Get your patient on Orquidea - Norethindrone tablet (Norethindrone)
Orquidea - Norethindrone tablet prescribing information
Cigarette smoking greatly increases the possibility of suffering heart attacks and strokes. Women who use oral contraceptives are strongly advised not to smoke.
INDICATIONS AND USAGE
1. Indications. Progestin-only oral contraceptives are indicated for the prevention of pregnancy.
2. Efficacy. If used perfectly, the first-year failure rate for progestin-only oral contraceptives is 0.5%. However, the typical failure rate is estimated to be closer to 5%, due to late or omitted pills. The following table lists the pregnancy rates for users of all major methods of contraception.
Table 2: Percentage of Women Experiencing an Unintended Pregnancy During the First Year of Typical Use and the First Year of Perfect Use of Contraception and the Percentage Continuing Use at the End of the First Year. United States.
| Emergency Contraceptive Pills: Treatment initiated within 72 hours after unprotected intercourse reduces the risk of pregnancy by at least 75%. 9 | |||
| Lactational Amenorrhea Method: LAM is a highly effective, temporary method of contraception. 10 | |||
| Source: Trussell, J, Contraceptive Efficacy. In: Hatcher RA, Trussell J, Stewart F, Cates W, Stewart GK, Kowal D, Guest F, Contraceptive Technology: Seventeenth Revised Edition. New York NY: Irvington Publishers, 1998. | |||
| 1.000000000000000e+00 Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any reason. | |||
| 2.000000000000000e+00 Among couples who initiate use of a method (not necessarily for the first time), and who use it perfectly (both consistently and correctly), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason. | |||
| 3.000000000000000e+00 Among couples attempting to avoid pregnancy, the percentage who continue to use a method for one year. | |||
| 4.000000000000000e+00 The percentage of women becoming pregnant noted in columns (2) and (3) are based on data from populations where contraception is not used and from women who cease using contraception in order to become pregnant. Among such populations, about 89% become pregnant within one year. This estimate was lowered slightly (to 85%) to represent the percentage that would become pregnant within one year among women now relying on reversible methods of contraception if they abandoned contraception altogether. | |||
| 5.000000000000000e+00 Foams, creams, gels, vaginal suppositories, and vaginal film. | |||
| 6.000000000000000e+00 Cervical mucus (ovulation) method supplemented by calendar in the pre-ovulatory and basal body temperature in the post-ovulatory phases. | |||
| 7.000000000000000e+00 With spermicidal cream or jelly. | |||
| 8.000000000000000e+00 Without spermicides. | |||
| 9.000000000000000e+00 The treatment schedule is one dose within 72 hours after unprotected intercourse, and a second dose 12 hours after the first dose. The Food and Drug Administration has declared the following brands of oral contraceptives to be safe and effective for emergency contraception: Ovral ® (1 dose is 2 white pills), Alesse ® (1 dose is 5 pink pills), Nordette ® or Levlen ® (1 dose is 4 yellow pills). | |||
| 1.000000000000000e+01 However, to maintain effective protection against pregnancy, another method of contraception must be used as soon as menstruation resumes, the frequency or duration of breastfeeds is reduced, bottle feeds are introduced, or the baby reaches 6 months of age. | |||
% of Women Experiencing an Unintended Pregnancy within the First Year of Use | % of Women Continuing Use at One Year 3 | ||
Method (1) | Typical Use 1 (2) | Perfect Use 2 (3) | (4) |
Chance 4 | 85 | 85 | |
Spermicides 5 | 26 | 6 | 40 |
Periodic abstinence | 25 | 63 | |
Calendar | 9 | ||
Ovulation Method | 3 | ||
Sympto-Thermal 6 | 2 | ||
Post-Ovulation | 1 | ||
Cap 7 | |||
Parous Women | 40 | 26 | 42 |
Nulliparous Women | 20 | 9 | 56 |
Sponge | |||
Parous Women | 40 | 20 | 42 |
Nulliparous Women | 20 | 9 | 56 |
Diaphragm 7 | 20 | 6 | 56 |
Withdrawal | 19 | 4 | |
Condom 8 | |||
Female (Reality) | 21 | 5 | 56 |
Male | 14 | 3 | 61 |
Pill | 5 | 71 | |
Progestin only | 0.5 | ||
Combined | 0.1 | ||
IUDs | |||
Progesterone T | 2.0 | 1.5 | 81 |
Copper T380A | 0.8 | 0.6 | 78 |
LNg 20 | 0.1 | 0.1 | 81 |
Depo-Provera ® | 0.3 | 0.3 | 70 |
Levonorgestrel Implants (Norplant ® ) | 0.05 | 0.05 | 88 |
Female Sterilization | 0.5 | 0.5 | 100 |
Male Sterilization | 0.15 | 0.10 | 100 |
DOSAGE AND ADMINISTRATION
To achieve maximum contraceptive effectiveness, Orquidea must be taken exactly as directed. One tablet is taken every day, at the same time. Administration is continuous, with no interruption between pill packs. See PATIENT LABELING for detailed instructions.
