Get your patient on Metformin Er 500 Mg - Metformin Er 500 Mg tablet (Metformin Er 500 Mg)
Metformin Er 500 Mg - Metformin Er 500 Mg tablet prescribing information
BOXED WARNING
WARNING: LACTIC ACIDOSIS Postmarketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin associated lactic acidosis was characterized by elevated blood lactate levels (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio; and metformin plasma levels generally >5 mcg/mL [see Warnings and Precautions (5.1) ]. Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g. carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment. Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided [see Dosage and Administration (2) , Contraindications (4) , Warnings and Precautions (5.1) ]. If metformin-associated lactic acidosis is suspected, immediately discontinue metformin hydrochloride extended-release tablets and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1) ].
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
Metformin hydrochloride extended-release tablets are indicated as an adjunct to diet and exercise to improve glycemic control in adults and pediatric patients 10 years of age and older with type 2 diabetes mellitus.
DOSAGE AND ADMINISTRATION
NA
Recommended Dosage for Metformin Hydrochloride Extended-Release Tablets in Adults and Pediatric Patients Aged 10 Years and Older With Type 2 Diabetes Mellitus
• Swallow metformin hydrochloride extended-release tablets whole and never crush, cut or chew. • The recommended starting dosage of metformin hydrochloride extended-release is 500 mg orally once daily with the evening meal. • Increase the dosage in increments of 500 mg weekly on the basis of glycemic control and tolerability, up to a maximum of 2,000 mg once daily with the evening meal. • If glycemic control is not achieved with metformin hydrochloride extended-release tablets 2,000 mg once daily, consider a trial of metformin hydrochloride extended-release tablets 1,000 mg twice daily. • If a dose of metformin hydrochloride extended-release tablets is missed, instruct patients not to take two doses the same day and to resume their usual dose of metformin hydrochloride extended-release tablets with the next schedule dose.
Switching Between Metformin Hydrochloride Immediate-Release Tablets and Metformin Hydrochloride Extended-Release Tablets
• Patients may switch between metformin hydrochloride immediate-release tablets and metformin hydrochloride extended-release tablets at the same total daily dosage. • Patients receiving metformin hydrochloride extended-release tablets who require dosages higher than 2,000 mg daily may switch to metformin hydrochloride immediate-release tablets and titrate to the maximum total daily dosage of 2,550 mg.
Recommended Dosage in Patients With Renal Impairment
• Assess renal function prior to initiation of metformin hydrochloride extended-release tablets and periodically thereafter. • Metformin hydrochloride extended-release tablets are contraindicated in patients with an estimated glomerular filtration rate (eGFR) below 30 mL/minute/1.73 m 2 . • Initiation of metformin hydrochloride extended-release tablets in patients with an eGFR between 30 to 45 mL/minute/1.73 m 2 is not recommended. • In patients taking metformin hydrochloride extended-release tablets whose eGFR later falls below 45 mL/min/1.73 m2, assess the benefit risk of continuing therapy. • Discontinue metformin hydrochloride extended-release tablets if the patient’s eGFR later falls below 30 mL/minute/1.73 m 2 [see Warnings and Precautions (5.1) ].
Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue metformin hydrochloride extended-release tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m 2 ; in patients with a history of liver disease, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart metformin hydrochloride extended-release tablets if renal function is stable.
DOSAGE FORMS AND STRENGTHS
Metformin hydrochloride extended-release tablets, USP are available as: • Extended-release tablets: 500 mg white to off-white uncoated, modified capsule shaped tablets debossed with "G7" on one side and plain on other side.
• Extended-release tablets: 750 mg white to off white uncoated, modified capsule shaped tablets debossed with "G8" on one side and plain on other side.
USE IN SPECIFIC POPULATIONS
NA
Pregnancy
Risk Summary Limited data with metformin hydrochloride extended-release tablets in pregnant women are not sufficient to determine a drug-associated risk for major birth defects or miscarriage. Published studies with metformin use during pregnancy have not reported a clear association with metformin and major birth defect or miscarriage risk [see Data ]. There are risks to the mother and fetus associated with poorly controlled diabetes mellitus in pregnancy [see Clinical Considerations ].
No adverse developmental effects were observed when metformin was administered to pregnant Sprague Dawley rats and rabbits during the period of organogenesis at doses up to 2-and 5-times, respectively, a 2,550 mg clinical dose, based on body surface area [see Data ].
