Get your patient on Ceftriaxone Sodium - Ceftriaxone Sodium injection, Powder, For Solution (Ceftriaxone Sodium)
Ceftriaxone Sodium - Ceftriaxone Sodium injection, Powder, For Solution prescribing information
INDICATIONS AND USAGE
Before instituting treatment with ceftriaxone for injection, USP, appropriate specimens should be obtained for isolation of the causative organism and for determination of its susceptibility to the drug. Therapy may be instituted prior to obtaining results of susceptibility testing.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ceftriaxone for injection, USP and other antibacterial drugs, ceftriaxone for injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Ceftriaxone for injection, USP is indicated for the treatment of the following infections when caused by susceptible organisms:
Lower Respiratory Tract Infections
Caused by Streptococcus pneumoniae, Staphylococcus aureus, Haemophilus influenzae, Haemophilus parainfluenzae, Klebsiella pneumoniae, Escherichia coli, Enterobacter aerogenes, Proteus mirabilis or Serratia marcescens.
Acute Bacterial Otitis Media
Caused by Streptococcus pneumoniae, Haemophilus influenzae (including beta-lactamase producing strains) or Moraxella catarrhalis (including beta-lactamase producing strains).
NOTE: In one study lower clinical cure rates were observed with a single dose of ceftriaxone compared to 10 days of oral therapy. In a second study comparable cure rates were observed between single dose ceftriaxone and the comparator. The potentially lower clinical cure rate of ceftriaxone should be balanced against the potential advantages of parenteral therapy (see CLINICAL STUDIES ).
Skin and Skin Structure Infections
Caused by Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes , Viridans group streptococci, Escherichia coli, Enterobacter cloacae, Klebsiella oxytoca, Klebsiella pneumoniae, Proteus mirabilis, Morganella morganii•, Pseudomonas aeruginosa, Serratia marcescens, Acinetobacter calcoaceticus, Bacteroides fragilis• or Peptostreptococcus species.
Urinary Tract Infections (complicated and uncomplicated)
Caused by Escherichia coli, Proteus mirabilis, Proteus vulgaris, Morganella morganii or Klebsiella pneumoniae.
Uncomplicated Gonorrhea (cervical/urethral and rectal)
Caused by Neisseria gonorrhoeae , including both penicillinase- and nonpenicillinase-producing strains, and pharyngeal gonorrhea caused by nonpenicillinase-producing strains of Neisseria gonorrhoeae .
Pelvic Inflammatory Disease
Caused by Neisseria gonorrhoeae . Ceftriaxone sodium, like other cephalosporins, has no activity against Chlamydia trachomatis . Therefore, when cephalosporins are used in the treatment of patients with pelvic inflammatory disease and Chlamydia trachomatis is one of the suspected pathogens, appropriate antichlamydial coverage should be added.
Bacterial Septicemia
Caused by Staphylococcus aureus, Streptococcus pneumoniae, Escherichia coli, Haemophilus influenzae or Klebsiella pneumoniae.
Bone and Joint Infections
Caused by Staphylococcus aureus, Streptococcus pneumoniae, Escherichia coli, Proteus mirabilis, Klebsiella pneumoniae or Enterobacter species.
Intra-abdominal Infections
Caused by Escherichia coli, Klebsiella pneumoniae, Bacteroides fragilis, Clostridium species (Note: most strains of Clostridium difficile are resistant) or Peptostreptococcus species.
Meningitis
Caused by Haemophilus influenzae, Neisseria meningitidis or Streptococcus pneumoniae . Ceftriaxone has also been used successfully in a limited number of cases of meningitis and shunt infection caused by Staphylococcus epidermidis• and Escherichia coli• .
•Efficacy for this organism in this organ system was studied in fewer than ten infections.
