proportions vs PBO + 26 weeks taper (99.5% CI) | N/A | N/A | 42.0% (18.0, 66.0) | 39.1% (12.5 , 65.7) |
| Unadjusted difference in proportions vs PBO + 52 weeks taper (99.5% CI) | N/A | N/A | 38.4% (14.4, 62.3) | 35.4% (8.6, 62.2) |
| Components of Sustained Remission | ||||
| Sustained absence of GCA signs and symptoms Patients who did not have any signs or symptoms of GCA recorded from Week 12 up to Week 52. , n (%) | 20 (40.0%) | 23 (45.1%) | 69 (69.0%) | 28 (57.1%) |
| Sustained ESR<30 mm/hr Patients who did not have an elevated ESR ≥30 mm/hr which was classified as attributed to GCA from Week 12 up to Week 52. , n (%) | 20 (40.0%) | 22 (43.1%) | 83 (83.0%) | 37 (75.5%) |
| Sustained CRP normalization Patients who did not have two or more consecutive CRP records of ≥ 1mg/dL from Week 12 up to Week 52. , n (%) | 17 (34.0%) | 13 (25.5%) | 72 (72.0%) | 34 (69.4%) |
| Successful prednisone tapering Patients who did not enter escape therapy and received ≤ 100mg of additional concomitant prednisone from Week 12 up to Week 52. , n (%) | 10 (20.0%) | 20 (39.2%) | 60 (60.0%) | 28 (57.1%) |
The estimated annual cumulative prednisone dose was lower in the two ACTEMRA dose groups (medians of 1887 mg and 2207 mg on ACTEMRA QW and Q2W, respectively) relative to the placebo arms (medians of 3804 mg and 3902 mg on placebo + 26 weeks prednisone and placebo + 52 weeks prednisone taper, respectively).
Giant Cell Arteritis – Intravenous Administration
Intravenously administered ACTEMRA in patients with GCA was assessed in WP41152 (NCT03923738), an open-label PK-PD and safety study to determine the appropriate intravenous dose of ACTEMRA that achieved comparable PK-PD profiles to the ACTEMRA-SC regimen.
At enrollment, all patients (n=24) were in remission on ACTEMRA-IV. In Period 1, all patients received open-label ACTEMRA-IV 7 mg/kg every 4 weeks for 20 weeks. Patients who completed Period 1 and remained in remission (n=22) were eligible to enter Period 2, and received open-label ACTEMRA-IV 6 mg/kg every 4 weeks for 20 weeks.
The efficacy of intravenous ACTEMRA 6 mg/kg in adult patients with GCA is based on pharmacokinetic exposure and extrapolation to the efficacy established for subcutaneous ACTEMRA in patients with GCA [see Clinical Pharmacology (12.3) and Clinical Studies (14.3) ] .
Systemic Sclerosis-Associated Interstitial Lung Disease– Subcutaneous Administration
The clinical efficacy of ACTEMRA was assessed in a phase 3 multicenter, randomized, double-blind, placebo controlled study in patients with SSc (Study WA29767). A phase 2/3 multicenter, randomized, double-blind, placebo controlled study in patients with SSc (Study WA27788) provided supportive information. Study WA29767 (NCT02453256) enrolled adult patients with SSc defined by the 2013 American College of Rheumatology/European League Against Rheumatism classification criteria for SSc, with onset of disease (first non-Raynaud symptom) of ≤ 5 years, modified Rodnan Skin Score (mRSS) of ≥ 10 and ≤ 35 at screening, elevated inflammatory markers (or platelets), and active disease based on at least one of the following: disease duration ≤ 18 months, increase in mRSS ≥ 3 units over 6 months, involvement of one new body area and an increase in mRSS of ≥ 2 over 6 months, or involvement of two new body areas within the previous 6 months, or presence of at least one tendon friction rub. Study WA27788 (NCT01532869) enrolled adult patients with SSc with onset of disease ≤ 5 years, mRSS of ≥ 15 and ≤ 40 at screening, active disease, and elevated inflammatory markers or platelets. Patients in both studies were not permitted to use biologic agents (such as TNF antagonists), alkylating agents, or cyclophosphamide.
In Study WA29767, 212 patients were randomized in a 1:1 ratio to receive weekly SC injections of 162 mg of ACTEMRA or placebo during the 48-week, double-blinded, placebo controlled period. Rescue treatment was allowed during the treatment period after 16 weeks for >10% percent predicted FVC (ppFVC) decline or after 24 weeks for worsening skin fibrosis. The primary efficacy endpoint was change from baseline at Week 48 in mRSS. Change from baseline in FVC at Week 48 was a key secondary endpoint.
In the overall population of Study WA29767, there was not a statistically significant difference in the mean change from baseline to Week 48 in mRSS (primary endpoint) in patients receiving ACTEMRA compared to placebo (difference: -1.73; 95% CI: -3.78, 0.32). There also was not a statistically significant effect on the primary endpoint of mRSS in Study WA27788.
