Get your patient on Doxycycline Hyclate - Doxycycline Hyclate tablet, Film Coated (Doxycycline Hyclate)
Doxycycline Hyclate - Doxycycline Hyclate tablet, Film Coated prescribing information
INDICATIONS AND USAGE
Doxycycline hyclate tablets are indicated for use as an adjunct to scaling and root planing to promote attachment level gain and to reduce pocket depth in patients with adult periodontitis.
DOSAGE AND ADMINISTRATION
THE DOSAGE OF DOXYCYCLINE HYCLATE TABLETS DIFFERS FROM THAT OF DOXYCYCLINE USED TO TREAT INFECTIONS. EXCEEDING THE RECOMMENDED DOSAGE MAY RESULT IN AN INCREASED INCIDENCE OF SIDE EFFECTS INCLUDING THE DEVELOPMENT OF RESISTANT MICROORGANISMS.
Doxycycline hyclate tablets 20 mg twice daily as an adjunct following scaling and root planing may be administered for up to 9 months. Doxycycline hyclate tablets should be taken twice daily at 12 hour intervals, usually in the morning and evening. It is recommended that if Doxycycline hyclate tablet is taken close to meal times, allow at least one hour prior to or two hours after meals. Safety beyond 12 months and efficacy beyond 9 months have not been established.
Administration of adequate amounts of fluid along with the tablets is recommended to wash down the drug and reduce the risk of esophageal irritation and ulceration. (See ADVERSE REACTIONS section).
CONTRAINDICATIONS
This drug is contraindicated in persons who have shown hypersensitivity to doxycycline or any of the other tetracyclines.
ADVERSE REACTIONS
Adverse Reactions in Clinical Trials of a bioequivalent form of doxycycline hyclate capsules
In clinical trials of adult patients with periodontal disease 213 patients received 20 mg BID over a 9 to 12 month period. The most frequent adverse reactions occurring in studies involving treatment with a bioequivalent form of doxycycline hyclate capsules or placebo are listed below:
Incidence (%) of Adverse Reactions in Clinical Trials of Doxycycline Hyclate Capsules, 20 mg (Bioequivalent to Doxycycline Hyclate Tablets, 20 mg) vs. Placebo
| Adverse Reaction | Doxycycline Hyclate Capsules 20 mg BID (n=213) | Placebo (n=215) |
| Headache | 55 (26%) | 56 (26%) |
| Common Cold | 47 (22%) | 46 (21%) |
| Flu Symptoms | 24 (11%) | 40 (19%) |
| Tooth Ache | 14 (7%) | 28 (13%) |
| Periodontal Abscess | 8 (4%) | 21 (10%) |
| Tooth Disorder | 13 (6%) | 19 (9%) |
| Nausea | 17 (8%) | 12 (6%) |
| Sinusitis | 7 (3%) | 18 (8%) |
| Injury | 11 (5%) | 18 (8%) |
| Dyspepsia | 13 (6%) | 5 (2%) |
| Sore Throat | 11 (5%) | 13 (6%) |
| Joint Pain | 12 (6%) | 8 (4%) |
| Diarrhea | 12 (6%) | 8 (4%) |
| Sinus Congestion | 11 (5%) | 11 (5%) |
| Coughing | 9 (4%) | 11 (5%) |
| Sinus Headache | 8 (4%) | 8 (4%) |
| Rash | 8 (4%) | 6 (3%) |
| Back Pain | 7 (3%) | 8 (4%) |
| Back Ache | 4 (2%) | 9 (4%) |
| Menstrual Cramp | 9 (4%) | 5 (2%) |
| Acid Indigestion | 8 (4%) | 7 (3%) |
| Pain | 8 (4%) | 5 (2%) |
| Infection | 4 (2%) | 6 (3%) |
| Gum Pain | 1(<1%) | 6 (3%) |
| Bronchitis | 7 (3%) | 5 (2%) |
| Muscle Pain | 2 (1%) | 6 (3%) |
Note: Percentages are based on total number of study participants in each treatment group.
