Get your patient on Dyural 40 Kit (Methylprednisolone Acetate, Lidocaine Hydrochloride, Bupivacaine Hydrochloride, Povidine Iodine, Sodium Chloride, Isopropyl Alcohol)
Dyural 40 Kit prescribing information
WARNING: RISK OF CARDIAC ARREST WITH USE OF BUPIVACAINE HYDROCHLORIDE INJECTION IN OBSTETRICAL ANESTHESIA
There have been reports of cardiac arrest with difficult resuscitation or death during use of Bupivacaine Hydrochloride Injection for epidural anesthesia in obstetrical patients. In most cases, this has followed use of the 0.75% (7.5 mg/mL) concentration. Resuscitation has been difficult or impossible despite apparently adequate preparation and appropriate management. Cardiac arrest has occurred after convulsions resulting from systemic toxicity, presumably following unintentional intravascular injection. The 0.75% (7.5 mg/mL) concentration of Bupivacaine Hydrochloride Injection is not recommended for obstetrical anesthesia and should be reserved for surgical procedures where a high degree of muscle relaxation and prolonged effect are necessary [see Warnings and Precautions (5.1) ].
INDICATIONS AND USAGE
Bupivacaine Hydrochloride Injection is indicated in adults for the production of local or regional anesthesia or analgesia for surgery, dental and oral surgery procedures, diagnostic and therapeutic procedures, and for obstetrical procedures. Specific concentrations and presentations of Bupivacaine Hydrochloride Injection are recommended for each type of block indicated to produce local or regional anesthesia or analgesia [see Dosage and Administration (2.2) ].
Limitations of Use
Not all blocks are indicated for use with Bupivacaine Hydrochloride Injection given clinically significant risks associated with use [see Dosage and Administration (2.2) , Contraindications (4) , Warnings and Precautions (5.1 , 5.4 , 5.5 , 5.7 , 5.9) ] .
DOSAGE AND ADMINISTRATION
- Not for intrathecal use. (2.1 )
- Avoid use of solutions containing antimicrobial preservatives (i.e., multiple-dose vials) for epidural or caudal anesthesia. (2.1 , 5.4 )
- Three mL of Bupivacaine Hydrochloride and Epinephrine Injection without antimicrobial preservative (0.5% bupivacaine with 1:200,000 epinephrine) is recommended for use as a test dose prior to caudal and lumbar epidural blocks when clinical conditions permit. (2.4 )
- See full prescribing information for:
- Recommended concentrations and dosages of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection according to type of block. (2.2 )
- Additional dosage and administration information pertaining to use in epidural anesthesia, test dose for caudal and lumbar epidural blocks, use in dentistry, and use in ophthalmic surgery. (2.3 , 2.4 , 2.5 , 2.6 )
Important Dosage and Administration Information
- Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection is not for intrathecal use.
- Avoid use of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection solutions containing antimicrobial preservatives (i.e., multiple-dose vials) for epidural or caudal anesthesia [see Warnings and Precautions (5.4) ] .
- Discard unused portions of solution not containing preservatives, i.e., those supplied in single-dose vials, following initial use.
- Visually inspect this product for particulate matter and discoloration prior to administration whenever solution and container permit. Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection are clear, colorless solutions. Do not administer solutions which are discolored or contain particulate matter.
- Mixing or the prior or intercurrent use of any other local anesthetic with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection is not recommended because of insufficient data on the clinical use of such mixtures.
Administration Precautions
- Bupivacaine Hydrochloride Injection Injection/Bupivacaine Hydrochloride and Epinephrine Injection are to be administered in carefully adjusted dosages by or under the supervision of experienced clinicians who are well versed in the diagnosis and management of dose-related toxicity and other acute emergencies which might arise from the block to be employed.
- Use Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection only if the following are immediately available: oxygen, cardiopulmonary resuscitative equipment and drugs, and the personnel resources needed for proper management of toxic reactions and related emergencies [see Warnings and Precautions (5.2) , Adverse Reactions (6) , Overdosage (10) ] .
- The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects related to local anesthetic systemic toxicity when additional local anesthetics are administered with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection [see Warnings and Precautions (5.2) , Drug Interactions (7.1) , Overdosage (10) ] .
- Aspirate for blood or cerebrospinal fluid (where applicable) prior to injecting Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection, both the initial dose and all subsequent doses, to avoid intravascular or intrathecal injection. However, a negative aspiration for blood or cerebrospinal fluid does not ensure against an intravascular or intrathecal injection [see Warnings and Precautions (5.9) ] .
- Avoid rapid injection of a large volume of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection and use fractional (incremental) doses when feasible.
- During major regional nerve blocks, such as those of the brachial plexus or lower extremity, the patient should have an indwelling intravenous catheter to assure adequate intravenous access. The lowest dosage of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection that results in effective anesthesia should be used to avoid high plasma levels and serious adverse reactions.
- Perform careful and constant monitoring of cardiovascular and respiratory (adequacy of oxygenation and ventilation) vital signs and the patient's level of consciousness after each local anesthetic injection.
- Use Bupivacaine Hydrochloride and Epinephrine Injection in carefully restricted quantities in areas of the body supplied by end arteries or having otherwise compromised blood supply such as digits, nose, external ear, or penis [see Warnings and Precautions (5.12) ] .
Recommended Concentrations and Dosages of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection
The dosage of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection administered varies with the anesthetic procedure, the area to be anesthetized, the vascularity of the tissues, the number of neuronal segments to be blocked, the depth of anesthesia and degree of muscle relaxation required, the duration of anesthesia desired, individual tolerance, and the physical condition of the patient. Administer the smallest dosage and concentration required to produce the desired result.
