Get your patient on Methylphenidate Hydrochloride extended - Release - Methylphenidate Hydrochloride tablet, Extended Release (Methylphenidate Hydrochloride)
Methylphenidate Hydrochloride extended - Release - Methylphenidate Hydrochloride tablet, Extended Release prescribing information
WARNING: ABUSE, MISUSE, AND ADDICTION
Methylphenidate hydrochloride extended-release tablets have a high potential for abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release tablets, can result in overdose and death [see Overdosage (10 )] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing methylphenidate hydrochloride extended-release tablets, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks, proper storage of the drug, and proper disposal of any unused drug. Throughout methylphenidate hydrochloride extended-release tablets treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction [see Warnings and Precautions (5.1 ) and Drug Abuse and Dependence (9.2 )] .
INDICATIONS AND USAGE
Methylphenidate hydrochloride extended-release tablets are indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in children 6 years of age and older, adolescents, and adults up to the age of 65 [see Clinical Studies (14 )] .
DOSAGE AND ADMINISTRATION
Pretreatment Screening
Prior to treating patients with methylphenidate hydrochloride extended-release tablets, assess:
- for the presence of cardiac disease (i.e., perform a careful history, family history of sudden death or ventricular arrhythmia, and physical exam) [see Warnings and Precautions (5.2 )].
- the family history and clinically evaluate patients for motor or verbal tics or Tourette’s syndrome before initiating methylphenidate hydrochloride extended-release tablets [see Warnings and Precautions (5.13 )].
Recommended Dosage
Methylphenidate hydrochloride extended-release tablets should be administered orally once daily in the morning with or without food.
Methylphenidate hydrochloride extended-release tablets must be swallowed whole with the aid of liquids, and must not be chewed, divided, or crushed [see Patient Counseling Information (17 )] .
Patients New to Methylphenidate
The recommended starting dose of methylphenidate hydrochloride extended-release tablets for patients who are not currently taking methylphenidate or stimulants other than methylphenidate is 18 mg once daily for children and adolescents and 18 or 36 mg once daily for adults (see Table 1).
Patient Age | Recommended Starting Dose | Dose Range |
Children 6-12 years of age | 18 mg/day | 18 mg - 54 mg/day |
Adolescents 13-17 years of age | 18 mg/day | 18 mg - 72 mg/day not to exceed 2 mg/kg/day |
Adults 18-65 years of age | 18 or 36 mg/day | 18 mg - 72 mg/day |
Patients Currently Using Methylphenidate
The recommended dose of methylphenidate hydrochloride extended-release tablets for patients who are currently taking methylphenidate twice daily or three times daily at doses of 10 to 60 mg/day is provided in Table 2. Dosing recommendations are based on current dose regimen and clinical judgment. Conversion dosage should not exceed 72 mg daily.
Previous Methylphenidate Daily Dose | Recommended Methylphenidate Hydrochloride Extended-Release Tablets Starting Dose |
5 mg Methylphenidate twice daily or three times daily | 18 mg every morning |
10 mg Methylphenidate twice daily or three times daily | 36 mg every morning |
15 mg Methylphenidate twice daily or three times daily | 54 mg every morning |
20 mg Methylphenidate twice daily or three times daily | 72 mg every morning |
Other methylphenidate regimens: Clinical judgment should be used when selecting the starting dose.
Dose Titration
Doses may be increased in 18 mg increments at weekly intervals for patients who have not achieved an optimal response at a lower dose. Daily dosages above 54 mg in children and 72 mg in adolescents have not been studied and are not recommended. Daily dosages above 72 mg in adults are not recommended.
A 27 mg dosage strength is available for physicians who wish to prescribe between the 18 mg and 36 mg dosages.
Dosage Reduction and Discontinuation
If paradoxical aggravation of symptoms or other adverse reactions occur, reduce dosage or, if necessary, discontinue methylphenidate hydrochloride extended-release tablets.
If improvement is not observed after appropriate dosage adjustment over a one-month period, discontinue methylphenidate hydrochloride extended-release tablets.
DOSAGE FORMS AND STRENGTHS
Strength | Color | Tablet Description |
| 27 mg | Gray | capsule shaped, imprinted with “M” in a box and “27” on one side in black ink |
| 36 mg | White | capsule shaped, imprinted with “M” in a box and “36” on one side in black ink |
| 54 mg | Red | capsule shaped, imprinted with “M” in a box and “54” on one side in black ink |
USE IN SPECIFIC POPULATIONS
Pregnancy
Pregnancy Category C Methylphenidate has been shown to have teratogenic effects in rabbits when given in doses of 200 mg/kg/day, which is approximately 100 times and 40 times the maximum recommended human dose on a mg/kg and mg/m 2 basis, respectively.
A reproduction study in rats revealed no evidence of harm to the fetus at oral doses up to 30 mg/kg/day, approximately 15-fold and 3-fold the maximum recommended human dose of methylphenidate hydrochloride extended-release tablets on a mg/kg and mg/m 2 basis, respectively. The approximate plasma exposure to methylphenidate plus its main metabolite PPAA in pregnant rats was 1 to 2 times that seen in trials in volunteers and patients with the maximum recommended dose of methylphenidate hydrochloride extended-release tablets based on the AUC.