Surgical Prophylaxis
The preoperative administration of a single 1 g dose of ceftriaxone may reduce the incidence of postoperative infections in patients undergoing surgical procedures classified as contaminated or potentially contaminated (e.g., vaginal or abdominal hysterectomy or cholecystectomy for chronic calculous cholecystitis in high-risk patients, such as those over 70 years of age, with acute cholecystitis not requiring therapeutic antimicrobials, obstructive jaundice or common duct bile stones) and in surgical patients for whom infection at the operative site would present serious risk (e.g., during coronary artery bypass surgery). Although ceftriaxone has been shown to have been as effective as cefazolin in the prevention of infection following coronary artery bypass surgery, no placebo-controlled trials have been conducted to evaluate any cephalosporin antibiotic in the prevention of infection following coronary artery bypass surgery.
When administered prior to surgical procedures for which it is indicated, a single 1 g dose of ceftriaxone provides protection from most infections due to susceptible organisms throughout the course of the procedure.
DOSAGE AND ADMINISTRATION
Ceftriaxone in the ADD-Vantage ® Vial is intended for intravenous infusion only, after dilution with appropriate volume of ADD-Vantage ® diluent solution.
Do not use diluents containing calcium, such as Ringer’s solution or Hartmann’s solution, to reconstitute ceftriaxone vials or to further dilute a reconstituted vial for IV administration because a precipitate can form. Precipitation of ceftriaxone-calcium can also occur when ceftriaxone is mixed with calcium-containing solutions in the same IV administration line.
Ceftriaxone must not be administered simultaneously with calcium-containing IV solutions, including continuous calcium-containing infusions such as parenteral nutrition via a Y-site. However, in patients other than neonates, ceftriaxone and calcium-containing solutions may be administered sequentially of one another if the infusion lines are thoroughly flushed between infusions with a compatible fluid (see WARNINGS ).
There have been no reports of an interaction between ceftriaxone and oral calcium-containing products or interaction between intramuscular ceftriaxone and calcium-containing products (IV or oral).
Neonates
Hyperbilirubinemic neonates, especially prematures, should not be treated with ceftriaxone for injection. Ceftriaxone is contraindicated in premature neonates (see CONTRAINDICATIONS ).
Ceftriaxone is contraindicated in neonates (≤ 28 days) if they require (or are expected to require) treatment with calcium-containing IV solutions, including continuous calcium-containing infusions such as parenteral nutrition because of the risk of precipitation of ceftriaxone-calcium (see CONTRAINDICATIONS ).
Intravenous doses should be given over 60 minutes in neonates to reduce the risk of bilirubin encephalopathy.
Pediatric Patients
For the treatment of skin and skin structure infections, the recommended total daily dose is 50 to 75 mg/kg given once a day (or in equally divided doses twice a day). The total daily dose should not exceed 2 grams.
For the treatment of acute bacterial otitis media, a single intramuscular dose of 50 mg/kg (not to exceed 1 gram) is recommended (see INDICATIONS AND USAGE ). (The above is for informational purposes only. Ceftriaxone in ADD-Vantage ® vials is not intended for intramuscular use.)
For the treatment of serious miscellaneous infections other than meningitis, the recommended total daily dose is 50 to 75 mg/kg, given in divided doses every 12 hours. The total daily dose should not exceed 2 grams.
In the treatment of meningitis, it is recommended that the initial therapeutic dose be 100 mg/kg (not to exceed 4 grams). Thereafter, a total daily dose of 100 mg/kg/day (not to exceed 4 grams daily) is recommended. The daily dose may be administered once a day (or in equally divided doses every 12 hours). The usual duration of therapy is 7 to 14 days.
Adults
The usual adult daily dose is 1 to 2 grams given once a day (or in equally divided doses twice a day) depending on the type and severity of infection. The total daily dose should not exceed 4 grams.
If Chlamydia trachomatis is a suspected pathogen, appropriate antichlamydial coverage should be added, because ceftriaxone sodium has no activity against this organism.
For the treatment of uncomplicated gonococcal infections, a single intramuscular dose of 250 mg is recommended. (The above is for informational purposes only. Ceftriaxone in ADD-Vantage ® vials is not intended for intramuscular use.)
For preoperative use (surgical prophylaxis), a single dose of 1 gram administered intravenously ½ to 2 hours before surgery is recommended.
