Get your patient on Clemastine Fumarate - Clemastine Fumarate tablet (Clemastine Fumarate)
Clemastine Fumarate - Clemastine Fumarate tablet prescribing information
INDICATIONS AND USAGE
Clemastine Fumarate Tablets USP, 2.68 mg are indicated for the relief of symptoms associated with allergic rhinitis such as sneezing, rhinorrhea, pruritus, and lacrimation. Clemastine Fumarate Tablets USP, 2.68 mg are also indicated for the relief of mild, uncomplicated allergic skin manifestations of urticaria and angioedema. It should be noted that clemastine fumarate is indicated for the dermatologic indications at the 2.68 mg dosage level only.
DOSAGE AND ADMINISTRATION
DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND RESPONSE OF THE PATIENT.
Clemastine Fumarate Tablets 2.68 mg
The maximum recommended dosage is one tablet three times daily. Many patients respond favorably to a single dose which may be repeated as required, but not to exceed three tablets daily.
CONTRAINDICATIONS
Usage in Nursing Mothers
Because of the higher risk of antihistamines for infants generally and for newborns and prematures in particular, antihistamine therapy is contraindicated in nursing mothers.
Usage in Lower Respiratory Disease
Antihistamines should not be used to treat lower respiratory tract symptoms including asthma. Antihistamines are also contraindicated in the following conditions:
Hypersensitivity to clemastine fumarate or other antihistamines of similar chemical structure.
Monoamine oxidase inhibitor therapy (see Drug Interactions section).
ADVERSE REACTIONS
Transient drowsiness, the most common adverse reaction associated with clemastine fumarate, occurs relatively frequently and may require discontinuation of therapy in some instances.
Antihistaminic Compounds
It should be noted that the following reactions have occurred with one or more antihistamines and, therefore, should be kept in mind when prescribing drugs belonging to this class, including clemastine. The most frequent adverse reactions are italicized.
- General: Urticaria, drug rash, anaphylactic shock, photosensitivity, excessive perspiration, chills, dryness of the mouth, nose, and throat.
- Cardiovascular System: Hypotension, headache, palpitations, tachycardia, extrasystoles.
- Hematologic System: Hemolytic anemia, thrombocytopenia, agranulocytosis.
- Nervous System: Sedation, sleepiness, dizziness, disturbed coordination , fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paresthesias, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, hysteria, neuritis, convulsions.
- GI System: Epigastric distress, anorexia, nausea, vomiting, diarrhea, constipation.
- GU System: Urinary frequency, difficult urination, urinary retention, early menses.
- Respiratory System: Thickening of bronchial secretions, tightness of chest and wheezing, nasal stuffiness.
Drug Interactions
MAO inhibitors prolong and intensify the anticholinergic (drying) effects of antihistamines.
DESCRIPTION
Clemastine belongs to the benzhydryl ether group of antihistaminic compounds. The chemical name is (+)-(2 R )-2-[2-[[( R )- p -Chloro-α-methyl-α-phenylbenzyl]-oxy]ethyl]-1-methylpyrrolidine fumarate (1:1).
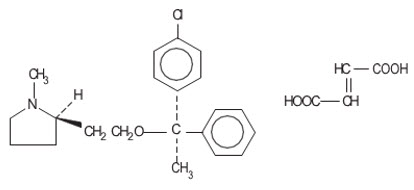 | |
| C 21 H 26 C1NO∙C 4 H 4 O 4 | M.W. 459.97 |
Each tablet for oral administration contains 2.68 mg of clemastine fumarate, USP.
Inactive ingredients: colloidal silicon dioxide, corn starch, lactose monohydrate, povidone, pregelatinized starch and stearic acid.
CLINICAL PHARMACOLOGY
Clemastine fumarate is an antihistamine with anticholinergic (drying) and sedative side effects. Antihistamines appear to compete with histamine for cell receptor sites on effector cells. The inherently long duration of antihistaminic effects of clemastine fumarate has been demonstrated in wheal and flare studies. In normal human subjects who received histamine injections over a 24-hour period, the antihistaminic activity of clemastine reached a peak at 5 to 7 hours, persisted for 10 to 12 hours and, in some cases, for as long as 24 hours. Pharmacokinetic studies in man utilizing 3 H and 14 C labeled compound demonstrates that: clemastine is rapidly and nearly completely absorbed from the gastrointestinal tract, peak plasma concentrations are attained in 2 to 4 hours, and urinary excretion is the major mode of elimination.
HOW SUPPLIED
Clemastine Fumarate Tablets USP, 2.68 mg are white, round, scored tablets debossed with "NH" and "268". Tablets are packaged in bottles of 30 (NDC 64950-268-03) and 100 (NDC 64950-268-10)
Store at 20° to 25°C (68° to 77°F)
[See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).