The safety of methylphenidate for use during human pregnancy has not been established. There are no adequate and well-controlled studies in pregnant women. Methylphenidate hydrochloride extended-release tablets should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Labor and Delivery
The effect of methylphenidate hydrochloride extended-release tablets on labor and delivery in humans is unknown.
Nursing Mothers
It is not known whether methylphenidate is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised if methylphenidate hydrochloride extended-release tablets are administered to a nursing woman.
In lactating female rats treated with a single oral dose of 5 mg/kg radiolabeled methylphenidate, radioactivity (representing methylphenidate and/or its metabolites) was observed in milk and levels were generally similar to those in plasma.
Pediatric Use
Methylphenidate hydrochloride extended-release tablets should not be used in children under six years, since safety and efficacy in this age group have not been established. Long-term effects of methylphenidate in children have not been well established.
Geriatric Use
Methylphenidate hydrochloride extended-release tablets have not been studied in patients greater than 65 years of age.
CONTRAINDICATIONS
Hypersensitivity to Methylphenidate
Hypersensitivity reactions, such as angioedema and anaphylactic reactions, have been observed in patients treated with methylphenidate hydrochloride extended-release tablets. Therefore, methylphenidate hydrochloride extended-release tablets are contraindicated in patients known to be hypersensitive to methylphenidate or other components of the product [see Adverse Reactions (6.5 )] .
Monoamine Oxidase Inhibitors
Methylphenidate hydrochloride extended-release tablets are contraindicated during treatment with monoamine oxidase (MAO) inhibitors, and also within a minimum of 14 days following discontinuation of a MAO inhibitor (hypertensive crises may result) [see Drug Interactions (7.1 )] .
WARNINGS AND PRECAUTIONS
Abuse, Misuse, and Addiction
Methylphenidate hydrochloride extended-release tablets have a high potential for abuse and misuse. The use of methylphenidate hydrochloride extended-release tablets exposes individuals to the risks of abuse and misuse, which can lead to the development of a substance use disorder, including addiction. Methylphenidate hydrochloride extended-release tablets can be diverted for non-medical use into illicit channels or distribution [see Drug Abuse and Dependence (9.2 )] . Misuse and abuse of CNS stimulants, including methylphenidate hydrochloride extended-release tablets, can result in overdose and death [see Overdosage (10 )] , and this risk is increased with higher doses or unapproved methods of administration, such as snorting or injection.
Before prescribing methylphenidate hydrochloride extended-release tablets, assess each patient’s risk for abuse, misuse, and addiction. Educate patients and their families about these risks and proper disposal of any unused drug. Advise patients to store methylphenidate hydrochloride extended-release tablets in a safe place, preferably locked, and instruct patients to not give methylphenidate hydrochloride extended-release tablets to anyone else. Throughout methylphenidate hydrochloride extended-release tablets treatment, reassess each patient’s risk of abuse, misuse, and addiction and frequently monitor for signs and symptoms of abuse, misuse, and addiction.
Risks to Patients with Serious Cardiac Disease
Sudden death has been reported in patients with structural cardiac abnormalities or other serious cardiac disease who were treated with CNS stimulants at the recommended ADHD dosage.
Avoid methylphenidate hydrochloride extended-release tablets use in patients with known structural cardiac abnormalities, cardiomyopathy, serious cardiac arrhythmia, coronary artery disease, or other serious cardiac disease.
Increased Blood Pressure and Heart Rate
CNS stimulants may cause an increase in blood pressure (mean increase approximately 2 to 4 mmHg) and heart rate (mean increase approximately 3 to 6 bpm). Some patients may have larger increases.
Monitor all methylphenidate hydrochloride extended-release tablets-treated patients for hypertension and tachycardia.
Psychiatric Adverse Reactions
Exacerbation of Pre-Existing Psychosis CNS stimulants may exacerbate symptoms of behavior disturbance and thought disorder in patients with a pre-existing psychotic disorder.
Induction of a Manic Episode in Patients with Bipolar Disorder CNS stimulants may induce a manic or mixed episode in patients. Prior to initiating methylphenidate hydrochloride extended-release tablets treatment, screen patients for risk factors for developing a manic episode (e.g., comorbid or history of depressive symptoms or a family history of suicide, bipolar disorder, or depression).
New Psychotic or Manic Symptoms CNS stimulants, at the recommended dosage, may cause psychotic or manic symptoms (e.g., hallucinations, delusional thinking, or mania) in patients without a prior history of psychotic illness or mania. In a pooled analysis of multiple short-term, placebo-controlled studies of CNS stimulants, psychotic or manic symptoms occurred in approximately 0.1% of CNS stimulant-treated patients, compared with 0% of placebo-treated patients. If such symptoms occur, consider discontinuing methylphenidate hydrochloride extended-release tablets.
Seizures
There is some clinical evidence that stimulants may lower the convulsive threshold in patients with prior history of seizures, in patients with prior EEG abnormalities in absence of seizures, and, very rarely, in patients without a history of seizures and no prior EEG evidence of seizures. In the presence of seizures, the drug should be discontinued.
Priapism
Prolonged and painful erections, sometimes requiring surgical intervention, have been reported with methylphenidate use in both adult and pediatric male patients [see Adverse Reactions (6.5 )] . Although priapism was not reported with methylphenidate initiation, it developed after some time on methylphenidate, often subsequent to an increase in dosage. Priapism also occurred during methylphenidate withdrawal (drug holidays or during discontinuation).