In the overall population of Study WA29767, patients treated with ACTEMRA, as compared to placebo treated patients, were observed to have less decline from baseline in ppFVC and observed FVC at 48 weeks. FVC results from Study WA27788 were similar.
Of the 212 patients who were randomized in Study WA29767, 68 patients (65%) in the ACTEMRA arm and 68 patients (64%) in the placebo arm had SSc-ILD at baseline, as confirmed by a visual read of high resolution computed tomograph (HRCT) by blinded thoracic radiologists. The mean ppFVC at baseline for patients with SSc-ILD identified by HRCT was 79.6% (median 80.5%). Post-hoc analyses were performed to evaluate results within the subgroups of patients with and without SSc-ILD.
Table 10 shows results from Study WA29767 for the changes from baseline to Week 48 in ppFVC, observed FVC, and mRSS both in the overall population and within subgroups based on SSc-ILD status at baseline. The ppFVC and observed FVC results in the overall population were primarily driven by results in the SSc-ILD subgroup. In the SSc-ILD subgroup, the differences in mean changes from baseline to Week 48 for ACTEMRA, as compared to placebo, were 6.47% and 241 mL for ppFVC and observed FVC, respectively. Figure 2 illustrates the mean change from baseline through Week 48 in observed FVC in patients with SSc-ILD.
The results of the key FVC secondary endpoints from Study WA29767 support a conclusion of effectiveness of ACTEMRA in reducing the rate of progressive loss of lung function in the study population. However, in settings where a trial does not provide evidence of an effect on the primary endpoint, the estimated magnitude of effect on other endpoints should be interpreted with caution, and comparisons to results of other products and studies may be misleading.
| Overall Population | Subgroup Without SSc-ILD Post-hoc results are shown for these subgroups. Four patients had ILD status missing at baseline. | SSc-ILD Subgroup | ||||
|---|---|---|---|---|---|---|
| PBO QW | TCZ 162 mg QW | PBO QW | TCZ 162 mg QW | PBO QW | TCZ 162 mg QW | |
| Number of patients | 106 | 104 | 36 | 34 | 68 | 68 |
| PBO=placebo; TCZ=tocilizumab; ppFVC = percent predicted forced vital capacity; LSM=least squares mean; mRSS = modified Rodnan skin score; CI=confidence interval | ||||||
| Change from Baseline in mRSS at Week 48 | ||||||
| LSM | -4.41 | -6.14 | -6.16 | -8.56 | -3.77 | -5.88 |
| Difference in LSM, TCZ-placebo (95% CI) | -1.73 (-3.78, 0.32) | -2.40 (-5.59, 0.79) | -2.11 (-4.89, 0.67) | |||
| Change from Baseline in ppFVC at Week 48 | ||||||
| LSM | -4.58 | -0.38 | -0.82 | -0.32 | -6.40 | 0.07 |
| Difference in LSM, TCZ-placebo (95% CI) | 4.20 (2.00, 6.40) | 0.50 (-2.27, 3.27) | 6.47 (3.43, 9.50) | |||
| Change from Baseline in Observed FVC (mL) at Week 48 | ||||||
| LSM | -190 | -24 | -53 | -11 | -255 | -14 |
| Difference in LSM, TCZ-placebo (95% CI) | 167 (83, 250) | 43 (-60, 145) | 241 (124, 358) | |||
Figure 2 Mean Change from Baseline to Week 48 in Observed Forced Vital Capacity in SSc-ILD Patients from Study WA29767
| PBO = placebo; TCZ = tocilizumab; QW = every week dose |
 |
Polyarticular Juvenile Idiopathic Arthritis – Intravenous Administration
The efficacy of ACTEMRA was assessed in a three-part study, WA19977 (NCT00988221), including an open-label extension in children 2 to 17 years of age with active polyarticular juvenile idiopathic arthritis (PJIA), who had an inadequate response to methotrexate or inability to tolerate methotrexate. Patients had at least 6 months of active disease (mean disease duration of 4.2 ± 3.7 years), with at least five joints with active arthritis (swollen or limitation of movement accompanied by pain and/or tenderness) and/or at least 3 active joints having limitation of motion (mean, 20 ± 14 active joints). The patients treated had subtypes of JIA that at disease onset included Rheumatoid Factor Positive or Negative Polyarticular JIA, or Extended Oligoarticular JIA. Treatment with a stable dose of methotrexate was permitted but was not required during the study. Concurrent use of disease modifying antirheumatic drugs (DMARDs), other than methotrexate, or other biologics (e.g., TNF antagonists or T cell costimulation modulator) were not permitted in the study.
