Get your patient on Podofilox - Podofilox gel (Podofilox)
Podofilox - Podofilox gel prescribing information
INDICATIONS AND USAGE
Podofilox gel is indicated for the topical treatment of anogenital warts (external genital warts and perianal warts). This product is not indicated in the treatment of mucous membrane warts (see PRECAUTIONS ).
Diagnosis
Although anogenital warts have a characteristic appearance, histopathologic confirmation should be obtained if there is any doubt of the diagnosis. Differentiating warts from squamous cell carcinoma and "Bowenoid papulosis" is of particular concern. Squamous cell carcinoma may also be associated with human papillomavirus which should not be treated with podofilox gel.
DOSAGE AND ADMINISTRATION
The prescriber should ensure that the patient is fully aware of the correct method of therapy and identify which specific warts should be treated.
Apply twice daily for 3 consecutive days, then discontinue for 4 consecutive days. This one-week cycle of treatment may be repeated until there is no visible wart tissue or for a maximum of four cycles. If there is incomplete response after four treatment cycles, discontinue treatment and consider alternative treatment. Safety and effectiveness of more than four treatment cycles has not been established. There is no evidence to suggest that more frequent application will increase efficacy, but additional applications would be expected to increase the rate of local adverse reactions and systemic absorption.
Podofilox gel should be applied to the warts with the applicator tip or finger. Application on the surrounding normal tissue should be minimized. Treatment should be limited to 10 cm 2 or less of wart tissue and to no more than 0.5 gram of the gel per day.
Care should be taken to allow the gel to dry before allowing the return of opposing skin surfaces to their normal positions. Patients should be instructed to wash their hands thoroughly before and after each application.
CONTRAINDICATIONS
Podofilox gel is contraindicated for patients who develop hypersensitivity or intolerance to any components of the formulation.
ADVERSE REACTIONS
In clinical trials with podofilox gel, the following local adverse reactions were reported during the treatment of anogenital warts. The severity of local adverse reactions were predominantly mild or moderate and did not increase during the treatment period. Severe reactions were most frequent within the first 2 weeks of treatment.
Adverse Reaction | Mild | Moderate | Severe |
Inflammation | 32.2% | 30.4% | 9.3% |
Burning | 37.1% | 25.9% | 11.5% |
Erosion | 27.0% | 20.8% | 8.9% |
Pain | 23.7% | 20.4% | 11.5% |
Itching | 32.2% | 16.0% | 7.8% |
Bleeding | 19.2% | 3.0% | 0.7% |
Other local adverse reactions reported included stinging (7%), and erythema (5%); less commonly reported local adverse events included desquamation, scabbing, discoloration, tenderness, dryness, crusting, fissures, soreness, ulceration, swelling/edema, tingling, rash, and blisters.
The most common systemic adverse event reported during the clinical studies was headache (7%).
To report SUSPECTED ADVERSE REACTIONS, contact Padagis at 1-866-634-9120 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Podofilox is an antimitotic drug which can be chemically synthesized or purified from the plant families Coniferae and Berberidaceae (e.g. species of Juniperus and Podophyllum ). Podofilox gel is formulated for topical administration. Each gram of gel contains 5 mg of podofilox in a buffered alcoholic gel containing alcohol (81% v/v), butylated hydroxytoluene, glycerin, hydroxypropyl cellulose, lactic acid, and sodium lactate.
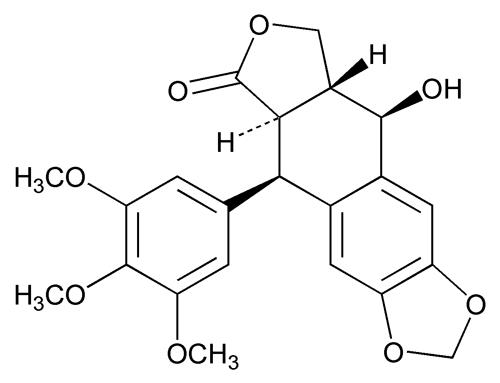
Podofilox has a molecular weight of 414.4 daltons, and is soluble in alcohol and sparingly soluble in water. Its chemical name is [5R,-(5α, 5aβ, 8aα, 9α]-5,8,8a,9-tetrahydro-9-hydroxy- 5-(3,4,5-trimethoxyphenyl) furo[3',4':6,7]naphtho-[2,3,-d]-1,3-dioxol-6(5aH)-one.
Podofilox has the following structural formula:
CLINICAL PHARMACOLOGY
Mechanism of Action
Treatment of anogenital warts with podofilox results in necrosis of visible wart tissue. The exact mechanism of action is unknown.
Pharmacokinetics
In systemic absorption studies in 52 patients, topical application of 0.05 mL of an ethanolic solution containing 0.5% podofilox to external genitalia did not result in detectable serum levels. Applications of 0.1 to 1.5 mL resulted in peak serum levels of 1 to 17 ng/mL one to two hours after application. The elimination half-life ranged from 1.0 to 4.5 hours. The drug was not found to accumulate after multiple treatments 1 .
CLINICAL STUDIES
In the first multicenter clinical study in 326 patients with anogenital warts, podofilox gel and its vehicle were applied in a double-blind fashion to comparable patient groups. Of the 260 patients with efficacy data, 176 were treated with podofilox gel. Patients applied podofilox gel twice daily for three consecutive days followed by a 4 day “rest” period.
At the end of 4 weeks, 38.4% of the patients had complete clearing of the wart tissue when treated with podofilox gel.
In the second multicenter clinical trial in 108 evaluable patients with anogenital warts, podofilox topical solution was compared with podofilox gel for efficacy. As in the first clinical trial, patients applied podofilox gel twice daily for three consecutive days followed by a four day “rest” period.
Similar clearance rates were observed. At the end of 4 weeks, 25.6% of the patients had complete clearing of the wart tissue when treated with podofilox gel.
HOW SUPPLIED
Podofilox Gel 0.5% is supplied as 3.5 grams of clear gel in aluminum tubes with an applicator tip. NDC 0574-0621-05. Store at 20-25°C (68-77°F). [See USP controlled room temperature.] Avoid excessive heat. Do not freeze.
Keep out of reach of children.
Rx only