Clonazepam - Clonazepam tablet prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of these drugs for patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS and PRECAUTIONS ).
- The use of benzodiazepines, including clonazepam, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing clonazepam and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (see WARNINGS ).
- The continued use of benzodiazepines, including clonazepam, may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of clonazepam after continued use may precipitate acute withdrawal reactions, which can be life-threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue clonazepam or reduce the dosage (see DOSAGE AND ADMINISTRATION and WARNINGS ).
INDICATIONS AND USAGE
Seizure Disorders
Clonazepam tablets, are useful alone or as an adjunct in the treatment of the Lennox-Gastaut syndrome (petit mal variant), akinetic and myoclonic seizures. In patients with absence seizures (petit mal) who have failed to respond to succinimides, clonazepam tablets, may be useful.
Some loss of effect may occur during the course of clonazepam treatment (see PRECAUTIONS: Loss of Effect ).
Panic Disorder
Clonazepam tablets, are indicated for the treatment of panic disorder, with or without agoraphobia, as defined in DSM-V. Panic disorder is characterized by the occurrence of unexpected panic attacks and associated concern about having additional attacks, worry about the implications or consequences of the attacks, and/or a significant change in behavior related to the attacks.
The efficacy of clonazepam tablets, was established in two 6- to 9-week trials in panic disorder patients whose diagnoses corresponded to the DSM-IIIR category of panic disorder (see CLINICAL PHARMACOLOGY: Clinical Trials ).
Panic disorder (DSM-V) is characterized by recurrent unexpected panic attacks, i.e., a discrete period of intense fear or discomfort in which four (or more) of the following symptoms develop abruptly and reach a peak within 10 minutes: (1) palpitations, pounding heart or accelerated heart rate; (2) sweating; (3) trembling or shaking; (4) sensations of shortness of breath or smothering; (5) feeling of choking; (6) chest pain or discomfort; (7) nausea or abdominal distress; (8) feeling dizzy, unsteady, lightheaded or faint; (9) derealization (feelings of unreality) or depersonalization (being detached from oneself); (10) fear of losing control; (11) fear of dying; (12) paresthesias (numbness or tingling sensations); (13) chills or hot flushes.
The effectiveness of clonazepam tablets, in long-term use, that is, for more than 9 weeks, has not been systematically studied in controlled clinical trials. The physician who elects to use clonazepam tablets, for extended periods should periodically reevaluate the long-term usefulness of the drug for the individual patient (see DOSAGE AND ADMINISTRATION ).
DOSAGE AND ADMINISTRATION
Clonazepam is available as a tablet. The tablets should be administered with water by swallowing the tablet whole.
Seizure Disorders
The use of multiple anticonvulsants may result in an increase of CNS depressant adverse effects. This should be considered before adding clonazepam to an existing anticonvulsant regimen.
Adults
The initial dose for adults with seizure disorders should not exceed 1.5 mg/day divided into three doses. Dosage may be increased in increments of 0.5 to 1 mg every 3 days until seizures are adequately controlled or until side effects preclude any further increase. Maintenance dosage must be individualized for each patient depending upon response. Maximum recommended daily dose is 20 mg.
Pediatric Patients
Clonazepam tablets are administered orally. In order to minimize drowsiness, the initial dose for infants and children (up to 10 years of age or 30 kg of body weight) should be between 0.01 and 0.03 mg/kg/day but not to exceed 0.05 mg/kg/day given in two or three divided doses. Dosage should be increased by no more than 0.25 to 0.5 mg every third day until a daily maintenance dose of 0.1 to 0.2 mg/kg of body weight has been reached, unless seizures are controlled or side effects preclude further increase. Whenever possible, the daily dose should be divided into three equal doses. If doses are not equally divided, the largest dose should be given before retiring.
Geriatric Patients
There is no clinical trial experience with clonazepam tablets in seizure disorder patients 65 years of age and older. In general, elderly patients should be started on low doses of clonazepam tablets and observed closely (see PRECAUTIONS: Geriatric Use ).
