Get your patient on Dapsone - Dapsone tablet (Dapsone)
Dapsone - Dapsone tablet prescribing information
CONTRAINDICATIONS
Hypersensitivity to Dapsone and/or its derivatives.
ADVERSE REACTIONS
In addition to the warnings listed above, the following syndromes and serious reactions have been reported in patients on Dapsone.
Hematologic Effects: Dose-related hemolysis is the most common adverse effect and is seen in patients with or without G6PD deficiency. Almost all patients demonstrate the inter-related changes of a loss of 1 to 2g of hemoglobin, an increase in the reticulocytes (2 to 12%), a shortened red cell life span and a rise in methemoglobin. G6PD deficient patients have greater responses.
Nervous System Effects: Peripheral neuropathy is a definite but unusual complication of Dapsone therapy in non-leprosy patients. Motor loss is predominant. If muscle weakness appears, Dapsone should be withdrawn. Recovery on withdrawal is usually substantially complete. The mechanism of recovery is reported by axonal regeneration. Some recovered patients have tolerated retreatment at reduced dosage. In leprosy this complication may be difficult to distinguish from a leprosy reactional state.
Falsely Reduced HbA1c
Falsely reduced HbA1c measurements have been reported with dapsone use. Alternate measures of glycemic control (e.g., fructosamine and/or more frequent blood glucose monitoring) are recommended when a discordance between HbA1c and blood glucose concentrations are observed or suspected. Falsely reduced HbA1c may occur without overt evidence of hemolysis or anemia.
Body As A Whole: In addition to the warnings and adverse effects reported above, additional adverse reactions include: nausea, vomiting, abdominal pains, pancreatitis, vertigo, blurred vision, tinnitus, insomnia, fever, headache, psychosis, phototoxicity, pulmonary eosinophilia, tachycardia, albuminuria, the nephrotic syndrome, hypoalbuminemia without proteinuria, renal papillary necrosis, male infertility, drug-induced Lupus erythematosus and an infectious mononucleosis-like syndrome. In general, with the exception of the complications of severe anoxia from overdosage (retinal and optic nerve damage, etc.) these adverse reactions have regressed off drug.
DESCRIPTION
Dapsone-USP, 4,4'-diaminodiphenylsulfone (DDS), is a primary treatment for Dermatitis herpetiformis. It is an antibacterial drug for susceptible cases of leprosy. It is a white to yellow crystalline powder. Sparingly soluble in alcohol; Soluble in acetone and in dilute mineral acids; practically insoluble in water.
Dapsone is issued on prescription in tablets of 25 and 100 mg for oral use. 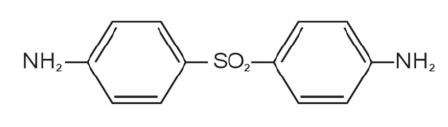
Inactive Ingredients: Colloidal Silicon Dioxide, Corn Starch, Magnesium Stearate and Microcrystalline Cellulose.
USP Dissolution Test Pending.
CLINICAL PHARMACOLOGY
Actions: The mechanism of action in Dermatitis herpetiformis has not been established. By the kinetic method in mice, Dapsone is bactericidal as well as bacteriostatic against Mycobacterium leprae .
Absorption and Excretion: Dapsone, when given orally, is rapidly and almost completely absorbed. About 85 percent of the daily intake is recoverable from the urine mainly in the form of water-soluble metabolites. Excretion of the drug is slow and a constant blood level can be maintained with the usual dosage.
Blood Levels: Detected a few minutes after ingestion, the drug reaches peak concentration in 4 to 8 hours. Daily administration for at least eight days is necessary to achieve a plateau level. With doses of 200 mg daily, this level averaged 2.3 mcg/ml with a range of 0.1 to 7.0 mcg/ml. The half-life in the plasma in different individuals varies from ten hours to fifty hours and averages twenty-eight hours. Repeat tests in the same individual are constant. Daily administration (50 to 100 mg) in leprosy patients will provide blood levels in excess of the usual minimum inhibitory concentration even for patients with a short Dapsone half-life.
HOW SUPPLIED
Dapsone Tablets USP, 25 mg are available as white to creamy white, uncoated round shaped tablets, debossed with “N” above the bisect and “135” below the bisect and plain on other side.
Bottle of 30 tablets NDC 70954-135-10
Bottle of 100 tablets NDC 70954-135-20
Dapsone Tablets USP, 100 mg are available as white to creamy white, uncoated round shaped tablets, debossed with “N” above the bisect and “136” below the bisect and plain on other side.
Bottle of 30 tablets NDC 70954-136-10
Bottle of 100 tablets NDC 70954-136-20