Get your patient on Enalapril Maleate - Enalapril Maleate tablet (Enalapril Maleate)
Enalapril Maleate - Enalapril Maleate tablet prescribing information
WARNING: FETAL TOXICITY See full prescribing information for complete boxed warning.
- When pregnancy is detected, discontinue enalapril as soon as possible.
- Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus. (See WARNINGS, Fetal Toxicity.)
INDICATIONS AND USAGE
Hypertension Enalapril maleate tablets, USP are indicated for the treatment of hypertension.
Enalapril maleate tablets, USP are effective alone or in combination with other antihypertensive agents, especially thiazide-type diuretics. The blood pressure lowering effects of enalapril maleate tablets, USP and thiazides are approximately additive.
Heart Failure Enalapril maleate tablets, USP are indicated for the treatment of symptomatic congestive heart failure, usually in combination with diuretics and digitalis. In these patients enalapril maleate tablets, USP improve symptoms, increase survival, and decrease the frequency of hospitalization (see CLINICAL PHARMACOLOGY, Heart Failure, Mortality Trials for details and limitations of survival trials).
Asymptomatic Left Ventricular Dysfunction In clinically stable asymptomatic patients with left ventricular dysfunction (ejection fraction ≤35 percent), enalapril maleate tablets, USP decrease the rate of development of overt heart failure and decrease the incidence of hospitalization for heart failure (see CLINICAL PHARMACOLOGY, Heart Failure, Mortality Trials for details and limitations of survival trials).
In using enalapril maleate tablets, USP consideration should be given to the fact that another angiotensin-converting enzyme inhibitor, captopril, has caused agranulocytosis, particularly in patients with renal impairment or collagen vascular disease, and that available data are insufficient to show that enalapril maleate tablets, USP do not have a similar risk (see WARNINGS, Neutropenia/Agranulocytosis ).
In considering use of enalapril maleate tablets, USP, it should be noted that in controlled clinical trials ACE inhibitors have an effect on blood pressure that is less in black patients than in non-blacks. In addition, it should be noted that black patients receiving ACE inhibitors have been reported to have a higher incidence of angioedema compared to nonblacks (see WARNINGS, Head and Neck Angioedema ).
DOSAGE AND ADMINISTRATION
Hypertension In patients who are currently being treated with a diuretic, symptomatic hypotension occasionally may occur following the initial dose of enalapril maleate. The diuretic should, if possible, be discontinued for two to three days before beginning therapy with enalapril maleate to reduce the likelihood of hypotension (see WARNINGS, Hypotension ). If the patients blood pressure is not controlled with enalapril maleate alone, diuretic therapy may be resumed.
If the diuretic cannot be discontinued an initial dose of 2.5 mg should be used under medical supervision for at least two hours and until blood pressure has stabilized for at least an additional hour (see WARNINGS, Hypotension and PRECAUTIONS, Drug Interactions ).
The recommended initial dose in patients not on diuretics is 5 mg once a day. Dosage should be adjusted according to blood pressure response. The usual dosage range is 10 to 40 mg per day administered in a single dose or two divided doses. In some patients treated once daily, the antihypertensive effect may diminish toward the end of the dosing interval. In such patients, an increase in dosage or twice daily administration should be considered. If blood pressure is not controlled with enalapril maleate alone, a diuretic may be added. Concomitant administration of enalapril maleate with potassium supplements, potassium salt substitutes, or potassium-sparing diuretics may lead to increases of serum potassium (see PRECAUTIONS ).