The types of block and recommended Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection concentrations are shown in Table 1.
| Type of Block | Bupivacaine Hydrochloride | Bupivacaine Hydrochloride and Epinephrine | |||
|---|---|---|---|---|---|
| 0.25% (2.5 mg/mL) | 0.5% (5 mg/mL) | 0.75% (7.5 mg/mL) Bupivacaine Hydrochloride Injection 0.75% (7.5 mg/mL) is not recommended for nonobstetrical surgical procedures in pregnant patients. | 0.25% (2.5 mg/mL) | 0.5% (5 mg/mL) | |
| ✓= indicated use [see Warnings and Precautions (5.1) ]. | |||||
| Local infiltration | ✓ | ✓ | |||
| Peripheral nerve block | ✓ | ✓ | ✓ | ✓ | |
| Retrobulbar block | ✓ | ||||
| Sympathetic block | ✓ | ||||
| Caudal block Avoid use of multiple-dose vials of Bupivacaine Hydrochloride Injection and Bupivacaine Hydrochloride and Epinephrine Injection for caudal or epidural anesthesia [see Warnings and Precautions (5.4)] . | ✓ | ✓ | ✓ | ✓ | |
| Lumbar epidural block | ✓ | ✓ | ✓ (not for obstetrical anesthesia) | ✓ | ✓ |
| Epidural test dose | ✓ | ||||
| Dental block | ✓ | ||||
At recommended dosages, Bupivacaine Hydrochloride/Bupivacaine Hydrochloride and Epinephrine produces complete sensory block, but the effect on motor function differs among the three concentrations. Table 2 provides information on the expected effect on motor function for the three concentrations.
| Bupivacaine Hydrochloride Injection Concentration | Motor Function |
|---|---|
| 0.25% (2.5 mg/mL) These products include Bupivacaine Hydrochloride Injection and Bupivacaine Hydrochloride and Epinephrine Injection [the epinephrine concentration (1:200,000) is not included in the table]. | When used for caudal, epidural, or peripheral nerve block, produces incomplete motor block. Should be used for operations in which muscle relaxation is not important, or when another means of providing muscle relaxation is used concurrently. Onset of action may be slower than with the 0.5% (5 mg/mL) or 0.75% (7.5 mg/mL) solutions. |
| 0.5% (5 mg/mL) | Provides motor blockade for caudal, epidural, or nerve block, but muscle relaxation may be inadequate for operations in which complete muscle relaxation is essential. |
| 0.75% (7.5 mg/mL) These are only Bupivacaine Hydrochloride Injection products [there is no 0.75% (7.5 mg/mL) concentration for Bupivacaine Hydrochloride and Epinephrine Injection]. | Produces complete motor block. Most useful for epidural block in abdominal operations requiring complete muscle relaxation, and for retrobulbar anesthesia. Not for obstetrical anesthesia. |
The duration of anesthesia with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection is such that for most indications, a single-dose is sufficient.
The maximum dosage limit within the recommended dosage range must be individualized in each case after evaluating the size and physical status of the patient, as well as the anticipated rate of systemic absorption from a particular injection site.
The dosages in Table 3 are recommended as a guide for use in the average adult. These doses may be repeated once every three hours. Do not exceed a total daily dosage of 400 mg in 24 hours. The duration of anesthetic effect may be prolonged by the addition of epinephrine.
| Type of Block | Concentration of Bupivacaine Hydrochloride Injection | Each Dose | Motor Block With continuous (intermittent) techniques, repeat doses increase the degree of motor block. The first repeat dose of 0.5% (5 mg/mL) may produce complete motor block. Intercostal nerve block with 0.25% (2.5 mg/mL) also may produce complete motor block for intra-thoracic and upper intra-abdominal surgery. | |
|---|---|---|---|---|
| mL | mg of Bupivacaine Hydrochloride Injection | |||
| Local infiltration | 0.25% (2.5 mg/mL) Solutions with or without epinephrine (i.e., applies to Bupivacaine Hydrochloride Injection and Bupivacaine Hydrochloride and Epinephrine Injection). The Bupivacaine Hydrochloride and Epinephrine Injection products include epinephrine (1:200,000). | Up to 70 (without epinephrine) | Up to 175 (without epinephrine) | ― |
| Up to 90 (with epinephrine) | Up to 225 (with epinephrine) | |||
| Peripheral nerve block | 0.5% (5 mg/mL) | 5–35 (without epinephrine) | 25–175 (without epinephrine) | moderate to complete |
| 5–45 (with epinephrine) | 25–225 (with epinephrine) | |||
| 0.25% (2.5 mg/mL) | 5–70 (without epinephrine) | 12.5–175 (without epinephrine) | moderate to complete | |
| 5–90 (with epinephrine) | 12.5–225 (with epinephrine) | |||
| Retrobulbar block [see Dosage and Administration (2.6) ] | 0.75% (7.5 mg/mL) | 2–4 | 15–30 | complete |
| Sympathetic block | 0.25% (2.5 mg/mL) | 20–50 | 50–125 | ― |
| Caudal block [see Dosage and Administration (2.4) ] | 0.5% (5 mg/mL) | 15–30 | 75–150 | moderate to complete |
| 0.25% (2.5 mg/mL) | 15–30 | 37.5–75 | moderate | |
| Lumbar epidural block [see Dosage and Administration (2.3) ] | 0.75% (7.5 mg/mL) For single-dose use; not for intermittent epidural technique. Not for obstetrical anesthesia. | 10–20 | 75–150 | complete |
| 0.5% (5 mg/mL) | 10–20 | 50–100 | moderate to complete | |
| 0.25% (2.5 mg/mL) | 10–20 | 25–50 | partial to moderate | |
| Epidural test dose [see Dosage and Administration (2.4) ] | 0.5% (5 mg/mL) with epinephrine | 2–3 | 10–15 (10–15 micrograms epinephrine) | ― |
| Dental [see Dosage and Administration (2.5) ] | 0.5% (5 mg/mL) with epinephrine | 1.8–3.6 per site | 9–18 per site | ― |
Use in Epidural Anesthesia
During the administration of epidural anesthesia, it is recommended that a test dose of Bupivacaine Hydrochloride and Epinephrine Injection without antimicrobial preservative (0.5% bupivacaine with 1:200,000 epinephrine) be administered initially and the effects monitored before the full dose is given. When using a "continuous" catheter technique, test doses should be given prior to both the initial and all supplemental doses, because a catheter in the epidural space can migrate into a blood vessel or through the dura [see Dosage and Administration (2.4) ] .
During epidural administration, administer Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection, 0.5% (5 mg/mL) and Bupivacaine Hydrochloride Injection 0.75% (7.5 mg/mL) solutions in incremental doses of 3 mL to 5 mL with sufficient time between doses to detect toxic manifestations of unintentional intravascular or intrathecal injection. Administer injections slowly, with frequent aspirations before and during the injection to avoid intravascular injection. Perform syringe aspirations before and during each supplemental injection in continuous (intermittent) catheter techniques. In obstetrics, use ONLY the 0.5% (5 mg/mL) and 0.25% (2.5 mg/mL) concentrations of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection [see Warnings and Precautions (5.1) ]; incremental doses of 3 mL to 5 mL of the 0.5% (5 mg/mL) solution not exceeding 50 mg to 100 mg at any dosing interval are recommended. Repeat doses should be preceded by a test dose containing epinephrine if not clinically contraindicated. Use only the single-dose vials for caudal or epidural anesthesia; avoid use of the multiple-dose vials for these procedures, which contain a preservative [see Dosage and Administration (2.1 , 2.4) , Warnings and Precautions (5.4 , 5.9) ] .
