Get your patient on Combipatch (estradiol/norethindrone Acetate Transdermal System)- Estradiol/norethindrone Acetate Transdermal System patch, Extended Release (Estradiol/Norethindrone Acetate Transdermal System)
Combipatch (Estradiol/Norethindrone Acetate Transdermal System)- Estradiol/Norethindrone Acetate Transdermal System patch, Extended Release prescribing information
WARNING: CARDIOVASCULAR DISORDERS, BREAST CANCER, ENDOMETRIAL CANCER, AND PROBABLE DEMENTIA
Estrogen Plus Progestin TherapyCardiovascular Disorders and Probable Dementia
Estrogen plus progestin therapy should not be used for the prevention of cardiovascular disease or dementia. (See CLINICAL STUDIES and WARNINGS , Cardiovascular Disorders and Probable Dementia ).
The Women’s Health Initiative (WHI) estrogen plus progestin substudy reported an increased risk of deep vein thrombosis (DVT), pulmonary embolism (PE), stroke and myocardial infarction (MI) in postmenopausal women (50 to 79 years of age) during 5.6 years of treatment with daily oral conjugated estrogens (CE) [0.625 mg] combined with medroxyprogesterone acetate (MPA) [2.5 mg], relative to placebo. (See CLINICAL STUDIES and WARNINGS , Cardiovascular Disorders ).
The WHI Memory Study (WHIMS) estrogen plus progestin ancillary study of the WHI reported an increased risk of developing probable dementia in postmenopausal women 65 years of age or older during 4 years of treatment with daily CE (0.625 mg) combined with MPA (2.5 mg), relative to placebo. It is unknown whether this finding applies to younger postmenopausal women. (See CLINICAL STUDIES and WARNINGS , Probable Dementia and PRECAUTIONS , Geriatric Use ).
Breast Cancer
The WHI estrogen plus progestin substudy demonstrated an increased risk of invasive breast cancer. (See CLINICAL STUDIES and WARNINGS , Malignant Neoplasms , Breast Cancer ).
In the absence of comparable data, these risks should be assumed to be similar for other doses of CE and MPA, and other combinations and dosage forms of estrogens and progestins. Estrogens with or without progestins should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
Estrogen-Alone TherapyEndometrial Cancer
There is an increased risk of endometrial cancer in a woman with a uterus who uses unopposed estrogens. Adding a progestin to estrogen therapy has been shown to reduce the risk of endometrial hyperplasia, which may be a precursor to endometrial cancer. Adequate diagnostic measures, including directed or random endometrial sampling when indicated, should be undertaken to rule out malignancy in postmenopausal women with undiagnosed persistent or recurring abnormal genital bleeding. (See WARNINGS , Malignant Neoplasms , Endometrial Cancer ).
Cardiovascular Disorders and Probable Dementia
Estrogen-alone therapy should not be used for the prevention of cardiovascular disease or dementia (See CLINICAL STUDIES and WARNINGS , Cardiovascular Disorders and Probable Dementia ).
The WHI estrogen-alone substudy reported increased risks of stroke and DVT in postmenopausal women (50 to 79 years of age) during 7.1 years of treatment with daily oral CE (0.625 mg)-alone, relative to placebo. (See CLINICAL STUDIES and WARNINGS , Cardiovascular Disorders ).
The WHIMS estrogen-alone ancillary study of the WHI reported an increased risk of developing probable dementia in postmenopausal women 65 years of age or older during 5.2 years of treatment with daily CE (0.625 mg)-alone, relative to placebo. It is unknown whether this finding applies to younger postmenopausal women. (See CLINICAL STUDIES and WARNINGS , Probable Dementia and PRECAUTIONS , Geriatric Use ).
In the absence of comparable data, these risks should be assumed to be similar for other doses of CE and other dosage forms of estrogens. Estrogens with or without progestins should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman.
INDICATIONS AND USAGE
CombiPatch is indicated in a woman with a uterus for:
- Treatment of moderate to severe vasomotor symptoms due to menopause.
- Treatment of moderate to severe symptoms of vulvar and vaginal atrophy due to menopause. When prescribing solely for the treatment of symptoms of vulvar and vaginal atrophy, topical vaginal products should be considered.
- Treatment of hypoestrogenism due to hypogonadism, castration, or primary ovarian failure.
DOSAGE AND ADMINISTRATION
Generally, when estrogen therapy is prescribed for a postmenopausal woman with a uterus, a progestin should be considered to reduce the risk of endometrial cancer. A woman without a uterus generally does not need a progestin. In some cases, however, hysterectomized women with a history of endometriosis may need a progestin. Use of estrogen-alone or in combination with a progestin, should be with the lowest effective dose and for the shortest duration consistent with treatment goals and risks for the individual woman. Postmenopausal women should be reevaluated periodically as clinically appropriate to determine whether treatment is still necessary. Adequate diagnostic measures, such as directed or random endometrial sampling, when indicated, should be undertaken to rule out malignancy in a postmenopausal woman with a uterus with undiagnosed persistent or recurring abnormal genital bleeding.
Initiation of Therapy
Patients should be started at the lowest dose. Estrogens with or without progestins should be prescribed at the lowest effective doses and for the shortest duration consistent with treatment goals and risks for the individual woman. The lowest effective dose of CombiPatch has not been determined in clinical trials.
Women not currently using continuous estrogen or combination estrogen plus progestin therapy may start therapy with CombiPatch at any time. However, women currently using continuous estrogen or combination estrogen plus progestin therapy should complete the current cycle of therapy, before initiating CombiPatch therapy.
Women often experience withdrawal bleeding at the completion of the cycle. The first day of this bleeding would be an appropriate time to begin CombiPatch therapy.
