Get your patient on Oxazepam - Oxazepam capsule (Oxazepam)
Oxazepam - Oxazepam capsule prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; AND DEPENDENCE AND WITHDRAWAL REACTIONS
• Concomitant use of benzodiazepines and opioids may result in profound sedati on, respiratory depression, coma, and death
Reserve concomitant prescribing of these drugs in patients for whom alternati ve treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS and PRECAUTIONS ).
• The use of benzodiazepines, including Oxazepam, exposes users to risks of abuse, misu se, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing Oxazepam and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (see WARNINGS ).
• The continued use of benzodiazepines, including Oxazepam, may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of Oxazepam after continued use may precipitate acute withdrawa l reactions, which can be life-threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue Oxazepam or reduce the dosage (see DOSAGE AND ADMINISTRATION and WARNINGS ).
DOSAGE AND ADMINISTRATION
Because of the flexibility of this product and the range of emotional disturbances responsive to it, dosage should be individualized for maximum beneficial effects.
| OXAZEPAM | Usual Dose |
| Mild-to-moderate anxiety, with associated tension, irritability, agitation, or related symptoms of functional origin secondary to organic | 10 to 15 mg. 3 or 4 times daily |
| Severe anxiety syndromes, agitation, or anxiety associated with depression | 15 to 30 mg. 3 or 4 times daily |
| Older patients with anxiety, tension, irritability, and agitation | Initial dosage: 10 mg, 3 times daily. If necessary, increase cautiously to 15 mg, 3 or 4 times daily. |
| Alcoholics with acute inebriation, tremulousness, or anxiety on withdrawal | 15 to 30 mg, 3 or 4 times daily |
This product is not indicated in pediatric patients under 6 years of age. Absolute dosage for pediatric patients 6 to 12 years of age is not established.
Discontinuation or Dosage Reduction of Oxazepam
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue Oxazepam or reduce the dosage. If a patient develops withdrawal reactions, consider pausing the taper or increasing the dosage to the previous tapered dosage level. Subsequently decrease the dosage more slowly (see WARNINGS : Dependence and Withdrawal Reactions and DRUG ABUSE AND DEPENDENCE: Dependence ).
CONTRAINDICATIONS
History of previous hypersensitivity reaction to oxazepam. Oxazepam is not indicated in psychoses.
ADVERSE REACTIONS
The necessity for discontinuation of therapy due to undesirable effects has been rare. Transient, mild drowsiness is commonly seen in the first few days of therapy. If it persists, the dosage should be reduced. In few instances, dizziness, vertigo, headache, and rarely syncope have occurred either alone or together with drowsiness. Mild paradoxical reactions, i.e., excitement, stimulation of affect, have been reported in psychiatric patients; these reactions may be secondary to relief of anxiety and usually appear in the first two weeks of therapy.
Other side effects occurring during oxazepam therapy include rare instances of minor diffuse skin rashes-morbilliform, urticarial, and maculopapular nausea, lethargy, edema, slurred speech, tremor and altered libido. Such side effects have been infrequent and are generally controlled with reduction of dosage. A case of an extensive fixed drug eruption also has been reported.
Although rare, leukopenia and hepatic dysfunction including jaundice have been reported during therapy. Periodic blood counts and liver-function tests are advisable. Ataxia with oxazepam has been reported in rare instances and does not appear to be specifically related to dose or age.
Although the following side reactions have not as yet been reported with oxazepam, they have occurred with related compounds (chlordiazepoxide and diazepam): paradoxical excitation with severe rage reactions, hallucinations, menstrual irregularities, change in EEG pattern, blood dyscrasias including agranulocytosis, blurred vision, diplopia, incontinence, stupor, disorientation, fever, and euphoria.
Transient amnesia or memory impairment has been reported in association with the use of benzodiazepines.
Drug Interactions:
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABAA sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid- related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and monitor patients closely for respiratory depression and sedation.
DESCRIPTION
Oxazepam, USP is the first of a chemical series of compounds known as the 3- hydroxybenzodiazepinones. A therapeutic agent providing versatility and flexibility in control of common emotional disturbances, this product exerts prompt action in a wide variety of disorders associated with anxiety, tension, agitation, and irritability, and anxiety associated with depression. In tolerance and toxicity studies on several animal species, this product reveals significantly greater safety factors than related compounds (chlordiazepoxide and diazepam) and manifests a wide separation of effective doses and doses inducing side effects.
Oxazepam, is 7-chloro-1,3-dihydro-3-hydroxy-5-phenyl-2 H -1,4-benzodiazepin-2-one and has the following structural formula:
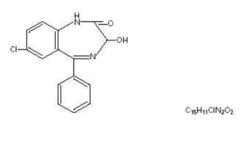 MW 286.72
MW 286.72
Oxazepam is a white-crystalline powder.
Each capsule for oral administration contain 10 mg, 15 mg or 30 mg of oxazepam.
Inactive ingredients: croscarmellose sodium, D&C yellow #10, FD&C blue #1, FD&C blue # 2, FD&C red # 40, ferric oxide black, gelatin, lactose monohydrate, magnesium stearate, microcrystalline cellulose, n-butyl alcohol, propylene glycol, sda-3a alcohol, shellac, sodium lauryl sulfate, titanium dioxide and other inert ingredients.
The 15 mg capsule also contains: FD&C yellow # 6.
The 30 mg capsule also contains: D&C red # 28.
CLINICAL PHARMACOLOGY
Pharmacokinetic testing in 12 volunteers demonstrated that a single 30 mg dose of a capsule, tablet or suspension will result in equivalent extent of absorption.
For the capsule and tablet, peak plasma levels averaged 450 mg/mL and were observed to occur about 3 hours after dosing. The mean elimination half-life for oxazepam was approximately 8.2 hours (range 5.7 to 10.9 hours).
This product has a single, major inactive metabolite in man, a glucuronide excreted in the urine.
Age (<80 years old) does not appear to have a clinically significant effect on oxazepam kinetics. A statistically significant increase in elimination half-life in the very elderly (> 80 years of age) as compared to younger subjects has been reported, due to a 30% increase in volume of distribution, as well as a 50% reduction in unbound clearance of oxazepam in the very elderly (see PRECAUTIONS, Geriatric Use ) .
HOW SUPPLIED
Oxazepam capsules, USP are available as:
10 mg - Each #4 white opaque/white opaque gelatin capsule, printed FBP660 in black ink on the cap and body contains 10 mg of Oxazepam, USP. Oxazepam Capsules are supplied in bottles of 100 (NDC 52817-290-10)
15 mg - Each #4 red opaque/red opaque gelatin capsule, printed FBP661in black ink on the cap and body, contains 15 mg of Oxazepam, USP. Oxazepam Capsules are supplied in bottles of 100 (NDC 52817-291-10)
30 mg - Each #4 maroon opaque/maroon opaque gelatin capsule, printed FBP662 in black ink on the cap and body, contains 30 mg of Oxazepam, USP. Oxazepam Capsules are supplied in bottles of 100 (NDC 52817-292-10)
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture.
Dispense in a tight, light-resistant container.