Generally, ceftriaxone therapy should be continued for at least 2 days after the signs and symptoms of infection have disappeared. The usual duration of therapy is 4 to 14 days; in complicated infections, longer therapy may be required.
When treating infections caused by Streptococcus pyogenes , therapy should be continued for at least 10 days.
No dosage adjustment is necessary for patients with impairment of renal or hepatic function (see PRECAUTIONS ).
The dosages recommended for adults require no modification in elderly patients, up to 2 g per day, provided there is no severe renal and hepatic impairment (see PRECAUTIONS ).
Directions for Use
Intravenous Administration
Ceftriaxone should be administered intravenously by infusion over a period of 30 minutes, except in neonates where administration over 60 minutes is recommended to reduce the risk of bilirubin encephalopathy.
ADD-Vantage ® Vials for IV Use Only contain ceftriaxone sodium equivalent to 1 g or 2 g ceftriaxone to be used with 50 mL or 100 mL of 5% Dextrose Injection, USP, or 0.9% Sodium Chloride Injection, USP, in the ADD-Vantage ® Flexible Diluent Container (see Instructions for Use of the ADD-Vantage® System at the end of this package insert).
Compatibility and Stability
Do not use diluents containing calcium, such as Ringer’s solution or Hartmann’s solution, to reconstitute ceftriaxone for injection vials or to further dilute a reconstituted vial for IV administration. Particulate formation can result.
Ceftriaxone has been shown to be compatible with Flagyl® IV (metronidazole hydrochloride). The concentration should not exceed 5 to 7.5 mg/mL metronidazole hydrochloride with ceftriaxone 10 mg/mL as an admixture. The admixture is stable for 24 hours at room temperature only in 0.9% sodium chloride injection or 5% dextrose in water (D5W). No compatibility studies have been conducted with the Flagyl® IV RTU® (metronidazole) formulation or using other diluents. Metronidazole at concentrations greater than 8 mg/mL will precipitate. Do not refrigerate the admixture as precipitation will occur.
Vancomycin, amsacrine, aminoglycosides, and fluconazole are incompatible with ceftriaxone in admixtures. When any of these drugs are to be administered concomitantly with ceftriaxone by intermittent intravenous infusion, it is recommended that they be given sequentially, with thorough flushing of the intravenous lines (with one of the compatible fluids) between the administrations.
Ceftriaxone for injection solutions should not be physically mixed with or piggybacked into solutions containing other antimicrobial drugs or into diluent solutions other than those listed above, due to possible incompatibility (see WARNINGS ).
Ceftriaxone sodium sterile powder should be stored at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature] and protected from light. After reconstitution, protection from normal light is not necessary. The color of solutions ranges from light yellow to amber, depending on the length of storage, concentration and diluent used.
After the indicated stability time periods, unused portions of solutions should be discarded.
NOTE: Parenteral drug products should be inspected visually for particulate matter before administration. ADD-Vantage ® Vials – Ceftriaxone for Injection Units: See Instructions for Use of the ADD-Vantage® System at the end of this package insert.
CONTRAINDICATIONS
Hypersensitivity
Ceftriaxone for injection is contraindicated in patients with known hypersensitivity to ceftriaxone, any of its excipients or to any other cephalosporin. Patients with previous hypersensitivity reactions to penicillin and other beta lactam antibacterial agents may be at greater risk of hypersensitivity to ceftriaxone (see WARNINGS – Hypersensitivity Reactions ).
Neonates
Premature neonates
Ceftriaxone for injection is contraindicated in premature neonates up to a postmenstrual age of 41 weeks (gestational age + chronological age).
Hyperbilirubinemic neonates
Hyperbilirubinemic neonates should not be treated with ceftriaxone for injection. Ceftriaxone can displace bilirubin from its binding to serum albumin, leading to a risk of bilirubin encephalopathy in these patients.