CONTRAINDICATIONS
Progestin-only oral contraceptives tablets should not be used by women who currently have the following conditions:
Known or suspected pregnancy
Known or suspected carcinoma of the breast
Undiagnosed abnormal genital bleeding
Hypersensitivity to any component of this product
Benign or malignant liver tumors
Acute liver disease
ADVERSE REACTIONS
- Menstrual irregularity is the most frequently reported side effect.
- Frequent and irregular bleeding are common, while long duration of bleeding episodes and amenorrhea are less likely.
- Headache, breast tenderness, nausea, and dizziness are increased among progestin-only oral contraceptive users in some studies.
- Androgenic side effects such as acne, hirsutism, and weight gain occur rarely.
DESCRIPTION
Each peach Orquidea tablet provides a continuous oral contraceptive regimen of 0.35 mg norethindrone daily, and the inactive ingredients include corn starch, FD&C Yellow No. 6 Aluminum Lake, lactose monohydrate, magnesium stearate, and povidone.
The chemical name for norethindrone is 17-Hydroxy-19-Nor-17-alpha-pregn-4-en-20-yn-3-one. The structural formula follows:
Therapeutic class = oral contraceptive.
CLINICAL PHARMACOLOGY
Mode of Action
Norethindrone tablets progestin-only oral contraceptives prevent conception by suppressing ovulation in approximately half of users, thickening the cervical mucus to inhibit sperm penetration, lowering the mid-cycle LH and FSH peaks, slowing the movement of the ovum through the fallopian tubes, and altering the endometrium.
Pharmacokinetics
Absorption: Norethindrone is rapidly absorbed with maximum plasma concentrations occurring within 1 to 2 hours after norethindrone tablets administration (see Table 1 ). Norethindrone appears to be completely absorbed following oral administration; however, it is subject to first pass metabolism resulting in an absolute bioavailability of approximately 65%.
Figure 1: Mean ± SD Norethindrone Plasma Concentrations Following Norethindrone Tablets Administration.
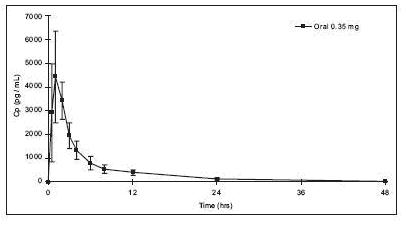
Peak plasma concentrations occur approximately 1 hour after administration (mean T max 1.2 hours). The mean (SD) C max was 4816.8 (1532.6) pg/mL and generally occurred within 1 hour (mean) of tablet administration, ranging from 0.5 to 2 hours. The mean (SD) C avg was 885 (250) pg/mL, however, the mean concentration at 24 hrs was 130 (47) pg/mL.
Table 1 provides summary statistics of the pharmacokinetic parameters associated with single dose norethindrone tablets administration.
Table 1: Mean ± SD Pharmacokinetic Parameters Following Single Dose Administration of Norethindrone Tablets in 12 Healthy Female Subjects Under Fasting Conditions
Pharmacokinetic Parameter | Norethindrone 0.35 mg |
T max (hr) | 1.2 ± 0.5 |
C max (pg/mL) | 4817 ± 1533 |
AUC (0-48) (pg·h/mL) | 21233 ± 6002 |
t½ (h) | 7.7 ± 0.5 |
The food effect on the rate and extent of norethindrone absorption after norethindrone tablets administration have not been evaluated.
Distribution: Following oral administration, norethindrone is 36% bound to sex hormone-binding globulin (SHBG) and 61% bound to albumin. Volume of distribution of norethindrone is approximately 4 L/kg.
Metabolism: Norethindrone undergoes extensive biotransformation, primarily via reduction, followed by sulfate and glucuronide conjugation; less than 5% of a norethindrone dose is excreted unchanged; greater than 50% and 20 to 40% of a dose is excreted in urine and feces, respectively. The majority of metabolites in the circulation are sulfate, with glucuronides accounting for most of the urinary metabolites.
Excretion: Plasma clearance rate for norethindrone has been estimated to be approximately 600 L/day. Norethindrone is excreted in both urine and feces, primarily as metabolites. The mean terminal elimination half-life of norethindrone following single dose administration of norethindrone tablet is approximately 8 hours.
HOW SUPPLIED
Orquidea (norethindrone tablets USP, 0.35 mg) is available in a blister pack containing 28 peach, round, flat faced, beveled edge tablets, debossed 213 on one side and other side plain.
Carton of 3 tri-laminated aluminium pouches (NDC 70700-316-85), each pouch contains one blister pack of 28 tablets.
STORAGE
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]