Test Dose for Caudal and Lumbar Epidural Blocks
Three mL of Bupivacaine Hydrochloride and Epinephrine Injection without antimicrobial preservative (0.5% bupivacaine with 1:200,000 epinephrine) is recommended for use as a test dose prior to caudal and lumbar epidural blocks when clinical conditions permit. This test dose may serve as a warning of unintended intravascular or intrathecal injection. Closely monitor for early clinical signs of toxicity following each test dose [see Warnings and Precautions (5.9) ] . Allot adequate time for onset of spinal block to detect possible intrathecal injection. An intravascular or intrathecal injection is still possible even if results of the test dose are negative. The test dose itself may produce a systemic toxic reaction, high spinal, or cardiovascular effects from the epinephrine [see Warnings and Precautions (5.2 , 5.9) , Overdosage (10) ].
Use in Dentistry
Bupivacaine Hydrochloride and Epinephrine Injection 0.5% (5 mg/mL) is recommended for infiltration and block injection in the maxillary and mandibular area when a longer duration of local anesthesia is desired, such as for procedures generally associated with significant postoperative pain. The average dose of 1.8 mL (9 mg) per injection site will usually suffice; an occasional second dose of 1.8 mL (9 mg) may be used if necessary to produce adequate anesthesia after allowing 2 to 10 minutes for block onset [see Clinical Pharmacology (12.2) ]. Use the lowest effective dose and allow time between injections; it is recommended that the total dose for all injection sites, spread out over a single dental sitting, not exceed 90 mg for a healthy adult patient (ten 1.8 mL injections of 0.5% (5 mg/mL) Bupivacaine Hydrochloride and Epinephrine Injection). Inject slowly and with frequent aspirations.
Use in Ophthalmic Surgery
When Bupivacaine Hydrochloride Injection 0.75% (7.5 mg/mL) is used for retrobulbar block, complete corneal anesthesia usually precedes onset of clinically acceptable external ocular muscle akinesia. Therefore, presence of akinesia rather than anesthesia alone should determine readiness of the patient for surgery [see Warnings and Precautions (5.15) ] .
DOSAGE FORMS AND STRENGTHS
Bupivacaine Hydrochloride Injection, USP is a clear, colorless solution available as:
- 0.25% (25 mg/10 mL) (2.5 mg/mL) in single-dose teartop vials.
- 0.25% (75 mg/30 mL) (2.5 mg/mL) in single-dose teartop vials.
- 0.25% (125 mg/50 mL) (2.5 mg/mL) in multiple-dose fliptop vials.
- 0.5% (50 mg/10 mL) (5 mg/mL) in single-dose teartop vials.
- 0.5% (150 mg/30 mL) (5 mg/mL) in single-dose teartop vials.
- 0.5% (250 mg/50 mL) (5 mg/mL) in multiple-dose fliptop vials.
- 0.75% (75 mg/10 mL) (7.5 mg/mL) in single-dose teartop vials.
- 0.75% (225 mg/30 mL) (7.5 mg/mL) in single-dose teartop vials.
USE IN SPECIFIC POPULATIONS
- Pediatric Use : Administration of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection in pediatric patients younger than 12 years is not recommended. (8.4 )
- Geriatric Use : Patients 65 years and over, particularly those with hypertension, may be at increased risk for developing hypotension while undergoing anesthesia with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection. (8.5 )
- Moderate to Severe Hepatic Impairment : Consider increased monitoring for bupivacaine systemic toxicity. (8.6 )
Pregnancy
Risk Summary
Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection is contraindicated for obstetrical paracervical block anesthesia. Its use in this technique has resulted in fetal bradycardia and death [see Contraindications (4) , Warnings and Precautions (5.1) ].
There are no available data on use of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection in pregnant women to inform a drug-associated risk of adverse developmental outcomes.
In animal studies, embryo-fetal lethality was noted when bupivacaine was administered subcutaneously to pregnant rabbits during organogenesis at clinically relevant doses. Decreased pup survival was observed in a rat pre- and post-natal developmental study (dosing from implantation through weaning) at a dose level comparable to the daily maximum recommended human dose (MRHD) on a body surface area (BSA) basis. Based on animal data, advise pregnant women of the potential risks to a fetus (see Data ).
Local anesthetics rapidly cross the placenta, and when used for epidural, caudal, or pudendal block anesthesia, can cause varying degrees of maternal, fetal, and neonatal toxicity [see Clinical Pharmacology (12.3) ]. The incidence and degree of toxicity depend upon the procedure performed, the type, and amount of drug used, and the technique of drug administration. Adverse reactions in the parturient, fetus, and neonate involve alterations of the CNS, peripheral vascular tone, and cardiac function.
If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, inform the patient of the potential hazard to the fetus. The estimated background risk of major birth defects and miscarriage for the indicated populations are unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2–4% and 15–20%, respectively.
Clinical Considerations
Maternal Adverse Reactions
Maternal hypotension has resulted from regional anesthesia. Local anesthetics produce vasodilation by blocking sympathetic nerves. The supine position is dangerous in pregnant women at term because of aortocaval compression by the gravid uterus. Therefore, during treatment of systemic toxicity, maternal hypotension or fetal bradycardia following regional block, the parturient should be maintained in the left lateral decubitus position if possible, or manual displacement of the uterus off the great vessels be accomplished. Elevating the patient's legs will also help prevent decreases in blood pressure. The fetal heart rate also should be monitored continuously and electronic fetal monitoring is highly advisable.
Labor or Delivery
Epidural, caudal, or pudendal anesthesia may alter the forces of parturition through changes in uterine contractility or maternal expulsive efforts. Epidural anesthesia has been reported to prolong the second stage of labor by removing the parturient's reflex urge to bear down or by interfering with motor function. The use of obstetrical anesthesia may increase the need for forceps assistance.
The use of some local anesthetic drug products during labor and delivery may be followed by diminished muscle strength and tone for the first day or two of life. This has not been reported with bupivacaine.
It is extremely important to avoid aortocaval compression by the gravid uterus during administration of regional block to parturients. To do this, the patient must be maintained in the left lateral decubitus position or a blanket roll or sandbag may be placed beneath the right hip and gravid uterus displaced to the left.