The estimated background risk of major birth defects is 6 to 10% in women with pre-gestational diabetes mellitus with an HbA1C >7 and has been reported to be as high as 20 to 25% in women with a HbA1C >10. The estimated background risk of miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations Disease-associated maternal and/or embryo/fetal risk
Poorly-controlled diabetes mellitus in pregnancy increases the maternal risk for diabetic ketoacidosis, pre-eclampsia, spontaneous abortions, preterm delivery, stillbirth and delivery complications. Poorly controlled diabetes mellitus increases the fetal risk for major birth defects, stillbirth, and macrosomia related morbidity.
Data Human Data Published data from post-marketing studies have not reported a clear association with metformin and major birth defects, miscarriage, or adverse maternal or fetal outcomes when metformin was used during pregnancy. However, these studies cannot definitely establish the absence of any metformin-associated risk because of methodological limitations, including small sample size and inconsistent comparator groups.
Animal Data Metformin hydrochloride did not adversely affect development outcomes when administered to pregnant rats and rabbits at doses up to 600 mg/kg/day. This represents an exposure of about 2 and 5 times a 2,550 mg clinical dose based on body surface area comparisons for rats and rabbits, respectively. Determination of fetal concentrations demonstrated a partial placental barrier to metformin.
Lactation
Risk Summary Limited published studies report that metformin is present in human milk [see Data ]. However, there is insufficient information to determine the effects of metformin on the breastfed infant and no available information on the effects of metformin on milk production. Therefore, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for metformin hydrochloride extended-release tablets and any potential adverse effects on the breastfed child from metformin hydrochloride extended-release tablets or from the underlying maternal condition.
Data Published clinical lactation studies report that metformin is present in human milk which resulted in infant doses approximately 0.11% to 1% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 0.13 and 1. However, the studies were not designed to definitely establish the risk of use of metformin during lactation because of small sample size and limited adverse event data collected in infants.
Females and Males of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as therapy with metformin hydrochloride extended-release tablets may result in ovulation in some anovulatory women.
Pediatric Use
Safety and effectiveness of metformin hydrochloride extended-release tablets as an adjunct to diet and exercise to improve glycemic control in pediatric patients 10 years of age and older with type 2 diabetes mellitus have been established. Use of metformin hydrochloride extended-release tablets for this indication is supported by evidence from adequate and well-controlled trials of metformin hydrochloride immediate-release tablets in adults with additional data from a controlled clinical trial of metformin hydrochloride immediate-release tablets in pediatric patients 10 to 16 years old with type 2 diabetes mellitus.
The safety and effectiveness of metformin extended-release tablets for glycemic control in pediatric patients less than 10 years of age with type 2 diabetes mellitus have not been established.
Geriatric Use
Controlled clinical studies of metformin hydrochloride extended-release tablets did not include sufficient numbers of elderly patients to determine whether they respond differently from younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy and the higher risk of lactic acidosis. Assess renal function more frequently in elderly patients [see Warnings and Precautions (5.1) ].
Renal Impairment
Metformin is substantially excreted by the kidney, and the risk of metformin accumulation and lactic acidosis increases with the degree of renal impairment. Metformin hydrochloride extended-release tablets are contraindicated in severe renal impairment, patients with an estimated glomerular filtration rate (eGFR) below 30 mL/min/1.73 m 2 [see Dosage and Administration (2.4) , Contraindications (4) , Warnings and Precautions (5.1) , and Clinical Pharmacology (12.3) ].
Hepatic Impairment
Use of metformin in patients with hepatic impairment has been associated with some cases of lactic acidosis. Metformin hydrochloride extended-release tablets are not recommended in patients with hepatic impairment. [see Warnings and Precautions (5.1) ].
CONTRAINDICATIONS
Metformin hydrochloride extended-release tablets, USP are contraindicated in patients with:
- Severe renal impairment (eGFR below 30 mL/min/1.73 m 2 ) [see Warnings and Precautions (5.1)].
- Hypersensitivity to metformin.
- Acute or chronic metabolic acidosis, including diabetic ketoacidosis, with or without coma.