Part I consisted of a 16-week active ACTEMRA treatment lead-in period (n=188) followed by Part II, a 24-week randomized double-blind placebo-controlled withdrawal period, followed by Part III, a 64-week open-label period. Eligible patients weighing at or above 30 kg received ACTEMRA at 8 mg/kg intravenously once every four weeks. Patients weighing less than 30 kg were randomized 1:1 to receive either ACTEMRA 8 mg/kg or 10 mg/kg intravenously every four weeks. At the conclusion of the open-label Part I, 91% of patients taking background MTX in addition to tocilizumab and 83% of patients on tocilizumab monotherapy achieved an ACR 30 response at week 16 compared to baseline and entered the blinded withdrawal period (Part II) of the study. The proportions of patients with JIA ACR 50/70 responses in Part I were 84.0%, and 64%, respectively for patients taking background MTX in addition to tocilizumab and 80% and 55% respectively for patients on tocilizumab monotherapy.
In Part II, patients (ITT, n=163) were randomized to ACTEMRA (same dose received in Part I) or placebo in a 1:1 ratio that was stratified by concurrent methotrexate use and concurrent corticosteroid use. Each patient continued in Part II of the study until Week 40 or until the patient satisfied JIA ACR 30 flare criteria (relative to Week 16) and qualified for escape.
The primary endpoint was the proportion of patients with a JIA ACR 30 flare at week 40 relative to week 16. JIA ACR 30 flare was defined as 3 or more of the 6 core outcome variables worsening by at least 30% with no more than 1 of the remaining variables improving by more than 30% relative to Week 16.
ACTEMRA treated patients experienced significantly fewer disease flares compared to placebo-treated patients (26% [21/82] versus 48% [39/81]; adjusted difference in proportions -21%, 95% CI: -35%, -8%).
During the withdrawal phase (Part II), more patients treated with ACTEMRA showed JIA ACR 30/50/70 responses at Week 40 compared to patients withdrawn to placebo.
Polyarticular Juvenile Idiopathic Arthritis – Subcutaneous Administration
Subcutaneously administered ACTEMRA in pediatric patients with polyarticular juvenile idiopathic arthritis (PJIA) was assessed in WA28117 (NCT01904279), a 52-week, open-label, multicenter, PK-PD and safety study to determine the appropriate subcutaneous dose of ACTEMRA that achieved comparable PK/PD profiles to the ACTEMRA-IV regimen. PJIA patients aged 1 to 17 years with an inadequate response or inability to tolerate MTX, including patients with well-controlled disease on treatment with ACTEMRA-IV and ACTEMRA-naïve patients with active disease, were treated with subcutaneous ACTEMRA based on body weight.
Patients weighing at or above 30 kg (n = 25) were treated with 162 mg of ACTEMRA-SC every 2 weeks and patients weighing less than 30 kg (n = 27) received 162 mg of ACTEMRA-SC every 3 weeks for 52 weeks. Of these 52 patients, 37 (71%) were naive to ACTEMRA and 15 (29%) had been receiving ACTEMRA-IV and switched to ACTEMRA-SC at baseline.
The efficacy of subcutaneous ACTEMRA in children 2 to 17 years of age is based on pharmacokinetic exposure and extrapolation of the established efficacy of intravenous ACTEMRA in polyarticular JIA patients and subcutaneous ACTEMRA in patients with RA [see Clinical Pharmacology (12.3) and Clinical Studies (14.2 and 14.6) ] .
Systemic Juvenile Idiopathic Arthritis – Intravenous Administration
The efficacy of ACTEMRA for the treatment of active SJIA was assessed in WA18221 (NCT00642460), a 12-week randomized, double blind, placebo-controlled, parallel group, 2-arm study. Patients treated with or without MTX, were randomized (ACTEMRA:placebo = 2:1) to one of two treatment groups: 75 patients received ACTEMRA infusions every two weeks at either 8 mg per kg for patients at or above 30 kg or 12 mg per kg for patients less than 30 kg and 37 were randomized to receive placebo infusions every two weeks. Corticosteroid tapering could occur from week six for patients who achieved a JIA ACR 70 response. After 12 weeks or at the time of escape, due to disease worsening, patients were treated with ACTEMRA in the open-label extension phase at weight appropriate dosing.
The primary endpoint was the proportion of patients with at least 30% improvement in JIA ACR core set (JIA ACR 30 response) at Week 12 and absence of fever (no temperature at or above 37.5°C in the preceding 7 days). JIA ACR (American College of Rheumatology) responses are defined as the percentage improvement (e.g., 30%, 50%, 70%) in 3 of any 6 core outcome variables compared to baseline, with worsening in no more than 1 of the remaining variables by 30% or more. Core outcome variables consist of physician global assessment, parent per patient global assessment, number of joints with active arthritis, number of joints with limitation of movement, erythrocyte sedimentation rate (ESR), and functional ability (childhood health assessment questionnaire-CHAQ).