Therapeutic Regimens
Combination estrogen plus progestin regimens are indicated for women with an intact uterus. Two CombiPatch (estradiol/NETA) transdermal delivery systems are available: 0.05 mg estradiol with 0.14 mg NETA per day (9 cm 2 ) and 0.05 mg estradiol with 0.25 mg NETA per day (16 cm 2 ). The lowest effective dose should be used. For all regimens, women should be reevaluated at 3- to 6-month intervals to determine if changes in hormone therapy or if continued hormone therapy is appropriate.
Continuous Combined Regimen
CombiPatch 0.05 mg estradiol/0.14 mg NETA per day (9 cm 2 ) matrix transdermal system is used for continuous uninterrupted treatment applied twice weekly on the lower abdomen. A new system should be applied to the skin every 3 to 4 days (twice weekly) during a 28-day cycle.
Additionally, a dose of 0.05 mg estradiol/0.25 mg NETA (16 cm 2 system) is available if a greater progestin dose is desired. Irregular bleeding may occur particularly in the first six months, but generally decreases with time, and often to an amenorrheic state.
Continuous Sequential Regimen
CombiPatch can be applied as a sequential regimen in combination with an estradiol-only transdermal delivery system.
In this treatment regimen, a 0.05 mg per day (nominal delivery rate) estradiol transdermal system (Vivelle-Dot ® ) is worn for the first 14 days of a 28-day cycle, replacing the system every 3 to 4 days (twice weekly) according to product directions.
For the remaining 14 days of the 28-day cycle, CombiPatch 0.05 mg estradiol/0.14 mg NETA per day (9 cm 2 ) transdermal system should be worn continuously on the lower abdomen. The CombiPatch system should be replaced every 3 to 4 days (twice weekly) during this 14-day period in the 28-day cycle.
Additionally, a dose of 0.05 mg estradiol/0.25 mg NETA (16 cm 2 system) is available if a greater progestin dose is desired. Women should be advised that monthly withdrawal bleeding often occurs.
Application of the System
Site Selection
CombiPatch should be placed on a smooth (fold-free), clean, dry area of the skin on the lower abdomen. CombiPatch should not be applied to or near the breasts. The area selected should not be oily (which can impair adherence of the system), damaged, or irritated. The waistline should be avoided, since tight clothing may rub the system off or modify drug delivery. The sites of application must be rotated, with an interval of at least one week allowed between applications to the same site.
Application
After opening the pouch, remove 1 side of the protective liner, taking care not to touch the adhesive part of the transdermal delivery system with the fingers. Immediately apply the transdermal delivery system to a smooth (fold-free) area of skin on the lower abdomen. Remove the second side of the protective liner and press the system firmly in place with the hand for at least 10 seconds, making sure there is good contact, especially around the edges.
Care should be taken that the system does not become dislodged during bathing and other activities. If a system should fall off, the same system may be reapplied to another area of the lower abdomen. If necessary, a new transdermal system may be applied, in which case, the original treatment schedule should be continued. Only 1 system should be worn at any 1 time during the 3- to 4-day dosing interval.
Once in place, the transdermal system should not be exposed to the sun for prolonged periods of time.
Removal of the System
Removal of the system should be done carefully and slowly to avoid irritation of the skin. Should any adhesive remain on the skin after removal of the system, allow the area to dry for 15 minutes. Then gently rub the area with an oil-based cream or lotion to remove the adhesive residue.
CONTRAINDICATIONS
CombiPatch is contraindicated in women with any of the following conditions:
- Undiagnosed abnormal genital bleeding.
- Known, suspected, or history of breast cancer.
- Known or suspected estrogen-dependent neoplasia.
- Active DVT, PE, or history of these conditions.
- Active arterial thromboembolic disease (for example, stroke and MI), or a history of these conditions.
- Known anaphylactic reaction or angioedema or hypersensitivity with CombiPatch.
- Known liver impairment or disease.
- Known protein C, protein S, or antithrombin deficiency, or other known thrombophilic disorders.
- Known or suspected pregnancy.
ADVERSE REACTIONS
See BOXED WARNING , WARNINGS , and PRECAUTIONS .
