Get your patient on Noxivent 102- Noxivent 102 gas (Noxivent 102)
Noxivent 102- Noxivent 102 gas prescribing information
INDICATIONS AND USAGE
Noxivent™ is a vasodilator indicated to improve oxygenation and reduce the need for extracorporeal membrane oxygenation in term and near-term (>34 weeks gestation) neonates with hypoxic respiratory failure associated with clinical or echocardiographic evidence of pulmonary hypertension in conjunction with ventilatory support and other appropriate agents.
DOSAGE FORMS AND STRENGTHS
Noxivent™ (nitric oxide) is a gas available in 100 ppm and 800 ppm concentrations (3 ).
USE IN SPECIFIC POPULATIONS
CONTRAINDICATIONS
Neonates dependent on right-to-left shunting of blood (4 ).
WARNINGS AND PRECAUTIONS
Rebound: Abrupt discontinuation of Noxivent™ may lead to worsening oxygenation and increasing pulmonary artery pressure (5.1 ).
Methemoglobinemia: Methemoglobin increases with the dose of nitric oxide; following discontinuation or reduction of nitric oxide, methemoglobin levels return to baseline over a period of hours (5.2 ).
Elevated NO 2 Levels: Monitor NO 2 levels (5.3 ).
Heart Failure: In patients with pre-existing left ventricular dysfunction, Noxivent™ may increase pulmonary capillary wedge pressure leading to pulmonary edema (5.4 ).
ADVERSE REACTIONS
The most common adverse reaction is hypotension. (6 ).
To report SUSPECTED ADVERSE REACTIONS, contact Linde, Inc. at 1-800-772-9247 and www.linde.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DRUG INTERACTIONS
Nitric oxide donor compounds may increase the risk of developing methemoglobinemia (7 ).
Revised: 08/2023
DESCRIPTION
Noxivent™ (nitric oxide gas) is a drug administered by inhalation. Nitric oxide, the active substance in Noxivent™, is a pulmonary vasodilator. Noxivent™ is a gaseous blend of nitric oxide and nitrogen (0.08% and 99.92%, respectively for 800 ppm; 0.01% and 99.99%, respectively for 100 ppm). Noxivent™ is supplied in aluminum cylinders as a compressed gas under high pressure (2000 pounds per square inch gauge [psig]).
The structural formula of nitric oxide (NO) is shown below:
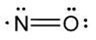
CLINICAL PHARMACOLOGY
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
No evidence of a carcinogenic effect was apparent, at inhalation exposures up to the recommended dose (20 ppm), in rats for 20 hr/day for up to two years. Higher exposures have not been investigated.
Nitric oxide has demonstrated genotoxicity in Salmonella (Ames Test), human lymphocytes, and after in vivo exposure in rats. There are no animal or human studies to evaluate nitric oxide for effects on fertility.
CLINICAL STUDIES
HOW SUPPLIED/STORAGE AND HANDLING
Noxivent™ (nitric oxide) is available in the following sizes:
| Size AD | Portable aluminum cylinders containing 362 liters at STP of nitric oxide gas in 800 ppm concentration in nitrogen (delivered volume 323 liters) (NDC 59579-102-02) |
| Size AQ | Aluminum cylinders containing 2154 liters at STP of nitric oxide gas in 800 ppm concentration in nitrogen (delivered volume 2082 liters) (NDC 59579-102-01) |
Store at 25°C (77°F) with excursions permitted between 15-30°C (59-86°F) [see USP Controlled Room Temperature].
All regulations concerning handling of pressure vessels must be followed.
Protect the cylinders from shocks, falls, oxidizing and flammable materials, moisture, and sources of heat or ignition.
Use of Noxivent cylinders in the MRI suite may create a projectile hazard.
Occupational Exposure
The exposure limit set by the Occupational Safety and Health Administration (OSHA) for nitric oxide is 25 ppm, and for NO 2 the limit is 5 ppm.