Primary endpoint result and JIA ACR response rates at Week 12 are shown in Table 11 .
| ACTEMRA N=75 | Placebo N=37 | |
|---|---|---|
| Primary Endpoint: JIA ACR 30 response + absence of fever | ||
| Responders | 85% | 24% |
| Weighted difference (95% CI) | 62 (45, 78) | - |
| JIA ACR Response Rates at Week 12 | ||
| JIA ACR 30 | ||
| Responders | 91% | 24% |
| Weighted difference The weighted difference is the difference between the ACTEMRA and Placebo response rates, adjusted for the stratification factors (weight, disease duration, background oral corticosteroid dose and background methotrexate use). (95% CI) CI: confidence interval of the weighted difference. | 67 (51, 83) | - |
| JIA ACR 50 | ||
| Responders | 85% | 11% |
| Weighted difference (95% CI) | 74 (58, 90) | - |
| JIA ACR 70 | ||
| Responders | 71% | 8% |
| Weighted difference (95% CI) | 63 (46, 80) | - |
The treatment effect of ACTEMRA was consistent across all components of the JIA ACR response core variables. JIA ACR scores and absence of fever responses in the open label extension were consistent with the controlled portion of the study (data available through 44 weeks).
Systemic Features
Of patients with fever or rash at baseline, those treated with ACTEMRA had fewer systemic features; 35 out of 41 (85%) became fever free (no temperature recording at or above 37.5°C in the preceding 14 days) compared to 5 out of 24 (21%) of placebo-treated patients, and 14 out of 22 (64%) became free of rash compared to 2 out of 18 (11%) of placebo-treated patients. Responses were consistent in the open label extension (data available through 44 weeks).
Corticosteroid Tapering
Of the patients receiving oral corticosteroids at baseline, 8 out of 31 (26%) placebo and 48 out of 70 (69%), ACTEMRA patients achieved a JIA ACR 70 response at week 6 or 8 enabling corticosteroid dose reduction. Seventeen (24%) ACTEMRA patients versus 1 (3%) placebo patient were able to reduce the dose of corticosteroid by at least 20% without experiencing a subsequent JIA ACR 30 flare or occurrence of systemic symptoms to week 12. In the open label portion of the study, by week 44, there were 44 out of 103 (43%) ACTEMRA patients off oral corticosteroids. Of these 44 patients 50% were off corticosteroids 18 weeks or more.
Health Related Outcomes
Physical function and disability were assessed using the Childhood Health Assessment Questionnaire Disability Index (CHAQ-DI). Seventy-seven percent (58 out of 75) of patients in the ACTEMRA treatment group achieved a minimal clinically important improvement in CHAQ-DI (change from baseline of ≥ 0.13 units) at week 12 compared to 19% (7 out of 37) in the placebo treatment group.
Systemic Juvenile Idiopathic Arthritis – Subcutaneous Administration
Subcutaneously administered ACTEMRA in pediatric patients with systemic juvenile idiopathic arthritis (SJIA) was assessed in WA28118 (NCT01904292), a 52-week, open-label, multicenter, PK-PD and safety study to determine the appropriate subcutaneous dose of ACTEMRA that achieved comparable PK/PD profiles to the ACTEMRA-IV regimen.
Eligible patients received ACTEMRA subcutaneously dosed according to body weight, with patients weighing at or above 30 kg (n = 26) dosed with 162 mg of ACTEMRA every week and patients weighing below 30 kg (n = 25) dosed with 162 mg of ACTEMRA every 10 days (n=8) or every 2 weeks (n=17) for 52 weeks. Of these 51 patients, 26 (51%) were naive to subcutaneous ACTEMRA and 25 (49%) had been receiving ACTEMRA intravenously and switched to subcutaneous ACTEMRA at baseline.
The efficacy of subcutaneous ACTEMRA in children 2 to 17 years of age is based on pharmacokinetic exposure and extrapolation of the established efficacy of intravenous ACTEMRA in systemic JIA patients [see Clinical Pharmacology (12.3) and Clinical Studies (14.8) ] .
Cytokine Release Syndrome – Intravenous Administration
The efficacy of ACTEMRA for the treatment of CRS was assessed in a retrospective analysis of pooled outcome data from clinical trials of CAR T-cell therapies for hematological malignancies. Evaluable patients had been treated with tocilizumab 8 mg/kg (12 mg/kg for patients < 30 kg) with or without additional high-dose corticosteroids for severe or life-threatening CRS; only the first episode of CRS was included in the analysis. The study population included 24 males and 21 females (total 45 patients) of median age 12 years (range, 3–23 years); 82% were Caucasian. The median time from start of CRS to first dose of tocilizumab was 4 days (range, 0-18 days). Resolution of CRS was defined as lack of fever and off vasopressors for at least 24 hours. Patients were considered responders if CRS resolved within 14 days of the first dose of tocilizumab, if no more than 2 doses of tocilizumab were needed, and if no drugs other than tocilizumab and corticosteroids were used for treatment. Thirty-one patients (69%; 95% CI: 53%–82%) achieved a response. Achievement of resolution of CRS within 14 days was confirmed in a second study using an independent cohort that included 15 patients (range: 9–75 years old) with CAR T cell-induced CRS.