WARNINGS AND PRECAUTIONS
NA
Lactic Acidosis
There have been postmarketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypotension and resistant bradyarrhythmias have occurred with severe acidosis. Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (>5 mmol/L), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate: pyruvate ratio; metformin plasma levels were generally >5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of metformin hydrochloride extended-release tablets. In metformin hydrochloride extended-release tablets treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin hydrochloride is dialyzable with a clearance of up to 170 mL/min under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and, if these symptoms occur, instruct them to discontinue metformin hydrochloride extended-release tablets and report these symptoms to their healthcare provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below: • Renal impairment — The postmarketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient’s renal function include [see Dosage and Administration (2.2), Clinical Pharmacology (12.3) ]: o Before initiating metformin hydrochloride extended-release tablets, obtain an estimated glomerular filtration rate (eGFR). o Metformin hydrochloride extended-release tablets are contraindicated in patients with an eGFR less than 30 mL/min/1.73 m 2 [see Contraindications (4) ]. o Initiation of metformin hydrochloride extended-release tablets are not recommended in patients with eGFR between 30 to 45 mL/min/1.73 m 2 . o Obtain an eGFR at least annually in all patients taking metformin hydrochloride extended-release tablets. In patients at risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently. o In patients taking metformin hydrochloride extended-release tablets whose eGFR falls below 45 mL/min/1.73 m 2 , assess the benefit and risk of continuing therapy. • Drug interactions — The concomitant use of metformin hydrochloride extended-release tablets with specific drugs may increase the risk of metformin-associated lactic acidosis: those that impair renal function, result in significant hemodynamic change, interfere with acid-base balance, or increase metformin accumulation. Consider more frequent monitoring of patients. • Age 65 or greater — The risk of metformin-associated lactic acidosis increases with the patient’s age because elderly patients have a greater likelihood of having hepatic, renal, or cardiac impairment than younger patients. Assess renal function more frequently in elderly patients. • Radiologic studies with contrast — Administration of intravascular iodinated contrast agents in metformin-treated patients has led to an acute decrease in renal function and the occurrence of lactic acidosis. Stop metformin hydrochloride extended-release tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m 2 ; in patients with a history of hepatic impairment, alcoholism or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure, and restart metformin hydrochloride extended-release tablets if renal function is stable. • Surgery and other procedures — Withholding of food and fluids during surgical or other procedures may increase the risk for volume depletion, hypotension, and renal impairment. Metformin hydrochloride extended-release tablets should be temporarily discontinued while patients have restricted food and fluid intake. • Hypoxic states — Several of the postmarketing cases of metformin-associated lactic acidosis occurred in the setting of acute congestive heart failure (particularly when accompanied by hypoperfusion and hypoxemia). Cardiovascular collapse (shock), acute myocardial infarction, sepsis, and other conditions associated with hypoxemia have been associated with lactic acidosis and may cause prerenal azotemia. When such an event occurs, discontinue metformin hydrochloride extended-release tablets. • Excessive alcohol intake — Alcohol potentiates the effect of metformin on lactate metabolism. Patients should be warned against excessive alcohol intake while receiving metformin hydrochloride extended-release tablets. • Hepatic impairment — Patients with hepatic impairment have developed cases of metformin-associated lactic acidosis. This may be due to impaired lactate clearance resulting in higher lactate blood levels. Therefore, avoid use of metformin hydrochloride extended-released tablets in patients with clinical or laboratory evidence of hepatic disease.
Vitamin B 12 Deficiency
In metformin hydrochloride tablets clinical trials of 29-week duration, a decrease to subnormal levels of previously normal serum vitamin B 12 levels was observed in approximately 7% of patients. Such decrease, possibly due to interference with B 12 absorption from the B 12 -intrinsic factor complex, may be associated with anemia but appears to be rapidly reversible with discontinuation of metformin hydrochloride tablets or vitamin B 12 supplementation. Certain individuals (those with inadequate vitamin B 12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B 12 levels. Measure hematologic parameters on an annual basis and vitamin B 12 at 2 to 3 year intervals in patients on metformin hydrochloride extended-release tablets and manage any abnormalities [see Adverse Reactions (6.1) ].
Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues
Insulin and insulin secretagogues (e.g., sulfonylurea) are known to cause hypoglycemia. Metformin hydrochloride extended-release tablets may increase the risk of hypoglycemia when combined with insulin and/or an insulin secretagogue. Therefore, a lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with metformin hydrochloride extended-release tablets [see Drug Interactions (7) ].