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
| 46% | 48% | 41% | |
| Abdominal Pain | 7% | 6% | 4% |
| Accidental Injury | 4% | 5% | 8% |
| Asthenia | 8% | 12% | 4% |
| Back Pain | 11% | 9% | 5% |
| Flu Syndrome | 9% | 5% | 7% |
| Headache | 18% | 20% | 20% |
| Pain | 6% | 4% | 9% |
| 19% | 23% | 24% | |
| Diarrhea | 4% | 5% | 7% |
| Dyspepsia | 1% | 5% | 5% |
| Flatulence | 4% | 5% | 4% |
| Nausea | 11% | 8% | 7% |
| 16% | 28% | 28% | |
| Depression | 3% | 5% | 9% |
| Insomnia | 3% | 6% | 7% |
| Nervousness | 3% | 5% | 1% |
| 24% | 38% | 26% | |
| Pharyngitis | 4% | 10% | 2% |
| Respiratory Disorder | 7% | 12% | 7% |
| Rhinitis | 7% | 13% | 9% |
| Sinusitis | 4% | 9% | 9% |
| 8% | 17% | 16% | |
| Application Site Reaction• | 2% | 6% | 4% |
| 54% | 63% | 28% | |
| Breast Pain | 25% | 31% | 7% |
| Dysmenorrhea | 20% | 21% | 5% |
| Leukorrhea | 5% | 5% | 3% |
| Menstrual Disorder | 6% | 12% | 2% |
| Papanicolaou Smear Suspicious | 8% | 4% | 5% |
| Vaginitis | 6% | 13% | 5% |
| †Represents milligrams of estradiol/NETA delivered daily by each system. | |||
| •Application site reactions includes localized bleeding, bruising, burning, discomfort, dryness, eczema, edema, erythema, inflammation, irritation, pain, papules, paresthesia, pruritus, rash, skin discoloration, skin pigmentation, swelling, urticaria, and vesicles. | |||
| 61% | 60% | 59% | |
| Abdominal Pain | 12% | 14% | 16% |
| Accidental Injury | 10% | 11% | 8% |
| Asthenia | 10% | 13% | 11% |
| Back Pain | 15% | 14% | 13% |
| Flu Syndrome | 14% | 10% | 7% |
| Headache | 25% | 17% | 21% |
| Infection | 5% | 3% | 3% |
| Pain | 19% | 15% | 13% |
| 42% | 32% | 31% | |
| Constipation | 2% | 5% | 3% |
| Diarrhea | 14% | 9% | 7% |
| Dyspepsia | 8% | 6% | 5% |
| Flatulence | 7% | 5% | 6% |
| Nausea | 8% | 12% | 11% |
| Tooth Disorder | 6% | 4% | 1% |
| 12% | 13% | 11% | |
| Peripheral Edema | 6% | 6% | 5% |
| 17% | 17% | 15% | |
| Arthralgia | 6% | 6% | 5% |
| 33% | 30% | 28% | |
| Depression | 8% | 9% | 8% |
| Dizziness | 6% | 7% | 5% |
| Insomnia | 8% | 6% | 4% |
| Nervousness | 5% | 6% | 3% |
| 45% | 43% | 40% | |
| Bronchitis | 5% | 3% | 4% |
| Pharyngitis | 9% | 9% | 8% |
| Respiratory Disorder | 13% | 9% | 13% |
| Rhinitis | 19% | 22% | 17% |
| Sinusitis | 10% | 12% | 12% |
| 38% | 37% | 31% | |
| Acne | 4% | 5% | 4% |
| Application Site Reaction• | 20% | 23% | 17% |
| Rash | 6% | 5% | 3% |
| 71% | 79% | 74% | |
| Breast Enlargement | 2% | 7% | 2% |
| Breast Pain | 34% | 48% | 40% |
| Dysmenorrhea | 30% | 31% | 19% |
| Leukorrhea | 10% | 8% | 9% |
| Menorrhagia | 2% | 5% | 9% |
| Menstrual Disorder | 17% | 19% | 14% |
| Vaginal Hemorrhage | 3% | 6% | 12% |
| Vaginitis | 9% | 13% | 13% |
| †Represents milligrams of estradiol/NETA delivered daily by each system. | |||
| •Application site reactions includes localized bleeding, bruising, burning, discomfort, dryness, eczema, edema, erythema, inflammation, irritation, pain, papules, paresthesia, pruritus, rash, skin discoloration, skin pigmentation, swelling, urticaria, and vesicles. | |||
The following additional adverse reactions have been identified during post-approval use of CombiPatch. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Genitourinary SystemEndometrial hyperplasia, endocervical polyp, uterine leiomyomata, fallopian tube cyst, uterine spasms.
BreastBreast cancer.
CardiovascularHypertension, varicose veins.
GastrointestinalJaundice cholestatic, cholelithiasis, gall bladder disorder, transaminases increased.
SkinSkin discoloration.
Central Nervous SystemAffect lability, libido disorder, migraine, vertigo, paresthesia.
MiscellaneousAngioedema, hypersensitivity, weight increased.
Drug Interactions
No drug interaction studies have been conducted with CombiPatch. In vitro and in vivo studies have shown that estrogens are metabolized partially by cytochrome P450 3A4 (CYP3A4). Therefore, inducers or inhibitors of CYP3A4 may affect estrogen drug metabolism. Inducers of CYP3A4 such as St. John’s wort ( Hypericum perforatum ) preparations, anticonvulsants (e.g., phenobarbital, phenytoin and carbamazepine), phenylbutazone, and anti-infectives (e.g., rifampin, rifabutin, nevirapine and efavirenz) may reduce plasma concentrations of estrogens, possibly resulting in a decrease in therapeutic effects and/or changes in the uterine bleeding profile. Inhibitors of CYP3A4 such as erythromycin, clarithromycin, ketoconazole, itraconazole, ritonavir, nelfinavir and grapefruit juice may increase plasma concentrations of estrogens and may result in side effects.
Adhesion
Averaging across 6 clinical trials lasting 3 months to 1 year, of 1,287 patients treated, CombiPatch transdermal systems completely adhered to the skin nearly 90 percent of the time over the 3- to 4-day wear period. Less than 2 percent of the patients required reapplication or replacement of systems due to lifting or detachment. Two patients (0.2 percent) discontinued therapy during clinical trials due to adhesion failure.
DESCRIPTION
CombiPatch ® (estradiol/norethindrone acetate transdermal system) is an adhesive-based matrix transdermal patch designed to release both estradiol, an estrogen, and norethindrone acetate (NETA), a progestational agent, continuously upon application to intact skin.
Two systems are available, providing the following in vivo delivery rates of estradiol and NETA.
| 1 NETA=norethindrone acetate. 2 Based on in vivo/in vitro flux data, delivery of both components per day via skin of average permeability (interindividual variation in skin permeability is approximately 20 percent). | |||
| System Size | Estradiol (mg) | NETA 1 (mg) | Nominal Delivery Rate 2 (mg per day) Estradiol / NETA |
|---|---|---|---|
| 9 cm 2 round | 0.62 | 2.7 | 0.05/0.14 |
| 16 cm 2 round | 0.51 | 4.8 | 0.05/0.25 |
Estradiol USP (estradiol) is a white to creamy white, odorless, crystalline powder, chemically described as estra-1,3,5(10)-triene-3,17 β -diol. The molecular weight of estradiol is 272.39 and the molecular formula is C 18 H 24 O 2 .