Methylphenidate hydrochloride extended-release tablets-treated patients who develop abnormally sustained or frequent and painful erections should seek immediate medical attention.
Peripheral Vasculopathy, including Raynaud’s Phenomenon
CNS stimulants, including methylphenidate hydrochloride extended-release tablets, used to treat ADHD are associated with peripheral vasculopathy, including Raynaud’s phenomenon. Signs and symptoms are usually intermittent and mild; however, sequelae have included digital ulceration and/or soft tissue breakdown. Effects of peripheral vasculopathy, including Raynaud’s phenomenon, were observed in postmarketing reports and at the therapeutic dosages of CNS stimulants in all age groups throughout the course of treatment. Signs and symptoms generally improved after dosage reduction or discontinuation of the CNS stimulant.
Careful observation for digital changes is necessary during methylphenidate hydrochloride extended-release tablets treatment. Further clinical evaluation (e.g., rheumatology referral) may be appropriate for methylphenidate hydrochloride extended-release tablets-treated patients who develop signs or symptoms of peripheral vasculopathy.
Long-Term Suppression of Growth in Pediatric Patients
CNS stimulants have been associated with weight loss and slowing of growth rate in pediatric patients.
Careful follow-up of weight and height in children ages 7 to 10 years who were randomized to either methylphenidate or nonmedication treatment groups over 14 months, as well as in naturalistic subgroups of newly methylphenidate-treated and nonmedication-treated children over 36 months (to the ages of 10 to 13 years), suggests that pediatric patients who received methylphenidate for 7 days per week throughout the year had a temporary slowing in growth rate (on average, a total of about 2 cm less growth in height and 2.7 kg less growth in weight over 3 years), without evidence of growth rebound during this development period. Closely monitor growth (weight and height) in methylphenidate hydrochloride extended-release tablets-treated pediatric patients. Pediatric patients who are not growing or gaining height or weight as expected may need to have their treatment interrupted.
Potential for Gastrointestinal Obstruction
Because the methylphenidate hydrochloride extended-release tablets are nondeformable and do not appreciably change in shape in the GI tract, methylphenidate hydrochloride extended-release tablets should not ordinarily be administered to patients with pre-existing severe gastrointestinal narrowing (pathologic or iatrogenic, for example: esophageal motility disorders, small bowel inflammatory disease, “short gut” syndrome due to adhesions or decreased transit time, past history of peritonitis, cystic fibrosis, chronic intestinal pseudo-obstruction, or Meckel’s diverticulum). There have been rare reports of obstructive symptoms in patients with known strictures in association with the ingestion of drugs in nondeformable controlled-release formulations. Due to the controlled-release design of the tablet, methylphenidate hydrochloride extended-release tablets should be used only in patients who are able to swallow the tablet whole [see Patient Counseling Information (17 )] .
Hematologic Monitoring
Periodic CBC, differential, and platelet counts are advised during prolonged therapy.
Acute Angle Closure Glaucoma
There have been rare reports of angle closure glaucoma associated with methylphenidate treatment.
Although the mechanism is not clear, methylphenidate hydrochloride extended-release tablets-treated patients considered at risk for acute angle closure glaucoma (e.g., patients with significant hyperopia) should be evaluated by an ophthalmologist.
Increased Intraocular Pressure and Glaucoma
There have been reports of an elevation of intraocular pressure (IOP) associated with methylphenidate treatment [see Adverse Reactions (6.5 )].
Prescribe methylphenidate hydrochloride extended-release tablets to patients with open-angle glaucoma or abnormally increased IOP only if the benefit of treatment is considered to outweigh the risk. Closely monitor methylphenidate hydrochloride extended-release tablets-treated patients with a history of abnormally increased IOP or open-angle glaucoma.
Motor and Verbal Tics, and Worsening of Tourette’s Syndrome
CNS stimulants, including methylphenidate, have been associated with the onset or exacerbation of motor and verbal tics [see Adverse Reactions (6.2 , 6.5 )] . Worsening of Tourette’s syndrome has also been reported .
Before initiating methylphenidate hydrochloride extended-release tablets, assess the family history and clinically evaluate patients for tics or Tourette’s syndrome. Regularly monitor methylphenidate hydrochloride extended-release tablets-treated patients for the emergence or worsening of tics or Tourette’s syndrome, and discontinue treatment if clinically appropriate.