Dosage Adjustment in Hypertensive Patients with Renal Impairment
The usual dose of enalapril is recommended for patients with a creatinine clearance more than 30 mL/min (serum creatinine of up to approximately 3 mg/dL). For patients with creatinine clearance less than or equal to 30 mL/min (serum creatinine more than or equal to 3 mg/dL), the first dose is 2.5 mg once daily. The dosage may be titrated upward until blood pressure is controlled or to a maximum of 40 mg daily.
| Renal Status | Creatinine Clearance mL/min | Initial Dose mg/day |
| Normal Renal Function | >80 mL/min | 5 mg |
| Mild Impairment | ≤80 >30 mL/min | 5 mg |
| Moderate to Severe Impairment | ≤30 mL/min | 2.5 mg |
| Dialysis Patients 1 | - | 2.5 mg on dialysis days 2 |
1 See WARNINGS, Anaphylactoid Reactions during membrane exposure.
2 Dosage on nondialysis days should be adjusted depending on the blood pressure response.
Heart Failure
Enalapril maleate is indicated for the treatment of symptomatic heart failure, usually in combination with diuretics and digitalis. In the placebocontrolled studies that demonstrated improved survival, patients were titrated as tolerated up to 40 mg, administered in two divided doses.
The recommended initial dose is 2.5 mg. The recommended dosing range is 2.5 to 20 mg given twice a day. Doses should be titrated upward, as tolerated, over a period of a few days or weeks. The maximum daily dose administered in clinical trials was 40 mg in divided doses.
After the initial dose of enalapril maleate, the patient should be observed under medical supervision for at least two hours and until blood pressure has stabilized for at least an additional hour (see WARNINGS and PRECAUTIONS, Drug Interactions ). If possible, the dose of any concomitant diuretic should be reduced which may diminish the likelihood of hypotension. The appearance of hypotension after the initial dose of enalapril maleate does not preclude subsequent careful dose titration with the drug, following effective management of the hypotension.
Asymptomatic Left Ventricular Dysfunction In the trial that demonstrated efficacy, patients were started on 2.5 mg twice daily and were titrated as tolerated to the targeted daily dose of 20 mg (in divided doses).
After the initial dose of enalapril maleate, the patient should be observed under medical supervision for at least two hours and until blood pressure has stabilized for at least an additional hour (see WARNINGS and PRECAUTIONS, Drug Interactions ). If possible, the dose of any concomitant diuretic should be reduced which may diminish the likelihood of hypotension. The appearance of hypotension after the initial dose of enalapril maleate does not preclude subsequent careful dose titration with the drug, following effective management of the hypotension.
Dosage Adjustment in Patients with Heart Failure and Renal Impairment or Hyponatremia
In patients with heart failure who have hyponatremia (serum sodium less than 130 mEq/L) or with serum creatinine greater than 1.6 mg/dL, therapy should be initiated at 2.5 mg daily under close medical supervision (see DOSAGE AND ADMINISTRATION, Heart Failure, WARNINGS and PRECAUTIONS, Drug Interactions ). The dose may be increased to 2.5 mg b.i.d., then 5 mg b.i.d. and higher as needed, usually at intervals of four days or more if at the time of dosage adjustment there is not excessive hypotension or significant deterioration of renal function. The maximum daily dose is 40 mg.
Pediatric Hypertensive Patients The usual recommended starting dose is 0.08 mg/kg (up to 5 mg) once daily. Dosage should be adjusted according to blood pressure response. Doses above 0.58 mg/kg (or in excess of 40 mg) have not been studied in pediatric patients (see C LINICAL PHARMACOLOGY, Clinical Pharmacology in Pediatric Patients ).
Enalapril maleate is not recommended in neonates and in pediatric patients with glomerular filtration rate less than 30 mL/min/1.73 m 2 , as no data are available.
Preparation of Suspension (for 200 mL of a 1.0 mg/mL suspension)
Add 50 mL of sodium citrate and citric acid oral solution, USP to a polyethylene terephthalate (PET) bottle containing ten 20 mg enalapril maleate tablets and shake for at least 2 minutes. Let concentrate stand for 60 minutes. Following the 60-minute hold time, shake the concentrate for an additional minute. Add 150 mL of Ora-Sweet SF to the concentrate in the PET bottle and shake the suspension to disperse the ingredients. The suspension should be refrigerated at 2°C to 8°C (36°F to 46°F) and can be stored for up to 30 days. Shake the suspension before each use.