Data
Animal Data
Bupivacaine hydrochloride produced developmental toxicity when administered subcutaneously to pregnant rats and rabbits at clinically relevant doses.
Bupivacaine hydrochloride was administered subcutaneously to rats at doses of 4.4, 13.3, & 40 mg/kg and to rabbits at doses of 1.3, 5.8, & 22.2 mg/kg during the period of organogenesis (implantation to closure of the hard palate). The high doses are comparable to the daily MRHD of 400 mg/day on a mg/m 2 BSA basis. No embryo-fetal effects were observed in rats at the high dose which caused increased maternal lethality. An increase in embryo-fetal deaths was observed in rabbits at the high dose in the absence of maternal toxicity with the fetal No Observed Adverse Effect Level representing approximately 0.3 times the MRHD on a BSA basis.
In a rat pre-and post-natal developmental study (dosing from implantation through weaning) conducted at subcutaneous doses of 4.4, 13.3, & 40 mg/kg, decreased pup survival was observed at the high dose. The high dose is comparable to the daily MRHD of 400 mg/day on a BSA basis.
Lactation
Risk Summary
Lactation studies have not been conducted with bupivacaine. Bupivacaine has been reported to be excreted in human milk suggesting that the nursing infant could be theoretically exposed to a dose of the drug. Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection should be administered to lactating women only if clearly indicated. Studies assessing the effects of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection in breastfed children have not been performed. Studies to assess the effect of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection on milk production or excretion have not been performed. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for bupivacaine and any potential adverse effects on the breastfed child from bupivacaine or from the underlying maternal condition.
Pediatric Use
Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection is approved for use in adults. Administration of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection in pediatric patients younger than 12 years is not recommended.
Continuous infusions of bupivacaine in pediatric patients have been reported to result in high systemic levels of bupivacaine and seizures; high plasma levels may also be associated with cardiovascular abnormalities.
Geriatric Use
Patients 65 years and over, particularly those with hypertension, may be at increased risk for developing hypotension while undergoing anesthesia with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection.
In clinical studies of bupivacaine, elderly patients reached the maximal spread of analgesia and maximal motor blockade more rapidly than younger adult patients.
Differences in various pharmacokinetic parameters have been observed between elderly and younger adult patients [see Clinical Pharmacology (12.3) ].
This product is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function. Elderly patients may require lower doses of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection.
Hepatic Impairment
Amide-type local anesthetics, such as bupivacaine, are metabolized by the liver. Patients with severe hepatic impairment, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations, and potentially local anesthetic systemic toxicity. Therefore, consider reduced dosing and increased monitoring for local anesthetic systemic toxicity in patients with moderate to severe hepatic impairment treated with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection, especially with repeat doses [see Warnings and Precautions (5.10) ] .
Renal Impairment
Bupivacaine is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with renal impairment. This should be considered when selecting the Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection dosage [see Use in Specific Populations (8.5) ] .
CONTRAINDICATIONS
Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection is contraindicated in:
- obstetrical paracervical block anesthesia. Its use in this technique has resulted in fetal bradycardia and death.
- intravenous regional anesthesia (Bier Block) [see Warnings and Precautions (5.7) ].
- patients with a known hypersensitivity to bupivacaine or to any local anesthetic agent of the amide-type or to other components of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection.
WARNINGS AND PRECAUTIONS
- Dose-Related Toxicity : Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after injection of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection. (5.2 )
- Methemoglobinemia : Cases of methemoglobinemia have been reported in association with local anesthetic use. See full prescribing information for more detail on managing these risks. (5.3 )
- Chondrolysis with Intra-Articular Infusion : Intra-articular infusions of local anesthetics including Bupivacaine Hydrochloride Injection following arthroscopic and other surgical procedures is an unapproved use, and there have been post-marketing reports of chondrolysis in patients receiving such infusions. (5.5 )
- Risk of Cardiac Arrest with Intravenous Regional Anesthesia Use (Bier Block) : There have been reports of cardiac arrest and death during the use of bupivacaine for intravenous regional anesthesia (Bier Block). (5.7 )
- Allergic-Type Reactions to Sulfites in Bupivacaine Hydrochloride and Epinephrine Injection : Bupivacaine Hydrochloride and Epinephrine Injection contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. (5.8 )
- Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection : Unintended intravascular or intrathecal injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progressing ultimately to respiratory arrest. Aspirate for blood or cerebrospinal fluid (where applicable) prior to each dose and consider using a test dose of Bupivacaine Hydrochloride and Epinephrine Injection. (5.9 )
Risk of Cardiac Arrest with Use of Bupivacaine Hydrochloride Injection in Obstetrical Anesthesia
There have been reports of cardiac arrest with difficult resuscitation or death during use of Bupivacaine Hydrochloride Injection for epidural anesthesia in obstetrical patients. In most cases, this has followed use of the 0.75% (7.5 mg/mL) concentration. Resuscitation has been difficult or impossible despite apparently adequate preparation and appropriate management. Cardiac arrest has occurred after convulsions resulting from systemic toxicity, presumably following unintentional intravascular injection. The 0.75% (7.5 mg/mL) concentration of Bupivacaine Hydrochloride Injection is not recommended for obstetrical anesthesia and should be reserved for surgical procedures where a high degree of muscle relaxation and prolonged effect are necessary.
Dose-Related Toxicity
The safety and effectiveness of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after injection of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection solutions.
Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
During major regional nerve blocks, such as those of the brachial plexus or lower extremity, the patient should have an indwelling intravenous catheter to assure adequate intravenous access. Use the lowest dosage of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection that results in effective anesthesia to avoid high plasma levels and serious adverse effects. Avoid rapid injection of a large volume of Bupivacaine. Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection solution and administer fractional (incremental) doses when feasible.
Injection of repeated doses of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection may cause significant increases in plasma levels with each repeated dose due to slow accumulation of the drug or its metabolites, or to slow metabolic degradation. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients and acutely ill patients should be given reduced doses commensurate with their age and physical status.
Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition [see Drug Interactions (7.5) ] . If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious CNS and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
Antimicrobial Preservatives in Multiple-Dose Vials
Avoid use of Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection solutions containing antimicrobial preservatives, i.e., those supplied in multiple-dose vials, for epidural or caudal anesthesia because safety has not been established with such use.
