Get your patient on Fluphenazine Hydrochloride - Fluphenazine Hydrochloride injection, Solution (Fluphenazine Hydrochloride)
Fluphenazine Hydrochloride - Fluphenazine Hydrochloride injection, Solution prescribing information
WARNING
Increased Mortality in Elderly Patients with Dementia-Related Psychosis –
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical, antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Fluphenazine Hydrochloride Injection, USP is not approved for the treatment of patients with dementia-related psychosis (see WARNINGS ).
INDICATIONS AND USAGE:
Fluphenazine Hydrochloride Injection, USP is indicated in the management of manifestations of psychotic disorders.
Fluphenazine hydrochloride has not been shown effective in the management of behavioral complications in patients with mental retardation.
DOSAGE AND ADMINISTRATION
The average well tolerated starting dose for adult psychotic patients is 1.25 mg (0.5 mL) intramuscularly. Depending on the severity and duration of symptoms, initial total daily dosage may range from 2.5 to 10 mg and should be divided and given at six to eight hour intervals.
The smallest amount that will produce the desired results must be carefully determined for each individual, since optimal dosage levels of this potent drug vary from patient to patient. In general, the parenteral dose for fluphenazine has been found to be approximately 1/3 to 1/2 the oral dose. Treatment may be instituted with a low initial dosage, which may be increased, if necessary, until the desired clinical effects are achieved. Dosages exceeding 10 mg daily should be used with caution.
When symptoms are controlled, oral maintenance therapy can generally be instituted often with single daily doses. Continued treatment by the oral route, if possible, is needed to achieve maximum therapeutic benefits; further adjustments in dosage may be necessary during the course of therapy to meet the patient’s requirements.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS:
Phenothiazines are contraindicated in patients with suspected or established subcortical brain damage, in patients receiving large doses of hypnotics and in comatose or severely depressed states. The presence of blood dyscrasia or liver damage precludes the use of fluphenazine hydrochloride. Fluphenazine hydrochloride is contraindicated in patients who have shown hypersensitivity to fluphenazine; cross-sensitivity to phenothiazine derivatives may occur.
ADVERSE REACTIONS:
Central Nervous System
The side effects most frequently reported with phenothiazine compounds are extrapyramidal symptoms including pseudoparkinsonism, dystonia, dyskinesia, akathisia, oculogyric crises, opisthotonos and hyperreflexia. Most often these extrapyramidal symptoms are reversible; however, they may be persistent (see below). With any given phenothiazine derivative, the incidence and severity of such reactions depend more on individual patient sensitivity than on other factors, but dosage level and patient age are also determinants.
Extrapyramidal reactions may be alarming, and the patient should be forewarned and reassured. These reactions can usually be controlled by administration of antiparkinsonian drugs such as Benztropine Mesylate or Intravenous Caffeine and Sodium Benzoate Injection, and by subsequent reductions in dosage.
Dystonia
Class effect: Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Tardive Dyskinesia
See WARNINGS . The syndrome is characterized by involuntary choreoathetoid movements which variously involve the tongue, face, mouth, lips or jaw (e.g., protrusion of the tongue, puffing of cheeks, puckering of the mouth, chewing movements), trunk and extremities. The severity of the syndrome and the degree of impairment produced vary widely.
The syndrome may become clinically recognizable either during treatment, upon dosage reduction, or upon withdrawal of treatment. Early detection of tardive dyskinesia is important. To increase the likelihood of detecting the syndrome at the earliest possible time, the dosage of the neuroleptic drug should be reduced periodically (if clinically possible) and the patient observed for signs of the disorder. This maneuver is critical, since neuroleptic drugs may mask the signs of the syndrome.
Other Central Nervous System Effects
Occurrences of neuroleptic malignant syndrome (NMS) have been reported in patients on neuroleptic therapy (see WARNINGS , Neuroleptic Malignant Syndrome ); leukocytosis, elevated CPK, liver function abnormalities and acute renal failure may also occur with NMS.
Drowsiness or lethargy, if they occur, may necessitate a reduction in dosage; the induction of a catatonic-like state has been known to occur with dosages of fluphenazine far in excess of the recommended amounts. As with other phenothiazine compounds, reactivation or aggravation of psychotic processes may be encountered.
Phenothiazine derivatives have been known to cause, in some patients, restlessness, excitement or bizarre dreams.
Autonomic Nervous System
Hypertension and fluctuation in blood pressure have been reported with fluphenazine hydrochloride.
Hypotension has rarely presented a problem with fluphenazine. However, patients with pheochromocytoma, cerebral vascular or renal insufficiency or a severe cardiac reserve deficiency such as mitral insufficiency appear to be particularly prone to hypotensive reactions with phenothiazine compounds, and should therefore be observed closely when the drug is administered. If severe hypotension should occur, supportive measures including the use of intravenous vasopressor drugs should be instituted immediately. Norepinephrine Bitartrate Injection is the most suitable drug for this purpose; epinephrine should not be used since phenothiazine derivatives have been found to reverse its action, resulting in a further lowering of blood pressure.
