Get your patient on Rectiv (Nitroglycerin)
Rectiv prior authorization resources
Most recent state uniform prior authorization forms
Dosage & administration
2 DOSAGE AND ADMINISTRATION
Apply 1 inch of ointment (375 mg of ointment equivalent to 1.5 mg of nitroglycerin) intra-anally every 12 hours for up to 3 weeks. A finger covering, such as plastic-wrap, disposable surgical glove or a finger cot, should be placed on the finger to apply the ointment. To obtain a 1.5 mg dose of nitroglycerin, the covered finger is laid alongside the 1 inch dosing line on the carton.
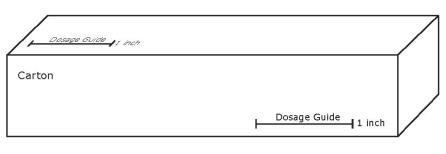
Refer to carton for accurate dosage guide.
The tube is gently squeezed until a line of ointment the length of the measuring line is expressed onto the covered finger. The ointment is gently inserted into the anal canal using the covered finger no further than to the first finger joint and the ointment is applied around the side of the anal canal. If this cannot be achieved due to pain, application of the ointment should be made directly to the outside of the anus. Treatment may be continued for up to three weeks.
RECTIV ointment is not for oral, ophthalmic, or intravaginal use. Hands should be washed after application of the ointment [see Patient Instruction ] .

Rectiv prescribing information
1 INDICATIONS AND USAGE
RECTIV ® (nitroglycerin) Ointment 0.4% is indicated for the treatment of moderate to severe pain associated with chronic anal fissure.
2 DOSAGE AND ADMINISTRATION
Apply 1 inch of ointment (375 mg of ointment equivalent to 1.5 mg of nitroglycerin) intra-anally every 12 hours for up to 3 weeks. A finger covering, such as plastic-wrap, disposable surgical glove or a finger cot, should be placed on the finger to apply the ointment. To obtain a 1.5 mg dose of nitroglycerin, the covered finger is laid alongside the 1 inch dosing line on the carton.

Refer to carton for accurate dosage guide.
The tube is gently squeezed until a line of ointment the length of the measuring line is expressed onto the covered finger. The ointment is gently inserted into the anal canal using the covered finger no further than to the first finger joint and the ointment is applied around the side of the anal canal. If this cannot be achieved due to pain, application of the ointment should be made directly to the outside of the anus. Treatment may be continued for up to three weeks.
RECTIV ointment is not for oral, ophthalmic, or intravaginal use. Hands should be washed after application of the ointment [see Patient Instruction ] .
3 DOSAGE FORMS AND STRENGTHS
Ointment, 0.4% w/w (4 mg /1 g) in 30 g tubes.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no data on the use of nitroglycerin ointment intra-anally during pregnancy to determine a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes. In animal reproduction studies, no malformations were observed in offspring of pregnant rats and rabbits administered nitroglycerin by topical or dietary route during the period of organogenesis (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Animal reproduction studies in rats and rabbits were conducted with topically applied nitroglycerin ointment at doses up to 80 mg/kg/day and 240 mg/kg/day, respectively. No toxic effects on dams or fetuses were seen at any dose tested.
An animal reproduction study was conducted in rats with nitroglycerin administered in the diet at levels up to 1% content (approximately 430 mg/kg/day) on days 6 to 15 of gestation. In offspring of the high-dose group, an increased but not statistically significant incidence of diaphragmatic hernias was noted together with decreased hyoid bone ossification. The latter finding probably reflects delayed development, thus indicating no clear evidence of a potential teratogenic effect of nitroglycerin.
8.000000000000000e+00 2 Lactation
Risk Summary
There are no data on the presence of nitroglycerin in either human or animal milk following topical administration, or the effects of nitroglycerin on milk production. A publication describing 40 lactating patients who used nitroglycerin ointment to treat chronic anal fissures for varied durations did not report signs of adverse effects in their breastfed infants, however, the dosing regimens were not described. The developmental and health benefits of breastfeeding should be considered along with the clinical need for RECTIV and any potential adverse effects on the breastfed infant from RECTIV or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of RECTIV in pediatric patients under 18 years of age have not been established.
8.5 Geriatric Use
Clinical studies of RECTIV did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Clinical data from the published literature indicate that the elderly demonstrate increased sensitivity to nitrates, which may be therapeutic but also manifest by more frequent or severe hypotension and related dizziness or fainting. Increased sensitivity may reflect the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
4 CONTRAINDICATIONS
- Use of PDE5 inhibitors (e.g. sildenafil, vardenafil and tadalafil) as these are shown to potentiate the hypotensive effects of organic nitrates. (4.1 ).