NETA USP is a white to creamy white, odorless, crystalline powder, chemically described as 17-hydroxy-19-nor-17α-pregn-4-en-20-yn-3-one acetate. The molecular weight of NETA is 340.47 and the molecular formula is C 22 H 28 O 3 .
The structural formulas for estradiol and NETA are:
CombiPatch is comprised of 3 layers. Proceeding from the visible surface toward the surface attached to the skin, these layers are (1) a translucent polyolefin film backing, (2) an adhesive layer containing estradiol, NETA, acrylic adhesive, silicone adhesive, oleic acid NF, povidone USP and dipropylene glycol, and (3) a polyester release protective liner, which is attached to the adhesive surface and must be removed before the system can be used.
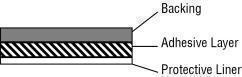
The active components of the system are estradiol USP and NETA USP. The remaining components of the system are pharmacologically inactive.
CLINICAL PHARMACOLOGY
Endogenous estrogens are largely responsible for the development and maintenance of the female reproductive system and secondary sexual characteristics. Although circulating estrogens exist in a dynamic equilibrium of metabolic interconversions, estradiol is the principal intracellular human estrogen and is substantially more potent than its metabolites, estrone and estriol at the receptor level.
The primary source of estrogen in normally cycling adult women is the ovarian follicle, which secretes 70 to 500 mcg of estradiol daily, depending on the phase of the menstrual cycle. After menopause, most endogenous estrogen is produced by conversion of androstenedione, secreted by the adrenal cortex, to estrone in the peripheral tissues. Thus, estrone and the sulfate conjugated form, estrone sulfate, are the most abundant circulating estrogens in postmenopausal women.
Estrogens act through binding to nuclear receptors in estrogen-responsive tissues. To date, 2 estrogen receptors have been identified. These vary in proportion from tissue to tissue.
Circulating estrogens modulate the pituitary secretion of the gonadotropins, luteinizing hormone (LH), and follicle stimulating hormone (FSH) through a negative feedback mechanism. Estrogens act to reduce the elevated levels of these hormones seen in postmenopausal women.
Pharmacokinetics
Absorption
Estradiol: Estrogens used in hormone therapy are well absorbed through the skin, mucous membranes, and gastrointestinal tract. Administration of CombiPatch every 3 to 4 days in postmenopausal women produces average steady-state estradiol serum concentrations of 45 to 50 pg/mL, which are equivalent to the normal ranges observed at the early follicular phase in premenopausal women. These concentrations are achieved within 12 to 24 hours following CombiPatch application. Minimal fluctuations in serum estradiol concentrations are observed following CombiPatch application, indicating consistent hormone delivery over the application interval.
In 1 study, serum concentrations of estradiol were measured in 40 healthy, postmenopausal women throughout 3 consecutive CombiPatch applications to the abdomen (each dose was applied for three 3.5-day periods). The corresponding pharmacokinetic parameters are summarized in Table 1.
| Estradiol | ||||
|---|---|---|---|---|
| System Size | Dose Estradiol/NETA (mg per day) | C max | C min | C avg |
| 9 cm 2 | 0.05/0.14 | 71 (32) | 27 (17) | 45 (21) |
| 16 cm 2 | 0.05/0.25 | 71 (30) | 37 (17) | 50 (21) |
| Estrone | ||||
| 9 cm 2 | 0.05/0.14 | 72 (23) | 49 (19) | 54 (19) |
| 16 cm 2 | 0.05/0.25 | 78 (22) | 58 (22) | 60 (18) |
Norethindrone: Progestins used in hormone therapy are well absorbed through the skin, mucous membranes, and gastrointestinal tract. Norethindrone steady-state concentrations are attained within 24 hours of application of the CombiPatch transdermal delivery systems. Minimal fluctuations in serum norethindrone concentrations are observed following CombiPatch treatment, indicating consistent hormone delivery over the application interval. Serum concentrations of norethindrone increase linearly with increasing doses of NETA.
In 1 study, serum concentrations of norethindrone were measured in 40 healthy, postmenopausal women throughout 3 consecutive CombiPatch applications to the abdomen (each dose was applied for three 3.5-day periods). The corresponding pharmacokinetic parameters are summarized in Table 2.
| System Size | Dose Estradiol/NETA (mg per day) | C max | C min | C avg |
|---|---|---|---|---|
| 9 cm 2 | 0.05/0.14 | 617 (341) | 386 (137) | 489 (244) |
| 16 cm 2 | 0.05/0.25 | 1060 (543) | 686 (306) | 840 (414) |
Distribution
Estradiol: The distribution of exogenous estrogens is similar to that of endogenous estrogens. Estrogens are widely distributed in the body and are generally found in higher concentrations in the sex hormone target organs. Estrogens circulate in the blood largely bound to sex hormone-binding globulin (SHBG) and albumin.
Norethindrone: In plasma, norethindrone is bound approximately 90 percent to SHBG and albumin.
Metabolism
Estradiol: Exogenous estrogens are metabolized in the same manner as endogenous estrogens. Circulating estrogens exist in a dynamic equilibrium of metabolic interconversions. These transformations take place mainly in the liver. Estradiol is converted reversibly to estrone, and both can be converted to estriol, which is a major urinary metabolite. Estrogens also undergo enterohepatic recirculation via sulfate and glucuronide conjugation in the liver, biliary secretion of conjugates into the intestine, and hydrolysis in the intestine followed by reabsorption. In postmenopausal women a significant portion of the circulating estrogens exist as sulfate conjugates, especially estrone sulfate, which serves as a circulating reservoir for the formation of more active estrogens.