Neonates Requiring Calcium Containing IV Solutions
Ceftriaxone for injection is contraindicated in neonates (≤ 28 days) if they require (or are expected to require) treatment with calcium-containing IV solutions, including continuous calcium-containing infusions such as parenteral nutrition because of the risk of precipitation of ceftriaxone-calcium (see CLINICAL PHARMACOLOGY , WARNINGS and DOSAGE AND ADMINISTRATION ).
Cases of fatal outcomes in which a crystalline material was observed in the lungs and kidneys at autopsy have been reported in neonates receiving ceftriaxone for injection and calcium-containing fluids.
In some of these cases, the same intravenous infusion line was used for both ceftriaxone for injection and calcium-containing fluids and in some a precipitate was observed in the intravenous infusion line. There have been no similar reports in patients other than neonates.
Lidocaine
Intravenous administration of ceftriaxone solutions containing lidocaine is contraindicated. When lidocaine solution is used as a solvent with ceftriaxone for intramuscular injection, exclude all contraindications to lidocaine. Refer to the prescribing information of lidocaine.
ADVERSE REACTIONS
Ceftriaxone is generally well tolerated. In clinical trials, the following adverse reactions, which were considered to be related to ceftriaxone therapy or of uncertain etiology, were observed:
Local Reactions
Pain, induration and tenderness was 1% overall. Phlebitis was reported in <1% after IV administration. The incidence of warmth, tightness or induration was 17% (3/17) after IM administration of 350 mg/mL and 5% (1/20) after IM administration of 250 mg/mL.
General Disorders and Administration Site Conditions
Injection site pain (0.6%)
Hypersensitivity
Rash (1.7%). Less frequently reported (<1%) were pruritus, fever or chills.
Infections and Infestations
Genital fungal infection (0.1%)
Hematologic
Eosinophilia (6%), thrombocytosis (5.1%) and leukopenia (2.1%). Less frequently reported (<1%) were anemia, hemolytic anemia, neutropenia, lymphopenia, thrombocytopenia and prolongation of the prothrombin time.
Blood and Lymphatic Disorders
Granulocytopenia (0.9%), coagulopathy (0.4%)
Gastrointestinal
Diarrhea/loose stools (2.7%). Less frequently reported (<1%) were nausea or vomiting, and dysgeusia. The onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment (see WARNINGS ).
Hepatic
Elevations of aspartate aminotransferase (AST) (3.1%) or alanine aminotransferase (ALT) (3.3%). Less frequently reported (<1%) were elevations of alkaline phosphatase and bilirubin.
Renal
Elevations of the BUN (1.2%). Less frequently reported (<1%) were elevations of creatinine and the presence of casts in the urine.
Central Nervous System
Headache or dizziness were reported occasionally (<1%).
Genitourinary
Moniliasis or vaginitis were reported occasionally (<1%).
Miscellaneous
Diaphoresis and flushing were reported occasionally (<1%).
Investigations
Blood creatinine increased (0.6%).
Other rarely observed adverse reactions (<0.1%) include abdominal pain, agranulocytosis, allergic pneumonitis, anaphylaxis, basophilia, biliary lithiasis, bronchospasm, colitis, dyspepsia, epistaxis, flatulence, gallbladder sludge, glycosuria, hematuria, jaundice, leukocytosis, lymphocytosis, monocytosis, nephrolithiasis, palpitations, a decrease in the prothrombin time, renal precipitations, seizures, and serum sickness.
Post-Marketing Experience
In addition to the adverse reactions reported during clinical trials, the following adverse experiences have been reported during clinical practice in patients treated with ceftriaxone. Data are generally insufficient to allow an estimate of incidence or to establish causation.
A small number of cases of fatal outcomes in which a crystalline material was observed in the lungs and kidneys at autopsy have been reported in neonates receiving ceftriaxone and calcium-containing fluids. In some of these cases, the same intravenous infusion line was used for both ceftriaxone and calcium-containing fluids and in some a precipitate was observed in the intravenous infusion line. At least one fatality has been reported in a neonate in whom ceftriaxone and calcium-containing fluids were administered at different time points via different intravenous lines; no crystalline material was observed at autopsy in this neonate. There have been no similar reports in patients other than neonates.