ADVERSE REACTIONS
The following are discussed in more detail in other sections of the labeling:
Abuse, Misuse, and Addiction [see Boxed Warning, Warnings and Precautions (5.1 )]
Hypersensitivity to Methylphenidate [see Contraindications (4.1 )]
Monoamine Oxidase Inhibitors [see Contraindications (4.2 ) and Drug Interactions (7.1 )]
Risks to Patients with Serious Cardiac Disease [see Warnings and Precautions (5.2 )]
Increased Blood Pressure and Heart Rate [see Warnings and Precautions (5.3 )]
Psychiatric Adverse Reactions [see Warnings and Precautions (5.4 )]
Seizures [see Warnings and Precautions (5.5 )]
Priapism [see Warnings and Precautions (5.6 )]
Peripheral Vasculopathy, including Raynaud’s Phenomenon [see Warnings and Precautions (5.7 )]
Long-Term Suppression of Growth in Pediatric Patients [see Warnings and Precautions (5.8 )]
Potential for Gastrointestinal Obstruction [see Warnings and Precautions (5.9 )]
Hematologic Monitoring [see Warnings and Precautions (5.10 )]
Acute Angle Closure Glaucoma [see Warnings and Precautions (5.11 )]
Increased Intraocular Pressure and Glaucoma [see Warnings and Precautions (5.12 )]
Motor and Verbal Tics, and Worsening of Tourette’s Syndrome [see Warnings and Precautions (5.13 )]
The most common adverse reaction in double-blind clinical trials (>5%) in pediatric patients (children and adolescents) was abdominal pain upper. The most common adverse reactions in double-blind clinical trials (>5%) in adult patients were decreased appetite, headache, dry mouth, nausea, insomnia, anxiety, dizziness, weight decreased, irritability, and hyperhidrosis [see Adverse Reactions (6.1 )] .
The most common adverse reactions associated with discontinuation (≥1%) from either pediatric or adult clinical trials were anxiety, irritability, insomnia, and blood pressure increased [see Adverse Reactions (6.3 )] .
The development program for methylphenidate hydrochloride extended-release tablets included exposures in a total of 3906 participants in clinical trials. Children, adolescents, and adults with ADHD were evaluated in 6 controlled clinical studies and 11 open-label clinical studies (see Table 3). Safety was assessed by collecting adverse events, vital signs, weights, and ECGs, and by performing physical examinations and laboratory analyses.
| Patient Population | N | Dose Range |
| Children | 2216 | 18 to 54 mg once daily |
| Adolescents | 502 | 18 to 72 mg once daily |
| Adults | 1188 | 18 to 108 mg once daily |
Adverse events during exposure were obtained primarily by general inquiry and recorded by clinical investigators using their own terminology. Consequently, to provide a meaningful estimate of the proportion of individuals experiencing adverse events, events were grouped in standardized categories using MedDRA terminology.
The stated frequencies of adverse events represent the proportion of individuals who experienced, at least once, a treatment-emergent adverse event of the type listed. An event was considered treatment-emergent if it occurred for the first time or worsened while receiving therapy following baseline evaluation.
Throughout this section, adverse reactions are reported. Adverse reactions are adverse events that were considered to be reasonably associated with the use of methylphenidate hydrochloride extended-release tablets based on the comprehensive assessment of the available adverse event information. A causal association for methylphenidate hydrochloride extended-release tablets often cannot be reliably established in individual cases. Further, because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in clinical trials of another drug and may not reflect the rates observed in clinical practice.
The majority of adverse reactions were mild to moderate in severity.
Commonly Observed Adverse Reactions in Double-Blind, Placebo-Controlled Clinical Trials
Adverse reactions in either the pediatric or adult double-blind adverse reactions tables may be relevant for both patient populations.
Children and Adolescents Table 4 lists the adverse reactions reported in 1% or more of methylphenidate hydrochloride extended-release tablets-treated children and adolescent subjects in 4 placebo-controlled, double-blind clinical trials.
System/Organ Class Adverse Reaction | Methylphenidate Hydrochloride Extended-Release Tablets (n=321) % | Placebo (n=318) % |
Gastrointestinal Disorders | ||
Abdominal pain upper | 6.2 | 3.8 |
Vomiting General Disorders and Administration Site Conditions | 2.8 | 1.6 |
Pyrexia Infections and Infestations | 2.2 | 0.9 |
Nasopharyngitis Nervous System Disorders | 2.8 | 2.2 |
Dizziness Psychiatric Disorders | 1.9 | 0 |
Insomnia Terms of Initial insomnia (methylphenidate hydrochloride extended-release tablets=0.6%) and Insomnia (methylphenidate hydrochloride extended-release tablets=2.2%) are combined into Insomnia. Respiratory, Thoracic and Mediastinal Disorders | 2.8 | 0.3 |
Cough | 1.9 | 0.9 |
Oropharyngeal pain | 1.2 | 0.9 |
The majority of adverse reactions were mild to moderate in severity.
Adults Table 5 lists the adverse reactions reported in 1% or more of methylphenidate hydrochloride extended-release tablets-treated adults in 2 placebo-controlled, double-blind clinical trials.
