Get your patient on Granisetron Hydrochloride - Granisetron Hydrochloride tablet (Granisetron Hydrochloride)
Granisetron Hydrochloride - Granisetron Hydrochloride tablet prescribing information
INDICATIONS AND USAGE
Granisetron hydrochloride tablets USP are indicated for the prevention of:
Nausea and vomiting associated with initial and repeat courses of emetogenic cancer therapy, including high-dose cisplatin.
Nausea and vomiting associated with radiation, including total body irradiation and fractionated abdominal radiation.
DOSAGE AND ADMINISTRATION
Emetogenic Chemotherapy
The recommended adult dosage of oral granisetron hydrochloride tablets USP is 2 mg once daily or 1 mg twice daily. In the 2 mg once-daily regimen, two 1 mg tablets are given up to 1 hour before chemotherapy. In the 1 mg twice-daily regimen, the first 1 mg tablet is given up to 1 hour before chemotherapy, and the second tablet, 12 hours after the first. Either regimen is administered only on the day(s) chemotherapy is given. Continued treatment, while not on chemotherapy, has not been found to be useful.
Use in the Elderly, Renal Failure Patients or Hepatically Impaired Patients
No dosage adjustment is recommended (see CLINICAL PHARMACOLOGY, Pharmacokinetics ) .
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Radiation (Either Total Body Irradiation or Fractionated Abdominal Radiation)
The recommended adult dosage of oral granisetron hydrochloride tablets USP is 2 mg once daily.
Two 1 mg tablets are taken within 1 hour of radiation.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Use in the Elderly
No dosage adjustment is recommended.
CONTRAINDICATIONS
Granisetron hydrochloride tablets are contraindicated in patients with known hypersensitivity to the drug or any of its components.
ADVERSE REACTIONS
QT prolongation has been reported with granisetron hydrochloride (see PRECAUTIONS and Drug Interactions ) .
Chemotherapy-Induced Nausea and Vomiting
Over 3700 patients have received granisetron hydrochloride tablets in clinical trials with emetogenic cancer therapies consisting primarily of cyclophosphamide or cisplatin regimens.
In patients receiving granisetron hydrochloride tablets 1 mg bid for 1, 7 or 14 days, or 2 mg daily for 1 day, adverse experiences reported in more than 5% of the patients with comparator and placebo incidences are listed in Table 4 .
| Percent of Patients With Event | ||||
|---|---|---|---|---|
| Granisetron Hydrochloride Tablets Adverse events were recorded for 7 days when granisetron hydrochloride tablets were given on a single day and for up to 28 days when granisetron hydrochloride tablets were administered for 7 or 14 days. 1 mg twice a day (N = 978) | Granisetron Hydrochloride Tablets 2 mg once a day (N = 1450) | Comparator Metoclopramide/dexamethasone; phenothiazines/dexamethasone; dexamethasone alone; prochlorperazine. (N = 599) | Placebo (N = 185) | |
| Headache Usually mild to moderate in severity. | 21% | 20% | 13% | 12% |
| Constipation | 18% | 14% | 16% | 8% |
| Asthenia | 14% | 18% | 10% | 4% |
| Diarrhea | 8% | 9% | 10% | 4% |
| Abdominal Pain | 6% | 4% | 6% | 3% |
| Dyspepsia | 4% | 6% | 5% | 4% |
Other adverse events reported in clinical trials were:
Gastrointestinal: In single-day dosing studies in which adverse events were collected for 7 days, nausea (20%) and vomiting (12%) were recorded as adverse events after the 24 hour efficacy assessment period.
Hepatic: In comparative trials, elevation of AST and ALT (> 2 times the upper limit of normal) following the administration of granisetron hydrochloride tablets occurred in 5% and 6% of patients, respectively. These frequencies were not significantly different from those seen with comparators (AST: 2%; ALT: 9%).
Cardiovascular: Hypertension (1%); hypotension, angina pectoris, atrial fibrillation, and syncope have been observed rarely.
Central Nervous System: Dizziness (5%), insomnia (5%), anxiety (2%), somnolence (1%). One case compatible with, but not diagnostic of, extrapyramidal symptoms has been reported in a patient treated with granisetron hydrochloride tablets.
Hypersensitivity: Rare cases of hypersensitivity reactions, sometimes severe (e.g., anaphylaxis, shortness of breath, hypotension, urticaria) have been reported.
Other: Fever (5%). Events often associated with chemotherapy also have been reported: leukopenia (9%), decreased appetite (6%), anemia (4%), alopecia (3%), thrombocytopenia (2%).
Over 5000 patients have received injectable granisetron hydrochloride in clinical trials.