Chondrolysis with Intra-Articular Infusion
Intra-articular infusions of local anesthetics including Bupivacaine Hydrochloride Injection following arthroscopic and other surgical procedures is an unapproved use, and there have been post-marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in pediatric and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are associated with chondrolysis. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2 nd month after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
Risk of Adverse Reactions Due to Drug Interactions with Bupivacaine Hydrochloride and Epinephrine Injection
Risk of Severe, Persistent Hypertension Due to Drug Interactions Between Bupivacaine Hydrochloride and Epinephrine Injection and Monoamine Oxidase Inhibitors and Tricyclic Antidepressants
Administration of Bupivacaine Hydrochloride and Epinephrine Injection (containing a vasoconstrictor) in patients receiving monoamine oxidase inhibitors (MAOI), or tricyclic antidepressants may result in severe, prolonged hypertension. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's hemodynamic status is essential [see Drug Interactions (7.2) ].
Risk of Severe, Persistent Hypertension or Cerebrovascular Accidents Due to Drug Interactions Between Bupivacaine Hydrochloride and Epinephrine Injection and Ergot-Type Oxytocic Drugs
Concurrent administration of Bupivacaine Hydrochloride and Epinephrine Injection and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. Avoid use of Bupivacaine Hydrochloride and Epinephrine Injection concomitantly with ergot-type oxytocic drugs [see Drug Interactions (7.3) ] .
Risk of Hypertension and Bradycardia Due to Drug Interactions Between Bupivacaine Hydrochloride and Epinephrine Injection and Nonselective Beta-Adrenergic Antagonists
Administration of Bupivacaine Hydrochloride and Epinephrine Injection (containing a vasoconstrictor) in patients receiving nonselective beta-adrenergic antagonists may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's blood pressure and heart rate is essential [see Drug Interactions (7.4) ] .
Risk of Cardiac Arrest with Intravenous Regional Anesthesia Use (Bier Block)
There have been reports of cardiac arrest and death during the use of bupivacaine for intravenous regional anesthesia (Bier Block). Information on safe dosages and techniques of administration of Bupivacaine Hydrochloride Injection in this procedure is lacking. Therefore, Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection is contraindicated for use with this technique [see Contraindications (4) ] .
Allergic-Type Reactions to Sulfites in Bupivacaine Hydrochloride and Epinephrine Injection
Bupivacaine Hydrochloride and Epinephrine Injection contains sodium metabisulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people. Bupivacaine Hydrochloride Injection without epinephrine does not contain sodium metabisulfite.
Risk of Systemic Toxicities with Unintended Intravascular or Intrathecal Injection
Unintended intravascular or intrathecal injection of. Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection may be associated with systemic toxicities, including CNS or cardiorespiratory depression and coma, progressing ultimately to respiratory arrest. Unintentional intrathecal injection during the intended performance of caudal or lumbar epidural block or nerve blocks near the vertebral column has resulted in underventilation or apnea ("Total or High Spinal"). A high spinal has been characterized by paralysis of the legs, loss of consciousness, respiratory paralysis, and bradycardia [see Adverse Reactions (6) ] .
Aspirate for blood or cerebrospinal fluid (where applicable) before injecting Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection, both the initial dose and all subsequent doses, to avoid intravascular or intrathecal injection. However, a negative aspiration for blood or cerebrospinal fluid does not ensure against an intravascular or intrathecal injection.
Use of Test Dose with Epidural Anesthesia
To serve as a warning of unintended intravascular or intrathecal injection, 3 mL of Bupivacaine Hydrochloride and Epinephrine Injection without antimicrobial preservative (0.5% bupivacaine with 1:200,000 epinephrine) may be used as a test dose prior to administration of the full dose in caudal and lumbar epidural blocks [see Dosage and Administration (2.4) ] . Three mL of Bupivacaine Hydrochloride and Epinephrine Injection without antimicrobial preservative (0.5% bupivacaine with 1:200,000 epinephrine) contains 15 mg bupivacaine and 15 mcg epinephrine. An intravascular or intrathecal injection is still possible even if results of the test dose are negative.
Signs/symptoms of unintended intravascular or intrathecal injection of the test dose of Bupivacaine Hydrochloride and Epinephrine Injection and monitoring recommendations are described below.
- Unintended intravascular injection: Likely to produce a transient "epinephrine response" within 45 seconds, consisting of an increase in heart rate and/or systolic blood pressure, circumoral pallor, palpitations, and nervousness in the unsedated patient. The sedated patient may exhibit only a pulse rate increase of 20 or more beats per minute for 15 or more seconds. Therefore, following the test dose, the heart rate should be monitored for increases. Patients on beta-blockers may not manifest changes in heart rate, but blood pressure monitoring can detect a transient rise in systolic blood pressure.
- Unintended intrathecal injection: Evidenced within a few minutes by signs of spinal block (e.g., decreased sensation of the buttocks, paresis of the legs, or, in the sedated patient, absent knee jerk).
The test dose itself may produce a systemic toxic reaction, high spinal or epinephrine-induced cardiovascular effects [see Overdosage (10) ] .
Risk of Toxicity in Patients with Hepatic Impairment
Because amide local anesthetics such as bupivacaine are metabolized by the liver, consider reduced dosing and increased monitoring for bupivacaine systemic toxicity in patients with moderate to severe hepatic impairment who are treated Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection, especially with repeat doses [see Use in Specific Populations (8.6) ].
Risk of Use in Patients with Impaired Cardiovascular Function
Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection should be given in reduced doses in patients with impaired cardiovascular function (e.g., hypotension, heartblock) because they may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection. Monitor patients closely for blood pressure, heart rate, and ECG changes.
Risk of Ischemic Injury or Necrosis in Body Areas with Limited Blood Supply
Use Bupivacaine Hydrochloride and Epinephrine Injection in carefully restricted quantities in areas of the body supplied by end arteries or having otherwise compromised blood supply such as digits, nose, external ear, or penis. Patients with hypertensive vascular disease may exhibit exaggerated vasoconstrictor response. Ischemic injury or necrosis may result.
Risk of Cardiac Arrhythmias with Concomitant Use of Potent Inhalation Anesthetics
Serious dose-related cardiac arrhythmias may occur if preparations containing a vasoconstrictor such as epinephrine (e.g., Bupivacaine Hydrochloride and Epinephrine Injection) are used in patients during or following the administration of potent inhalation anesthetics [see Drug Interactions (7.6) ] . In deciding whether to concurrently use Bupivacaine Hydrochloride and Epinephrine Injection with potent inhalation anesthetics in the same patient, the combined action of both agents upon the myocardium, the concentration and volume of vasoconstrictor used, and the time since injection, when applicable, should be taken into account.