COVID-19 – Intravenous Administration
The efficacy of ACTEMRA for the treatment of COVID-19 in adults was based on RECOVERY (NCT04381936), a randomized, controlled, open-label, platform study, and supported by the results from EMPACTA (NCT04372186), a randomized, double-blind, placebo-controlled study. Results of two other randomized, double-blind, placebo-controlled studies, COVACTA (NCT04320615) and REMDACTA (NCT04409262 ) , which evaluated the efficacy of ACTEMRA for the treatment of COVID-19 are also summarized.
RECOVERY (Randomised Evaluation of COVID-19 Therapy) Collaborative Group Study in Hospitalized Adults Diagnosed with COVID-19
RECOVERY was a randomized, controlled, open-label, multicenter platform study conducted in the United Kingdom to evaluate the efficacy and safety of potential treatments in hospitalized adult patients with severe COVID-19 pneumonia. Eligible patients for the ACTEMRA portion of the study had clinically suspected or laboratory-confirmed SARS-CoV-2 infection and no medical contraindications to any of the treatments and had clinical evidence of progressive COVID-19 (defined as oxygen saturation <92% on room air or receiving oxygen therapy, and CRP ≥75 mg/L). Patients were then randomized to receive either standard of care (SoC) or intravenous ACTEMRA at a weight-tiered dosing comparable to the recommended dosage [see Clinical Pharmacology (12.3) ] in addition to SoC.
Efficacy analyses were performed in the intent-to-treat (ITT) population comprising 4116 adult patients who were randomized to the ACTEMRA + SoC arm (n=2022) or to the SoC arm (n=2094). The mean age of participants was 64 years (range: 20 to 101), and patients were 67% male, 76% White, 11% Asian, 3% Black or African American, and 1% mixed race. At baseline, 0.2% of patients were not on supplemental oxygen, 45% of patients required low flow oxygen, 41% of patients required non-invasive ventilation or high-flow oxygen, and 14% of patients required invasive mechanical ventilation; 82% of patients were reported to be receiving systemic corticosteroids.
The primary efficacy endpoint was time to death through Day 28. The results for the overall population and the subgroups of patients who were or were not receiving systemic corticosteroids at time of randomization are summarized in Table 12 .
| ACTEMRA+ SoC N=2022 n (%) P-value reflects that the RECOVERY trial primary analysis results were statistically significant at the two-sided significance level of α = 0.05. | SoC N=2094 n (%) | Hazard Ratio (95% CI) | Risk Difference (95% CI) | |
|---|---|---|---|---|
| Mortality | 621 (30.7%) | 729 (34.9%) | 0.85 (0.76, 0.94) p= 0.0028 | -4.1% (-7.0, -1.3) |
| By baseline receipt of corticosteroid use | ||||
| Mortality for patients receiving systemic corticosteroids at randomization Probabilities of dying by Day 28 were estimated by the Kaplan-Meier method. | 482/1664 (29.0%) | 600/1721 (34.9%) | 0.79 (0.70, 0.89) | -5.9% (-9.1, -2.8) |
| Mortality for patients not receiving systemic corticosteroids at randomization | 139/357 (39.0%) | 127/367 (34.6%) | 1.16 (0.91, 1.48) | 4.4% (-2.6, 11.5) |
EMPACTA
EMPACTA was a randomized, double-blind, placebo-controlled, multicenter study to evaluate intravenous ACTEMRA 8 mg/kg in combination with SoC in hospitalized, non-ventilated adult patients with COVID-19 pneumonia. Eligible patients were at least 18 years of age, had confirmed SARS-CoV-2 infection by a positive reverse-transcriptase polymerase chain reaction (RT-PCR) result, had pneumonia confirmed by radiography, and had SpO2 < 94% on ambient air.
Of the 389 patients randomized, efficacy analyses were performed in the modified intent-to-treat (mITT) population comprising 377 patients who were randomized and received study medication (249 in the ACTEMRA arm; 128 in the placebo arm). The mean age of participants was 56 years (range: 20 to 95); 59% of patients were male, 56% were of Hispanic or Latino ethnicity, 53% were White, 20% were American Indian/Alaska Native, 15% were Black/African American and 2% were Asian. At baseline, 9% patients were not on supplemental oxygen, 64% patients required low flow oxygen, 27% patients required high-flow oxygen, and 73% were on corticosteroids.
The primary efficacy endpoint evaluated time to progression to mechanical ventilation or death through Day 28. The hazard ratio comparing ACTEMRA to placebo was 0.56 (95% CI, 0.33 to 0.97), a statistically significant result (log-rank, p-value = 0.036). The cumulative proportion of patients who required mechanical ventilation or died by Day 28 was 12.0% (95% CI, 8.5% to 16.9%) in the ACTEMRA arm and 19.3% (95% CI, 13.3% to 27.4%) in the placebo arm.
Mortality at Day 28 was 10.4% in the ACTEMRA arm versus 8.6% in the placebo arm (weighted difference (ACTEMRA arm - placebo arm): 2.0% [95% CI, -5.2% to 7.8%]).