Norethindrone: NETA is hydrolyzed to the active moiety, norethindrone, in most tissues including skin and blood. Norethindrone is primarily metabolized in the liver.
Excretion
Estradiol: Estradiol, estrone, and estriol are excreted in the urine along with glucuronide and sulfate conjugates. Estradiol has a short elimination half-life of approximately 2 to 3 hours; therefore, a rapid decline in serum levels is observed after the CombiPatch estradiol/NETA transdermal system is removed. Within 4 to 8 hours serum estradiol concentrations return to untreated, postmenopausal levels (less than 20 pg/mL).
Concentration data from clinical trials indicate that the pharmacokinetics of estradiol did not change over time, suggesting no evidence of the accumulation of estradiol following extended patch wear periods (up to 1 year).
Norethindrone: The elimination half-life of norethindrone is reported to be 6 to 8 hours. Norethindrone serum concentrations diminish rapidly and are less than 50 pg/mL within 48 hours after removal of the CombiPatch transdermal delivery system.
Concentration data from clinical trials indicate that the pharmacokinetics of norethindrone did not change over time, suggesting no evidence of the accumulation of norethindrone following extended patch wear periods (up to 1 year).
Special Populations
No pharmacokinetic studies were conducted in special populations, including patients with renal or hepatic impairment.
CLINICAL STUDIES
Effects on Vasomotor Symptoms
In 2 clinical trials designed to assess the degree of relief of moderate to severe vasomotor symptoms in postmenopausal women (n=332), CombiPatch was administered for 3 28-day cycles in Continuous Combined or Continuous Sequential treatment regimens versus placebo. In the Continuous Combined regimen, CombiPatch was applied throughout the 3 cycles, replacing the system twice weekly. In the Continuous Sequential regimen, an estradiol-only transdermal system (Vivelle ® 0.05 mg) was applied twice weekly during the first 14 days of a 28-day cycle; CombiPatch was applied for the remaining 14 days of the cycle and replaced twice weekly, as well. The mean number of hot flushes at baseline were 10 to 11 per day and 11 to 12 per day in the Continuous Combined and Continuous Sequential regimen trials, respectively. The mean number and intensity of daily hot flushes (intent-to-treat population) was significantly reduced from baseline to endpoint with either the Continuous Combined or Continuous Sequential administration of CombiPatch at all doses as compared to placebo (intent-to-treat population). (See Tables 3 and 4 )
| 1 Means were adjusted for imbalance among treatment groups and investigators (least squares mean from ANOVA). 2 Represents the milligrams of estradiol/NETA delivered daily by each system. 3 Population represents those patients who had baseline and endpoint observations. 4 The intensity of hot flushes was evaluated on a scale of 0 to 9 (none=0, mild=1-3, moderate= 4-6, severe=7-9). 5 P-value versus placebo = <0.001. 6 Total number of patients with available data is 56. 7 Total number of patients with available data is 50. | |||
| CombiPatch Continuous Combined | Placebo|||
|---|---|---|---|
| Adjusted Mean Change from Baseline 1 | 0.05/0.14 mg per day 2 n=57 | 0.05/0.25 mg per day 2 n=52 | n=51 |
| Number of Hot Flushes 3 | -9.3 5 | -8.9 5 | -6.2 |
| Daily Intensity of Hot Flushes 3,4 | -4.6 5,6 | -5.0 5 | -2.8 7 |
| 1 Means were adjusted for imbalance among treatment groups and investigators (least squares mean from ANOVA). 2 Represents the milligrams of estradiol/NETA delivered daily by each system. 3 Population represents those patients who had baseline and endpoint observations. 4 The intensity of hot flushes was evaluated on a scale of 0 to 9 (none=0, mild=1-3, moderate= 4-6, severe=7-9). 5 P-value versus placebo = <0.001. | |||
| CombiPatch ® Continuous Combined | Placebo|||
|---|---|---|---|
| Adjusted Mean Change from Baseline 1 | 0.05/0.14 mg per day 2 n=54 | 0.05/0.25 mg per day 2 n=59 | n=53 |
| Number of Hot Flushes 3 | -9.3 5 | -9.5 5 | -5.5 |
| Daily Intensity of Hot Flushes 3,4 | -4.4 5 | -4.5 5 | -2.1 |
Effects on the Endometrium
The use of unopposed estrogen therapy has been associated with an increased risk of endometrial hyperplasia, a possible precursor of endometrial adenocarcinoma. Progestins counter the estrogenic effects by decreasing the number of nuclear estradiol receptors and suppressing epithelial DNA synthesis in endometrial tissue.
Clinical studies indicate that the addition of a progestin to an estrogen regimen at least 12 days per cycle reduces the incidence of endometrial hyperplasia and the potential risk of adenocarcinoma in women with intact uteri. The addition of a progestin to an estrogen regimen has not been shown to interfere with the efficacy of estrogen therapy for its approved indications.
