Get your patient on Lincomycin - Lincomycin injection, Solution (Lincomycin)
Lincomycin - Lincomycin injection, Solution prescribing information
WARNING
Clostridioides difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including lincomycin and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
Because lincomycin therapy has been associated with severe colitis which may end fatally, it should be reserved for serious infections where less toxic antimicrobial agents are inappropriate, as described in the INDICATIONS AND USAGE section. It should not be used in patients with nonbacterial infections such as most upper respiratory tract infections.
C. diffficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile , and surgical evaluation should be instituted as clinically indicated.
INDICATIONS AND USAGE
Lincomycin injection is indicated in the treatment of serious infections due to susceptible strains of streptococci, pneumococci, and staphylococci. Its use should be reserved for penicillin-allergic patients or other patients for whom, in the judgment of the physician, a penicillin is inappropriate. Because of the risk of CDAD, as described in the BOXED WARNING , before selecting lincomycin the physician should consider the nature of the infection and the suitability of other alternatives.
Indicated surgical procedures should be performed in conjunction with antibacterial therapy.
Lincomycin injection may be administered concomitantly with other antimicrobial agents when indicated.
Lincomycin is not indicated in the treatment of minor bacterial infections or viral infections.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of lincomycin and other antibacterial drugs, lincomycin should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
If significant diarrhea occurs during therapy, lincomycin should be discontinued. (see BOXED WARNING )
INTRAMUSCULAR - Adults: Serious infections- 600 mg (2 mL) intramuscularly every 24 hours. More severe infections- 600 mg (2 mL) intramuscularly every 12 hours or more often. Pediatric patients over 1 month of age: Serious infections- one intramuscular injection of 10 mg/kg (5 mg/lb) every 24 hours. More severe infections- one intramuscular injection of 10 mg/kg (5 mg/lb) every 12 hours or more often.
INTRAVENOUS - Adults: The intravenous dose will be determined by the severity of the infection. For serious infections doses of 600 mg of lincomycin (2 mL of lincomycin) to 1 gram are given every 8 to 12 hours. For more severe infections these doses may have to be increased. In life-threatening situations daily intravenous doses of as much as 8 grams have been given. Intravenous doses are given on the basis of 1 gram of lincomycin diluted in not less than 100 mL of appropriate solution (see PHYSICAL COMPATIBILITIES ) and infused over a period of not less than one hour.
| Dose | Vol. Diluent | Time |
600 mg | 100 mL | 1 hr |
1 gram | 100 mL | 1 hr |
2 grams | 200 mL | 2 hr |
3 grams | 300 mL | 3 hr |
4 grams | 400 mL | 4 hr |
These doses may be repeated as often as required to the limit of the maximum recommended daily dose of 8 grams of lincomycin.
Pediatric patients over 1 month of age: 10 to 20 mg/kg/day (5 to 10 mg/lb/day) depending on the severity of the infection may be infused in divided doses as described above for adults.
NOTE: Severe cardiopulmonary reactions have occurred when lincomycin has been given at greater than the recommended concentration and rate (see PRECAUTIONS ).
SUBCONJUNCTIVAL INJECTION - 0.25 mL (75 mg) injected subconjunctivally will result in ocular fluid concentrations of antibacterial (lasting for at least 5 hours) sufficient for most susceptible pathogens.
Patients with Renal Impairment When therapy with lincomycin is required in individuals with severe renal impairment, an appropriate dose is 25 to 30% of that recommended for patients with normally functioning kidneys (see PRECAUTIONS ).
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
Lincomycin injection is contraindicated in patients previously found to be hypersensitive to lincomycin or clindamycin.
ADVERSE REACTIONS
The following adverse reactions have been reported with the use of lincomycin.
Gastrointestinal disorders Diarrhea, nausea, vomiting, glossitis, stomatitis, abdominal pain, abdominal discomfort † , anal pruritus
Skin and subcutaneous tissue disorders Toxic epidermal necrolysis, Stevens-Johnson syndrome, acute generalized exanthematous pustulosis, dermatitis bullous, dermatitis exfoliative, erythema multiforme (see WARNINGS ) , rash, urticaria, pruritus
Infections and infestations Vaginal infection, pseudomembranous colitis, Clostridioides difficile colitis (see WARNINGS )
Blood and lymphatic system disorders Pancytopenia, agranulocytosis, aplastic anemia, leukopenia, neutropenia, thrombocytopenic purpura
Immune system disorders Anaphylactic reaction (see WARNINGS ), angioedema, serum sickness
Hepatobiliary disorders Jaundice, liver function test abnormal, transaminases increased
Renal and urinary disorders Renal impairment, oliguria, proteinuria, azotemia
Cardiac disorders Cardio-respiratory arrest (see DOSAGE AND ADMINISTRATION )
Vascular disorders Hypotension (see DOSAGE AND ADMINISTRATION ), thrombophlebitis †
Ear and labyrinth disorders Vertigo, tinnitus
Neurologic disorder Headache, dizziness, somnolence
General disorders and administration site conditions Injection site abscess sterile ‡ , injection site induration ‡ , injection site pain ‡ , injection site irritation ‡
† Event has been reported with intravenous injection. ‡ Reported with intramuscular injection.
To report SUSPECTED ADVERSE REACTIONS, contact Micro Labs USA, Inc. at 1-855-839-8195 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Lincomycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents; therefore, it should be used with caution in patients receiving such agents.