COVACTA
COVACTA was a randomized, double-blind, placebo-controlled, multicenter study to evaluate intravenous ACTEMRA 8 mg/kg in combination with SoC for the treatment of adult patients hospitalized with severe COVID-19 pneumonia. The study randomized 452 patients who were at least 18 years of age with confirmed SARS-CoV-2 infection by a positive RT-PCR result, had pneumonia confirmed by radiography, and had oxygen saturation of 93% or lower on ambient air or a ratio of arterial oxygen partial pressure to fractional inspired oxygen of 300 mmHg or less. At baseline, 3% of patients were not on supplemental oxygen, 28% were on low flow oxygen, 30% were on non-invasive ventilation or high flow oxygen, 38% were on invasive mechanical ventilation, and 22% were on corticosteroids. The primary efficacy endpoint was clinical status on Day 28 assessed on a 7-category ordinal scale that ranged from "discharged" to "death." There were no statistically significant differences observed in the distributions of clinical status on the 7-category ordinal scale at Day 28 when comparing the ACTEMRA arm to the placebo arm.
Mortality at Day 28 was 19.7% in the ACTEMRA arm versus 19.4% in the placebo arm (weighted difference (ACTEMRA arm - placebo arm): 0.3% [95% CI, -7.6 to 8.2]).
REMDACTA
REMDACTA was a randomized, double-blind, placebo-controlled, multicenter study to evaluate intravenous ACTEMRA 8 mg/kg in combination with intravenous remdesivir (RDV) 200 mg on Day 1 followed by 100 mg once daily for a total of 10 days in hospitalized adult patients with severe COVID-19 pneumonia. The study randomized 649 adult patients with SARS-CoV-2 infection confirmed by a positive polymerase chain reaction (PCR) result, pneumonia confirmed by radiography, and who required supplemental oxygen > 6 L/min to maintain SpO2 >93%. At baseline, 7% of patients were on low flow oxygen, 80% were on non-invasive ventilation or high flow oxygen, 14% were on invasive mechanical ventilation, and 84% were on corticosteroids.
The primary efficacy endpoint was time from randomization to hospital discharge or 'ready for discharge' up to Day 28. There was no statistically significant difference between the treatment arms with respect to time to hospital discharge or "ready for discharge" through Day 28.
Mortality at Day 28 was 18.1% in the ACTEMRA+ RDV arm versus 19.5% in the placebo +RDV arm (weighted difference (ACTEMRA arm - placebo arm): -1.3% [95% CI, -7.8% to 5.2%]).
Mortality Across Studies in Patients Receiving Baseline Corticosteroids
A study-level meta-analysis was conducted on EMPACTA, COVACTA, REMDACTA and RECOVERY studies. For each of the four studies, the risk difference through Day 28 was estimated by the Kaplan-Meier method in the subgroup of patients receiving baseline corticosteroids, summarized in Figure 3 . Patients from the RECOVERY trial represent 78.8% of the total sample size in this meta-analysis. The combined risk difference showed that ACTEMRA treatment (n=2261) resulted in a 4.61% absolute reduction in the risk of death at Day 28 (risk difference=-4.6%; 95% CI: -7.3% to -1.9%) compared to SoC (n=2034).
Figure 3 Risk Differences Through Day 28 for Baseline Corticosteroid Use Subpopulation in RECOVERY, EMPACTA, COVACTA and REMDACTA studies

HOW SUPPLIED/STORAGE AND HANDLING
For Intravenous Infusion
ACTEMRA (tocilizumab) injection is a preservative-free, sterile clear, colorless to pale yellow solution. ACTEMRA is supplied as 80 mg/4 mL (NDC 50242-135-01), 200 mg/10 mL (NDC 50242-136-01), and 400 mg/20 mL (NDC 50242-137-01) individually packaged 20 mg/mL single-dose vials for further dilution prior to intravenous infusion.
For Subcutaneous Injection
ACTEMRA (tocilizumab) injection is supplied as a preservative-free, sterile, clear, colorless to slightly yellowish solution for subcutaneous administration. The following packaging configurations are available:
- Each single-dose prefilled syringe delivers 162 mg/0.9 mL (NDC 50242-138-01).
- Each single-dose ACTPen ® autoinjector delivers 162 mg/0.9 mL (NDC 50242-143-01).
Storage and Handling: Do not use beyond expiration date on the container, package, prefilled syringe, or autoinjector. ACTEMRA must be refrigerated at 36°F to 46°F (2ºC to 8ºC). Do not freeze. Protect the vials, syringes, and autoinjectors from light by storage in the original package until time of use, and keep syringes and autoinjectors dry. Once removed from the refrigerator, the prefilled syringe and autoinjector can be stored up to 2 weeks at or below 86°F (30°C). The prefilled syringe and autoinjector must always be kept in the carton.