CONTRAINDICATIONS
Enalapril maleate is contraindicated in patients who are hypersensitive to this product and in patients with a history of angioedema related to previous treatment with an angiotensin-converting enzyme inhibitor and in patients with hereditary or idiopathic angioedema.
Do not coadminister aliskiren with enalapril maleate in patients with diabetes (see PRECAUTIONS, Drug Interactions ).
Enalapril maleate is contraindicated in combination with a neprilysin inhibitor (e.g., sacubitril). Do not administer enalapril maleate within 36 hours of switching to or from sacubitril/valsartan, a neprilysin inhibitor (see WARNINGS, Head and Neck Angioedema ).
ADVERSE REACTIONS
Enalapril maleate has been evaluated for safety in more than 10,000 patients, including over 1000 patients treated for one year or more. Enalapril maleate has been found to be generally well tolerated in controlled clinical trials involving 2987 patients. For the most part, adverse experiences were mild and transient in nature. In clinical trials, discontinuation of therapy due to clinical adverse experiences was required in 3.3 percent of patients with hypertension and in 5.7 percent of patients with heart failure. The frequency of adverse experiences was not related to total daily dosage within the usual dosage ranges. In patients with hypertension the overall percentage of patients treated with enalapril maleate reporting adverse experiences was comparable to placebo.
Hypertension
Adverse experiences occurring in greater than one percent of patients with hypertension treated with enalapril maleate in controlled clinical trials are shown below. In patients treated with enalapril maleate, the maximum duration of therapy was three years; in placebo-treated patients the maximum duration of therapy was 12 weeks.
Enalapril Maleate (n = 2314) Incidence (discontinuation) | Placebo (n = 230) Incidence | |
Body As A Whole Fatigue Orthostatic EffectsAsthenia | 3.0 (<0.1) 1.2 (<0.1) 1.1 (0.1) | 2.6 0.0 0.9 |
| Digestive Diarrhea Nausea | 1.4 (<0.1) 1.4 (0.2) | 1.7 1.7 |
| Nervous/Psychiatric Headache Dizziness | 5.2 (0.3) 4.3 (0.4) | 9.1 4.3 |
| Respiratory Cough | 1.3 (0.1) | 0.9 |
| Skin Rash | 1.4 (0.4) | 0.4 |
Heart Failure Adverse experiences occurring in greater than one percent of patients with heart failure treated with enalapril maleate are shown below. The incidences represent the experiences from both controlled and uncontrolled clinical trials (maximum duration of therapy was approximately one year). In the placebo-treated patients, the incidences reported are from the controlled trials (maximum duration of therapy is 12 weeks). The percentage of patients with severe heart failure (NYHA Class IV) was 29 percent and 43 percent for patients treated with enalapril maleate and placebo, respectively.
Drug Interactions
Neprilysin Inhibitors Patients taking concomitant neprilysin inhibitors may be at increased risk for angioedema (see WARNINGS ).
Dual Blockade of the Renin-Angiotensin System (RAS)
Dual blockade of the RAS with angiotensin receptor blockers, ACE inhibitors, or aliskiren is associated with increased risks of hypotension, hyperkalemia, and changes in renal function (including acute renal failure) compared to monotherapy. Most patients receiving the combination of two RAS inhibitors do not obtain any additional benefit compared to monotherapy. In general, avoid combined use of RAS inhibitors. Closely monitor blood pressure, renal function, and electrolytes in patients on enalapril maleate and other agents that affect the RAS.
Do not coadminister aliskiren with enalapril maleate in patients with diabetes. Avoid use of aliskiren with enalapril maleate in patients with renal impairment (GFR <60 mL/min).