Macrovascular Outcomes
There have been no clinical studies establishing conclusive evidence of macrovascular risk reduction with metformin hydrochloride extended-release tablets.
ADVERSE REACTIONS
The following adverse reactions are also discussed elsewhere in the labeling:
- Lactic Acidosis [see Boxed Warning and Warnings and Precautions (5.1) ]
- Vitamin B 12 Deficiency [see Warnings and Precautions (5.2) ]
- Hypoglycemia [see Warnings and Precautions (5.3) ]
Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Metformin Hydrochloride Extended-Release Tablets In placebo-controlled trials, in adults with type 2 diabetes mellitus, 781 patients were administered metformin hydrochloride extended-release tablets. Adverse reactions reported in greater than 5% of the metformin hydrochloride extended-release tablets patients, and that were more common in metformin hydrochloride extended-release tablets - than placebo-treated patients, are listed in Table 1.
Table 1: Adverse Reactions from Clinical Trials of Metformin Hydrochloride Extended-Release Tablets Occurring >5% and More Common than Placebo in Adults with Type 2 Diabetes Mellitus
| Metformin Hydrochloride Extended- Released Tablets (n=781) | Placebo (n=195) | |
| Diarrhea | 10% | 3% |
| Nausea/Vomiting | 7% | 2% |
Diarrhea led to discontinuation of metformin hydrochloride extended-release tablets in 0.6% of patients. Additionally, the following adverse reactions were reported in ≥1% to ≤5% of metformin hydrochloride extended-release tablets patients and were more commonly reported with metformin hydrochloride extended-release tablets than placebo: abdominal pain, constipation, distention abdomen, dyspepsia/heartburn, flatulence, dizziness, headache, upper respiratory infection, taste disturbance.
Postmarketing Experience
The following adverse reactions have been identified during post approval use of metformin. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cholestatic, hepatocellular, and mixed hepatocellular liver injury have been reported with postmarketing use of metformin.
DRUG INTERACTIONS
Table 2 presents clinically significant drug interactions with metformin hydrochloride extended-release tablets. Table 2: Clinically Significant Drug Interactions with Metformin Hydrochloride Extended-Release Tablets
| Carbonic Anhydrase Inhibitors | |
| Clinical Impact: | Carbonic anhydrase inhibitors frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with metformin hydrochloride extended-release tablets may increase the risk for lactic acidosis. |
| Intervention: | Consider more frequent monitoring of these patients. |
| Examples: | Topiramate, zonisamide, acetazolamide or dichlorphenamide. |
| Drugs that Reduce Metformin Hydrochloride Extended-release Tablets Clearance | |
| Clinical Impact: | Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3) ]. |
| Intervention: | Consider the benefits and risks of concomitant use with metformin hydrochloride extended-release tablets. |
| Examples: | Ranolazine, vandetanib, dolutegravir, and cimetidine. |
| Alcohol | |
| Clinical Impact: | Alcohol is known to potentiate the effect of metformin on lactate metabolism. |
| Intervention: | Warn patients against excessive alcohol intake while receiving metformin hydrochloride extended-release tablets. |
| Insulin Secretagogues or Insulin | |
| Clinical Impact: | Coadministration of metformin hydrochloride extended-release tablets with an insulin secretagogue (e.g., sulfonylurea) or insulin may increase the risk of hypoglycemia. |
| Intervention: | Patients receiving an insulin secretagogue or insulin may require lower doses of the insulin secretagogue or insulin. |
| Drugs Affecting Glycemic Control | |
| Clinical Impact: | Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. |
| Intervention: | When such drugs are administered to a patient receiving metformin hydrochloride extended-release tablets, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving metformin hydrochloride extended-release tablets, observe the patient closely for hypoglycemia. |
| Examples: | Thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blockers, and isoniazid. |
DESCRIPTION
Metformin hydrochloride extended-release tablets, USP contain the antihyperglycemic agent metformin, which is a biguanide, in the form of monohydrochloride. The chemical name of metformin hydrochloride is N,N-dimethylimidodicarbonimidic diamide hydrochloride . The structural formula is as shown below:
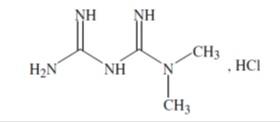
Metformin hydrochloride, USP is a white to off-white crystalline compound with a molecular formula of C 4 H 11 N 5 • HCl and a molecular weight of 165.62. Metformin hydrochloride is freely soluble in water, slightly soluble in ethanol, practically insoluble in acetone and in methylene chloride. The pK a of metformin is 12.4. The pH of a 1% aqueous solution of metformin hydrochloride is 6.35. Metformin hydrochloride extended-release tablets USP, contains 500 mg or 750 mg of metformin hydrochloride, which is equivalent to 389.93 mg, 584.90 mg metformin base, respectively. Metformin hydrochloride extended-release tablets USP, 500 mg tablets contain the inactive ingredients hypromellose, magnesium stearate, and polyvinyl pyrrolidone Metformin hydrochloride extended-release tablets USP, 750 mg tablets contain the inactive ingredients hypromellose, magnesium stearate, and polyvinyl pyrrolidone Meets USP Dissolution Test 10
CLINICAL PHARMACOLOGY
NA
Mechanism of Action
Metformin is an antihyperglycemic agent which improves glucose tolerance in patients with type 2 diabetes mellitus, lowering both basal and postprandial plasma glucose. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may decrease.