Autonomic reactions including nausea and loss of appetite, salivation, polyuria, perspiration, dry mouth, headache and constipation may occur. Autonomic effects can usually be controlled by reducing or temporarily discontinuing dosage.
In some patients, phenothiazine derivatives have caused blurred vision, glaucoma, bladder paralysis, fecal impaction, paralytic ileus, tachycardia or nasal congestion.
Metabolic and Endocrine
Weight change, peripheral edema, abnormal lactation, gynecomastia, menstrual irregularities, false results on pregnancy tests, impotency in men and increased libido in women have all been known to occur in some patients on phenothiazine therapy.
Allergic Reactions
Skin disorders such as itching, erythema, urticaria, seborrhea, photosensitivity, eczema and even exfoliative dermatitis have been reported with phenothiazine derivatives. The possibility of anaphylactoid reactions occurring in some patients should be borne in mind.
Hematologic
Routine blood counts are advisable during therapy since blood dyscrasias including leukopenia, agranulocytosis, thrombocytopenic or nonthrombocytopenic purpura, eosinophilia and pancytopenia have been observed with phenothiazine derivatives. Furthermore, if any soreness of the mouth, gums or throat, or any symptoms of upper respiratory infection occur and confirmatory leukocyte count indicates cellular depression, therapy should be discontinued and other appropriate measures instituted immediately.
Hepatic
Liver damage as manifested by cholestatic jaundice may be encountered, particularly during the first months of therapy; treatment should be discontinued if this occurs. An increase in cephalin flocculation, sometimes accompanied by alterations in other liver function tests, have been reported in patients receiving fluphenazine hydrochloride who have had no clinical evidence of liver damage.
Others
Sudden, unexpected and unexplained deaths have been reported in hospitalized psychotic patients receiving phenothiazines. Previous brain damage or seizures may be predisposing factors; high doses should be avoided in known seizure patients. Several patients have shown sudden flare-ups of psychotic behavior patterns shortly before death. Autopsy findings have usually revealed acute fulminating pneumonia or pneumonitis, aspiration of gastric contents or intramyocardial lesions.
Although this is not a general feature of fluphenazine, potentiation of CNS depressants (opiates, analgesics, antihistamines, barbiturates, alcohol) may occur.
The following adverse reactions have also occurred with phenothiazine derivatives; systemic lupus erythematosus-like syndrome; hypotension severe enough to cause fatal cardiac arrest; altered electrocardiographic and electroencephalographic tracings; altered cerebrospinal fluid proteins; cerebral edema; asthma, laryngeal edema and angioneurotic edema; with long-term use—skin pigmentation and lenticular and corneal opacities.
DESCRIPTION:
Fluphenazine Hydrochloride Injection, USP is a sterile, nonpyrogenic solution of fluphenazine hydrochloride in Water for Injection, for intramuscular use for the management of schizophrenia. Fluphenazine hydrochloride is a trifluoromethyl phenothiazine derivative and the chemical name is 1-Piperazineethanol, 4-[3-[2-(trifluoromethyl)-10H-phenothiazin-10-yl] propyl]-,dihydrochloride and has the following structural formula:
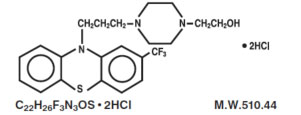
Each mL contains: Fluphenazine hydrochloride 2.5 mg; sodium chloride 9 mg to render the solution isotonic; methylparaben 1 mg and propylparaben 0.1 mg as preservatives; Water for Injection q.s. Hydrochloric acid and/or sodium hydroxide may have been added for pH adjustment (4.8 to 5.2).
CLINICAL PHARMACOLOGY:
Fluphenazine hydrochloride has activity at all levels of the central nervous system (CNS) as well as on multiple organ systems. The mechanism whereby its therapeutic action is exerted is unknown.
HOW SUPPLIED:
| Product No. | NDC No. | |
| 28110 | 63323-281-10 | Fluphenazine Hydrochloride Injection, USP 25 mg per 10 mL (2.5 mg per mL) flip-top vial packaged individually. |
Solutions should be protected from exposure to light. Parenteral solutions may vary in color from essentially colorless to light amber. If a solution has become any darker than light amber or is discolored in any other way, it should not be used.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
PROTECT FROM LIGHT.
The container closure is not made with natural rubber latex.
Fluphenazine Hydrochloride - Fluphenazine Hydrochloride injection, Solution PubMed™ news
- Journal Article • 2026 AprBeyond medication choice: Predictors of psychiatric readmission after inpatient initiation of long-acting injectable antipsychotics.
- Journal Article • 2026 AprExtrapyramidal and anticholinergic reactions in an adolescent patient: a case for pharmacogenetic consideration.
- Journal Article • 2026 AprCongenital malformation risk following prenatal antipsychotic exposure: a systematic safety surveillance approach.
- Journal Article • 2026 MarNovel 8-trifluoromethylquinobenzothiazines-Synthesis and Evaluation for Antiproliferative and Antibacterial Activity.
- Journal Article • 2026 MarThe multifunctional RNA helicase DDX39A drives glioblastoma progression by modulating WISP1 alternative splicing that induces an immunosuppressive macrophage polarization.