- Severe anemia (4.2 )
- Increased intracranial pressure (4.3 )
- Known hypersensitivity to nitroglycerin, other nitrates and nitrites, or any components of the ointment. (4.4 )
4.1 PDE5 inhibitor use
Administration of RECTIV is contraindicated in patients who are using a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5), such as sildenafil, vardenafil, and tadalafil, as these are shown to potentiate the hypotensive effects of organic nitrates [see DRUG INTERACTIONS (7.1 ) ] .
4.2 Severe anemia
RECTIV is contraindicated in patients with severe anemia.
4.3 Increased intracranial pressure
RECTIV is contraindicated in patients with increased intracranial pressure.
4.4 Hypersensitivity
RECTIV is contraindicated in patients who have shown hypersensitivity to it or to other nitrates or nitrites. Skin reactions consistent with hypersensitivity have been observed with organic nitrates.
5 WARNINGS AND PRECAUTIONS
- Cardiovascular Disorders: Venous and arterial dilatation as a consequence of nitroglycerin treatment can result in hypotension. Exercise caution when treating patients with any of the following conditions: blood volume depletion, existing hypotension, cardiomyopathies, congestive heart failure, acute myocardial infarction, or poor cardiac function for other reasons (5.1 ).
- Headache: Nitroglycerin produces dose-related headaches which may be severe (5.2 )
5.000000000000000e+00 1 Cardi ovascular disorders
Venous and arterial dilatation as a consequence of nitroglycerin treatment including RECTIV, can decrease venous blood returning to the heart and reduce arterial vascular resistance and systolic pressure. Exercise caution when treating patients with any of the following conditions: blood volume depletion, existing hypotension, cardiomyopathies, congestive heart failure, acute myocardial infarction, or poor cardiac function for other reasons. If patients with any of these conditions are treated with RECTIV, monitor cardiovascular status and clinical condition. The adverse reactions of RECTIV are likely to be more pronounced in the elderly.
5.000000000000000e+00 2 Headache
RECTIV produces dose-related headaches, which may be severe. Tolerance to headaches occurs.
6 ADVERSE REACTIONS
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reaction of RECTIV ointment applied to the anal canal is headache.
Headache may be recurrent following each dose. Headaches are typically of short duration and can be treated with an analgesic, e.g. acetaminophen, and are reversible upon discontinuation of treatment.
In Study REC-C-001, a double-blind, placebo-controlled trial in patients with a painful chronic anal fissure, the most frequent (≥ 2%) adverse reactions reported were as follows (Table 1):
| RECTIV N = 123 | Placebo N = 124 | |||
| System Organ Class Preferred term | Patients n (%) | Events n | Patients n (%) | Events n |
| Nervous system disorders | ||||
| Headache | 79 (64) | 938 | 51 (41) | 225 |
| Dizziness | 6 (5) | 26 | 0 | 0 |
Hypotension
Transient episodes of light-headedness, occasionally related to blood pressure changes, also may occur. Hypotension (including orthostatic hypotension) occurs infrequently, but in some patients may be severe enough to warrant discontinuation of therapy.
Allergic Reactions
Flushing, allergic reactions and application site reactions (including drug rash and exfoliative dermatitis) have been reported rarely.
Methemoglobinemia
In rare cases, therapeutic doses of organic nitrates have caused methemoglobinemia [ see OVERDOSAGE (10 ) ] .
7 DRUG INTERACTIONS
- PDE5 inhibitors: potentiation of hypotensive effects of organic nitrates; concomitant use is contraindicated. (4.1 , 7.1 )
- Antihypertensives: possible additive hypotensive effects. (7.2 )
- Aspirin: increased nitroglycerin levels. (7.3 )
- Tissue-type Plasminogen Activator (t-PA): decreased thrombolytic effect. (7.4 )
- Heparin: anticoagulant effect of heparin may be reduced. Monitor APTT. (7.5 )
- Ergotamine: increased bioavailability of ergotamine. (7.6 )
- Alcohol: Additive vasodilatory effects to nitroglycerin. Consumption of alcohol should be avoided. (7.7 )
7.1 PDE5 inhibitors
Phosphodiesterase type 5 (PDE5) inhibitors such as sildenafil, vardenafil, and tadalafil have been shown to potentiate the hypotensive effects of organic nitrates.