Risk of Adverse Reactions with Use in Head and Neck Area
Small doses of local anesthetics (e.g., Bupivacaine Hydrochloride Injection) injected into the head and neck area, including retrobulbar, dental, and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. The injection procedures require the utmost care. Confusion, convulsions, respiratory depression, and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. They may also be due to puncture of the dural sheath of the optic nerve during retrobulbar block with diffusion of any local anesthetic along the subdural space to the midbrain. Monitor circulation and respiration and constantly observe patients receiving Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection blocks. Resuscitative equipment and drugs, and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded [see Dosage and Administration (2.2) ].
Risk of Respiratory Arrest with Use in Ophthalmic Surgery
Clinicians who perform retrobulbar blocks should be aware that there have been reports of respiratory arrest following local anesthetic injection. Prior to retrobulbar block (e.g., with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection), as with all other regional procedures, resuscitative equipment and drugs, and personnel to manage respiratory arrest or depression, convulsions, and cardiac stimulation or depression should be immediately available [see Warnings and Precautions (5.14) ]. As with other anesthetic procedures, patients should be constantly monitored following ophthalmic blocks for signs of these adverse reactions, which may occur following relatively low total doses.
A concentration of 0.75% bupivacaine is indicated for retrobulbar block; however, this concentration is not indicated for any other peripheral nerve block, including the facial nerve, and not indicated for local infiltration, including the conjunctiva [see Indications and Usage (1) ].
Risk of Inadvertent Trauma to Tongue, Lips, and Buccal Mucosa in Dental Applications
Because of the long duration of anesthesia, when Bupivacaine Hydrochloride and Epinephrine Injection [0.5% (5 mg/mL) of bupivacaine] is used for dental injections, warn patients about the possibility of inadvertent trauma to tongue, lips, and buccal mucosa and advise them not to chew solid foods until sensation returns [see Patient Counseling Information (17) ] .
ADVERSE REACTIONS
The following clinically significant adverse reactions have been reported and described in the Warnings and Precautions section of the labeling:
- Cardiac Arrest in Obstetrical Anesthesia [see Warnings and Precautions (5.1) ]
- Dose-Related Toxicity [see Warnings and Precautions (5.2) ]
- Methemoglobinemia [see Warnings and Precautions (5.3) ]
- Chondrolysis with Intra-Articular Infusion [see Warnings and Precautions (5.5) ]
- Severe, Persistent Hypertension, Cerebrovascular Accidents, and Bradycardia Due to Drug Interactions [see Warnings and Precautions (5.6) ]
- Cardiac Arrest with Intravenous Regional Anesthesia Use [see Contraindications (4) , Warnings and Precautions (5.7) ]
- Allergic-Type Reactions [see Warnings and Precautions (5.8) ]
- Systemic Toxicities with Unintended Intravascular or Intrathecal Injection [see Warnings and Precautions (5.9) ]
- Respiratory Arrest Following Retrobulbar Block [see Warnings and Precautions (5.15) ]
The following adverse reactions from voluntary reports or clinical studies have been reported with bupivacaine or bupivacaine and epinephrine. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions to Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection are characteristic of those associated with other amide-type local anesthetics. A major cause of adverse reactions to this group of drugs is excessive plasma levels, which may be due to overdosage, unintentional intravascular injection, or slow metabolic degradation.
The most commonly encountered acute adverse reactions that demand immediate counter-measures were related to the CNS and the cardiovascular system. These adverse reactions were generally dose-related and due to high plasma levels which may have resulted from overdosage, rapid absorption from the injection site, diminished tolerance, or from unintentional intravascular injection of the local anesthetic solution. In addition to systemic dose-related toxicity, unintentional intrathecal injection of drug during the intended performance of caudal or lumbar epidural block or nerve blocks near the vertebral column (especially in the head and neck region) has resulted in underventilation or apnea ("Total or High Spinal"). Also, hypotension due to loss of sympathetic tone and respiratory paralysis or underventilation due to cephalad extension of the motor level of anesthesia have occurred. This has led to secondary cardiac arrest when untreated.
Nervous System Disorders
Adverse reactions were characterized by excitation and/or depression of the central nervous system and included restlessness, anxiety, dizziness, tinnitus, blurred vision, tremors, convulsions, drowsiness, unconsciousness, respiratory arrest, nausea, vomiting, chills, pupillary constriction.
In the practice of caudal or lumbar epidural block, unintentional penetration of the subarachnoid space by the catheter or needle has occurred. Subsequent adverse effects may have depended partially on the amount of drug administered intrathecally and the physiological and physical effects of a dural puncture. A high spinal has been characterized by paralysis of the legs, loss of consciousness, respiratory paralysis, and bradycardia.
Neurologic effects following epidural or caudal anesthesia have included spinal block of varying magnitude (including high or total spinal block); hypotension secondary to spinal block; urinary retention; fecal and urinary incontinence; loss of perineal sensation and sexual function; persistent anesthesia, paresthesia, weakness, paralysis of the lower extremities and loss of sphincter control, all of which had slow, incomplete, or no recovery; headache; backache; septic meningitis; meningismus; slowing of labor; increased incidence of forceps delivery; and cranial nerve palsies due to traction on nerves from loss of cerebrospinal fluid.
Neurologic effects following other procedures or routes of administration have included persistent anesthesia, paresthesia, weakness, paralysis, all with slow, incomplete, or no recovery.
Convulsions : Incidence varied with the procedure used and the total dose administered. In a survey of studies of epidural anesthesia, overt toxicity progressing to convulsions occurred in approximately 0.1% of local anesthetic administrations. The incidences of adverse neurologic reactions associated with the use of local anesthetics may be related to the total dose of local anesthetic administered and are also dependent upon the particular drug used, the route of administration, and the physical status of the patient.
Cardiac Disorders
High doses or unintentional intravascular injection have led to high plasma levels and related depression of the myocardium, decreased cardiac output, heartblock, hypotension, bradycardia, ventricular arrhythmias, including ventricular tachycardia and ventricular fibrillation, and cardiac arrest [see Warnings and Precautions (5.9) ].
Immune System Disorders
Allergic-type reactions have occurred as a result of sensitivity to bupivacaine or to other formulation ingredients, such as the antimicrobial preservative methylparaben contained in multiple-dose vials or sulfites in epinephrine-containing solutions. These reactions were characterized by signs such as urticaria, pruritus, erythema, angioneurotic edema (including laryngeal edema), tachycardia, sneezing, nausea, vomiting, dizziness, syncope, excessive sweating, elevated temperature, and severe hypotension. Cross sensitivity among members of the amide-type local anesthetic group has been reported [see Warnings and Precautions (5.8) ] .