Panic Disorder
Adults
The initial dose for adults with panic disorder is 0.25 mg twice daily. An increase to the target dose for most patients of 1 mg/day may be made after 3 days. The recommended dose of 1 mg/day is based on the results from a fixed dose study in which the optimal effect was seen at 1 mg/day. Higher doses of 2, 3 and 4 mg/day in that study were less effective than the 1 mg/day dose and were associated with more adverse effects. Nevertheless, it is possible that some individual patients may benefit from doses of up to a maximum dose of 4 mg/day, and in those instances, the dose may be increased in increments of 0.125 to 0.25 mg twice daily every 3 days until panic disorder is controlled or until side effects make further increases undesired. To reduce the inconvenience of somnolence, administration of one dose at bedtime may be desirable.
Treatment should be discontinued gradually, with a decrease of 0.125 mg twice daily every 3 days, until the drug is completely withdrawn.
There is no body of evidence available to answer the question of how long the patient treated with clonazepam should remain on it. Therefore, the physician who elects to use clonazepam tablets for extended periods should periodically reevaluate the long-term usefulness of the drug for the individual patient.
Pediatric Patients
There is no clinical trial experience with clonazepam tablets in panic disorder patients under 18 years of age.
Geriatric Patients
There is no clinical trial experience with clonazepam tablets in panic disorder patients 65 years of age and older. In general, elderly patients should be started on low doses of clonazepam tablets and observed closely (see PRECAUTIONS: Geriatric Use ).
Discontinuation or Dosage Reduction of Clonazepam
To reduce the risk of withdrawal reactions, increased seizure frequency, and status epilepticus, use a gradual taper to discontinue clonazepam or reduce the dosage. If a patient develops withdrawal reactions, consider pausing the taper or increasing the dosage to the previous tapered dosage level. Subsequently decrease the dosage more slowly (see WARNINGS: Dependence and Withdrawal Reactions and DRUG ABUSE AND DEPENDENCE: Dependence ).
CONTRAINDICATIONS
Clonazepam is contraindicated in patients with the following conditions:
- History of sensitivity to benzodiazepines
- Clinical or biochemical evidence of significant liver disease
- Acute narrow angle glaucoma (it may be used in patients with open angle glaucoma who are receiving appropriate therapy).
ADVERSE REACTIONS
The adverse experiences for clonazepam are provided separately for patients with seizure disorders and with panic disorder.
Seizure Disorders
The most frequently occurring side effects of clonazepam are referable to CNS depression. Experience in treatment of seizures has shown that drowsiness has occurred in approximately 50% of patients and ataxia in approximately 30%. In some cases, these may diminish with time; behavior problems have been noted in approximately 25% of patients. Others, listed by system, including those identified during postapproval use of clonazepam are:
Cardiovascular : Palpitations
Dermatologic : Hair loss, hirsutism, skin rash, ankle and facial edema
Gastrointestinal : Anorexia, coated tongue, constipation, diarrhea, dry mouth, encopresis, gastritis, increased appetite, nausea, sore gums
Genitourinary : Dysuria, enuresis, nocturia, urinary retention
Hematopoietic : Anemia, leukopenia, thrombocytopenia, eosinophilia
Hepatic : Hepatomegaly, transient elevations of serum transaminases and alkaline phosphatase
Musculoskeletal : Muscle weakness, pains
Miscellaneous : Dehydration, general deterioration, fever, lymphadenopathy, weight loss or gain
Neurologic : Abnormal eye movements, aphonia, choreiform movements, coma, diplopia, dysarthria, dysdiadochokinesis, “glassy-eyed” appearance, headache, hemiparesis, hypotonia, nystagmus, respiratory depression, slurred speech, tremor, vertigo
Psychiatric : Confusion, depression, amnesia, hysteria, increased libido, insomnia, psychosis (the behavior effects are more likely to occur in patients with a history of psychiatric disturbances).
The following paradoxical reactions have been observed: irritability, aggression, agitation, nervousness, hostility, anxiety, sleep disturbances, nightmares, abnormal dreams, hallucinations.