Adverse Reactions for Tetracyclines
The following adverse reactions have been observed in patients receiving tetracyclines:
Gastrointestinal: anorexia, nausea, vomiting, diarrhea, glossitis, dysphagia, enterocolitis, and inflammatory lesions (with vaginal candidiasis) in the anogenital region. Hepatotoxicity has been reported rarely. Rare instances of esophagitis and esophageal ulcerations have been reported in patients receiving the capsule forms of the drugs in the tetracycline class. Most of these patients took medications immediately before going to bed. (See DOSAGE AND ADMINISTRATION section).
Skin: maculopapular and erythematous rashes. Exfoliative dermatitis has been reported but is uncommon. Photosensitivity is discussed above. (See WARNINGS section). Renal toxicity: Rise in BUN has been reported and is apparently dose related. (See WARNINGS section). Hypersensitivity reactions: urticaria, angioneurotic edema, anaphylaxis, anaphylactoid purpura, serum sickness, pericarditis, and exacerbation of systemic lupus erythematosus. Blood: Hemolytic anemia, thrombocytopenia, neutropenia, and eosinophilia have been reported. Psychiatric: Depression, anxiety, suicidal ideation, insomnia, abnormal dreams, hallucination.
To report SUSPECTED ADVERSE REACTIONS, contact Alembic Pharmaceuticals Limited at 1-866-210-9797 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Because tetracyclines have been shown to depress plasma prothrombin activity, patients who are on anticoagulant therapy may require downward adjustment of their anticoagulant dosage. Since bacterial antibiotics, such as the tetracycline class of antibiotics, may interfere with the bactericidal action of members of the β-lactam (e.g. penicillin) class of antibiotics, it is not advisable to administer these antibiotics concomitantly. Absorption of tetracyclines is impaired by antacids containing aluminum, calcium, or magnesium, and iron-containing preparations, and by bismuth subsalicylate. Barbiturates, carbamazepine, and phenytoin decrease the half-life of doxycycline. The concurrent use of tetracycline and methoxyflurane has been reported to result in fatal renal toxicity. Concurrent use of tetracyclines may render oral contraceptives less effective.
DESCRIPTION
Doxycycline hyclate tablets, USP are available as a 20 mg formulation of doxycycline for oral administration. The structural formula of doxycycline hyclate is: 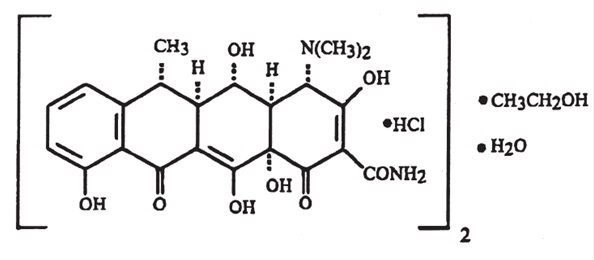 with an empirical formula of (C 22 H 24 N 2 O 8 •HCl) 2 •C 2 H 6 O•H 2 O and a molecular weight of 1025.89.
with an empirical formula of (C 22 H 24 N 2 O 8 •HCl) 2 •C 2 H 6 O•H 2 O and a molecular weight of 1025.89.
The chemical designation for doxycycline is 4-(dimethylamino)-1,4,4a,5,5a,6,11,12a–octahydro-3,5,10,12,12a–pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamide monohydrochloride, compound with ethyl alcohol (2:1), monohydrate.
Doxycycline hyclate, USP is a yellow to light-yellow powder which is freely soluble in water and in methanol; sparingly soluble in alcohol; practically insoluble in chloroform and in ether. It dissolves in aqueous solutions of alkali hydroxides and carbonates.
Inert ingredients in the formulation are: colloidal silicon dioxide, croscarmellose sodium, lactose anhydrous, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. Each tablet contains 23 mg of doxycycline hyclate equivalent to 20 mg of doxycycline.