System/Organ Class Adverse Reaction | Methylphenidate Hydrochloride Extended-Release Tablets (n=415) % | Placebo (n=212) % |
Cardiac Disorders | ||
Tachycardia | 4.8 | 0 |
Palpitations Ear and Labyrinth Disorders | 3.1 | 0.9 |
Vertigo Eye Disorders | 1.7 | 0 |
Vision blurred Gastrointestinal Disorders | 1.7 | 0.5 |
Dry mouth | 14.0 | 3.8 |
Nausea | 12.8 | 3.3 |
Dyspepsia | 2.2 | 0.9 |
Vomiting | 1.7 | 0.5 |
Constipation General Disorders and Administration Site Conditions | 1.4 | 0.9 |
Irritability Infections and Infestations | 5.8 | 1.4 |
Upper respiratory tract infection Investigations Weight decreased | 2.2 6.5 | 0.9 3.3 |
Metabolism and Nutrition Disorders | ||
Decreased appetite | 25.3 | 6.6 |
Anorexia Musculoskeletal and Connective Tissue Disorders | 1.7 | 0 |
Muscle tightness Nervous System Disorders | 1.9 | 0 |
Headache | 22.2 | 15.6 |
Dizziness | 6.7 | 5.2 |
Tremor | 2.7 | 0.5 |
Paresthesia | 1.2 | 0 |
Sedation | 1.2 | 0 |
Tension headache Psychiatric Disorders | 1.2 | 0.5 |
Insomnia | 12.3 | 6.1 |
Anxiety | 8.2 | 2.4 |
Initial insomnia | 4.3 | 2.8 |
Depressed mood | 3.9 | 1.4 |
Nervousness | 3.1 | 0.5 |
Restlessness | 3.1 | 0 |
Agitation | 2.2 | 0.5 |
Aggression | 1.7 | 0.5 |
Bruxism | 1.7 | 0.5 |
Depression | 1.7 | 0.9 |
Libido decreased | 1.7 | 0.5 |
Affect lability | 1.4 | 0.9 |
Confusional state | 1.2 | 0.5 |
Tension Respiratory, Thoracic and Mediastinal Disorders | 1.2 | 0.5 |
Oropharyngeal pain Skin and Subcutaneous Tissue Disorders | 1.7 | 1.4 |
Hyperhidrosis | 5.1 | 0.9 |
The majority of ADRs were mild to moderate in severity.
Other Adverse Reactions Observed in Methylphenidate Hydrochloride Extended-Release Tablets Clinical Trials
This section includes adverse reactions reported by methylphenidate hydrochloride extended-release tablets-treated subjects in double-blind trials that do not meet the criteria specified for Table 4 or Table 5 and all adverse reactions reported by methylphenidate hydrochloride extended-release tablets-treated subjects who participated in open-label and postmarketing clinical trials.
Blood and Lymphatic System Disorders: Leukopenia
Eye Disorders: Accommodation disorder, Dry eye
Vascular Disorders: Hot flush
Gastrointestinal Disorders: Abdominal discomfort, Abdominal pain, Diarrhea
General Disorders and Administrative Site Conditions: Asthenia, Fatigue, Feeling jittery, Thirst
Infections and Infestations: Sinusitis
Investigations: Alanine aminotransferase increased, Blood pressure increased, Cardiac murmur, Heart rate increased
Musculoskeletal and Connective Tissue Disorders: Muscle spasms
Nervous System Disorders: Lethargy, Psychomotor hyperactivity, Somnolence
Psychiatric Disorders: Anger, Hypervigilance, Mood altered, Mood swings, Panic attack, Sleep disorder, Tearfulness, Tic
Reproductive System and Breast Disorders: Erectile dysfunction
Respiratory, Thoracic and Mediastinal Disorders: Dyspnea
Skin and Subcutaneous Tissue Disorders: Rash, Rash macular
Vascular Disorders: Hypertension
Discontinuation Due to Adverse Reactions
Adverse reactions in the 4 placebo-controlled studies of children and adolescents leading to discontinuation occurred in 2 methylphenidate hydrochloride extended-release tablets patients (0.6%) including depressed mood (1, 0.3%) and headache and insomnia (1, 0.3%), and 6 placebo patients (1.9%) including headache and insomnia (1, 0.3%), irritability (2, 0.6%), headache (1, 0.3%), psychomotor hyperactivity (1, 0.3%), and tic (1, 0.3%).
In the 2 placebo-controlled studies of adults, 25 methylphenidate hydrochloride extended-release tablets patients (6.0%) and 6 placebo patients (2.8%) discontinued due to an adverse reaction. Those events with an incidence of >0.5% in the methylphenidate hydrochloride extended-release tablets patients included anxiety (1.7%), irritability (1.4%), blood pressure increased (1.0%), and nervousness (0.7%). In placebo patients, blood pressure increased and depressed mood had an incidence of >0.5% (0.9%).
In the 11 open-label studies of children, adolescents, and adults, 266 methylphenidate hydrochloride extended-release tablets patients (7.0%) discontinued due to an adverse reaction. Those events with an incidence of >0.5% included insomnia (1.2%), irritability (0.8%), anxiety (0.7%), decreased appetite (0.7%), and tic (0.6%).
Blood Pressure and Heart Rate Increases
In the laboratory classroom clinical trials in children (Studies 1 and 2), both methylphenidate hydrochloride extended-release tablets once daily and methylphenidate three times daily increased resting pulse by an average of 2 to 6 bpm and produced average increases of systolic and diastolic blood pressure of roughly 1 to 4 mm Hg during the day, relative to placebo. In the placebo-controlled adolescent trial (Study 4), mean increases from baseline in resting pulse rate were observed with methylphenidate hydrochloride extended-release tablets and placebo at the end of the double-blind phase (5 and 3 beats/minute, respectively). Mean increases from baseline in blood pressure at the end of the double-blind phase for methylphenidate hydrochloride extended-release tablets and placebo-treated patients were 0.7 and 0.7 mm Hg (systolic) and 2.6 and 1.4 mm Hg (diastolic), respectively. In one placebo-controlled study in adults (Study 6), dose-dependent mean increases of 3.9 to 9.8 bpm from baseline in standing pulse rate were observed with methylphenidate hydrochloride extended-release tablets at the end of the double-blind treatment vs. an increase of 2.7 beats/minute with placebo. Mean changes from baseline in standing blood pressure at the end of double-blind treatment ranged from 0.1 to 2.2 mm Hg (systolic) and -0.7 to 2.2 mm Hg (diastolic) for methylphenidate hydrochloride extended-release tablets and was 1.1 mm Hg (systolic) and -1.8 mm Hg (diastolic) for placebo. In a second placebo-controlled study in adults (Study 5), mean changes from baseline in resting pulse rate were observed for methylphenidate hydrochloride extended-release tablets and placebo at the end of the double-blind treatment (3.6 and -1.6 beats/minute, respectively). Mean changes from baseline in blood pressure at the end of the double-blind treatment for methylphenidate hydrochloride extended-release tablets and placebo-treated patients were -1.2 and -0.5 mm Hg (systolic) and 1.1 and 0.4 mm Hg (diastolic), respectively [see Warnings and Precautions (5.3 )] .