Pharmacokinetics
Absorption
The absolute bioavailability of a metformin hydrochloride tablets, 500 mg tablet given under fasting conditions is approximately 50% to 60%. Studies using single oral doses of metformin hydrochloride tablets 500 to 1,500 mg and 850 to 2,550 mg, indicate that there is a lack of dose proportionality with increasing doses, which is due to decreased absorption rather than an alteration in elimination. At usual clinical doses and dosing schedules of metformin hydrochloride tablets, steady state plasma concentrations of metformin are reached within 24 to 48 hours and are generally <1 μg/mL.
Following a single oral dose of metformin hydrochloride extended-release tablets, C max is achieved with a median value of 7 hours and a range of 4 to 8 hours. Peak plasma levels are approximately 20% lower compared to the same dose of metformin hydrochloride tablets, however, the extent of absorption (as measured by AUC) is comparable to metformin hydrochloride tablets.
At steady state, the AUC and C max are less than dose proportional for metformin hydrochloride extended-release tablets within the range of 500 to 2,000 mg administered once daily. Peak plasma levels are approximately 0.6, 1.1, 1.4 and 1.8 mcg/mL for 500, 1,000, 1,500, and 2,000 mg once-daily doses, respectively. The extent of metformin absorption (as measured by AUC) from metformin hydrochloride extended-release tablets at a 2,000 mg once-daily dose is similar to the same total daily dose administered as metformin hydrochloride tablets 1,000 mg twice daily. After repeated administration of metformin hydrochloride extended-release tablets, metformin did not accumulate in plasma. Effect of food : Food decreases the extent of absorption and slightly delays the absorption of metformin, as shown by approximately a 40% lower mean peak plasma concentration (C max ), a 25% lower area under the plasma concentration versus time curve (AUC), and a 35-minute prolongation of time to peak plasma concentration (T max ) following administration of a single 850 mg tablet of metformin hydrochloride tablets with food, compared to the same tablet strength administered fasting.
Although the extent of metformin absorption (as measured by AUC) from the metformin hydrochloride extended-release tablet increased by approximately 50% when given with food, there was no effect of food on C max and T max of metformin. Both high and low fat meals had the same effect on the pharmacokinetics of metformin hydrochloride extended-release tablets. Distribution The apparent volume of distribution (V/F) of metformin following single oral doses of metformin hydrochloride tablets 850 mg averaged 654 ± 358 L. Metformin is negligibly bound to plasma proteins. Metformin partitions into erythrocytes, most likely as a function of time.
Metabolism Intravenous single-dose studies in normal subjects demonstrate that metformin is excreted unchanged in the urine and does not undergo hepatic metabolism (no metabolites have been identified in humans) nor biliary excretion.
Elimination Renal clearance (see Table 3) is approximately 3.5 times greater than creatinine clearance, which indicates that tubular secretion is the major route of metformin elimination. Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
Specific Populations Renal Impairment In patients with decreased renal function the plasma and blood half-life of metformin is prolonged and the renal clearance is decreased (see Table 3) [see Dosage and Administration (2.3), Contraindications (4) , Warnings and Precautions (5.1) and Use in Specific Populations (8.6)].
Hepatic Impairment No pharmacokinetic studies of metformin have been conducted in patients with hepatic impairment [see Warnings and Precautions (5.1) and Use in Specific Populations (8.7)].