DRUG INTERACTIONS
- Local Anesthetics : The toxic effects of local anesthetics are additive. Monitor for neurologic and cardiovascular effects when additional local anesthetics are administered. (7.1 )
- Monoamine Oxidase Inhibitors and Tricyclic Antidepressants : Administration of Bupivacaine Hydrochloride and Epinephrine Injection to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension. Concurrent use of these agents should generally be avoided. (5.6 , 7.2 )
- Ergot-Type Oxytocic Drugs : Concurrent administration of Bupivacaine Hydrochloride and Epinephrine Injection and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. (5.6 , 7.3 )
- Nonselective Beta-Adrenergic Antagonists : Administration of Bupivacaine Hydrochloride and Epinephrine Injection (containing a vasoconstrictor) in patients receiving nonselective beta-adrenergic antagonists may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. (5.6 , 7.4 )
- Drugs Associated with Methemoglobinemia : Patients are at increased risk of developing methemoglobinemia when concurrently exposed to nitrates, nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs. (7.5 )
- Potent Inhalation Anesthetics : Serious dose-related cardiac arrhythmias may occur if preparations containing a vasoconstrictor such as epinephrine are used in patients during or following the administration of potent inhalation anesthetics. (5.13 , 7.6 )
Local Anesthetics
The toxic effects of local anesthetics are additive. If coadministration of other local anesthetics with Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.1) , Warnings and Precautions (5.2) ] .
Monoamine Oxidase Inhibitors and Tricyclic Antidepressants
The administration Bupivacaine Hydrochloride and Epinephrine Injection to patients receiving monoamine oxidase inhibitors, or tricyclic antidepressants may produce severe, prolonged hypertension. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's hemodynamic status is essential [see Warnings and Precautions (5.6) ] .
Ergot-Type Oxytocic Drugs
Concurrent administration of Bupivacaine Hydrochloride and Epinephrine Injection and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents. Avoid use of Bupivacaine Hydrochloride and Epinephrine concomitantly with ergot-type oxytocic drugs [see Warnings and Precautions (5.6) ].
Nonselective Beta-Adrenergic Antagonists
Administration of Bupivacaine Hydrochloride and Epinephrine Injection (containing a vasoconstrictor) in patients receiving nonselective beta-adrenergic antagonists may cause severe hypertension and bradycardia. Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful monitoring of the patient's blood pressure and heart rate is essential [see Warnings and Precautions (5.6) ].
Drugs Associated with Methemoglobinemia
Patients who are administered Bupivacaine Hydrochloride Injection/Bupivacaine Hydrochloride and Epinephrine Injection are at increased risk of developing methemoglobinemia when concurrently exposed to following drugs, which could include other local anesthetics [see Warnings and Precautions (5.3) ] .
| Class | Examples |
|---|---|
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, isofamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
Potent Inhalation Anesthetics
Serious dose-related cardiac arrhythmias may occur if preparations containing a vasoconstrictor such as epinephrine (e.g., Bupivacaine Hydrochloride and Epinephrine Injection) are used in patients during or following the administration of potent inhalation anesthetics [see Warnings and Precautions (5.13) ] .
Phenothiazines and Butyrophenones
Phenothiazines and butyrophenones may reduce or reverse the pressor effect of epinephrine. Concurrent use of Bupivacaine Hydrochloride and Epinephrine Injection and these agents should generally be avoided. In situations when concurrent therapy is necessary, careful patient monitoring is essential.
DESCRIPTION
Bupivacaine Hydrochloride Injection contains bupivacaine hydrochloride, an amide local anesthetic, as the active pharmaceutical ingredient. The route of administration for Bupivacaine Hydrochloride Injection (without epinephrine) is by injection, for infiltration, perineural, caudal, epidural, or retrobulbar use. Multiple-dose vials contain methylparaben [see Warnings and Precautions (5.4) ] .
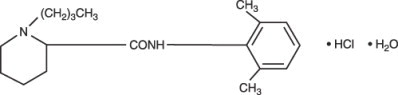
Bupivacaine hydrochloride is 2-piperidinecarboxamide, 1-butyl- N -(2,6-dimethylphenyl)-, monohydrochloride, monohydrate. It is a white crystalline powder that is freely soluble in 95 percent ethanol, soluble in water, and slightly soluble in chloroform or acetone. It has the following structural formula:
Bupivacaine Hydrochloride Injection, USP is a clear and colorless sterile isotonic solution. Each mL of single-dose vial contains 2.5 mg, 5 mg, or 7.5 mg of bupivacaine hydrochloride (equivalent to 2.22 mg, 4.44 mg, or 6.66 mg of bupivacaine, respectively), sodium chloride for isotonicity, sodium hydroxide or hydrochloric acid to adjust the pH between 4 and 6.5, in water for injection.
For the multiple-dose vials, each mL also contains 1 mg methylparaben as preservative.
CLINICAL PHARMACOLOGY
Mechanism of Action
Bupivacaine blocks the generation and the conduction of nerve impulses, presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse, and by reducing the rate of rise of the action potential. In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of affected nerve fibers. Clinically, the order of loss of nerve function is as follows: (1) pain, (2) temperature, (3) touch, (4) proprioception, and (5) skeletal muscle tone.
Epinephrine is a vasoconstrictor added to bupivacaine to slow absorption into the general circulation and thus prolong maintenance of an active tissue concentration.
Pharmacodynamics
Systemic absorption of bupivacaine produces effects on the cardiovascular system and CNS. At blood concentrations achieved with normal therapeutic doses, changes in cardiac conduction, excitability, refractoriness, contractility, and peripheral vascular resistance are minimal. However, toxic blood concentrations depress cardiac conduction and excitability, which may lead to atrioventricular block, ventricular arrhythmias, and cardiac arrest, sometimes resulting in fatalities. In addition, myocardial contractility is depressed and peripheral vasodilation occurs, leading to decreased cardiac output and arterial blood pressure. These cardiovascular changes are more likely to occur after unintended intravascular injection of bupivacaine [see Warnings and Precautions (5.9) ] .
Following systemic absorption, bupivacaine can produce CNS stimulation, CNS depression, or both. Apparent central stimulation is manifested as restlessness, tremors, and shivering, progressing to convulsions, followed by CNS depression and coma progressing ultimately to respiratory arrest. However, bupivacaine has a primary depressant effect on the medulla and on higher centers. The depressed stage may occur without a prior excited state.