Hypotension — Patients on Diuretic Therapy Patients on diuretics and especially those in whom diuretic therapy was recently instituted, may occasionally experience an excessive reduction of blood pressure after initiation of therapy with enalapril. The possibility of hypotensive effects with enalapril can be minimized by either discontinuing the diuretic or increasing the salt intake prior to initiation of treatment with enalapril. If it is necessary to continue the diuretic, provide close medical supervision after the initial dose for at least two hours and until blood pressure has stabilized for at least an additional hour (see WARNINGS, Hypotension and DOSAGE AND ADMINISTRATION ).
Agents Causing Renin Release The antihypertensive effect of enalapril maleate is augmented by antihypertensive agents that cause renin release (e.g., diuretics).
Nonsteroidal Anti-Inflammatory Agents Including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors)
In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, coadministration of NSAIDs, including selective COX-2 inhibitors, with ACE inhibitors, including enalapril, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically in patients receiving enalapril and NSAID therapy.
In a clinical pharmacology study, indomethacin or sulindac was administered to hypertensive patients receiving enalapril maleate. In this study there was no evidence of a blunting of the antihypertensive action of enalapril maleate. However, reports suggest that NSAIDs may diminish the antihypertensive effect of ACE inhibitors.
Other Cardiovascular Agents Enalapril maleate has been used concomitantly with beta adrenergicblocking agents, methyldopa, nitrates, calcium-blocking agents, hydralazine, prazosin and digoxin without evidence of clinically significant adverse interactions.
Agents Increasing Serum Potassium Enalapril maleate attenuates potassium loss caused by thiazide-type diuretics. Potassium-sparing diuretics (e.g., spironolactone, triamterene, or amiloride), potassium supplements, or potassium-containing salt substitutes may lead to significant increases in serum potassium. Therefore, if concomitant use of these agents is indicated because of demonstrated hypokalemia, they should be used with caution and with frequent monitoring of serum potassium. Potassium-sparing agents should generally not be used in patients with heart failure receiving enalapril maleate.
Lithium Lithium toxicity has been reported in patients receiving lithium concomitantly with drugs which cause elimination of sodium, including ACE inhibitors. A few cases of lithium toxicity have been reported in patients receiving concomitant enalapril maleate and lithium and were reversible upon discontinuation of both drugs. It is recommended that serum lithium levels be monitored frequently if enalapril is administered concomitantly with lithium.
Gold Nitritoid reactions (symptoms include facial flushing, nausea, vomiting and hypotension) have been reported rarely in patients on therapy with injectable gold (sodium aurothiomalate) and concomitant ACE inhibitor therapy including enalapril maleate.
mTOR (Mammalian Target of Rapamycin) Inhibitors Patients receiving coadministration of ACE inhibitor and mTOR inhibitor (e.g., temsirolimus, sirolimus, everolimus) therapy may be at increased risk for angioedema (see WARNINGS, Head and Neck Angioedema ).
DESCRIPTION
Enalapril maleate, USP is the maleate salt of enalapril, the ethyl ester of a long-acting angiotensin-converting enzyme inhibitor, enalaprilat. Enalapril maleate is chemically described as (S)-1-[ N -[1-(ethoxycarbonyl)-3-phenylpropyl]-L-alanyl]-L-proline, ( Z )-2-butenedioate salt (1:1). Its molecular formula is C 20 H 28 N 2 O 5 •C 4 H 4 O 4 , and its structural formula is: 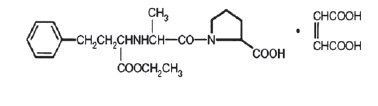
Enalapril maleate, USP is a white to off-white, crystalline powder with molecular weight of 492.53 g/mol. It is sparingly soluble in water, soluble in ethanol, and freely soluble in methanol.
Enalapril is a pro-drug; following oral administration, it is bioactivated by hydrolysis of the ethyl ester to enalaprilat, which is the active angiotensin-converting enzyme inhibitor.