Table 5 gives the comparative frequencies of the five commonly reported adverse events (≥ 3%) in patients receiving granisetron hydrochloride injection, 40 mcg/kg, in single-day chemotherapy trials. These patients received chemotherapy, primarily cisplatin, and intravenous fluids during the 24 hour period following granisetron hydrochloride injection administration.
| Percent of Patients with Event | ||
|---|---|---|
| Granisetron Hydrochloride Injection Adverse events were generally recorded over 7 days post-granisetron hydrochloride injection administration. 40 mcg/kg (N = 1268) | Comparator Metoclopramide/dexamethasone and phenothiazines/dexamethasone. (N = 422) | |
| Headache | 14% | 6% |
| Asthenia | 5% | 6% |
| Somnolence | 4% | 15% |
| Diarrhea | 4% | 6% |
| Constipation | 3% | 3% |
In the absence of a placebo group, there is uncertainty as to how many of these events should be attributed to granisetron hydrochloride, except for headache, which was clearly more frequent than in comparison groups.
Radiation-Induced Nausea and Vomiting
In controlled clinical trials, the adverse events reported by patients receiving granisetron hydrochloride tablets and concurrent radiation were similar to those reported by patients receiving granisetron hydrochloride tablets prior to chemotherapy. The most frequently reported adverse events were diarrhea, asthenia, and constipation. Headache, however, was less prevalent in this patient population.
Postmarketing Experience
QT prolongation has been reported with granisetron hydrochloride (see PRECAUTIONS and Drug Interactions ) .
Drug Interactions
Granisetron does not induce or inhibit the cytochrome P-450 drug-metabolizing enzyme system in vitro . There have been no definitive drug-drug interaction studies to examine pharmacokinetic or pharmacodynamic interaction with other drugs; however, in humans, granisetron hydrochloride injection has been safely administered with drugs representing benzodiazepines, neuroleptics, and anti-ulcer medications commonly prescribed with antiemetic treatments. Granisetron hydrochloride injection also does not appear to interact with emetogenic cancer chemotherapies. Because granisetron is metabolized by hepatic cytochrome P-450 drug-metabolizing enzymes, inducers or inhibitors of these enzymes may change the clearance and, hence, the half-life of granisetron. No specific interaction studies have been conducted in anesthetized patients. In addition, the activity of the cytochrome P-450 subfamily 3A4 (involved in the metabolism of some of the main narcotic analgesic agents) is not modified by granisetron in vitro .
In in vitro human microsomal studies, ketoconazole inhibited ring oxidation of granisetron. However, the clinical significance of in vivo pharmacokinetic interactions with ketoconazole is not known. In a human pharmacokinetic study, hepatic enzyme induction with phenobarbital resulted in a 25% increase in total plasma clearance of intravenous granisetron. The clinical significance of this change is not known.
QT prolongation has been reported with granisetron hydrochloride. Use of granisetron hydrochloride in patients concurrently treated with drugs known to prolong the QT interval and/or are arrhythmogenic may result in clinical consequences.
Serotonin syndrome (including altered mental status, autonomic instability, and neuromuscular symptoms) has been described following the concomitant use of 5-HT 3 receptor antagonists and other serotonergic drugs, including selective serotonin reuptake inhibitors (SSRIs) and serotonin and noradrenaline reuptake inhibitors (SNRIs) (see WARNINGS ) .
DESCRIPTION
Granisetron hydrochloride tablets USP contain granisetron hydrochloride USP, an antinauseant and antiemetic agent. Chemically it is endo -N-(9-methyl-9-azabicyclo [3.3.1] non-3-yl)-1-methyl-1H-indazole-3-carboxamide hydrochloride.
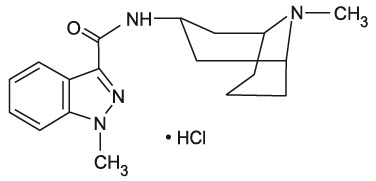
Granisetron hydrochloride is a white to off-white crystalline powder that is freely soluble in water and slightly soluble in methanol.
Tablets for Oral Administration
Each white, triangular, biconvex, film-coated granisetron hydrochloride tablet USP contains 1.12 mg granisetron hydrochloride equivalent to granisetron, 1 mg. Inactive ingredients are: hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, and titanium dioxide.
CLINICAL PHARMACOLOGY
Granisetron is a selective 5-hydroxytryptamine 3 (5-HT 3 ) receptor antagonist with little or no affinity for other serotonin receptors, including 5-HT 1 ; 5-HT 1A ; 5-HT 1B/C ; 5-HT 2 ; for alpha 1- , alpha 2- , or beta-adrenoreceptors; for dopamine-D 2 ; or for histamine-H 1 ; benzodiazepine; picrotoxin or opioid receptors.