CombiPatch was effective in reducing the incidence of estrogen-induced endometrial hyperplasia after 1 year of therapy in two clinical trials. Nine hundred fifty-five (955) postmenopausal women (with intact uteri) were treated with (i) a continuous regimen of CombiPatch alone ( Continuous Combined regimen), (ii) a sequential regimen with an estradiol-only (Vivelle 0.05 mg) transdermal system followed by a CombiPatch transdermal system ( Continuous Sequential regimen), or (iii) continuous regimen with an estradiol-only transdermal system (Vivelle 0.05 mg). The incidence of endometrial hyperplasia (primary endpoint) was significantly less after 1 year of therapy with either CombiPatch regimen than with the estradiol-only transdermal system. Tables 5 and 6 summarize these results (intent-to-treat populations).
| 1 Represents milligrams of estradiol/NETA delivered daily by each system. 2 Biopsy after 12 cycles of treatment or hyperplasia before cycle 12. 3 Comparison of continuous combined regimen versus estradiol-only patch was significant (p <0.001). 4 This patient had hyperplasia at baseline. 5 One of 39 patients had hyperplasia in an endometrial polyp. | |||
| CombiPatch Continuous Combined | Vivelle Continuous | ||
|---|---|---|---|
| 0.05/0.14 mg per day 1 | 0.05/0.25 mg per day 1 | 0.05 mg per day | |
| Number of Patients with Biopsies 2 | 123 | 98 | 103 |
| Number (%) of Patients with Hyperplasia | 1 (<1%) 3 | 1 (1%) 3,4 | 39 (38%) 5 |
| 1 Represents milligrams of estradiol/NETA delivered daily by each system. 2 Biopsy after 12 cycles of treatment or hyperplasia before cycle 12. 3 Comparison of continuous sequential regimen versus estradiol-only patch was significant (p <0.001). 4 This patient had hyperplasia at baseline. 5 This patient had hyperplasia in an endometrial polyp. | |||
| CombiPatch Continuous Sequential | Vivelle Continuous | ||
|---|---|---|---|
| 0.05/0.14 mg per day 1 | 0.05/0.25 mg per day 1 | 0.05 mg per day | |
| Number of Patients with Biopsies 2 | 117 | 114 | 115 |
| Number (%) of Patients with Hyperplasia | 1 (<1%) 3,4 | 1 (<1%) 3,5 | 23 (20%) |
Effects on Uterine Bleeding or Spotting
With the Continuous Combined regimen, of the women treated with CombiPatch and who completed the 1-year study, the incidence of cumulative amenorrhea (the absence of bleeding or spotting during a 28-day cycle and sustained to the end of the study) increased over time. The incidence of amenorrhea from cycle 10 through 12 was 53 percent and 39 percent for the CombiPatch 0.05/0.14 mg per day and CombiPatch 0.05/0.25 mg per day treatment groups, respectively. Women who experienced bleeding usually characterized it as light (intensity of 1.3 on a scale of 1 to 4) with a duration of 4 and 6 days for the CombiPatch 0.05/0.14 mg per day and CombiPatch 0.05/0.25 mg per day treatment groups, respectively. (See Figure 1 )
Figure 1. Incidence of Cumulative Amenorrhea• in CombiPatch Continuous Combined Transdermal Therapy by Cycle Over a 1-Year Period (Intent-to-Treat Population)
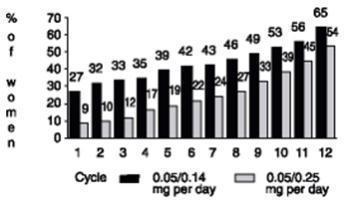
•Cumulative amenorrhea is defined as the absence of bleeding for the duration of a 28-day cycle and sustained to the end of the study.
Women’s Health Initiative Studies
The WHI enrolled approximately 27,000 predominantly healthy postmenopausal women in two substudies to assess the risks and benefits of daily oral CE (0.625 mg)-alone or in combination with MPA (2.5 mg) compared to placebo in the prevention of certain chronic diseases. The primary endpoint was the incidence of coronary heart disease (CHD) [defined as nonfatal MI, silent MI and CHD death], with invasive breast cancer as the primary adverse outcome. A “global index” included the earliest occurrence of CHD, invasive breast cancer, stroke, PE, endometrial cancer (only in the CE plus MPA substudy), colorectal cancer, hip fracture, or death due to other cause. These substudies did not evaluate the effects of CE plus MPA or CE-alone on menopausal symptoms.
WHI Estrogen Plus Progestin Substudy
The WHI estrogen plus progestin substudy was stopped early. According to the predefined stopping rule, after an average follow-up of 5.6 years of treatment, the increased risk of invasive breast cancer and cardiovascular events exceeded the specified benefits included in the “global index.” The absolute excess risk of events included in the “global index” was 19 per 10,000 women-years.
For those outcomes included in the WHI “global index” that reached statistical significance after 5.6 years of follow-up, the absolute excess risks per 10,000 women years in the group treated with CE plus MPA were 7 more CHD events, 8 more strokes, 10 more PEs, and 8 more invasive breast cancers, while the absolute risk reductions per 10,000 women-years were 6 fewer colorectal cancers and 5 fewer hip fractures.
Results of the estrogen plus progestin substudy, which included 16,608 women (average 63 years of age, range 50 to 79 years; 83.9 percent white, 6.8 percent black, 5.4 percent Hispanic, 3.9 percent Other), are presented in Table 7 . These results reflect centrally adjudicated data after an average follow-up of 5.6 years.