The duration of local anesthesia after administration of Bupivacaine Hydrochloride Injection is longer than that observed after administration of other commonly used short-acting local anesthetics. There appears to be period of analgesia that persists after the resolution of the block and return of sensation.
The onset of action following dental injections is usually 2 to 10 minutes and may last up to 7 hours. The duration of anesthetic effect is prolonged by the addition of epinephrine 1:200,000.
Pharmacokinetics
Systemic plasma levels of bupivacaine following administration of Bupivacaine Hydrochloride Injection do not correlate with local efficacy.
Absorption
The rate of systemic absorption of bupivacaine is dependent upon the total dose and concentration of drug administered, the route of administration, the vascularity of the administration site, and the presence or absence of epinephrine in the anesthetic solution. A dilute concentration of epinephrine (1:200,000) usually reduces the rate of absorption and peak plasma concentration of bupivacaine, permitting the use of moderately larger total doses and sometimes prolonging the duration of action [see Dosage and Administration (2) ] .
After injection of Bupivacaine Hydrochloride Injection for caudal, epidural, or peripheral nerve block, peak levels of bupivacaine in the blood are reached in 30 to 45 minutes, followed by a decline to insignificant levels during the next three to six hours.
Distribution
Bupivacaine appears to cross the placenta by passive diffusion. The rate and degree of diffusion is governed by (1) the degree of plasma protein binding, (2) the degree of ionization, and (3) the degree of lipid solubility. Fetal/maternal ratios of bupivacaine appear to be inversely related to the degree of plasma protein binding, because only the free, unbound drug is available for placental transfer. Bupivacaine with a high protein binding capacity (95%) has a low fetal/maternal ratio (0.2 to 0.4). The extent of placental transfer is also determined by the degree of ionization and lipid solubility of the drug. Lipid soluble, nonionized drugs readily enter the fetal blood from the maternal circulation.
Depending upon the route of administration, bupivacaine is distributed to some extent to all body tissues, with high concentrations found in highly perfused organs such as the liver, lungs, heart, and brain.
Pharmacokinetic studies on the plasma profile of bupivacaine after direct intravenous injection (Bupivacaine Hydrochloride Injection is not approved for intravenous use) suggest a three-compartment open model. The first compartment is represented by the rapid intravascular distribution of the drug. The second compartment represents the equilibration of the drug throughout the highly perfused organs such as the brain, myocardium, lungs, kidneys, and liver. The third compartment represents an equilibration of the drug with poorly perfused tissues, such as muscle and fat.
Elimination
The half-life of bupivacaine in adults is 2.7 hours.
Metabolism
Amide-type local anesthetics such as bupivacaine are metabolized primarily in the liver via conjugation with glucuronic acid. Pipecoloxylidine is the major metabolite of bupivacaine. The elimination of drug from tissue distribution depends largely upon the availability of binding sites in the circulation to carry it to the liver where it is metabolized.
Excretion
The kidney is the main excretory organ for most local anesthetics and their metabolites. Urinary excretion is affected by urinary perfusion and factors affecting urinary pH. Only 6% of bupivacaine is excreted unchanged in the urine.
Specific Populations
Geriatric Patients
Elderly patients exhibited higher peak plasma concentrations than younger patients following administration of Bupivacaine Hydrochloride Injection. The total plasma clearance was decreased in these patients [see Use in Specific Populations (8.5) ] .
Patients with Hepatic Impairment
Various pharmacokinetic parameters of the local anesthetics can be significantly altered by the presence of hepatic disease. Patients with hepatic disease, especially those with severe hepatic disease, may be more susceptible to the potential toxicities of the amide-type local anesthetics [see Use in Specific Populations (8.6) ] .
Patients with Renal Impairment
Various pharmacokinetic parameters of the local anesthetics can be significantly altered by the presence of renal disease, factors affecting urinary pH, and renal blood flow [see Use in Specific Populations (8.5 , 8.7) ] .
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term studies in animals to evaluate the carcinogenic potential of bupivacaine hydrochloride have not been conducted.
Mutagenesis
The mutagenic potential of bupivacaine hydrochloride has not been determined.
Impairment of Fertility
The effect of bupivacaine on fertility has not been determined.
HOW SUPPLIED/STORAGE AND HANDLING
Store at 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F). [See USP Controlled Room Temperature.]
Bupivacaine Hydrochloride Injection, USP ─ Solutions of bupivacaine hydrochloride that do not contain epinephrine may be autoclaved. Autoclave at 15-pound pressure, 121 °C (250 °F) for 15 minutes. This product is clear and colorless. Do not use the solution if it is discolored or if it contains a precipitate.
| Unit of Sale | Concentration |
|---|---|
| NDC 0409-1159-01 Tray of 25 single-dose teartop vials | 0.25% 25 mg/10 mL (2.5 mg/mL) |
| NDC 0409-1159-02 Tray of 25 single-dose teartop vials | 0.25% 75 mg/30 mL (2.5 mg/mL) |
| NDC 0409-1160-01 Tray of 25 multiple-dose fliptop vials | 0.25% 125 mg/50 mL (2.5 mg/mL) |
| NDC 0409-1163-01 Tray of 25 multiple-dose fliptop vials | 0.5% 250 mg/50 mL (5 mg/mL) |
| NDC 0409-1162-01 Tray of 25 single-dose teartop vials | 0.5% 50 mg/10 mL (5 mg/mL) |
| NDC 0409-1162-02 Tray of 25 single-dose teartop vials | 0.5% 150 mg/30 mL (5 mg/mL) |
| NDC 0409-1165-01 Tray of 25 single-dose teartop vials | 0.75% 75 mg/10 mL (7.5 mg/mL) |
| NDC 0409-1165-02 Tray of 25 single-dose teartop vials | 0.75% 225 mg/30 mL (7.5 mg/mL) |
For single-dose vials: Discard unused portion.
Mechanism of Action
Bupivacaine blocks the generation and the conduction of nerve impulses, presumably by increasing the threshold for electrical excitation in the nerve, by slowing the propagation of the nerve impulse, and by reducing the rate of rise of the action potential. In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of affected nerve fibers. Clinically, the order of loss of nerve function is as follows: (1) pain, (2) temperature, (3) touch, (4) proprioception, and (5) skeletal muscle tone.
Epinephrine is a vasoconstrictor added to bupivacaine to slow absorption into the general circulation and thus prolong maintenance of an active tissue concentration.