Enalapril maleate is supplied as 2.5 mg, 5 mg, 10 mg, and 20 mg tablets for oral administration. In addition to the active ingredient enalapril maleate, each tablet contains the following inactive ingredients: anhydrous lactose and zinc stearate.
CLINICAL PHARMACOLOGY
Mechanism of Action
Enalapril, after hydrolysis to enalaprilat, inhibits angiotensinconverting enzyme (ACE) in human subjects and animals. ACE is a peptidyl dipeptidase that catalyzes the conversion of angiotensin I to the vasoconstrictor substance, angiotensin II. Angiotensin II also stimulates aldosterone secretion by the adrenal cortex. The beneficial effects of enalapril in hypertension and heart failure appear to result primarily from suppression of the renin-angiotensin-aldosterone system. Inhibition of ACE results in decreased plasma angiotensin II, which leads to decreased vasopressor activity and to decreased aldosterone secretion. Although the latter decrease is small, it results in small increases of serum potassium. In hypertensive patients treated with enalapril maleate alone for up to 48 weeks, mean increases in serum potassium of approximately 0.2 mEq/L were observed. In patients treated with enalapril maleate plus a thiazide diuretic, there was essentially no change in serum potassium (see PRECAUTIONS ). Removal of angiotensin II negative feedback on renin secretion leads to increased plasma renin activity.
ACE is identical to kininase, an enzyme that degrades bradykinin. Whether increased levels of bradykinin, a potent vasodepressor peptide, play a role in the therapeutic effects of enalapril maleate remains to be elucidated.
While the mechanism through which enalapril maleate lowers blood pressure is believed to be primarily suppression of the reninangiotensin-aldosterone system, enalapril maleate is antihypertensive even in patients with low-renin hypertension. Although enalapril maleate was antihypertensive in all races studied, black hypertensive patients (usually a low-renin hypertensive population) had a smaller average response to enalapril monotherapy than non-black patients.
Pharmacokinetics and Metabolism
Following oral administration of enalapril maleate, peak serum concentrations of enalapril occur within about one hour. Based on urinary recovery, the extent of absorption of enalapril is approximately 60 percent. Enalapril absorption is not influenced by the presence of food in the gastrointestinal tract. Following absorption, enalapril is hydrolyzed to enalaprilat, which is a more potent angiotensin-converting enzyme inhibitor than enalapril; enalaprilat is poorly absorbed when administered orally. Peak serum concentrations of enalaprilat occur three to four hours after an oral dose of enalapril maleate. Excretion of enalapril maleate is primarily renal. Approximately 94 percent of the dose is recovered in the urine and feces as enalaprilat or enalapril. The principal components in urine are enalaprilat, accounting for about 40 percent of the dose, and intact enalapril. There is no evidence of metabolites of enalapril, other than enalaprilat.
The serum concentration profile of enalaprilat exhibits a prolonged terminal phase, apparently representing a small fraction of the administered dose that has been bound to ACE. The amount bound does not increase with dose, indicating a saturable site of binding. The effective half-life for accumulation of enalaprilat following multiple doses of enalapril maleate is 11 hours. The disposition of enalapril and enalaprilat in patients with renal insufficiency is similar to that in patients with normal renal function until the glomerular filtration rate is 30 mL/min or less. With glomerular filtration rate ≤30 mL/min, peak and trough enalaprilat levels increase, time to peak concentration increases and time to steady state may be delayed. The effective half-life of enalaprilat following multiple doses of enalapril maleate is prolonged at this level of renal insufficiency (see DOSAGE AND ADMINISTRATION ). Enalaprilat is dialyzable at the rate of 62 mL/min.
Studies in dogs indicate that enalapril crosses the blood-brain barrier poorly, if at all; enalaprilat does not enter the brain. Multiple doses of enalapril maleate in rats do not result in accumulation in any tissues. Milk of lactating rats contains radioactivity following administration of 14 C-enalapril maleate. Radioactivity was found to cross the placenta following administration of labeled drug to pregnant hamsters.