| a Adapted from numerous WHI publications. WHI publications can be viewed at www.nhlbi.nih.gov/whi. b Results are based on centrally adjudicated data. c Nominal confidence intervals (CI) unadjusted for multiple looks and multiple comparisons. d Not included in “global index”. e Includes metastatic and non-metastatic breast cancer, with the exception of in situ breast cancer. f All deaths, except from breast or colorectal cancer, definite or probable CHD, PE or cerebrovascular disease. g A subset of the events was combined in a “global index”, defined as the earliest occurrence of CHD events, invasive breast cancer, stroke, pulmonary embolism, colorectal cancer, hip fracture, or death due to other causes. | |||
| Absolute Risk per 10,000 Women-Years | |||
| CHD events | 1.23 (0.99–1.53) | 41 | 34 |
| Nonfatal MI | 1.28 (1.00–1.63) | 31 | 25 |
| CHD death | 1.10 (0.70–1.75) | 8 | 8 |
| All strokes | 1.31 (1.03–1.68) | 33 | 25 |
| Ischemic stroke | 1.44 (1.09–1.90) | 26 | 18 |
| Deep vein thrombosis d | 1.95 (1.43–2.67) | 26 | 13 |
| Pulmonary embolism | 2.13 (1.45–3.11) | 18 | 8 |
| Invasive breast cancer e | 1.24 (1.01–1.54) | 41 | 33 |
| Colorectal cancer | 0.61 (0.42–0.87) | 10 | 16 |
| Endometrial cancer d | 0.81 (0.48–1.36) | 6 | 7 |
| Cervical cancer d | 1.44 (0.47–4.42) | 2 | 1 |
| Hip fracture | 0.67 (0.47–0.96) | 11 | 16 |
| Vertebral fractures d | 0.65 (0.46–0.92) | 11 | 17 |
| Lower arm/wrist fractures d | 0.71 (0.59–0.85) | 44 | 62 |
| Total fractures d | 0.76 (0.69–0.83) | 152 | 199 |
| Overall mortality f | 1.00 (0.83–1.19) | 52 | 52 |
| Global Index g | 1.13 (1.02–1.25) | 184 | 165 |
Timing of the initiation of estrogen plus progestin therapy relative to the start of menopause may affect the overall risk benefit profile. The WHI estrogen plus progestin substudy stratified by age showed in women 50 to 59 years of age a nonsignificant trend toward reduced risk for overall mortality [hazard ratio (HR) 0.69 (95 percent CI 0.44 to 1.07)].
WHI Estrogen-Alone Substudy
The WHI estrogen-alone substudy was stopped early because an increased risk of stroke was observed, and it was deemed that no further information would be obtained regarding the risks and benefits of estrogen-alone in predetermined primary endpoints.
Results of the estrogen-alone substudy, which included 10,739 women (average 63 years of age, range 50 to 79 years; 75.3 percent white, 15.1 percent black, 6.1 percent Hispanic, 3.6 percent Other) after an average follow-up of 7.1 years, are presented in Table 8 .
| a Adapted from numerous WHI publications. WHI publications can be viewed at www.nhlbi.nih.gov/whi . b Nominal CI unadjusted for multiple looks and multiple comparisons. c Results are based on centrally adjudicated data for an average follow-up of 7.1 years. d Not included in “global index”. e Results are based on an average follow-up of 6.8 years. f All deaths, except from breast or colorectal cancer, definite or probable CHD, PE or cerebrovascular disease. g A subset of the events was combined in a “global index”, defined as the earliest occurrence of CHD events, invasive breast cancer, stroke, pulmonary embolism, colorectal cancer, hip fracture, or death due to other causes. | |||
| Absolute Risk per 10,000 Women-Years | |||
| CHD events c | 0.95 (0.78–1.16) | 54 | 57 |
| Nonfatal MI c | 0.91 (0.73–1.14) | 40 | 43 |
| CHD death c | 1.01 (0.71–1.43) | 16 | 16 |
| All strokes c | 1.33 (1.05–1.68) | 45 | 33 |
| Ischemic stroke c | 1.55 (1.19–2.01) | 38 | 25 |
| Deep vein thrombosis c,d | 1.47 (1.06–2.06) | 23 | 15 |
| Pulmonary embolism c | 1.37 (0.90–2.07) | 14 | 10 |
| Invasive breast cancer c | 0.80 (0.62–1.04) | 28 | 34 |
| Colorectal cancer e | 1.08 (0.75–1.55) | 17 | 16 |
| Hip fracture c | 0.65 (0.45–0.94) | 12 | 19 |
| Vertebral fractures c,d | 0.64 (0.44–0.93) | 11 | 18 |
| Lower arm/wrist fractures c,d | 0.58 (0.47–0.72) | 35 | 59 |
| Total fractures c,d | 0.71 (0.64–0.80) | 144 | 197 |
| Death due to other causes e,f | 1.08 (0.88–1.32) | 53 | 50 |
| Overall mortality c,d | 1.04 (0.88–1.22) | 79 | 75 |
| Global Index g | 1.02 (0.92–1.13) | 206 | 201 |
For those outcomes included in the WHI “global index” that reached statistical significance, the absolute excess risk per 10,000 women-years in the group treated with CE-alone was 12 more strokes, while the absolute risk reduction per 10,000 women-years was 7 fewer hip fractures. 9 The absolute excess risk of events included in the “global index” was a nonsignificant 5 events per 10,000 women-years. There was no difference between the groups in terms of all-cause mortality.
No overall difference for primary CHD events (nonfatal MI, silent MI and CHD death) and invasive breast cancer incidence in women receiving CE-alone compared with placebo was reported in final centrally adjudicated results from the estrogen-alone substudy, after an average follow-up of 7.1 years (see Table 8 ).
Centrally adjudicated results for stroke events from the estrogen-alone substudy, after an average follow-up of 7.1 years, reported no significant difference in distribution of stroke subtype or severity, including fatal strokes, in women receiving CE-alone compared to placebo. Estrogen-alone increased the risk for ischemic stroke, and this excess was present in all subgroups of women examined. 10 (see Table 8 ).
Timing of the initiation of estrogen therapy relative to the start of menopause may affect the overall risk benefit profile. The WHI estrogen-alone substudy stratified by age showed in women 50 to 59 years of age, a nonsignificant trend toward reduced risk for CHD [HR 0.63 (95 percent, CI 0.36 to 1.09)] and overall mortality [HR 0.71 (95 percent CI, 0.46 to 1.11)].