Respiratory : Chest congestion, rhinorrhea, shortness of breath, hypersecretion in upper respiratory passages
Panic Disorder : Adverse events during exposure to clonazepam were obtained by spontaneous report and recorded by clinical investigators using terminology of their own choosing. Consequently, it is not possible to provide a meaningful estimate of the proportion of individuals experiencing adverse events without first grouping similar types of events into a smaller number of standardized event categories. In the tables and tabulations that follow, CIGY dictionary terminology has been used to classify reported adverse events, except in certain cases in which redundant terms were collapsed into more meaningful terms, as noted below.
The stated frequencies of adverse events represent the proportion of individuals who experienced, at least once, a treatment-emergent adverse event of the type listed. An event was considered treatment-emergent if it occurred for the first time or worsened while receiving therapy following baseline evaluation.
Adverse Findings Observed in Short-Term, Placebo-Controlled Trials
Adverse Events Associated with Discontinuation of Treatment
Overall, the incidence of discontinuation due to adverse events was 17% in clonazepam compared to 9% for placebo in the combined data of two 6- to 9-week trials. The most common events (≥1%) associated with discontinuation and a dropout rate twice or greater for clonazepam than that of placebo included the following:
Table 2 Most Common Adverse Events (≥1%) Associated with Discontinuation of Treatment
Adverse Event | Clonazepam (N=574) | Placebo (N=294) |
Somnolence | 7% | 1% |
Depression | 4% | 1% |
Dizziness | 1% | <1% |
Nervousness | 1% | 0% |
Ataxia | 1% | 0% |
Intellectual Ability Reduced | 1% | 0% |
Adverse Events Occurring at an Incidence of 1% or More among Clonazepam-Treated Patients
Table 3 enumerates the incidence, rounded to the nearest percent, of treatment-emergent adverse events that occurred during acute therapy of panic disorder from a pool of two 6- to 9-week trials. Events reported in 1% or more of patients treated with clonazepam (doses ranging from 0.5 to 4 mg/day) and for which the incidence was greater than that in placebo-treated patients are included.
The prescriber should be aware that the figures in Table 3 cannot be used to predict the incidence of side effects in the course of usual medical practice where patient characteristics and other factors differ from those that prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses and investigators. The cited figures, however, do provide the prescribing physician with some basis for estimating the relative contribution of drug and nondrug factors to the side effect incidence in the population studied.
Table 3 Treatment-Emergent Adverse Event Incidence in 6- to 9-Week Placebo-Controlled Clinical Trials •
Clonazepam Maximum Daily Dose | ||||||
Adverse Event by Body System | < 1 mg n=96 % | 1- < 2 mg n=129 % | 2- < 3 mg n=113 % | ≥ 3 mg n=235 % | All Clonazepam Groups N=574 % | Placebo N=294 % |
Central & Peripheral Nervous System | ||||||
Somnolence † | 26 | 35 | 50 | 36 | 37 | 10 |
Dizziness | 5 | 5 | 12 | 8 | 8 | 4 |
Coordination Abnormal † | 1 | 2 | 7 | 9 | 6 | 0 |
Ataxia † | 2 | 1 | 8 | 8 | 5 | 0 |
Dysarthria † | 0 | 0 | 4 | 3 | 2 | 0 |
Psychiatric | ||||||
Depression | 7 | 6 | 8 | 8 | 7 | 1 |
Memory Disturbance | 2 | 5 | 2 | 5 | 4 | 2 |
Nervousness | 1 | 4 | 3 | 4 | 3 | 2 |
Intellectual Ability Reduced | 0 | 2 | 4 | 3 | 2 | 0 |
Emotional Lability | 0 | 1 | 2 | 2 | 1 | 1 |
Libido Decreased | 0 | 1 | 3 | 1 | 1 | 0 |
Confusion | 0 | 2 | 2 | 1 | 1 | 0 |
