Get your patient on Heparin Sodium - Heparin Sodium injection, Solution (Heparin Sodium)
Heparin Sodium - Heparin Sodium injection, Solution prescribing information
Warnings and Precautions, Hyperkalemia (5.9 ) 7/2024
Heparin sodium injection is an anticoagulant indicated for (1 )
- Prophylaxis and treatment of venous thrombosis and pulmonary embolism
- Prevention of postoperative deep venous thrombosis and pulmonary embolism in patients undergoing major abdominothoracic surgery or who, for other reasons, are at risk of developing thromboembolic disease
- Atrial fibrillation with embolization
- Treatment of acute and chronic consumptive coagulopathies (disseminated intravascular coagulation)
- Prevention of clotting in arterial and cardiac surgery
- Prophylaxis and treatment of peripheral arterial embolism
- Use as an anticoagulant in blood transfusions, extracorporeal circulation, and dialysis procedures
Recommended Adult Dosages:
- Therapeutic Anticoagulant Effect with Full-Dose Heparin† (2.3 )
Deep Subcutaneous (Intrafat) Injection Use a different site for each injection | Initial Dose | 5,000 units by intravenous injection followed by 10,000 to 20,000 units of a concentrated solution, subcutaneously |
Every 8 hours or Every 12 hours | 8,000 to 10,000 units of a concentrated solution 15,000 to 20,000 units of a concentrated solution | |
| Intermittent Intravenous Injection | Initial Dose | 10,000 units, either undiluted or in 50 to 100 mL of 0.9% Sodium Chloride Injection, USP |
| Every 4 to 6 hours | 5,000 to 10,000 units, either undiluted or in 50 to 100 mL of 0.9% Sodium Chloride Injection, USP | |
| Intravenous Infusion | Initial Dose | 5,000 units by intravenous injection |
| Continuous | 20,000 to 40,000 units/24 hours in 1,000 mL of 0.9% Sodium Chloride Injection, USP (or in any compatible solution) for infusion | |
| † Based on 150 lb (68 kg) patient. Adjust dose based on laboratory monitoring. | ||
Heparin sodium injection, USP contains parabens:
- 1 mL multiple-dose vial contains 1,000 USP units
- 1 mL multiple-dose vial contains 5,000 USP units
- 1 mL multiple-dose vial contains 10,000 USP units
- History of heparin-induced thrombocytopenia (HIT) or heparin-induced thrombocytopenia and thrombosis (HITTS) (4 )
- Known hypersensitivity to heparin or pork products (4 )
- In whom suitable blood coagulation tests cannot be performed at appropriate intervals (4 )
An uncontrolled bleeding state, except when this is due to disseminated intravascular coagulation (4 )
- Fatal Medication Errors: Confirm choice of correct strength prior to administration (5.1 )
- Hemorrhage: Hemorrhage, including fatal events, has occurred in patients receiving heparin. Use caution in conditions with increased risk of hemorrhage (5.2 )
HIT and HITTS: Monitor for signs and symptoms and discontinue if indicative of HIT and HITTS (5.3 )
Benzyl Alcohol Toxicity: Use preservative-free formulation in neonates and infants
Monitoring: Blood coagulation tests guide therapy for full-dose heparin. Periodically monitor platelet count, hematocrit, and occult blood in stool in all patients receiving heparin (5.5 , 5.6 )
Hyperkalemia: Measure blood potassium in patients at risk of hyperkalemia before starting heparin therapy and periodically in all patients. (5.9 )
Most common adverse reactions are hemorrhage, thrombocytopenia, HIT and HITTS, injection site irritation, general hypersensitivity reactions, and elevations of aminotransferase levels. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Northstar Rx LLC at 1-800-206-7821 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drugs that interfere with coagulation, platelet aggregation or drugs that counteract coagulation may induce bleeding (7.2 )
DESCRIPTION
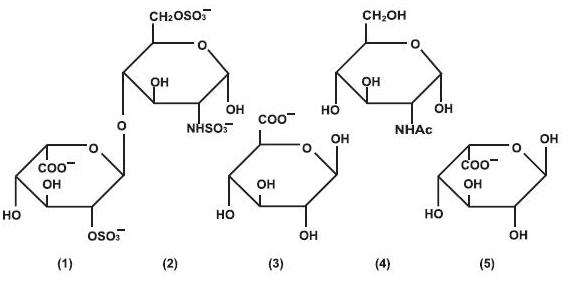
Heparin is a heterogeneous group of straight-chain anionic mucopolysaccharides, called glycosaminoglycans, having anticoagulant properties. Although others may be present, the main sugars occurring in heparin are: (1) α -L-iduronic acid 2-sulfate, (2) 2-deoxy-2-sulfamino- α -D-glucose 6-sulfate, (3) β -D-glucuronic acid, (4) 2-acetamido-2-deoxy- α -D-glucose and (5) α -L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2)> (1)> (4)> (3)> (5), and are joined by glycosidic linkages, forming polymers of varying sizes. Heparin is strongly acidic because of its content of covalently linked sulfate and carboxylic acid groups. In heparin sodium, the acidic protons of the sulfate units are partially replaced by sodium ions. Heparin Sodium Injection, USP is a sterile solution of heparin sodium derived from porcine intestinal mucosa, standardized for anticoagulant activity, in water for injection. It is to be administered by intravenous or deep subcutaneous routes. The potency is determined by a biological assay using a USP reference standard based on units of heparin activity per milligram. Structure of Heparin Sodium (representative subunits):
Heparin Sodium Injection, USP (porcine), preserved with parabens, is available as follows:
Each mL of the 1,000 units per mL preparation contains: 1,000 USP Heparin units (porcine); 9 mg sodium chloride; 1.5 mg methylparaben; 0.15 mg propylparaben; Water for Injection q.s. made isotonic with sodium chloride. Hydrochloric acid and/or sodium hydroxide may have been added for pH adjustment (5.0 to 7.5).