Serotonin receptors of the 5-HT 3 type are located peripherally on vagal nerve terminals and centrally in the chemoreceptor trigger zone of the area postrema. During chemotherapy that induces vomiting, mucosal enterochromaffin cells release serotonin, which stimulates 5-HT 3 receptors. This evokes vagal afferent discharge, inducing vomiting. Animal studies demonstrate that, in binding to 5-HT 3 receptors, granisetron blocks serotonin stimulation and subsequent vomiting after emetogenic stimuli such as cisplatin. In the ferret animal model, a single granisetron injection prevented vomiting due to high-dose cisplatin or arrested vomiting within 5 to 30 seconds.
In most human studies, granisetron has had little effect on blood pressure, heart rate or ECG. No evidence of an effect on plasma prolactin or aldosterone concentrations has been found in other studies.
Following single and multiple oral doses, granisetron hydrochloride tablets slowed colonic transit in normal volunteers. However, granisetron hydrochloride had no effect on oro-cecal transit time in normal volunteers when given as a single intravenous (IV) infusion of 50 mcg/kg or 200 mcg/kg.
Pharmacokinetics
In healthy volunteers and adult cancer patients undergoing chemotherapy, administration of granisetron hydrochloride tablets produced mean pharmacokinetic data shown in Table 1 .
| Peak Plasma Concentration (ng/mL) | Terminal Phase Plasma Half-Life (h) | Volume of Distribution (L/kg) | Total Clearance (L/h/kg) | |
|---|---|---|---|---|
| Cancer Patients 1 mg bid, 7 days (N = 27) | 5.99 [0.63 to 30.9] | N.D. Not determined after oral administration; following a single intravenous dose of 40 mcg/kg, terminal phase half-life was determined to be 8.95 hours. | N.D. | 0.52 [0.09 to 7.37] |
| Volunteers single 1 mg dose (N = 39) | 3.63 [0.27 to 9.14] | 6.23 [0.96 to 19.9] | 3.94 [1.89 to 39.4] | 0.41 [0.11 to 24.6] |
Absorption
When granisetron tablets were administered with food, AUC was decreased by 5% and C max increased by 30% in non-fasted healthy volunteers who received a single dose of 10 mg.
Distribution
Plasma protein binding is approximately 65% and granisetron distributes freely between plasma and red blood cells.
Metabolism
Granisetron metabolism involves N-demethylation and aromatic ring oxidation followed by conjugation. In vitro liver microsomal studies show that granisetron's major route of metabolism is inhibited by ketoconazole, suggestive of metabolism mediated by the cytochrome P-450 3A subfamily. Animal studies suggest that some of the metabolites may also have 5-HT 3 receptor antagonist activity.
Elimination
Clearance is predominantly by hepatic metabolism. In normal volunteers, approximately 11% of the orally administered dose is eliminated unchanged in the urine in 48 hours. The remainder of the dose is excreted as metabolites, 48% in the urine and 38% in the feces.
Subpopulations
Gender
The effects of gender on the pharmacokinetics of granisetron hydrochloride tablets have not been studied. However, after intravenous infusion of granisetron hydrochloride, no difference in mean AUC was found between males and females, although males had a higher C max generally.
In elderly and pediatric patients and in patients with renal failure or hepatic impairment, the pharmacokinetics of granisetron was determined following administration of intravenous granisetron.
Elderly
The ranges of the pharmacokinetic parameters in elderly volunteers (mean age 71 years), given a single 40 mcg/kg intravenous dose of granisetron hydrochloride injection, were generally similar to those in younger healthy volunteers; mean values were lower for clearance and longer for half-life in the elderly.
Renal Failure Patients
Total clearance of granisetron was not affected in patients with severe renal failure who received a single 40 mcg/kg intravenous dose of granisetron hydrochloride injection.
Hepatically Impaired Patients
A pharmacokinetic study with intravenous granisetron hydrochloride in patients with hepatic impairment due to neoplastic liver involvement showed that total clearance was approximately halved compared to patients without hepatic impairment. Given the wide variability in pharmacokinetic parameters noted in patients, dosage adjustment in patients with hepatic functional impairment is not necessary.
Pediatric Patients
A pharmacokinetic study in pediatric cancer patients (2 to 16 years of age), given a single 40 mcg/kg intravenous dose of granisetron hydrochloride injection, showed that volume of distribution and total clearance increased with age. No relationship with age was observed for peak plasma concentration or terminal phase plasma half-life. When volume of distribution and total clearance are adjusted for body weight, the pharmacokinetics of granisetron are similar in pediatric and adult cancer patients.
HOW SUPPLIED
Granisetron hydrochloride tablets USP are available as:
White colored, triangular shaped, biconvex, film-coated tablets with debossing of '1GN' on one side and plain surface on the other side.
Granisetron HCl tablets, USP are available as 20 Unit Dose tablets and 2's pack. NDC 51991-735-20-20'S Pack NDC 51991-735-32 - 2'S Pack
Storage
Store between 20° and 25°C (68° and 77°F) [see USP Controlled Room Temperature]. Keep container closed tightly.
Protect from light. Retain in carton until time of use.