Postmarketing Experience
The following additional adverse reactions have been identified during postapproval use of methylphenidate hydrochloride extended-release tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency:
Blood and Lymphatic System Disorders: Pancytopenia, Thrombocytopenia, Thrombocytopenic purpura
Cardiac Disorders: Angina pectoris, Bradycardia, Extrasystoles, Supraventricular tachycardia, Ventricular extrasystoles
Eye Disorders: Diplopia, Increased intraocular pressure, Mydriasis, Visual impairment
General Disorders: Chest pain, Chest discomfort, Drug effect decreased, Hyperpyrexia, Therapeutic response decreased
Hepatobiliary Disorders: Hepatocellular injury, Acute hepatic failure
Immune System Disorders: Hypersensitivity reactions such as Angioedema, Anaphylactic reactions, Auricular swelling, Bullous conditions, Exfoliative conditions, Urticarias, Pruritus NEC, Rashes, Eruptions, and Exanthemas NEC
Investigations: Blood alkaline phosphatase increased, Blood bilirubin increased, Hepatic enzyme increased, Platelet count decreased, White blood cell count abnormal
Musculoskeletal, Connective Tissue and Bone Disorders: Arthralgia, Myalgia, Muscle twitching, Rhabdomyolysis
Nervous System Disorders: Convulsion, Grand mal convulsion, Dyskinesia, Serotonin syndrome in combination with serotonergic drugs, Motor and verbal tics
Psychiatric Disorders: Disorientation, Hallucination, Hallucination auditory, Hallucination visual, Mania, Logorrhea, Libido changes
Reproductive System and Breast Disorders: Priapism
Skin and Subcutaneous Tissue Disorders: Alopecia, Erythema
Vascular Disorders: Raynaud’s phenomenon
DRUG INTERACTIONS
MAO Inhibitors
Methylphenidate hydrochloride extended-release tablets should not be used in patients being treated (currently or within the preceding 2 weeks) with MAO inhibitors [see Contraindications (4.2 )] .
Vasopressor Agents
Because of possible increases in blood pressure, methylphenidate hydrochloride extended-release tablets should be used cautiously with vasopressor agents [see Warnings and Precautions (5.3 )] .
Coumarin Anticoagulants, Antidepressants, and Selective Serotonin Reuptake Inhibitors
Human pharmacologic studies have shown that methylphenidate may inhibit the metabolism of coumarin anticoagulants, anticonvulsants (e.g., phenobarbital, phenytoin, primidone), and some antidepressants (tricyclics and selective serotonin reuptake inhibitors). Downward dose adjustment of these drugs may be required when given concomitantly with methylphenidate. It may be necessary to adjust the dosage and monitor plasma drug concentrations (or, in the case of coumarin, coagulation times), when initiating or discontinuing concomitant methylphenidate.
Halogenated Anesthetics
Concomitant use of halogenated anesthetics and methylphenidate hydrochloride extended-release tablets may increase the risk of sudden blood pressure and heart rate increase during surgery. Monitor blood pressure and avoid use of methylphenidate hydrochloride extended-release tablets in patients being treated with anesthetics on the day of surgery.
Risperidone
Combined use of methylphenidate with risperidone when there is a change, whether an increase or decrease, in dosage of either or both medications, may increase the risk of extrapyramidal symptoms (EPS). Monitor for signs of EPS.
DESCRIPTION
Methylphenidate Hydrochloride Extended-Release Tablets USP are a central nervous system (CNS) stimulant. Methylphenidate hydrochloride extended-release tablets are available in four tablet strengths. Each extended-release tablet for once-a-day oral administration contains 27, 36, or 54 mg of methylphenidate HCl USP and is designed to have a 12-hour duration of effect. Chemically, methylphenidate HCl is d,l (racemic) methyl α-phenyl-2-piperidineacetate hydrochloride.
Its structural formula is:
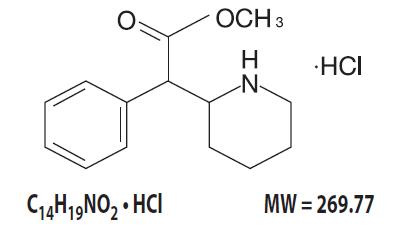
Methylphenidate HCl USP is a white, odorless crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone.
Methylphenidate hydrochloride extended-release tablets also contain the following inert ingredients: silicified microcrystalline cellulose, ethylcellulose, hydroxypropyl cellulose, hypromellose, magnesium stearate, medium-chain triglycerides, oleic acid, polyethylene glycol, polyvinyl alcohol, propylene glycol, talc, titanium dioxide, triacetin, shellac.