The time course of the interaction appears to be related to the half-life of the PDE5 inhibitor, however, the dose dependence of this interaction has not been studied. Use of RECTIV within a few days of PDE5 inhibitors is contraindicated.
7.2 Antihypertensives
Patients receiving antihypertensive drugs, beta-adrenergic blockers, and other nitrates should be observed for possible additive hypotensive effects when using RECTIV. Marked orthostatic hypotension has been reported when calcium channel blockers and organic nitrates were used concomitantly.
Beta-blockers blunt the reflex tachycardia produced by nitroglycerin without preventing its hypotensive effects. If beta-blockers are used with RECTIV in patients with angina pectoris, additional hypotensive effects may occur.
7.3 Aspirin
Coadministration of aspirin (at doses between 500 mg and 1000 mg) and nitroglycerin has been reported to result in increased nitroglycerin maximum concentrations by as much as 67% and AUC by 73% when administered as a single dose. The pharmacological effects of RECTIV may be enhanced by concomitant administration of aspirin.
7.4 Tissue-type Plasminogen Activator (t-PA)
Intravenous administration of nitroglycerin decreases the thrombolytic effect of tissue-type plasminogen activator (t-PA). Plasma levels of t-PA are reduced when coadministered with nitroglycerin. Therefore, caution should be observed in patients receiving RECTIV during t-PA therapy.
7.5 Heparin
Although an interaction has been reported between intravenous heparin and intravenous nitroglycerin (resulting in a decrease in the anticoagulant effect of heparin), the data are not consistent. If patients are to receive intravenous heparin and RECTIV concurrently, the anticoagulation status of the patient must be checked.
7.6 Ergotamine
Oral administration of nitroglycerin markedly decreases the first-pass metabolism of dihydroergotamine and consequently increases its oral bioavailability. Ergotamine is known to precipitate angina pectoris. Therefore the possibility of ergotism in patients receiving RECTIV should be considered.
7.7 Alcohol
The vasodilating effects of nitroglycerin have been shown to be additive to the effects observed with alcohol.
11 DESCRIPTION
Nitroglycerin is 1,2,3,-propanetriol trinitrate, an organic nitrate whose structural formula is as follows:
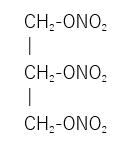
and whose molecular weight is 227.09. RECTIV (nitroglycerin) Ointment 0.4% contains 0.4% nitroglycerin w/w (4 mg nitroglycerin/1 g ointment), propylene glycol, lanolin, sorbitan sesquioleate, paraffin wax, and white petrolatum. RECTIV (nitroglycerin) Ointment 0.4% is available in tubes with a one-inch dosing line on the carton allowing the measurement of approximately 375 mg of nitroglycerin ointment 0.4% (1.5 mg nitroglycerin) for application.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Nitroglycerin forms free radical nitric oxide (NO), which activates guanylate cyclase, resulting in an increase of guanosine 3’,5’-monophosphate (cyclic GMP) in smooth muscle and other tissues. This leads to dephosphorylation of myosin light chains, which regulates the contractile state in smooth muscle and results in vasodilatation.
12.2 Pharmacodynamics
The principal pharmacological action of nitroglycerin is relaxation of vascular smooth muscle. Intra-anal application of nitroglycerin reduces sphincter tone and resting intra-anal pressure.
12.3 Pharmacokinetics
Absorption: In six healthy subjects, the average absolute bioavailability of nitroglycerin applied to the anal canal as a 0.2% w/w ointment was approximately 50% of the 0.75 mg nitroglycerin dose.
Distribution: The volume of distribution of nitroglycerin following intravenous administration is about 3 L/kg. At plasma concentrations between 50 and 500 ng/mL, the binding of nitroglycerin to plasma proteins is approximately 60%, while that of 1,2- and 1,3-dinitroglycerin is 60% and 30%, respectively.
Metabolism: Nitroglycerin is metabolized by a liver reductase enzyme to glycerol di- and mononitrate metabolites and ultimately to glycerol and organic nitrate. Known sites of extrahepatic metabolism include red blood cells and vascular walls. In addition to nitroglycerin, the two major metabolites, 1,2- and 1,3- dinitroglycerols are found in plasma. The contribution of metabolites to the relaxation of the internal anal sphincter is unknown. The dinitrates are further metabolized to nonvasoactive mononitrates and ultimately to glycerol and carbon dioxide.