Gastrointestinal
Pancreatitis, stomatitis and glossitis.
Genitourinary
Oliguria, ureteric obstruction, post-renal acute renal failure.
Dermatologic
Exanthema, allergic dermatitis, urticaria, edema; acute generalized exanthematous pustulosis (AGEP) and isolated cases of severe cutaneous adverse reactions (erythema multiforme, Stevens-Johnson syndrome or Lyell’s syndrome/toxic epidermal necrolysis) have been reported.
Hematological Changes
Isolated cases of agranulocytosis (< 500/mm 3 ) have been reported, most of them after 10 days of treatment and following total doses of 20 g or more.
Neurologic
Encephalopathy, seizures, myoclonus, and non-convulsive status epilepticus (see WARNINGS and PRECAUTIONS ).
Other, Adverse Reactions
Symptomatic precipitation of ceftriaxone calcium salt in the gallbladder, kernicterus, oliguria, and anaphylactic or anaphylactoid reactions.
Cephalosporin Class Adverse Reactions
In addition to the adverse reactions listed above which have been observed in patients treated with ceftriaxone, the following adverse reactions and altered laboratory test results have been reported for cephalosporin class antibiotics.
Adverse Reactions
Allergic reactions, drug fever, serum sickness-like reaction, renal dysfunction, toxic nephropathy, reversible hyperactivity, hypertonia, hepatic dysfunction including cholestasis, aplastic anemia, hemorrhage, and superinfection.
Altered Laboratory Tests
Positive direct Coombs’ test, false-positive test for urinary glucose, and elevated LDH (see PRECAUTIONS ).
Several cephalosporins have been implicated in triggering seizures, particularly in patients with renal impairment when the dosage was not reduced (see DOSAGE AND ADMINISTRATION ). If seizures associated with drug therapy occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
DESCRIPTION
Ceftriaxone for injection, USP is a sterile, semisynthetic, broad-spectrum cephalosporin antibiotic for intravenous or intramuscular administration. Ceftriaxone sodium is (6 R ,7 R )-7-[2-(2-Amino-4-thiazolyl)glyoxylamido]-8-oxo-3-[[(1,2,5,6-tetrahydro-2-methyl-5,6-dioxo- as -triazin-3-yl)thio]methyl]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7 2 -( Z )-(O-methyloxime), disodium salt, sesquaterhydrate.
The chemical formula of ceftriaxone sodium is C 18 H 16 N 8 Na 2 O 7 S 3 •3.5H 2 O. It has a calculated molecular weight of 661.60 and the following structural formula:
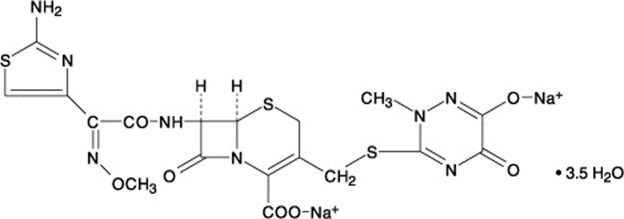
Ceftriaxone sodium is a white to yellowish crystalline powder which is readily soluble in water, sparingly soluble in methanol and very slightly soluble in ethanol. The pH of a 1% aqueous solution is approximately 6.7. The color of ceftriaxone sodium solutions ranges from light yellow to amber, depending on the length of storage, concentration and diluent used.
Ceftriaxone sodium contains approximately 83 mg (3.6 mEq) of sodium per gram of ceftriaxone activity.
CLINICAL PHARMACOLOGY
Average plasma concentrations of ceftriaxone following a single 30-minute intravenous (IV) infusion of a 0.5, 1 or 2 g dose and intramuscular (IM) administration of a single 0.5 (250 mg/mL or 350 mg/mL concentrations) or 1 g dose in healthy subjects are presented in Table 1 .