Respiratory System | ||||||
Upper Respiratory Tract Infection † | 10 | 10 | 7 | 6 | 8 | 4 |
Sinusitis | 4 | 2 | 8 | 4 | 4 | 3 |
Rhinitis | 3 | 2 | 4 | 2 | 2 | 1 |
Coughing | 2 | 2 | 4 | 0 | 2 | 0 |
Pharyngitis | 1 | 1 | 3 | 2 | 2 | 1 |
Bronchitis | 1 | 0 | 2 | 2 | 1 | 1 |
Gastrointestinal System | ||||||
Constipation † | 0 | 1 | 5 | 3 | 2 | 2 |
Appetite Decreased | 1 | 1 | 0 | 3 | 1 | 1 |
Abdominal Pain † | 2 | 2 | 2 | 0 | 1 | 1 |
Body as a Whole | ||||||
Fatigue | 9 | 6 | 7 | 7 | 7 | 4 |
Allergic Reaction | 3 | 1 | 4 | 2 | 2 | 1 |
Musculoskeletal | ||||||
Myalgia | 2 | 1 | 4 | 0 | 1 | 1 |
Resistance Mechanism Disorders | ||||||
Influenza | 3 | 2 | 5 | 5 | 4 | 3 |
Urinary System | ||||||
Micturition Frequency | 1 | 2 | 2 | 1 | 1 | 0 |
Urinary Tract Infection † | 0 | 0 | 2 | 2 | 1 | 0 |
Vision Disorders | ||||||
Blurred Vision | 1 | 2 | 3 | 0 | 1 | 1 |
Reproductive Disorders ‡ | ||||||
Female | ||||||
Dysmenorrhea | 0 | 6 | 5 | 2 | 3 | 2 |
Colpitis | 4 | 0 | 2 | 1 | 1 | 1 |
Male | ||||||
Ejaculation Delayed | 0 | 0 | 2 | 2 | 1 | 0 |
Impotence | 3 | 0 | 2 | 1 | 1 | 0 |
• Events reported by at least 1% of patients treated with clonazepam and for which the incidence was greater than that for placebo. † Indicates that the p-value for the dose-trend test (Cochran-Mantel-Haenszel) for adverse event incidence was ≤0.10. ‡ Denominators for events in gender-specific systems are: n=240 (clonazepam), 102 (placebo) for male, and 334 (clonazepam), 192 (placebo) for female.
Commonly Observed Adverse Events
Table 4 Incidence of Most Commonly Observed Adverse Events • in Acute Therapy in Pool of 6- to 9-Week Trials
Adverse Event | Clonazepam (N=574) | Placebo (N=294) |
Somnolence | 37% | 10% |
Depression | 7% | 1% |
Coordination Abnormal | 6% | 0% |
Ataxia | 5% | 0% |
• Treatment-emergent events for which the incidence in the clonazepam patients was ≥5% and at least twice that in the placebo patients.
Treatment-Emergent Depressive Symptoms
In the pool of two short-term placebo-controlled trials, adverse events classified under the preferred term "depression" were reported in 7% of clonazepam-treated patients compared to 1% of placebo-treated patients, without any clear pattern of dose relatedness. In these same trials, adverse events classified under the preferred term "depression" were reported as leading to discontinuation in 4% of clonazepam-treated patients compared to 1% of placebo-treated patients. While these findings are noteworthy, Hamilton Depression Rating Scale (HAM-D) data collected in these trials revealed a larger decline in HAM-D scores in the clonazepam group than the placebo group suggesting that clonazepam-treated patients were not experiencing a worsening or emergence of clinical depression.
Other Adverse Events Observed During the Premarketing Evaluation of Clonazepam in Panic Disorder
Following is a list of modified CIGY terms that reflect treatment-emergent adverse events reported by patients treated with clonazepam at multiple doses during clinical trials. All reported events are included except those already listed in Table 3 or elsewhere in labeling, those events for which a drug cause was remote, those event terms which were so general as to be uninformative, and events reported only once and which did not have a substantial probability of being acutely life-threatening. It is important to emphasize that, although the events occurred during treatment with clonazepam, they were not necessarily caused by it.
Events are further categorized by body system and listed in order of decreasing frequency. These adverse events were reported infrequently, which is defined as occurring in 1/100 to 1/1000 patients.