Elimination: Metabolism is the primary route of drug elimination. Nitroglycerin plasma concentrations decrease rapidly with a mean elimination half-life of two to three minutes. Half-life values range from 1.5 to 7.5 minutes. Clearance (13.6 L/min) greatly exceeds hepatic blood flow.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Animal carcinogenicity studies with topically applied nitroglycerin have not been performed.
Rats receiving up to 434 mg/kg/day of dietary nitroglycerin for 2 years developed dose-related fibrotic and neoplastic changes in liver, including carcinomas, and interstitial cell tumors in testes. At the highest dose, the incidence of hepatocellular carcinomas was 52% compared to 0% in untreated controls. Incidence of testicular tumors were 52% vs. 8% in controls. Lifetime dietary administration of up to 1058 mg/kg/day of nitroglycerin was not tumorigenic in mice.
Mutagenesis
Nitroglycerin was mutagenic in the in vitro bacterial reverse mutation (Ames) assay with Salmonella typhimurium . A similar mutation in this S. typhimurium was also reported with other NO donors. There was no evidence of clastogenic potential in multiple assays including a rodent dominant lethal assay, an in vitro Chinese Hamster Ovary assay that was conducted in the absence of metabolic activation, and several in vivo chromosomal aberration assays conducted in rats and dogs.
Impairment of Fertility
In a three-generation reproduction study, rats received dietary nitroglycerin at doses up to approximately 434 mg/kg/day for 6 months prior to mating of the F 0 generation with treatment continuing through successive F 1 and F 2 generations. The high dose was associated with decreased feed intake and body weight gain in both sexes at all matings. No specific effect on the fertility of the F 0 generation was seen. Infertility noted in subsequent generations, however, was attributed to increased interstitial cell tissue and aspermatogenesis in the high-dose males.
14 CLINICAL STUDIES
RECTIV ointment was evaluated in a 3-week double-blind, randomized, multi-center, placebo-controlled study. Patients with a painful chronic anal fissure for at least 6 weeks and moderate or severe pain prior to treatment (≥ 50 mm on the 100mm visual analog scale, VAS) were randomized to receive 0.4% (1.5mg) nitroglycerin or placebo ointment applied to the anal canal every 12 hours. Pain as assessed by the change in VAS from baseline to Days 14-18 was lower in patients receiving 0.4% ointment compared to placebo. The mean change from baseline was 44mm for RECTIV and 37mm for placebo. The difference in the mean change in pain between RECTIV and placebo was -7.0mm (95% Confidence Interval: -13.6 to -0.4mm).
16 HOW SUPPLIED/STORAGE AND HANDLING
RECTIV (nitroglycerin) Ointment 0.4% is available in 30 g (NDC 58914-301-80) aluminum tubes with polyethylene screw caps.
Store at 20°-25°C (68°-77°F); excursions permitted between 15°-30°C (59°-86°F). [See USP Controlled Room Temperature].
Keep the tube tightly closed. Use within 8 weeks of first opening.
Patient Instructions for Use
When do I apply the ointment?
Apply the ointment every 12 hours exactly as your doctor has told you to.
How do I apply the ointment?
Cover your finger with plastic-wrap, a disposable surgical glove or a finger cot.
Lay the covered finger alongside the 1 inch dosing line marked on the side of the medicine box (see figure below) so that the tip of your finger is at one end of the dosing line. Starting at the tip of the finger, squeeze the ointment onto your finger for the same length marked on the box.

Refer to carton for accurate dosage guide.
Gently insert the finger with the ointment into the anal canal, up to the first finger joint. Carefully smear the ointment around the inner sides of the anal canal. If this cannot be achieved due to pain, application of the ointment should be made directly to the outside of the anus.
What do I do after I have applied the ointment?
Throw away the finger covering in the garbage, out of the reach of children and pets. Wash your hands.
What are the ingredients in RECTIV ?
Active ingredient: nitroglycerin
Inactive ingredients: propylene glycol, lanolin, sorbitan sesquioleate, paraffin wax and white petrolatum.
Manufactured by: PHARBIL Waltrop GmbH Im Wirrigen 25 45731 Waltrop Germany
Distributed by: AbbVie Inc. North Chicago, IL 60064
© 2024 AbbVie. All rights reserved.
RECTIV and its design are trademarks of Allergan Sales, LLC, an AbbVie company.
12.1 Mechanism of Action
Nitroglycerin forms free radical nitric oxide (NO), which activates guanylate cyclase, resulting in an increase of guanosine 3’,5’-monophosphate (cyclic GMP) in smooth muscle and other tissues. This leads to dephosphorylation of myosin light chains, which regulates the contractile state in smooth muscle and results in vasodilatation.