Geriatrics Limited data from controlled pharmacokinetic studies of metformin hydrochloride tablets in healthy elderly subjects suggest that total plasma clearance of metformin is decreased, the half-life is prolonged, and C max is increased, compared to healthy young subjects. It appears that the change in metformin pharmacokinetics with aging is primarily accounted for by a change in renal function (see Table 3). [see Warnings and Precautions (5.1) and Use in Specific Populations (8.5)].
Table 3: Select Mean (±S.D.) Metformin Pharmacokinetic Parameters Following Single or Multiple Oral Doses of Metformin Hydrochloride Tablets
Subject Groups: Metformin Hydrochloride Tablets dose a (number of subjects) | C max b (mcg/mL) | T max c (hrs) | Renal Clearance (mL/min) |
Healthy, nondiabetic adults: | |||
500 mg single dose (24) | 1.03 (±0.33) | 2.75 (±0.81) | 600 (±132) |
850 mg single dose (74) d | 1.6 (±0.38) | 2.64 (±0.82) | 552 (±139) |
850 mg three times daily for 19 doses e (9) | 2.01 (±0.42) | 1.79 (±0.94) | 642 (±173) |
Adults with type 2 diabetes mellitus: | |||
850 mg single dose (23) | 1.48 (±0.5) | 3.32 (±1.08) | 491 (±138) |
850 mg three times daily for 19 doses e (9) | 1.9 (±0.62) | 2.01 (±1.22) | 550 (±160) |
Elderly f , healthy nondiabetic adults: 850 mg single dose (12) | 2.45 (±0.7) | 2.71 (±1.05) | 412 (±98) |
Renal-impaired adults: 850 mg single dose Mild (CL cr g 61 to 90 mL/min) (5) Moderate (CL cr 31 to 60 mL/min) (4) Severe (CL cr 10 to 30 mL/min) (6) | 1.86 (±0.52) | 3.2 (±0.45) | 384 (±122) |
4.12 (±1.83) | 3.75 (±0.5) | 108 (±57) | |
3.93 (±0.92) | 4.01 (±1.1) | 130 (±90) |
a All doses given fasting except the first 18 doses of the multiple dose studies b Peak plasma concentration c Time to peak plasma concentration d Combined results (average means) of five studies: mean age 32 years (range 23 to 59 years) e Kinetic study done following dose 19, given fasting f Elderly subjects, mean age 71 years (range 65 to 81 years) g CL cr = creatinine clearance normalized to body surface area of 1.73 m 2
Pediatric Patients After administration of a single oral metformin hydrochloride 500 mg tablet with food, geometric mean metformin C max and AUC differed less than 5% between pediatric patients (12 to 16 years of age) with type 2 diabetes mellitus and sex and weight-matched healthy adults (20 to 45 years of age), all with normal renal function.
Sex Metformin pharmacokinetic parameters did not differ significantly between normal subjects and patients with type 2 diabetes mellitus when analyzed according to sex (males=19, females=16).
Race No studies of metformin pharmacokinetic parameters according to race have been performed.