Table 1. Ceftriaxone Plasma Concentrations after Single Dose Administration
Dose/Route | Average Plasma Concentrations (mcg/mL) | ||||||||
0.5 hour | 1 hour | 2 hour | 4 hour | 6 hour | 8 hour | 12 hour | 16 hour | 24 hour | |
0.5 g IV• | 82 | 59 | 48 | 37 | 29 | 23 | 15 | 10 | 5 |
0.5 g IM 250 mg/mL | 22 | 33 | 38 | 35 | 30 | 26 | 16 | ND | 5 |
0.5 g IM 350 mg/mL | 20 | 32 | 38 | 34 | 31 | 24 | 16 | ND | 5 |
1 g IV• | 151 | 111 | 88 | 67 | 53 | 43 | 28 | 18 | 9 |
1 g IM | 40 | 68 | 76 | 68 | 56 | 44 | 29 | ND | ND |
2 g IV• | 257 | 192 | 154 | 117 | 89 | 74 | 46 | 31 | 15 |
ND = Not determined. | |||||||||
• IV doses were infused at a constant rate over 30 minutes.
Ceftriaxone was completely absorbed following IM administration with mean maximum plasma concentrations occurring between 2 and 3 hours post-dose. Multiple IV or IM doses ranging from 0.5 to 2 g at 12 to 24 hour intervals resulted in 15% to 36% accumulation of ceftriaxone above single dose values.
Ceftriaxone concentrations in urine are shown in Table 2 .
Table 2. Urinary Concentrations of Ceftriaxone after Single Dose Administration
Dose/Route | Average Urinary Concentrations (mcg/mL) | |||||
0 to 2 hour | 2 to 4 hour | 4 to 8 hour | 8 to 12 hour | 12 to 24 hour | 24 to 48 hour | |
0.5 g IV | 526 | 366 | 142 | 87 | 70 | 15 |
0.5 g IM | 115 | 425 | 308 | 127 | 96 | 28 |
1 g IV | 995 | 855 | 293 | 147 | 132 | 32 |
1 g IM | 504 | 628 | 418 | 237 | ND | ND |
2 g IV | 2692 | 1976 | 757 | 274 | 198 | 40 |
ND = Not determined. | ||||||
Thirty-three percent to 67% of a ceftriaxone dose was excreted in the urine as unchanged drug and the remainder was secreted in the bile and ultimately found in the feces as microbiologically inactive compounds. After a 1 g IV dose, average concentrations of ceftriaxone, determined from 1 to 3 hours after dosing, were 581 mcg/mL in the gallbladder bile, 788 mcg/mL in the common duct bile, 898 mcg/mL in the cystic duct bile, 78.2 mcg/g in the gallbladder wall and 62.1 mcg/mL in the concurrent plasma.
Over a 0.15 to 3 g dose range in healthy adult subjects, the values of elimination half-life ranged from 5.8 to 8.7 hours; apparent volume of distribution from 5.78 to 13.5 L; plasma clearance from 0.58 to 1.45 L/hour; and renal clearance from 0.32 to 0.73 L/hour. Ceftriaxone is reversibly bound to human plasma proteins, and the binding decreased from a value of 95% bound at plasma concentrations of <25 mcg/mL to a value of 85% bound at 300 mcg/mL. Ceftriaxone crosses the blood placenta barrier.
The average values of maximum plasma concentration, elimination half-life, plasma clearance and volume of distribution after a 50 mg/kg IV dose and after a 75 mg/kg IV dose in pediatric patients suffering from bacterial meningitis are shown in Table 3 . Ceftriaxone penetrated the inflamed meninges of infants and pediatric patients; CSF concentrations after a 50 mg/kg IV dose and after a 75 mg/kg IV dose are also shown in Table 3 .