Doxycycline hyclate tablets, USP meets USP Dissolution Test 3 .
CLINICAL PHARMACOLOGY
After oral administration, doxycycline hyclate is rapidly and nearly completely absorbed from the gastrointestinal tract. Doxycycline is eliminated with a half-life of approximately 18 hours by renal and fecal excretion of unchanged drug.
Mechanism of Action
Doxycycline has been shown to inhibit collagenase activity in vitro.1 Additional studies have shown that doxycycline reduces the elevated collagenase activity in the gingival crevicular fluid of patients with adult periodontitis. 2,3 The clinical significance of these findings is not known.
Microbiology
Doxycycline is a member of the tetracycline class of antibiotics. The dosage of doxycycline achieved with this product during administration is well below the concentration required to inhibit microorganisms commonly associated with adult periodontitis. Clinical studies with this product demonstrated no effect on total anaerobic and facultative bacteria in plaque samples from patients administered this dose regimen for 9 to 18 months.
This product should not be used for reducing the numbers of or eliminating those microorganisms associated with periodontitis.
Pharmacokinetics
The pharmacokinetics of doxycycline following oral administration of doxycycline hyclate tablets were investigated in 4 volunteer studies involving 107 adults. Additionally, doxycycline pharmacokinetics have been characterized in numerous scientific publications. 4 Pharmacokinetic parameters for doxycycline hyclate tablets following single oral doses and at steady-state in healthy subjects are presented as follows:
Pharmacokinetic Parameters for Doxycycline Hyclate Tablets
| n | C max • (ng/mL) | T max † (hr) | Cl/F • (L/hr) | t 1/2 • (hr) | |
| Single dose 20 mg (tablet) | 20 | 362 ± 101 | 1.4 (1 to 2.5) | 3.85 ± 1.3 | 18.1 ± 4.85 |
| Steady-State 20 mg BID ‡ | 30 | 790 ± 285 | 2 (0.98 to 12) | 3.76 ± 1.06 | Not Determined |
• Mean ± SD
† Mean and range
‡ Steady-State data were obtained from normal volunteers administered a bioequivalent formulation.
Absorption
Doxycycline is well absorbed after oral administration. In a single-dose study, concomitant administration of doxycycline hyclate tablets with a 1000 calorie, high-fat, high-protein meal which included dairy products, in healthy volunteers, resulted in a decrease in the rate and extent of absorption and delay in the time to maximum concentrations.
Distribution
Doxycycline is greater than 90% bound to plasma proteins. Its apparent volume of distribution is variously reported as between 52.6 and 134 L. 4,6
Metabolism
Major metabolites of doxycycline have not been identified. However, enzyme inducers such as barbiturates, carbamazepine, and phenytoin decrease the half-life of doxycycline.
Excretion
Doxycycline is excreted in the urine and feces as unchanged drug. It is variously reported that between 29% and 55.4% of an administered dose can be accounted for in the urine by 72 hours. 5,6 Half-life averaged 18 hours in subjects receiving a single 20 mg doxycycline dose.
Special Populations
Geriatric
Doxycycline pharmacokinetics have not been evaluated in geriatric patients.
Pediatric
Doxycycline pharmacokinetics have not been evaluated in pediatric patients (See WARNINGS section).
Gender
Doxycycline pharmacokinetics were compared in 9 men and 11 women under fed and fasted conditions. While female subjects had a higher rate (C max ) and extent of absorption (AUC), these differences are thought to be due to differences in body weight/lean body mass. Differences in other pharmacokinetic parameters were not significant.
Race
Differences in doxycycline pharmacokinetics among racial groups have not been evaluated.
Renal Insufficiency
Studies have shown no significant difference in serum half-life of doxycycline in patients with normal and severely impaired renal function. Hemodialysis does not alter the half-life of doxycycline.