Each tablet also contains the following additional inert ingredients: 27 mg : yellow iron oxide, red iron oxide, black iron oxide; 36 mg : red iron oxide, black iron oxide; 54 mg : red iron oxide, black iron oxide.
System Components and Performance
Methylphenidate hydrochloride extended-release tablets use a precisely designed diffusion process to deliver methylphenidate hydrochloride at a controlled rate. The system comprises a polymeric core surrounded by a diffusion controlling membrane with an immediate release drug overcoat. In an aqueous environment, such as the gastrointestinal tract, the drug overcoat dissolves within one hour, providing an initial dose of methylphenidate. Water enters the tablet core through the diffusion controlling membrane, and methylphenidate is released in a controlled fashion resulting in C max at 6 to 7 hours. The non-absorbed components of the tablet pass through the gastrointestinal tract and are eliminated in the stool. It is possible that methylphenidate hydrochloride extended-release tablets may be visible on abdominal x-rays under certain circumstances, especially when digital enhancing techniques are utilized.
Meets USP Dissolution Test 4.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis In a lifetime carcinogenicity study carried out in B6C3F1 mice, methylphenidate caused an increase in hepatocellular adenomas and, in males only, an increase in hepatoblastomas at a daily dose of approximately 60 mg/kg/day. This dose is approximately 30 times and 4 times the maximum recommended human dose of methylphenidate hydrochloride extended-release tablets on a mg/kg and mg/m 2 basis, respectively. Hepatoblastoma is a relatively rare rodent malignant tumor type. There was no increase in total malignant hepatic tumors. The mouse strain used is sensitive to the development of hepatic tumors, and the significance of these results to humans is unknown.
Methylphenidate did not cause any increases in tumors in a lifetime carcinogenicity study carried out in F344 rats; the highest dose used was approximately 45 mg/kg/day, which is approximately 22 times and 5 times the maximum recommended human dose of methylphenidate hydrochloride extended-release tablets on a mg/kg and mg/m 2 basis, respectively.
In a 24-week carcinogenicity study in the transgenic mouse strain p53+/-, which is sensitive to genotoxic carcinogens, there was no evidence of carcinogenicity. Male and female mice were fed diets containing the same concentration of methylphenidate as in the lifetime carcinogenicity study; the high-dose groups were exposed to 60 to 74 mg/kg/day of methylphenidate.
Mutagenesis Methylphenidate was not mutagenic in the in vitro Ames reverse mutation assay or the in vitro mouse lymphoma cell forward mutation assay. Sister chromatid exchanges and chromosome aberrations were increased, indicative of a weak clastogenic response, in an in vitro assay in cultured Chinese Hamster Ovary cells. Methylphenidate was negative in vivo in males and females in the mouse bone marrow micronucleus assay.
Impairment of Fertility Methylphenidate did not impair fertility in male or female mice that were fed diets containing the drug in an 18-week Continuous Breeding study. The study was conducted at doses up to 160 mg/kg/day, approximately 80-fold and 8-fold the highest recommended human dose of methylphenidate hydrochloride extended-release tablets on a mg/kg and mg/m 2 basis, respectively.
CLINICAL STUDIES
Methylphenidate hydrochloride extended-release tablets were demonstrated to be effective in the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in 4 randomized, double-blind, placebo-controlled studies in children and adolescents and 2 double-blind placebo-controlled studies in adults who met the Diagnostic and Statistical Manual 4 th edition (DSM-IV) criteria for ADHD.
Children
Three double-blind, active- and placebo-controlled studies were conducted in 416 children aged 6 to 12 years. The controlled studies compared methylphenidate hydrochloride extended-release tablets given once daily (18, 36, or 54 mg), methylphenidate given three times daily over 12 hours (15, 30, or 45 mg total daily dose), and placebo in two single-center, 3-week crossover studies (Studies 1 and 2) and in a multicenter, 4-week, parallel-group comparison (Study 3). The primary comparison of interest in all three trials was methylphenidate hydrochloride extended-release tablets versus placebo.
Symptoms of ADHD were evaluated by community schoolteachers using the Inattention/Overactivity with Aggression (IOWA) Conners scale. Statistically significant reduction in the Inattention/Overactivity subscale versus placebo was shown consistently across all three controlled studies for methylphenidate hydrochloride extended-release tablets. The scores for methylphenidate hydrochloride extended-release tablets and placebo for the three studies are presented in Figure 2.
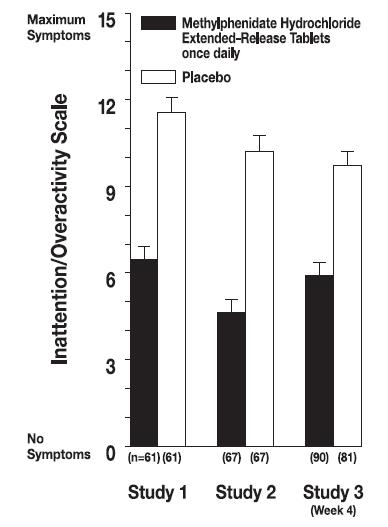
Figure 2. Mean Community School Teacher IOWA Conners Inattention/Overactivity Scores with methylphenidate hydrochloride extended-release tablets once-daily (18, 36, or 54 mg) and placebo. Studies 1 and 2 involved a 3-way crossover of 1 week per treatment arm. Study 3 involved 4 weeks of parallel-group treatments with a Last Observation Carried Forward analysis at week 4. Error bars represent the mean plus standard error of the mean.