Pharmacodynamics and Clinical Effects Hypertension Administration of enalapril maleate to patients with hypertension of severity ranging from mild to severe results in a reduction of both supine and standing blood pressure usually with no orthostatic component. Symptomatic postural hypotension is therefore infrequent, although it might be anticipated in volume-depleted patients (see WARNINGS, Hypotension ).
In most patients studied, after oral administration of a single dose of enalapril, onset of antihypertensive activity was seen at one hour with peak reduction of blood pressure achieved by four to six hours.
At recommended doses, antihypertensive effects have been maintained for at least 24 hours. In some patients the effects may diminish toward the end of the dosing interval (see DOSAGE AND ADMINISTRATION, Hypertension ).
In some patients achievement of optimal blood pressure reduction may require several weeks of therapy.
The antihypertensive effects of enalapril maleate have continued during long-term therapy. Abrupt withdrawal of enalapril maleate has not been associated with a rapid increase in blood pressure.
In hemodynamic studies in patients with essential hypertension, blood pressure reduction was accompanied by a reduction in peripheral arterial resistance with an increase in cardiac output and little or no change in heart rate. Following administration of enalapril maleate, there is an increase in renal blood flow; glomerular filtration rate is usually unchanged. The effects appear to be similar in patients with renovascular hypertension.
When given together with thiazide-type diuretics, the blood pressure lowering effects of enalapril maleate are approximately additive.
In a clinical pharmacology study, indomethacin or sulindac was administered to hypertensive patients receiving enalapril maleate. In this study there was no evidence of a blunting of the antihypertensive action of enalapril maleate (see PRECAUTIONS, Drug Interactions ).
Heart Failure In trials in patients treated with digitalis and diuretics, treatment with enalapril resulted in decreased systemic vascular resistance, blood pressure, pulmonary capillary wedge pressure and heart size, and increased cardiac output and exercise tolerance. Heart rate was unchanged or slightly reduced, and mean ejection fraction was unchanged or increased. There was a beneficial effect on severity of heart failure as measured by the New York Heart Association (NYHA) classification and on symptoms of dyspnea and fatigue. Hemodynamic effects were observed after the first dose and appeared to be maintained in uncontrolled studies lasting as long as four months. Effects on exercise tolerance, heart size, and severity and symptoms of heart failure were observed in placebo-controlled studies lasting from eight weeks to over one year.
HOW SUPPLIED
Enalapril maleate tablets, USP are supplied as follows:
2.5 mg tablets: White, round, biconvex tablet, debossed with “333” on one side and plain with a bisect on the other side; supplied in
Bottles of 100: NDC 50228-228-01 Bottles of 1000: NDC 50228-228-10
5 mg tablets: White, round, biconvex tablet, debossed with “334” on one side and plain with a bisect on the other side; supplied in
Bottles of 100: NDC 50228-229-01 Bottles of 1000: NDC 50228-229-10
10 mg tablets: White, round, biconvex tablet, debossed with “335” on one side and plain with a bisect on the other side; supplied in Bottles of 100: NDC 50228-230-01 Bottles of 1000: NDC 50228-230-10
20 mg tablets: White, round, biconvex tablet, debossed with “336” on one side and plain with a bisect on the other side; supplied in Bottles of 100: NDC 50228-231-01 Bottles of 1000: NDC 50228-231-10
Storage Store at 20°C to 25°C (68°F to 77°F); excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Keep container tightly closed. Protect from moisture.
Dispense in a tight container as per USP, if product package is subdivided.
Dispense the Prescribing Information at: https://sciegenpharm.com/medication-guide/
Manufactured by A&Z Pharmaceutical, Inc., 180 Oser Ave. Suite 300 Hauppauge, NY 11788
Manufactured for ScieGen Pharmaceuticals, Inc. Hauppauge, NY 11788