DESCRIPTION
Lincomycin injection, USP is a sterile solution containing lincomycin hydrochloride which is the monohydrated salt of lincomycin, a lincosamide antibacterial produced by the growth of a member of the lincolnensis group of Streptomyces lincolnensis (Fam. Streptomycetaceae ). The chemical name for lincomycin hydrochloride is Methyl 6,8-dideoxy-6-(1-methyl-trans-4-propyl-L2-pyrolidinecarboxamido)-1-thio-D-erythro-α-D-galacto-octopyranoside monohydrochloride monohydrate. The molecular formula of lincomycin hydrochloride is C 18 H 34 N 2 O 6 S.HCl.H 2 O and the molecular weight is 461.01.
The structural formula is represented below: 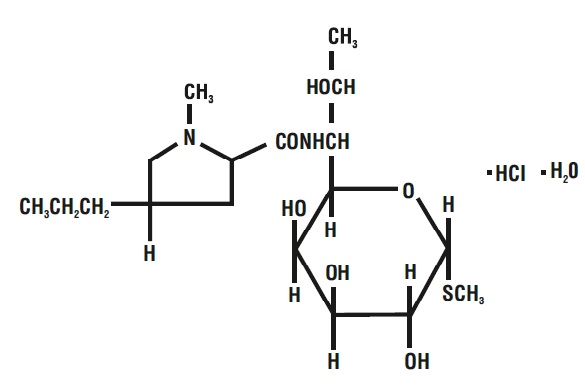
Lincomycin hydrochloride is a white or practically white, crystalline powder. Its solutions are acid and are dextrorotatory. Lincomycin hydrochloride is freely soluble in water; soluble in dimethylformamide and insoluble in acetone.
Lincomycin Injection, USP contains lincomycin hydrochloride in a sterile, clear, colorless to slightly yellow solution with benzyl alcohol used as a preservative 9.45 mg/mL, and water for injection.
Lincomycin injection, USP is a sterile solution for intramuscular and intravenous use. Lincomycin injection, USP is supplied in 2 mL and 10 mL multiple-dose vials containing 300 mg/mL of lincomycin (equivalent to 340 mg/mL of lincomycin hydrochloride, USP).
CLINICAL PHARMACOLOGY
Intramuscular administration of a single dose of 600 mg of lincomycin produces average peak serum concentrations of 11.6 mcg/mL at 60 minutes and maintains therapeutic concentrations for 17 to 20 hours for most susceptible gram-positive organisms. Urinary excretion after this dose ranges from 1.8 to 24.8 percent (mean: 17.3 percent).
A two hour intravenous infusion of 600 mg of lincomycin achieves average peak serum concentrations of 15.9 mcg/mL and maintains therapeutic concentrations for 14 hours for most susceptible gram-positive organisms. Urinary excretion ranges from 4.9 to 30.3 percent (mean: 13.8 percent).
The biological half-life after intramuscular or intravenous administration is 5.4 ± 1.0 hours. The serum half-life of lincomycin may be prolonged in patients with severe renal impairment compared to patients with normal renal function. In patients with hepatic impairment, serum half-life may be twofold longer than in patients with normal hepatic function. Hemodialysis and peritoneal dialysis are not effective in removing lincomycin from the serum.
Tissue distribution studies indicate that bile is an important route of excretion. Significant concentrations have been demonstrated in most body tissues. Although lincomycin appears to diffuse into cerebrospinal fluid (CSF), concentrations of lincomycin in the CSF appear inadequate for the treatment of meningitis.
Microbiology:
Mechanism of Action Lincomycin inhibits bacterial protein synthesis by binding to the 23S RNA of the 50S subunit of the bacterial ribosome. Lincomycin is predominantly bacteriostatic in vitro .
Resistance Cross resistance has been demonstrated between clindamycin and lincomycin. Resistance is most often due to methylation of specific nucleotides in the 23S RNA of the 50S ribosomal subunit, which can determine cross resistance to macrolides and streptogramins B (MLSB phenotype). Macrolide-resistant isolates of these organisms should be tested for inducible resistance to lincomycin/clindamycin using the D-zone test or other appropriate method.
Antimicrobial Activity Lincomycin has been shown to be active against most strains of the following bacteria both in vitro and in clinical infections: (see INDICATIONS AND USAGE ).
Staphylococcus aureus Streptococcus pneumoniae
The following in vitro data are available, but their clinical significance is unknown.
Lincomycin has been shown to be active in vitro against the following microorganisms; however, the safety and efficacy of lincomycin in treating clinical infections due to these organisms have not been established in adequate and well controlled trials.
Gram-positive bacteria: Corynebacterium diphtheriae Streptococcus pyogenes Viridans group streptococci
Anaerobic bacteria: Clostridium tetani Clostridium perfringens
Susceptibility Testing For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
HOW SUPPLIED
Lincomycin Injection, USP is a sterile, clear, colorless to slightly yellow solution, having a slight odor. Free from any visible particles available in the following strength and package sizes:
| Unit of Sale | Concentration |
| NDC 72485-115-01 10 mL multiple-dose vial | 3,000 mg/10 mL (300 mg/mL) |
| NDC 72485-115-10 10 x 10 mL carton multiple-dose vial |
Each mL of Lincomycin Injection, USP contains 300 mg lincomycin (equivalent to 340 mg lincomycin hydrochloride, USP); also benzyl alcohol, 9.45 mg added as preservative.
Store at controlled room temperature 20° to 25°C (68° to 77°F) [see USP].