Get your patient on Isoproterenol Hydrochloride - Isoproterenol Hydrochloride injection, Solution (Isoproterenol Hydrochloride)
Isoproterenol Hydrochloride - Isoproterenol Hydrochloride injection, Solution prescribing information
INDICATIONS AND USAGE
Isoproterenol hydrochloride injection is indicated:
- For mild or transient episodes of heart block that do not require electric shock or pacemaker therapy.
- For serious episodes of heart block and Adams-Stokes attacks (except when caused by ventricular tachycardia or fibrillation). (See CONTRAINDICATIONS.)
- For use in cardiac arrest until electric shock or pacemaker therapy, the treatments of choice, is available. (See CONTRAINDICATIONS.)
- For bronchospasm occurring during anesthesia.
- As an adjunct to fluid and electrolyte replacement therapy and the use of other drugs and procedures in the treatment of hypovolemic and septic shock, low cardiac output (hypoperfusion) states, congestive heart failure, and cardiogenic shock. (See WARNINGS.)
DOSAGE AND ADMINISTRATION
Start isoproterenol hydrochloride injection at the lowest recommended dose and increase the rate of administration gradually if necessary while carefully monitoring the patient. The usual route of administration is by intravenous infusion or bolus intravenous injection. In dire emergencies, the drug may be administered by intracardiac injection. If time is not of the utmost importance, initial therapy by intramuscular or subcutaneous injection is preferred.
Recommended dosage for adults with heart block, Adams-Stokes attacks, and cardiac arrest :
| Route of Administration | Preparation of Dilution | Initial Dose | Subsequent Dose Range• |
| Bolus intravenous injection | Dilute 1 mL (0.2 mg) in 9 mL of Sodium Chloride Injection, USP, or 5% Dextrose Injection, USP | 0.02 mg to 0.06 mg (1 mL to 3 mL of diluted solution) | 0.01 mg to 0.2 mg (0.5 mL to 10 mL of diluted solution) |
| Intravenous infusion | Dilute 10 mL (2 mg) in 500 mL of 5% Dextrose Injection, USP | 5 mcg/min. (1.25 mL of diluted solution per minute) | |
| Intramuscular | Use Solution undiluted | 0.2 mg (1 mL) | 0.02 mg to 1 mg (0.1 mL to 5 mL) |
| Subcutaneous | Use Solution undiluted | 0.2 mg (1 mL) | 0.15 mg to 0.2 mg (0.75 mL to 1 mL) |
| Intracardiac | Use Solution undiluted | 0.02 mg (0.1 mL) |
• Subsequent dosage and method of administration depend on the ventricular rate and the rapidity with which the cardiac pacemaker can take over when the drug is gradually withdrawn.
There are no well-controlled studies in children to establish appropriate dosing; however, the American Heart Association recommends an initial infusion rate of 0.1 mcg/kg/min, with the usual range being 0.1 mcg/kg/min to 1 mcg/kg/min.
Recommended dosage for adults with shock and hypoperfusion states:
| Route of Administration | Preparation of Dilution † | Infusion Rate †† |
| Intravenous infusion | Dilute 5 mL (1 mg) in 500 mL of 5% Dextrose Injection, USP | 0.5 mcg to 5 mcg per minute(0.25 mL to 2.5 mL of diluted solution) |
† Concentrations up to 10 times greater have been used when limitation of volume is essential.
†† Rates over 30 mcg per minute have been used in advanced stages of shock. The rate of infusion should be adjusted on the basis of heart rate, central venous pressure, systemic blood pressure, and urine flow. If the heart rate exceeds 110 beats per minute, it may be advisable to decrease or temporarily discontinue the infusion.
Recommended dosage for adults with bronchospasm occurring during anesthesia:
| Route of Administration | Preparation of Dilution | Initial Dose | Subsequent Dose |
| Bolus intravenous injection | Dilute 1 mL (0.2 mg) in 9 mL of Sodium Chloride Injection, USP, or 5% Dextrose Injection, USP | 0.01 mg to 0.02 mg (0.5 mL to 1 mL of diluted solution) | The initial dose may be repeated when necessary |
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Such solution should not be used.
CONTRAINDICATIONS
Use of isoproterenol hydrochloride injection is contraindicated in patients with tachyarrhythmias; tachycardia or heart block caused by digitalis intoxication; ventricular arrhythmias which require inotropic therapy; and angina pectoris.