Drug Interaction Studies In Vivo Assessment of Drug Interactions Table 4: Effect of Coadministered Drug on Plasma Metformin Systemic Exposure
Coadministered Drug | Dose of Coadministered Drug • | Dose of Metformin • | Geometric Mean Ratio (ratio with/without coadministered drug) No Effect = 1 | ||
AUC † | C max | ||||
No dosing adjustments required for the following: | |||||
Glyburide | 5 mg | 850 mg | metformin | 0.91 ‡ | 0.93 ‡ |
Furosemide | 40 mg | 850 mg | metformin | 1.09 ‡ | 1.22 ‡ |
Nifedipine | 10 mg | 850 mg | metformin | 1.16 | 1.21 |
Propranolol | 40 mg | 850 mg | metformin | 0.9 | 0.94 |
Ibuprofen | 400 mg | 850 mg | metformin | 1.05 ‡ | 1.07 ‡ |
Cationic drugs eliminated by renal tubular secretion may reduce metformin elimination [see Warnings and Precautions (5.9) and Drug Interactions (7.2). ] | |||||
Cimetidine | 400 mg | 850 mg | metformin | 1.4 | 1.61 |
Carbonic anhydrase inhibitors may cause metabolic acidosis [see Warnings and Precautions (5.1) and Drug Interactions (7.1). ] | |||||
Topiramate | 100 mg § | 500 mg § | metformin | 1.25 § | 1.17 |
• All metformin and coadministered drugs were given as single doses
† AUC = AUC (INF) ‡ Ratio of arithmetic means § At steady state with topiramate 100 mg every 12 hours and metformin 500 mg every 12 hours; AUC = AUC0-12h
Table 5: Effect of Metformin on Coadministered Drug Systemic Exposure
Coadministered Drug | Dose of Coadministered Drug • | Dose of Metformin • | Geometric Mean Ratio (ratio with/without metformin) No Effect = 1 | |||||
AUC ‡ | C max | |||||||
No dosing adjustments required for the following: | ||||||||
Glyburide | 5 mg | 850 mg | glyburide | 0.78 ‡ | 0.63 ‡ | |||
Furosemide | 40 mg | 850 mg | furosemide | 0.87 ‡ | 0.69 ‡ | |||
Nifedipine | 10 mg | 850 mg | nifedipine | 1.1 § | 1.08 | |||
Propranolol | 40 mg | 850 mg | propranolol | 1.01 § | 1.02 | |||
Ibuprofen | 400 mg | 850 mg | ibuprofen | 0.97 ¶ | 1.01 ¶ | |||
Cimetidine | 400 mg | 850 mg | cimetidine | 0.95 § | 1.01 | |||
• All metformin and coadministered drugs were given as single doses † AUC = AUC (INF) unless otherwise noted ‡ Ratio of arithmetic means, p-value of difference <0.05 § AUC (0-24 hr) reported ¶ Ratio of arithmetic means
NONCLINICAL TOXICOLOGY
NA
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies have been performed in rats (dosing duration of 104 weeks) and mice (dosing duration of 91 weeks) at doses up to and including 900 mg/kg/day and 1,500 mg/kg/day, respectively. These doses are both approximately 3 times the maximum recommended human daily dose of 2,550 mg based on body surface area comparisons. No evidence of carcinogenicity with metformin was found in either male or female mice. Similarly, there was no tumorigenic potential observed with metformin in male rats. There was, however, an increased incidence of benign stromal uterine polyps in female rats treated with 900 mg/kg/day.
There was no evidence of a mutagenic potential of metformin in the following in vitro tests: Ames test ( S. typhimurium ), gene mutation test (mouse lymphoma cells), or chromosomal aberrations test (human lymphocytes). Results in the in vivo mouse micronucleus test were also negative.
Fertility of male or female rats was unaffected by metformin when administered at doses as high as 600 mg/kg/day, which is approximately 2 times the maximum recommended human daily dose of 2,550 mg based on body surface area comparisons.
CLINICAL STUDIES
NA
Metformin Hydrochloride Extended-Release Tablets
A 24-week, double-blind, placebo-controlled trial of metformin hydrochloride extended-release tablets, taken once daily with the evening meal, was conducted in adults with type 2 diabetes mellitus who had failed to achieve glycemic control with diet and exercise. Patients entering the trial had a mean baseline HbA 1c of 8% and a mean baseline FPG of 176 mg/dL. The treatment dose was increased to 1,500 mg once daily if at Week 12 HbA 1c was ≥7% but <8% (patients with HbA 1c ≥8% were discontinued from the trial). At the final visit (24-week), mean HbA1c had increased 0.2% from baseline in placebo patients and decreased 0.6% with metformin hydrochloride extended-release tablets.
A 16-week, double-blind, placebo-controlled, dose-response trial of metformin hydrochloride extended-release tablets, taken once daily with the evening meal or twice daily with meals, was conducted in adults with type 2 diabetes mellitus who had failed to achieve glycemic control with diet and exercise. The results are shown in Table 6.