Table 3. Average Pharmacokinetic Parameters of Ceftriaxone in Pediatric Patients with Meningitis
50 mg/kg IV | 75 mg/kg IV | |
Maximum Plasma Concentrations (mcg/mL) | 216 | 275 |
Elimination Half-life (hour) | 4.6 | 4.3 |
Plasma Clearance (mL/hour/kg) | 49 | 60 |
Volume of Distribution (mL/kg) | 338 | 373 |
CSF Concentration – inflamed meninges (mcg/mL) | 5.6 | 6.4 |
Range (mcg/mL) | 1.3 to 18.5 | 1.3 to 44 |
Time after dose (hour) | 3.7 (± 1.6) | 3.3 (± 1.4) |
Compared to that in healthy adult subjects, the pharmacokinetics of ceftriaxone were only minimally altered in elderly subjects and in patients with renal impairment or hepatic dysfunction ( Table 4 ); therefore, dosage adjustments are not necessary for these patients with ceftriaxone dosages up to 2 g per day. Ceftriaxone was not removed to any significant extent from the plasma by hemodialysis; in six of 26 dialysis patients, the elimination rate of ceftriaxone was markedly reduced.
Table 4. Average Pharmacokinetic Parameters of Ceftriaxone in Humans
Subject Group | Elimination Half-Life (hour) | Plasma Clearance (L/hour) | Volume of Distribution (L) |
Healthy Subjects | 5.8 to 8.7 | 0.58 to 1.45 | 5.8 to 13.5 |
Elderly Subjects (mean age, 70.5 year) | 8.9 | 0.83 | 10.7 |
Patients With Renal Impairment | |||
Hemodialysis Patients (0 to 5 mL/min) • | 14.7 | 0.65 | 13.7 |
Severe (5 to 15 mL/min) | 15.7 | 0.56 | 12.5 |
Moderate (16 to 30 mL/min) | 11.4 | 0.72 | 11.8 |
Mild (31 to 60 mL/min) | 12.4 | 0.70 | 13.3 |
Patients With Liver Disease | 8.8 | 1.1 | 13.6 |
• Creatinine clearance.
The elimination of ceftriaxone is not altered when ceftriaxone is co-administered with probenecid.
Pharmacokinetics in the Middle Ear Fluid
In one study, total ceftriaxone concentrations (bound and unbound) were measured in middle ear fluid obtained during the insertion of tympanostomy tubes in 42 pediatric patients with otitis media. Sampling times were from 1 to 50 hours after a single intramuscular injection of 50 mg/kg of ceftriaxone. Mean (±SD) ceftriaxone levels in the middle ear reached a peak of 35 (±12) mcg/mL at 24 hours, and remained at 19 (±7) mcg/mL at 48 hours. Based on middle ear fluid ceftriaxone concentrations in the 23 to 25 hour and the 46 to 50 hour sampling time intervals, a half-life of 25 hours was calculated. Ceftriaxone is highly bound to plasma proteins. The extent of binding to proteins in the middle ear fluid is unknown.
Interaction with Calcium
Two in vitro studies, one using adult plasma and the other neonatal plasma from umbilical cord blood have been carried out to assess interaction of ceftriaxone and calcium. Ceftriaxone concentrations up to 1 mM (in excess of concentrations achieved in vivo following administration of 2 grams ceftriaxone infused over 30 minutes) were used in combination with calcium concentrations up to 12 mM (48 mg/dL). Recovery of ceftriaxone from plasma was reduced with calcium concentrations of 6 mM (24 mg/dL) or higher in adult plasma or 4 mM (16 mg/dL) or higher in neonatal plasma. This may be reflective of ceftriaxone-calcium precipitation.
Microbiology
Mechanism of Action
Ceftriaxone is a bactericidal agent that acts by inhibition of bacterial cell wall synthesis. Ceftriaxone has activity in the presence of some beta-lactamases, both penicillinases and cephalosporinases, of Gram-negative and Gram-positive bacteria.
Mechanism of Resistance
Resistance to ceftriaxone is primarily through hydrolysis by beta-lactamase, alteration of penicillin-binding proteins (PBPs), and decreased permeability.
