Get your patient on Kit For The Preparation Of Technetium Tc 99m Medronate - Tc 99m Medronate injection, Powder, Lyophilized, For Solution (Tc 99m Medronate)
Kit For The Preparation Of Technetium Tc 99m Medronate - Tc 99m Medronate injection, Powder, Lyophilized, For Solution prescribing information
INDICATIONS AND USAGE:
Technetium Tc 99m Medronate Injection may be used as a bone imaging agent to delineate areas of altered osteogenesis.
DOSAGES AND ADMINISTRATION:
Shielding should be utilized when preparing Technetium Tc 99m Medronate Injection.
After preparation with oxidant-free Sodium Pertechnetate Tc 99m Injection, the suggested dose range of Technetium Tc 99m Medronate Injection in the average ADULT patient (70 kg.) is:
- 370-740 megabecquerels: (10-20 millicuries) given intravenously.
- Imaging is optimal at 1 to 4 hours post Injection.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
The patient dose should be measured by a suitable radioactivity calibration system immediately prior to administration.
Radiation Dosimetry
The effective half-life was assumed to be the physical half-life for all calculated values. The estimated radiation absorbed doses to an average ADULT patient (70 kg) from an intravenous injection of a maximum of 740 megabecquerels (20 millicuries) of Technetium Tc 99m Medronate Injection are shown in Table 4.
Organ | (MGy/740 MBq) | (Rads / 20 mCi) | |
Total Body | 1.3 | 0.13 | |
Bone Total | 7.0 | 0.70 | |
Red Marrow | 5.6 | 0.56 | |
Kidneys | 8.0 | 0.80 | |
Liver | 0.6 | 0.06 | |
Bladder Wall | 2 hour void | 26 | 2.60 |
4.8 hour void | 62 | 6.20 | |
Ovaries | 2 hour void | 2.4 | 0.24 |
4.8 hour void | 3.4 | 0.34 | |
Testes | 2 hour void | 1.6 | 0.16 |
4.8 hour void | 2.2 | 0.22 | |
2 Method of calculation: "S" Absorbed Dose Per Unit Cumulated Activity for Selected Radionuclides and Organs, MIRD Pamphlet No 11 (1975)
CONTRAINDICATIONS:
None known.
ADVERSE REACTIONS:
Several adverse reactions due to Technetium Tc 99m Medronate Injection have been reported. These were usually hypersensitivity reactions characterized by itching, various skin rashes, hypotension, chills, nausea, and vomiting. There have also been rare cases of dizziness and asthenia associated with the use of Technetium Tc 99m Medronate.
Description
Kit for the Preparation of Technetium Tc 99m Medronate is a multidose reaction vial which contains the sterile, non-pyrogenic, non-radioactive ingredients necessary to produce Technetium Tc 99m Medronate Injection for diagnostic use by Intravenous injection.
Each 10mL multidose vial contains:
- Medronic acid: 20 mg
- Ascorbic acid: 1 mg
- Stannous fluoride, SnF 2 : 0.13 mg (minimum)
- Total tin (maximum, as stannous fluoride, SnF 2 ): 0.38 mg
The pH is adjusted to 6.5 (6.3 to 6.7) with sodium hydroxide and/or hydrochloric acid prior to lyophilization. No bacteriostatic preservative is present in the vial. The contents of the vial are lyophilized and sealed under nitrogen at the time of manufacture. The structural formula is:
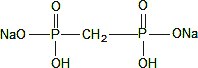
When a solution of sterile, non-pyrogenic, oxidant-free Sodium Pertechnetate Tc 99m Injection is added to the vial, the diagnostic agent, Technetium Tc 99m Medronate is formed for administration by intravenous injection. The pH of the reconstituted product is 5.4 to 6.8. The precise structure of Technetium Tc 99m Medronate Injection is not known at this time.
PHYSICAL CHARACTERISTICS:
Technetium Tc 99m decays by isomeric transition with a physical half-life of 6.02 hours 1 . The principal photon that is useful for detection and imaging studies is listed in Table 1.
Radiation | Mean % per Disintegration | Mean Energy (keV) |
Gamma-2 | 89.07 | 140.5 |
1 Kocher, DC: Radioactive Decay Data Tables, DOE/TIC-11026, 108, 1981 .
EXTERNAL RADIATION:
The specific gamma ray constant for Tc 99m is 0.78 R/millicurie-hr at 1 cm. The first half-value layer is 0.017 cm of lead (Pb). A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of the various thicknesses of Pb is shown in Table 2. To facilitate control of the radiation exposure from millicurie amounts of this radionuclide, the use of a 0.25 cm thickness of Pb will attenuate the radiation emitted by a factor of about 1,000.
Shield Thickness (Pb) cm | Coefficient of Attenuation |
0.017 | 0.5 |
0.08 | 10 -1 |
0.16 | 10 -2 |
0.25 | 10 -3 |
0.33 | 10 -4 |
To correct for physical decay of this radionuclide, the fractions that remain at selected intervals after the time of calibration are shown in Table 3.
Hours | Fraction Remaining | Hours | Fraction Remaining |
0• | 1.000 | 7 | 0.447 |
1 | 0.891 | 8 | 0.398 |
2 | 0.794 | 9 | 0.355 |
3 | 0.708 | 10 | 0.316 |
4 | 0.631 | 11 | 0.282 |
5 | 0.562 | 12 | 0.251 |
6 | 0.501 |
• Calibration Time
CLINICAL PHARMACOLOGY:
During the initial 24 hours following intravenous injection of Technetium Tc 99m Medronate, about 50% of each dose is retained in the skeleton, and about 50% is excreted in the urine. Upon intravenous injection, Technetium Tc 99m Medronate exhibits a specific affinity for areas of altered osteogenesis. In humans, blood levels fall to 4 to 10% of the injected dose by two hours post-injection and to 3 to 5% by three hours.
Uptake of Technetium Tc 99m Medronate Injection in bone appears to be related to osteogenic activity and to skeletal blood perfusion. The deposition in the skeleton is bilaterally symmetrical, with increased accumulation in the axial structure as compared to the appendicular skeleton. There is increased activity in the distal aspect on long bones as compared to the diaphyses.
HOW SUPPLIED:
Kit for the Preparation of Technetium Tc 99m Medronate Injection is supplied in kits of five (5) or thirty (30) sterile, non-pyrogenic vials. Each 10 mL multidose vial contains 20 mg medronic acid, 1 mg ascorbic acid, 0.13 mg minimum stannous fluoride (SnF 2 ) and 0.38 mg maximum total tin, as stannous fluoride, SnF 2 in lyophilized form. The pH is adjusted with sodium hydroxide and/or hydrochloric acid prior to lyophilization. The vial does not contain a preservative. The contents of the vial are lyophilized and sealed under nitrogen at the time of manufacture. The pH of the reconstituted product is 5.4 to 6.8.
Kit Contents
Included in each five (5) vial kit is one (1) package insert and ten (10) radiation labels. Included in each thirty (30) vial kit is one (1) package insert and sixty (60) radiation labels.
Storage
Store the product as supplied at 20-25°C (68-77°F) [See USP]. After reconstitution store at 20-25°C (68-77°F) [See USP] (see DOSAGE AND ADMINISTRATION).