Instructions for Use ACTEMRA ® (AC-TEM-RA) (tocilizumab) Injection, For Subcutaneous Use Single-dose Prefilled Syringe
Read and follow the Instructions for Use that come with your ACTEMRA prefilled syringe before you start using it and each time you get a prescription refill. Before you use ACTEMRA prefilled syringe for the first time, make sure your healthcare provider shows you the right way to use it.
- Do not remove the needle cap until you are ready to inject ACTEMRA.
- Do not try to take apart the syringe at any time.
- Do not reuse the same syringe.
Parts of your ACTEMRA Prefilled Syringe (See Figure A ).
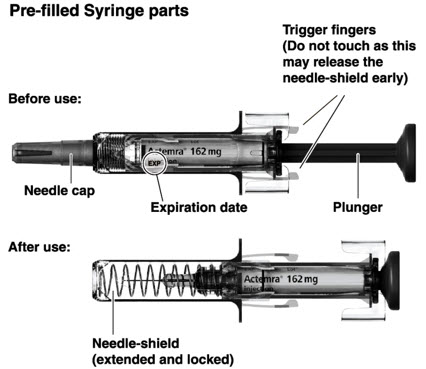
Supplies needed for your ACTEMRA Prefilled Syringe Injection (See Figure B ):
- ACTEMRA prefilled syringe
- alcohol pad
- sterile cotton ball or gauze
- puncture-resistant container or sharps container for safe disposal of needle cap and used syringe (See Step 4 "Dispose of the syringe" )
Step 1. Preparing for an ACTEMRA Injection
Find a comfortable space with a clean, flat, working surface.
- Take the box containing the syringe out of the refrigerator and open the box. Do not touch the trigger fingers on the syringe as this may damage the syringe.
- Remove 1 single-use ACTEMRA prefilled syringe from the box and let it warm up for 30 minutes to allow it to reach room temperature. If the syringe does not reach room temperature, this could cause your injection to feel uncomfortable and make it difficult to push the plunger in.
- Do not speed up the warming process in any way, such as using the microwave or placing the syringe in warm water.
- Check the expiration date on the ACTEMRA prefilled syringe (See Figure A ). Do not use it if the expiration date has passed because it may not be safe to use. If the expiration date has passed safely dispose of the syringe in a sharps container and get a new one.
Do not remove the needle cap while allowing your ACTEMRA prefilled syringe to reach room temperature.
- Keep your unused syringes in the original carton and keep in the refrigerator at 36°F to 46°F (2°C to 8°C). Do not freeze.
- Once removed from the refrigerator, your prefilled syringe can be stored up to 2 weeks at or below 86°F (30°C). Your prefilled syringe must always be kept in the original carton in order to protect from light and moisture. Hold your ACTEMRA prefilled syringe with the covered needle pointing down (See Figure C ).
- Check the liquid in the ACTEMRA prefilled syringe. It should be clear and colorless to pale yellow. Do not inject ACTEMRA if the liquid is cloudy, discolored, or has lumps or particles in it because it may not be safe to use. Safely dispose of the syringe in a sharps container and get a new one.
- Wash your hands well with soap and water.
Step 2. Choose and Prepare an Injection Site
Choose an Injection Site
- The front of your thigh and your abdomen except for the 2-inch area around your navel are the recommended injection sites (See Figure D ).
- The outer area of the upper arms may also be used only if the injection is being given by a caregiver. Do not attempt to use the upper arm area by yourself (See Figure D ).
Rotate Injection Site
- Choose a different injection site for each new injection at least 1-inch from the last area you injected.
- Do not inject into moles, scars, bruises, or areas where the skin is tender, red, hard or not intact.
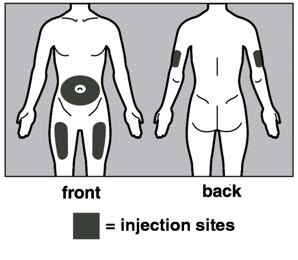
Prepare the Injection Site
- Wipe the injection site with an alcohol pad in a circular motion and let it air dry to reduce the chance of getting an infection. Do not touch the injection site again before giving the injection.
- Do not fan or blow on the clean area.
Step 3. Inject ACTEMRA
- Hold the ACTEMRA prefilled syringe with 1 hand and pull the needle cap straight off with your other hand (See Figure E ). Do not hold the plunger while you remove the needle cap. If you cannot remove the needle cap you should ask a caregiver for help or contact your healthcare provider.
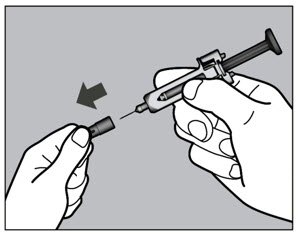
- Throw away the needle cap in a sharps container.
- There may be a small air bubble in the ACTEMRA prefilled syringe. You do not need to remove it.
- You may see a drop of liquid at the end of the needle. This is normal and will not affect your dose.
- Do not touch the needle or let it touch any surfaces.