Body as a Whole : weight increase, accident, weight decrease, wound, edema, fever, shivering, abrasions, ankle edema, edema foot, edema periorbital, injury, malaise, pain, cellulitis, inflammation localized
Cardiovascular Disorders : chest pain, hypotension postural
Central and Peripheral Nervous System Disorders : migraine, paresthesia, drunkenness, feeling of enuresis, paresis, tremor, burning skin, falling, head fullness, hoarseness, hyperactivity, hypoesthesia, tongue thick, twitching
Gastrointestinal System Disorders : abdominal discomfort, gastrointestinal inflammation, stomach upset, toothache, flatulence, pyrosis, saliva increased, tooth disorder, bowel movements frequent, pain pelvic, dyspepsia, hemorrhoids
Hearing and Vestibular Disorders : vertigo, otitis, earache, motion sickness
Heart Rate and Rhythm Disorders : palpitation
Metabolic and Nutritional Disorders : thirst, gout
Musculoskeletal System Disorders : back pain, fracture traumatic, sprains and strains, pain leg, pain nape, cramps muscle, cramps leg, pain ankle, pain shoulder, tendinitis, arthralgia, hypertonia, lumbago, pain feet, pain jaw, pain knee, swelling knee
Platelet, Bleeding and Clotting Disorders: bleeding dermal
Psychiatric Disorders : insomnia, organic disinhibition, anxiety, depersonalization, dreaming excessive, libido loss, appetite increased, libido increased, reactions decreased, aggression, apathy, disturbance in attention, excitement, anger, hunger abnormal, illusion, nightmares, sleep disorder, suicide ideation, yawning
Reproductive Disorders, Female : breast pain, menstrual irregularity
Reproductive Disorders, Male : ejaculation decreased
Resistance Mechanism Disorders : infection mycotic, infection viral, infection streptococcal, herpes simplex infection, infectious mononucleosis, moniliasis
Respiratory System Disorders : sneezing excessive, asthmatic attack, dyspnea, nosebleed, pneumonia, pleurisy
Skin and Appendages Disorders: acne flare, alopecia, xeroderma, dermatitis contact, flushing, pruritus, pustular reaction, skin burns, skin disorder
Special Senses Other, Disorders : taste loss
Urinary System Disorders : dysuria, cystitis, polyuria, urinary incontinence, bladder dysfunction, urinary retention, urinary tract bleeding, urine discoloration
Vascular (Extracardiac) Disorders: thrombophlebitis leg
Vision Disorders : eye irritation, visual disturbance, diplopia, eye twitching, styes, visual field defect, xerophthalmia
DESCRIPTION
Clonazepam Tablets, USP, a benzodiazepine, are available as round scored tablets containing 0.5 mg, 1 mg, or 2 mg clonazepam and microcrystalline cellulose, lactose monohydrate, corn starch, and magnesium stearate
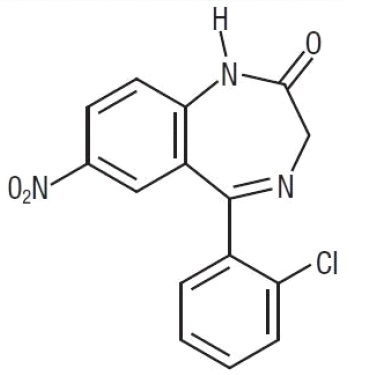
Chemically, clonazepam is 5-(2-chlorophenyl)-1, 3-dihydro-7-nitro-2 H -1, 4-benzodiazepin-2-one. It is a light yellow crystalline powder. It has a molecular weight of 315.72 and the following structural formula:
CLINICAL PHARMACOLOGY
Pharmacodynamics
The precise mechanism by which clonazepam exerts its antiseizure and antipanic effects is unknown, although it is believed to be related to its ability to enhance the activity of gamma aminobutyric acid (GABA), the major inhibitory neurotransmitter in the central nervous system.