Interaction with Other Antimicrobials
In an in vitro study antagonistic effects have been observed with the combination of chloramphenicol and ceftriaxone. Ceftriaxone has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE (1) section:
- Gram-negative Bacteria
Acinetobacter calcoaceticus
Enterobacter aerogenes
Enterobacter cloacae
Escherichia coli
Haemophilus influenzae
Haemophilus parainfluenzae
Klebsiella oxytoca
Klebsiella pneumoniae
Moraxella catarrhalis
Morganella morganii
Neisseria gonorrhoeae
Neisseria meningitidis
Proteus mirabilis
Proteus vulgaris
Pseudomonas aeruginosa
Serratia marcescens
- Gram-positive Bacteria
Staphylococcus aureus
Staphylococcus epidermidis
Streptococcus pneumoniae
Streptococcus pyogenes
Viridans group streptococci
- Anaerobic Bacteria
Bacteroides fragilis
Clostridium species
Peptostreptococcus species
The following in vitro data are available, but their clinical significance is unknown. At least 90 percent of the following microorganisms exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for ceftriaxone. However, the efficacy of ceftriaxone in treating clinical infections due to these microorganisms has not been established in adequate and well-controlled clinical trials.
- Gram-negative Bacteria
Citrobacter diversus
Citrobacter freundii
Providencia species (including Providencia rettgeri)
Salmonella species (including Salmonella typhi)
Shigella species
- Gram-positive Bacteria
Streptococcus agalactiae
- Anaerobic Bacteria
Porphyromonas (Bacteroides) melaninogenicus
Prevotella (Bacteroides) bivius
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
CLINICAL STUDIES
Clinical Trials in Pediatric Patients with Acute Bacterial Otitis Media
(The following is for informational purposes only. Ceftriaxone in ADD-Vantage ® vials is not intended for intramuscular use.) In two adequate and well-controlled US clinical trials a single IM dose of ceftriaxone was compared with a 10 day course of oral antibiotic in pediatric patients between the ages of 3 months and 6 years. The clinical cure rates and statistical outcome appear in the table below:
| Clinical Efficacy in Evaluable Population | ||||
|---|---|---|---|---|
| Study Day | Ceftriaxone Single Dose | Comparator- 10 Days of Oral Therapy | 95% Confidence Interval | Statistical Outcome |
Study 1 – US | amoxicillin/ clavulanate | Ceftriaxone is lower than control at study day 14 and 28. | ||
14 | 74% (220/296) | 82% (247/302) | (-14.4%, -0.5%) | |
28 | 58% (167/288) | 67% (200/297) | (-17.5%, -1.2%) | |
Study 2 – US | TMP-SMZ | Ceftriaxone is equivalent to control at study day 14 and 28. | ||
14 | 54% (113/210) | 60% (124/206) | (-16.4%, 3.6%) | |
28 | 35% (73/206) | 45% (93/205) | (-19.9%, 0.0%) | |
An open-label bacteriologic study of ceftriaxone without a comparator enrolled 108 pediatric patients, 79 of whom had positive baseline cultures for one or more of the common pathogens. The results of this study are tabulated as follows:
Week 2 and 4 Bacteriologic Eradication Rates in the Per Protocol Analysis in the Roche Bacteriologic Study by pathogen:
| Organism | Study Day 13 to 15 | Study Day 30+2 | ||
|---|---|---|---|---|
| No. Analyzed | No. Erad. (%) | No. Analyzed | No. Erad. (%) | |
Streptococcus pneumoniae | 38 | 32 (84) | 35 | 25 (71) |
Haemophilus influenzae | 33 | 28 (85) | 31 | 22 (71) |
Moraxella catarrhalis | 15 | 12 (80) | 15 | 9 (60) |
HOW SUPPLIED
Ceftriaxone for injection, USP is supplied as a sterile, white to yellowish crystalline powder in ADD-Vantage ® Vials as follows:
ADD-Vantage ® Vials containing 1 g equivalent to ceftriaxone. Box of 10 (NDC 0409-7333-49).
ADD-Vantage ® Vials containing 2 g equivalent to ceftriaxone. Box of 10 (NDC 0409-7336-49).
Storage Prior to Reconstitution
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from light.