- Do not use the prefilled syringe if it is dropped.
- If it is not used within 5 minutes of needle cap removal, the syringe should be disposed of in the puncture resistant container or sharps container and a new syringe should be used.
- Never reattach the needle cap after removal.
- Hold the ACTEMRA prefilled syringe in 1 hand between the thumb and index finger (See Figure F ).
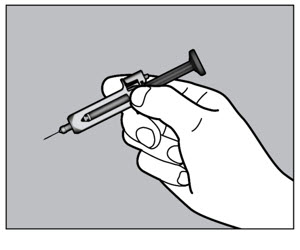
- Do not pull back on the plunger of the syringe.
- Use your other hand and gently pinch the area of skin you cleaned. Hold the pinched skin firmly. Pinching the skin is important to make sure that you inject under the skin (into fatty tissue) but not any deeper (into muscle). Injection into muscle could cause the injection to feel uncomfortable.
- Do not hold or push on the plunger while inserting the needle into the skin.
- Use a quick, dart-like motion to insert the needle all the way into the pinched skin at an angle between 45° to 90° (See Figure G ). It is important to use the correct angle to make sure the medicine is delivered under the skin (into fatty tissue), or the injection could be painful and the medicine may not work.
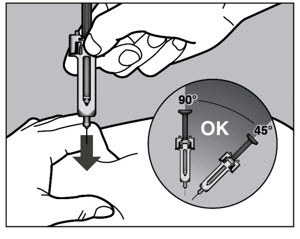
- Keep the syringe in position and let go of the pinch of skin.
- Slowly inject all of the medicine by gently pushing the plunger all the way down (See Figure H ). You must press the plunger all the way down to get the full dose of medicine and to ensure the trigger fingers are completely pushed to the side. If the plunger is not fully depressed the needle shield will not extend to cover the needle when it is removed. If the needle is not covered, carefully place the syringe into the puncture resistant container to avoid injury with the needle.
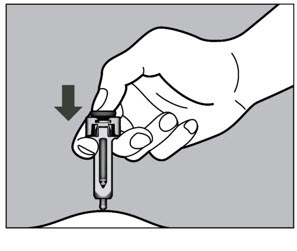
- After the plunger is pushed all the way down, keep pressing down on the plunger to be sure all of the medicine is injected before taking the needle out of the skin.
- Keep pressing down on the plunger while you take the needle out of the skin at the same angle as inserted (See Figure I ).
- After the needle is removed completely from the skin, release the plunger, allowing the needle-shield to protect the needle (See Figure J ).
After the Injection
- There may be a little bleeding at the injection site. You can press a cotton ball or gauze over the injection site.
- Do not rub the injection site.
- If needed, you may cover the injection site with a small bandage.
Step 4. Dispose of the syringe
- The ACTEMRA prefilled syringe should not be reused.
- Put the used syringe into your puncture resistant container (See " How do I throw away used syringes? ")
- Do not put the needle cap back on the needle.
- If your injection is given by another person, this person must also be careful when removing the syringe and disposing of the syringe to prevent accidental needle stick injury and passing infection.
How do I throw away used syringes?
- Put your used needles and syringes including ACTEMRA in a FDA-cleared sharps disposal container right away after use (See Figure K ). Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out
- upright stable during use
- leak-resistant
- properly labeled to warn of hazardous waste inside the container
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about the safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal .
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
- Keep ACTEMRA prefilled syringes and the disposal container out of the reach of children.
Record your Injection
- Write the date, time, and specific part of your body where you injected yourself. It may also be helpful to write any questions or concerns about the injection so you can ask your healthcare provider.
If you have questions or concerns about your ACTEMRA prefilled syringe, please contact your healthcare provider familiar with ACTEMRA or call 1-800-ACTEMRA.
This Medication Guide and Instructions for Use has been approved by the U.S. Food and Drug Administration. Medication Guide Revised: 08/2025 ACTEMRA is a registered trademark of Chugai Seiyaku Kabushiki Kaisha Corp., a member of the Roche Group.
Genentech, Inc. A Member of the Roche Group 1 DNA Way South San Francisco, CA 94080-4990
U.S. License No.: 1048 © 2025 Genentech, Inc. All rights reserved.
Mechanism of Action
Tocilizumab binds to both soluble and membrane-bound IL-6 receptors (sIL-6R and mIL-6R), and has been shown to inhibit IL-6-mediated signaling through these receptors. IL-6 is a pleiotropic pro-inflammatory cytokine produced by a variety of cell types including T- and B-cells, lymphocytes, monocytes and fibroblasts. IL-6 has been shown to be involved in diverse physiological processes such as T-cell activation, induction of immunoglobulin secretion, initiation of hepatic acute phase protein synthesis, and stimulation of hematopoietic precursor cell proliferation and differentiation. IL-6 is also produced by synovial and endothelial cells leading to local production of IL-6 in joints affected by inflammatory processes such as rheumatoid arthritis.