Pharmacokinetics
Clonazepam is rapidly and completely absorbed after oral administration. The absolute bioavailability of clonazepam is about 90%. Maximum plasma concentrations of clonazepam are reached within 1 to 4 hours after oral administration. Clonazepam is approximately 85% bound to plasma proteins. Clonazepam is highly metabolized, with less than 2% unchanged clonazepam being excreted in the urine. Biotransformation occurs mainly by reduction of the 7-nitro group to the 4-amino derivative. This derivative can be acetylated, hydroxylated and glucuronidated. Cytochrome P-450 including CYP3A, may play an important role in clonazepam reduction and oxidation. The elimination half-life of clonazepam is typically 30 to 40 hours. Clonazepam pharmacokinetics are dose-independent throughout the dosing range. There is no evidence that clonazepam induces its own metabolism or that of other drugs in humans.
Pharmacokinetics in Demographic Subpopulations and in Disease States
Controlled studies examining the influence of gender and age on clonazepam pharmacokinetics have not been conducted, nor have the effects of renal or liver disease on clonazepam pharmacokinetics been studied. Because clonazepam undergoes hepatic metabolism, it is possible that liver disease will impair clonazepam elimination. Thus, caution should be exercised when administering clonazepam to these patients (see CONTRAINDICATIONS ).
In children, clearance values of 0.42 ± 0.32 mL/min/kg (ages 2 – 18 years) and 0.88 ± 0.4 mL/min/kg (ages 7 – 12 years) were reported; these values decreased with increasing body weight. Ketogenic diet in children does not affect clonazepam concentrations.
Clinical Trials
Panic Disorder
Panic Disorder: The effectiveness of clonazepam in the treatment of panic disorder was demonstrated in two double-blind, placebo-controlled studies of adult outpatients who had a primary diagnosis of panic disorder (DSM-IIIR) with or without agoraphobia. In these studies, clonazepam was shown to be significantly more effective than placebo in treating panic disorder on change from baseline in panic attack frequency, the Clinician’s Global Impression Severity of Illness Score and the Clinician’s Global Impression Improvement Score.
Study 1 was a 9-week, fixed-dose study involving clonazepam doses of 0.5, 1, 2, 3 or 4 mg/day or placebo. This study was conducted in four phases: a 1-week placebo lead-in, a 3-week upward titration, a 6-week fixed dose, and a 7-week discontinuance phase. A significant difference from placebo was observed consistently only for the 1 mg/day group. The difference between the 1 mg dose group and placebo in reduction from baseline in the number of full panic attacks was approximately 1 panic attack per week. At endpoint, 74% of patients receiving clonazepam 1 mg/day were free of full panic attacks, compared to 56% of placebo-treated patients.
Study 2 was a 6-week, flexible-dose study involving clonazepam in a dose range of 0.5 to 4 mg/day or placebo. This study was conducted in three phases: a 1-week placebo lead-in, a 6-week optimal-dose, and a 6-week discontinuance phase. The mean clonazepam dose during the optimal dosing period was 2.3 mg/day. The difference between clonazepam and placebo in reduction from baseline in the number of full panic attacks was approximately 1 panic attack per week. At endpoint, 62% of patients receiving clonazepam were free of full panic attacks, compared to 37% of placebo-treated patients.
Subgroup analyses did not indicate that there were any differences in treatment outcomes as a function of race or gender.
HOW SUPPLIED
Clonazepam Tablets, USP, are supplied as 0.5 mg white to off white, round scored tablets, debossed with “CE” above bisect “192” on one side and plain on other side and packaged in
90 count bottles (NDC 62135-769-90) and
300 count bottles (NDC 62135-769-31).
1 mg white to off white, round scored tablets, debossed with “CE” above bisect “193” on one side and plain on other side and packaged in
90 count bottles (NDC 62135-770-90) and
300 count bottles (NDC 62135-770-31).
2 mg white to off white, round scored tablets, debossed with “CE” above bisect “194” on one side and plain on other side and packaged in
90 count bottles (NDC 62135-771-90) and
300 count bottles (NDC 62135-771-31).
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Dispense contents in a tight, light-resistant container as defined in the USP, with a child-resistant closure, as required.
KEEP TIGHTLY CLOSED.
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Manufactured for: Chartwell RX, LLC.
Congers, NY 10920
L71657
Rev. 10/2023
Print Medication Guide at: www.chartwellpharma.com/medguides .