In Studies 1 and 2, symptoms of ADHD were evaluated by laboratory schoolteachers using the SKAMP• laboratory school rating scale. The combined results from these two studies demonstrated statistically significant improvements in attention and behavior in patients treated with methylphenidate hydrochloride extended-release tablets versus placebo that were maintained through 12 hours after dosing. Figure 3 presents the laboratory schoolteacher SKAMP ratings for methylphenidate hydrochloride extended-release tablets and placebo.
•Swanson, Kotkin, Agler, M-Fynn and Pelham
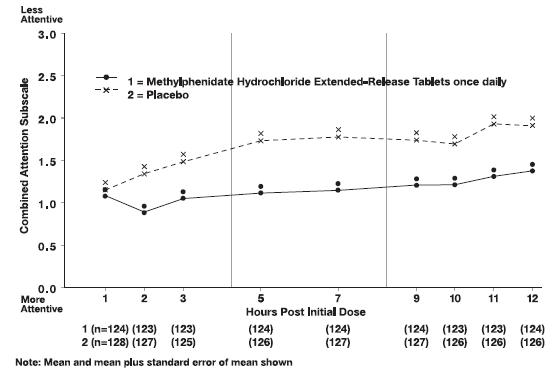
Figure 3. Laboratory School Teacher SKAMP Ratings: Mean (SEM) of Combined Attention (Studies 1 and 2)
Adolescents
In a randomized, double-blind, multicenter, placebo-controlled trial (Study 4) involving 177 patients, methylphenidate hydrochloride extended-release tablets were demonstrated to be effective in the treatment of ADHD in adolescents aged 13 to 18 years at doses up to 72 mg/day (1.4 mg/kg/day). Of 220 patients who entered an open 4-week titration phase, 177 were titrated to an individualized dose (maximum of 72 mg/day) based on meeting specific improvement criteria on the ADHD Rating Scale and the Global Assessment of Effectiveness with acceptable tolerability. Patients who met these criteria were then randomized to receive either their individualized dose of methylphenidate hydrochloride extended-release tablets (18 to 72 mg/day, n=87) or placebo (n=90) during a two-week double-blind phase. At the end of this phase, mean scores for the investigator rating on the ADHD Rating Scale demonstrated that methylphenidate hydrochloride extended-release tablets were statistically significantly superior to placebo.
Adults
Two double-blind, placebo-controlled studies were conducted in 627 adults aged 18 to 65 years. The controlled studies compared methylphenidate hydrochloride extended-release tablets administered once daily and placebo in a multicenter, parallel-group, 7-week dose-titration study (Study 5) (36 to 108 mg/day) and in a multicenter, parallel-group, 5-week, fixed-dose study (Study 6) (18, 36, and 72 mg/day).
Study 5 demonstrated the effectiveness of methylphenidate hydrochloride extended-release tablets in the treatment of ADHD in adults aged 18 to 65 years at doses from 36 mg/day to 108 mg/day based on the change from baseline to final study visit on the Adult ADHD Investigator Symptom Rating Scale (AISRS). Of 226 patients who entered the 7-week trial, 110 were randomized to methylphenidate hydrochloride extended-release tablets and 116 were randomized to placebo. Treatment was initiated at 36 mg/day and patients continued with incremental increases of 18 mg/day (36 to 108 mg/day) based on meeting specific improvement criteria with acceptable tolerability. At the final study visit, mean change scores (LS Mean, SEM) for the investigator rating on the AISRS demonstrated that methylphenidate hydrochloride extended-release tablets were statistically significantly superior to placebo.
Study 6 was a multicenter, double-blind, randomized, placebo-controlled, parallel-group, dose-response study (5-week duration) with 3 fixed-dose groups (18, 36, and 72 mg). Patients were randomized to receive methylphenidate hydrochloride extended-release tablets administered at doses of 18 mg (n=101), 36 mg (n=102), 72 mg/day (n=102), or placebo (n=96). All three doses of methylphenidate hydrochloride extended-release tablets were statistically significantly more effective than placebo in improving CAARS (Conners’ Adult ADHD Rating Scale) total scores at double-blind end point in adult subjects with ADHD.
HOW SUPPLIED/STORAGE AND HANDLING
Strength | Color | Tablet Description | Bottle Count | NDC |
27 mg | Gray | capsule shaped, imprinted with “M” in a box and “27” on one side in black ink | 100 | 0406-0127-01 |
36 mg | White | capsule shaped, imprinted with “M” in a box and “36” on one side in black ink | 100 | 0406-0136-01 |
54 mg | Red | capsule shaped, imprinted with “M” in a box and “54” on one side in black ink | 100 | 0406-0154-01 |
Storage and Handling Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Protect from humidity. Dispense in tight, light-resistant container with child-resistant closure.
Mechanism of Action
Methylphenidate HCl is a central nervous system (CNS) stimulant. The mode of therapeutic action in Attention Deficit Hyperactivity Disorder (ADHD) is not known. Methylphenidate is thought to block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.