Each mL of the 5,000 units per mL preparation contains: 5,000 USP Heparin units (porcine); 5 mg sodium chloride; 1.5 mg methylparaben; 0.15 mg propylparaben; Water for Injection q.s. Hydrochloric acid and/or sodium hydroxide may have been added for pH adjustment (5.0 to 7.5).
Each mL of the 10,000 units per mL preparation contains: 10,000 USP Heparin units (porcine); 1.5 mg methylparaben; 0.15 mg propylparaben; Water for Injection q.s. Hydrochloric acid and/or sodium hydroxide may have been added for pH adjustment (5.0 to 7.5).
CLINICAL PHARMACOLOGY
Mechanism of Action
Heparin interacts with the naturally occurring plasma protein, Antithrombin III, to induce a conformational change, which markedly enhances the serine protease activity of Antithrombin III, thereby inhibiting the activated coagulation factors involved in the clotting sequence, particularly Xa and IIa. Small amounts of heparin inhibit Factor Xa, and larger amounts inhibit thrombin (Factor IIa). Heparin also prevents the formation of a stable fibrin clot by inhibiting the activation of the fibrin stabilizing factor. Heparin does not have fibrinolytic activity; therefore, it will not lyse existing clots.
Pharmacodynamics
Various times (activated clotting time, activated partial thromboplastin time, prothrombin time, whole blood clotting time) are prolonged by full therapeutic doses of heparin; in most cases, they are not measurably affected by low doses of heparin. The bleeding time is usually unaffected by heparin.
Pharmacokinetics
Absorption
Heparin is not absorbed through the gastrointestinal tract and therefore administered via parenteral route. Peak plasma concentration and the onset of action are achieved immediately after intravenous administration.
Distribution
Heparin is highly bound to antithrombin, fibrinogens, globulins, serum proteases and lipoproteins. The volume of distribution is 0.07 L/kg.
Elimination
Metabolism
Heparin does not undergo enzymatic degradation.
Excretion
Heparin is mainly cleared from the circulation by liver and reticuloendothelial cells mediated uptake into extravascular space. Heparin undergoes biphasic clearance, a) rapid saturable clearance (zero order process due to binding to proteins, endothelial cells and macrophage) and b) slower first order elimination. The plasma half-life is dose-dependent and it ranges from 0.5 to 2 h.
Specific Populations
Geriatric patients
Patients over 60 years of age, following similar doses of heparin, may have higher plasma levels of heparin and longer activated partial thromboplastin times (aPTTs) compared with patients under 60 years of age [see Use in Specific Populations (8.5 )] .
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies in animals have been performed to evaluate carcinogenic potential of heparin. Also, no reproduction studies in animals have been performed concerning mutagenesis or impairment of fertility.
HOW SUPPLIED
Heparin Sodium Injection, USP (porcine) contains parabens and is available as follows:
| NDC Number | Concentration | Vial Fill Volume | Vial Type | Pack Size |
| 72603-250-25 | 1,000 USP Heparin Units/mL | 1 mL | Multiple-Dose Vial | Carton of 25 vials |
| 72603-456-25 | 5,000 USP Heparin Units/mL | 1 mL | Multiple-Dose Vial | Carton of 25 vials |
| 72603-579-25 | 10,000 USP Heparin Units/mL | 1 mL | Multiple-Dose Vial | Carton of 25 vials |
Use only if solution is clear and seal intact. Do not use if solution is discolored or contains a precipitate.
This container closure is not made with natural rubber latex.
STORAGE:
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].