Hepatic Insufficiency
Doxycycline pharmacokinetics have not been evaluated in patients with hepatic insufficiency.
Drug Interactions
(See PRECAUTIONS section)
Clinical Study
In a randomized, multi-centered, double-blind, 9-month Phase 3 study involving 190 adult patients with periodontal disease [at least two probing sites per quadrant of between 5 and 9 mm pocket depth (PD) and attachment level (ALv)], the effects of oral administration of 20 mg twice a day of doxycycline hyclate (using a bioequivalent capsule formulation) plus scaling and root planning (SRP) were compared to placebo control plus SRP. Both treatment groups were administered a course of scaling and root planing in 2 quadrants at Baseline. Measurements of ALv, PD and bleeding-on-probing (BOP) were obtained at Baseline, 3, 6, and 9 months from each site about each tooth in the two quadrants that received SRP using the UNC-15 manual probe. Each tooth site was categorized into one of three strata based on Baseline PD: 0 to 3 mm (no disease), 4 to 6 mm (mild/moderate disease), ≥ 7 mm (severe disease). For each stratum and treatment group, the following were calculated at month 3, 6, and 9: mean change in ALv from baseline, mean change in PD from baseline, mean percentage of tooth sites per patient exhibiting attachment loss of ≥ 2 mm from baseline, and percentage of tooth sites with bleeding on probing. The results are summarized in the following table.
Clinical Results at Nine Months of Doxycycline Hyclate Capsules, 20 mg, as an Adjunct to SRP (Bioequivalent to Doxycycline Hyclate Tablets, 20 mg)
| Parameter | Baseline Pocket Depth | ≥ 7 mm | |
| 0 to 3 mm | 4 to 6 mm | ||
| Number of Patients | |||
| (Doxycycline Hyclate Tablets 20 mg BID) | 90 | 90 | 79 |
| Number of Patients (Placebo) | 93 | 93 | 78 |
| Mean Gain (SD • ) in ALv † | |||
| Doxycycline Hyclate Tablets 20 mg BID | 0.25 (0.29) mm | 1.03 (0.47) mm ‡ | 1.55 (1.16) mm ‡ |
| Placebo | 0.2 (0.29) mm | 0.86 (0.48) mm | 1.17 (1.15) mm |
| Mean Decrease (SD • ) in PD § | |||
| Doxycycline Hyclate Tablets 20 mg BID | 0.16 (0.19) mm ¶ | 0.95 (0.47) mm ¶ | 1.68 (1.07) mm ¶ |
| Placebo | 0.05 (0.19) mm | 0.69 (0.48) mm | 1.2 (1.06) mm |
| % of Sites (SD • ) with loss of ALv † ≥ 2 mm | |||
| Doxycycline Hyclate Tablets 20 mg BID | 1.9 (4.2)% | 1.3 (4.5)% | 0.3 (9.4)% ‡ |
| Placebo | 2.2 (4.1)% | 2.4 (4.4)% | 3.6 (9.4)% |
| % of Sites (SD • ) with BOP # | |||
| Doxycycline Hyclate Tablets 20 mg BID | 39 (19)% ¶ | 64 (18)% ‡ | 75 (29)% |
| Placebo | 46 (19)% | 70 (18)% | 80 (29)% |
• SD=Standard Deviation
† ALv=Clinical Attachment Level
‡ p<0.05 vs. the placebo control group.
§ PD=Pocket Depth
¶ p<0.01 vs. the placebo control group.
# BOP=Bleeding on Probing
HOW SUPPLIED
Doxycycline hyclate tablets, USP are off white to pale yellow colored, mottled, round shape, biconvex, film-coated tablets debossed with “646” on one side and “L” on other side.
The tablets are available as:
NDC 62332-352-60 Bottle of 60 tablets
NDC 62332-352-31 Bottle of 100 tablets
NDC 62332-352-91 Bottle of 1000 tablets
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Dispense in a tight, light-resistant container using child-resistant closure as defined in the USP.