ADVERSE REACTIONS
The following reactions to isoproterenol hydrochloride injection have been reported:
CNS : Nervousness, headache, dizziness, nausea, visual blurring.
Cardiovascular : Tachycardia, palpitations, angina, Adams-Stokes attacks, pulmonary edema, hypertension, hypotension, ventricular arrhythmias, tachyarrhythmias.
In a few patients, presumably with organic disease of the AV node and its branches, isoproterenol hydrochloride injection has been reported to precipitate Adams-Stokes seizures during normal sinus rhythm or transient heart block.
Respiratory : Dyspnea.
Other : Flushing of the skin, sweating, mild tremors, weakness, pallor
Drug Interactions
Isoproterenol hydrochloride injection and epinephrine should not be administered simultaneously because both drugs are direct cardiac stimulants and their combined effects may induce serious arrhythmias. The drugs may, however, be administered alternately provided a proper interval has elapsed between doses.
Avoid isoproterenol hydrochloride injection when potent inhalational anesthetics such as halothane are employed because of potential to sensitize the myocardium to effects of sympathomimetic amines.
DESCRIPTION
Isoproterenol hydrochloride is 3,4-dihydroxy--[(isopropylamino)methyl] benzyl alcohol hydrochloride, a synthetic sympathomimetic amine that is structurally related to epinephrine but acts almost exclusively on beta receptors. The molecular formula is C 11 H 17 NO 3 HCl. It has a molecular weight of 247.72 and the following structural formula:
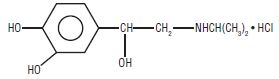
Isoproterenol hydrochloride is a racemic compound.
Each milliliter of the sterile solution contains: Isoproterenol hydrochloride injection, USP 0.2 mg Edetate Disodium (EDTA) 0.2 mg Sodium Chloride 7.0 mg Sodium Citrate, Dihydrate 2.07 mg Citric Acid, Anhydrous 2.5 mg Water for Injection 1.0 mL
The pH is adjusted between 2.5 and 4.5 with hydrochloric acid or sodium hydroxide. The sterile solution is nonpyrogenic and can be administered by the intravenous, intramuscular, subcutaneous, or intracardiac routes.
CLINICAL PHARMACOLOGY
Isoproterenol is a potent nonselective beta-adrenergic agonist with very low affinity for alpha-adrenergic receptors. Intravenous infusion of isoproterenol in man lowers peripheral vascular resistance, primarily in skeletal muscle but also in renal and mesenteric vascular beds. Diastolic pressure falls. Renal blood flow is decreased in normotensive subjects but is increased markedly in shock. Systolic blood pressure may remain unchanged or rise, although mean arterial pressure typically falls. Cardiac output is increased because of the positive inotropic and chronotropic effects of the drug in the face of diminished peripheral vascular resistance. The cardiac effects of isoproterenol may lead to palpitations, sinus tachycardia, and more serious arrhythmias; large doses of isoproterenol may cause myocardial necrosis in animals.
Isoproterenol relaxes almost all varieties of smooth muscle when the tone is high, but this action is most pronounced on bronchial and gastrointestinal smooth muscle. It prevents or relieves bronchoconstriction, but tolerance to this effect develops with overuse of the drug.
In man, isoproterenol causes less hyperglycemia than does epinephrine. Isoproterenol and epinephrine are equally effective in stimulating the release of free fatty acids and energy production.
Absorption, Fate, and Excretion. Isoproterenol is metabolized primarily in the liver and other tissues by COMT. Isoproterenol is a relatively poor substrate for MAO and is not taken up by sympathetic neurons to the same extent as are epinephrine and norepinephrine. The duration of action of isoproterenol may therefore be longer than that of epinephrine, but is still brief.
HOW SUPPLIED
| NDC | Container | Concentration | Fill | Quantity |
| 0548-9501-00 | Single-Dose Vial | 0.2 mg/mL | 1 mL | 10 × 1 mL Single Dose Vials |
| 0548-9502-00 | Single-Dose Vial | 1 mg/5 mL (0.2 mg/mL) | 5 mL | 10 × 5 mL Single Dose Vials |
Protect from light. Keep in opaque container until used.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Do not use if the injection is pinkish or darker than slightly yellow or contains a precipitate.