Table 6: Mean Changes from Baseline• in HbA1c and Fasting Plasma Glucose at Week 16 Comparing Metformin Extended-Release Tablets vs Placebo in Adults with Type 2 Diabetes Mellitus
Metformin Hydrochloride Extended-Release Tablets | Placebo | |||||
500 mg Once Daily | 1, 000 mg Once Daily | 1,500 mg Once Daily | 2,000 mg Once Daily | 1,000 mg Twice Daily | ||
Hemoglobin A 1c (%) | (n=115) | (n=115) | (n=111) | (n=125) | (n=112) | (n=111) |
Baseline | 8.2 | 8.4 | 8.3 | 8.4 | 8.4 | 8.4 |
Change at FINAL VISIT | –0.4 | –0.6 | –0.9 | –0.8 | –1.1 | 0.1 |
p-value a | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
FPG (mg/dL) | (n=126) | (n=118) | (n=120) | (n=132) | (n=122) | (n=113) |
Baseline | 182.7 | 183.7 | 178.9 | 181 | 181.6 | 179.6 |
Change at FINAL VISIT | –15.2 | –19.3 | –28.5 | –29.9 | –33.6 | 7.6 |
p-value a | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
a All comparisons versus Placebo
Mean baseline body weight was 193 lbs, 192 lbs, 188 lbs, 196 lbs, 193 lbs and 194 lbs in the metformin hydrochloride extended-release tablets 500 mg, 1,000 mg, 1,500 mg, and 2,000 mg once daily, 1,000 mg twice daily and placebo arms, respectively. Mean change in body weight from baseline to week 16 was -1.3 lbs, 1.3 lbs, -0.7 lbs, -1.5 lbs, -2.2 lbs and -1.8 lbs, respectively.
A 24-week, double-blind, randomized trial of metformin hydrochloride extended-release tablets, taken once daily with the evening meal, and metformin hydrochloride tablets, taken twice daily (with breakfast and evening meal), was conducted in adults with type 2 diabetes mellitus who had been treated with metformin hydrochloride tablets 500 mg twice daily for at least 8 weeks prior to study entry. The results are shown in Table 7.
Table 7: Mean Changes from Baseline• in HbA1c and Fasting Plasma Glucose at Week 24 Comparing Metformin Hydrochloride Extended-Release Tablets vs Metformin Hydrochloride Tablets in Adults with Type 2 Diabetes Mellitus
Metformin Hydrochloride Tablets 500 mg Twice Daily | Metformin Hydrochloride Extended – release tablets | ||
1 , 000 mg Once Daily | 1,500 mg Once Daily | ||
Hemoglobin A1c (%) | (n=67) | (n=72) | (n=66) |
Baseline | 7.06 | 6.99 | 7.02 |
Change at FINAL VISIT | 0.14 a | 0.27 | 0.13 |
(95% CI) | (–0.04, 0.31) | (0.11, 0.43) | (–0.02, 0.28) |
FPG (mg/dL) Baseline Change at FINAL VISIT (95% CI) | (n=69) | (n=72) | (n=70) |
127.2 | 131 | 131.4 | |
14 | 11.5 | 7.6 | |
(7, 21) | (4.4, 18.6) | (1, 14.2) | |
†a n=68
Mean baseline body weight was 210 lbs, 203 lbs and 193 lbs in the metformin hydrochloride tablets 500 mg twice daily, and metformin hydrochloride extended-release tablets 1,000 mg and 1,500 mg once daily arms, respectively. Mean change in body weight from baseline to week 24 was 0.9 lbs, 1.1 lbs and 0.9 lbs, respectively.
HOW SUPPLIED/STORAGE AND HANDLING
NA
How Supplied
Table 8: Metformin hydrochloride extended-release tablets, USP Available Strengths, Units, and Appearance
| Metformin hydrochloride extended-release tablets, USP | |||
| 500 mg | Bottles of 30 | NDC 62207-491-54 | White to off-white uncoated, modified capsule shaped tablets debossed with “G7” on one side and plain on other side. |
Bottles of 90 | NDC 62207-491-57 | ||
Bottles of 100 | NDC 62207-491-43 | ||
Bottles of 500 | NDC 62207-491-47 | ||
| Bottles of 1,000 | NDC 62207-491-49 | ||
| 750 mg | Bottles of 30 | NDC 62207-492-54 | White to off-white uncoated modified capsule shaped tablets debossed with “G8” on one side and plain on other side. |
Bottles of 90 | NDC 62207-492-57 | ||
Bottles of 100 | NDC 62207-492-43 | ||
Bottles of 500 | NDC 62207-492-47 | ||
Bottles of 1,000 | NDC 62207-492-49 | ||
Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Dispense in light-resistant container.
Mechanism of Action
Metformin is an antihyperglycemic agent which improves glucose tolerance in patients with type 2 diabetes mellitus, lowering both basal and postprandial plasma glucose. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. With metformin therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may decrease.