Women’s Health Initiative Memory Study
The WHIMS estrogen plus progestin ancillary study of WHI enrolled 4,532 predominantly healthy postmenopausal women 65 years of age and older (47 percent were 65 to 69 years of age; 35 percent were 70 to 74 years of age; 18 percent were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg) plus MPA (2.5 mg) on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 4 years, the relative risk of probable dementia for CE plus MPA versus placebo was 2.05 (95 percent CI, 1.21 to 3.48). The absolute risk of probable dementia for CE plus MPA versus placebo was 45 versus 22 cases per 10,000 women-years. Probable dementia as defined in this study included Alzheimer disease (AD), vascular dementia (VaD) and mixed type (having features of both AD and VaD). The most common classification of probable dementia in the treatment group and the placebo group was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women. (See WARNINGS , Probable Dementia and PRECAUTIONS , Geriatric Use )
The WHIMS estrogen-alone ancillary study of WHI enrolled 2,947 predominantly healthy hysterectomized postmenopausal women 65 to 79 years of age (45 percent were 65 to 69 years of age; 36 percent were 70 to 74 years of age; 19 percent were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg)-alone on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 5.2 years, the relative risk of probable dementia for CE-alone versus placebo was 1.49 (95 percent CI, 0.83 to 2.66). The absolute risk of probable dementia for CE-alone versus placebo was 37 versus 25 cases per 10,000 women-years. Probable dementia as defined in this study included AD, VaD and mixed type (having features of both AD and VaD). The most common classification of probable dementia in the treatment group and the placebo group was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women. (See WARNINGS , Probable Dementia and PRECAUTIONS , Geriatric Use )
When data from the two populations were pooled as planned in the WHIMS protocol, the reported overall relative risk for probable dementia was 1.76 (95 percent CI, 1.19 to 2.60). Differences between groups became apparent in the first year of treatment. It is unknown whether these findings apply to younger postmenopausal women. (See WARNINGS , Probable Dementia and PRECAUTIONS , Geriatric Use )
HOW SUPPLIED
CombiPatch estradiol/NETA transdermal delivery system is available in:
| 9 cm 2 | 0.05/0.14 mg per day | 8 systems per carton | 68968-0514-8 | CombiPatch 0.05/0.14 mg per day |
| 16 cm 2 | 0.05/0.25 mg per day | 8 systems per carton | 68968-0525-8 | CombiPatch 0.05/0.25 mg per day |
| • Nominal delivery rate described. See DESCRIPTION for more details regarding drug delivery. | ||||
Store CombiPatch in the refrigerator at 36℉ to 46℉ (2℃ to 8℃).
Store the systems in the sealed foil pouch.
Do not store the system in areas where extreme temperatures can occur.
Keep this and all medicines out of the reach of children.Vivelle® is a registered trademark of Novartis Corporation. & Vivelle-Dot ® is a registered trademark of Novartis AG.
Instructions for Use
CombiPatch (käm-bē `pach) (estradiol/norethindrone acetate transdermal system)
Step 1. Pick the days you will change your CombiPatch.
- You will need to change your patch every 3 to 4 days (twice weekly).
Step 2. Remove CombiPatch from the pouch.
- Tear open the protective pouch at the slit (do not use scissors) and remove the patch. See Figure A.
- The pouch should not be opened until you are ready to put the patch on.
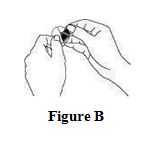
Step 3. Remove the adhesive liner.
- Peel off one side of the protective liner. See Figure B.
- Do not touch the sticky part of the patch with your fingers. See Figure B.
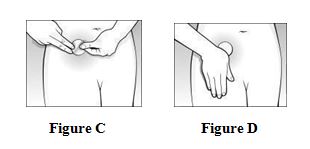
Step 4. Placing the CombiPatch on your skin.
- Put the sticky side of the patch on the lower abdomen (below the panty line). See Figure C.
- Peel off the second side of the protective liner. See Figure C.
- Press the patch firmly in place with your hand for about 10 seconds. See Figure D.
Note:
- Avoid the waistline, since clothing and belts may cause the CombiPatch to be rubbed off.
- Do not apply the CombiPatch to or near your breasts.
- Only apply the CombiPatch to skin that is clean, dry, and free of any powder, oil, or lotion.
- You should not apply the CombiPatch to injured, burned, or irritated skin, or areas with skin conditions (such as birth marks, tattoos, or that is very hairy).
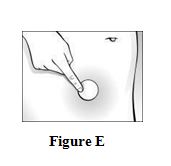
Step 5. Press the CombiPatch firmly onto your skin.
- Rub the edges of the CombiPatch with your fingers to make sure that it will stick to your skin. See Figure E.
Note:
- Bathing, swimming, or showering will not affect the CombiPatch.
- Once in place, the patch should not be exposed to the sun for prolonged periods of time.
- If your CombiPatch falls off reapply it. If you cannot reapply the CombiPatch, apply a new CombiPatch to another area (See Figures C , D and E ) and continue to follow your original placement schedule.
- If you stop using your CombiPatch or forget to apply a new CombiPatch as scheduled you may have spotting, or bleeding, and your symptoms may come back.
Step 6. Throwing away your used CombiPatch.
- When it is time to change your CombiPatch, remove the old CombiPatch before you apply a new one.
- To throw away the patch, fold the sticky side of the patch together, place it in a sturdy child-proof container, and place this container in the trash. Used patches should not be flushed in the toilet.
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Manufactured by: Noven Pharmaceuticals Inc. Miami, FL 33186
Distributed by: Noven Therapeutics, LLC Miami, FL 33186
For more information call 1-800-455-8070 or visit www.combipatch.com.
© 2024 Noven Pharmaceuticals, Inc.
102538-5 02/2024