Get your patient on Prazosin Hydrochloride - Prazosin Hydrochloride capsule (Prazosin Hydrochloride)
Prazosin Hydrochloride - Prazosin Hydrochloride capsule prescribing information
INDICATIONS & USAGE
Prazosin Hydrochloride Capsules, USP is indicated for the treatment of hypertension, to lower blood pressure. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes, including this drug.
Control of high blood pressure should be part of comprehensive cardiovascular risk management, including, as appropriate, lipid control, diabetes management, antithrombotic therapy, smoking cessation, exercise, and limited sodium intake. Many patients will require more than one drug to achieve blood pressure goals. For specific advice on goals and management, see published guidelines, such as those of the National High Blood Pressure Education Program's Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC).
Numerous antihypertensive drugs, from a variety of pharmacologic classes and with different mechanisms of action, have been shown in randomized controlled trials to reduce cardiovascular morbidity and mortality, and it can be concluded that it is blood pressure reduction, and not some other pharmacologic property of the drugs, that is largely responsible for those benefits. The largest and most consistent cardiovascular outcome benefit has been a reduction in the risk of stroke, but reductions in myocardial infarction and cardiovascular mortality also have been seen regularly.
Elevated systolic or diastolic pressure causes increased cardiovascular risk, and the absolute risk increase per mmHg is greater at higher blood pressures, so that even modest reductions of severe hypertension can provide substantial benefit. Relative risk reduction from blood pressure reduction is similar across populations with varying absolute risk, so the absolute benefit is greater in patients who are at higher risk independent of their hypertension (for example, patients with diabetes or hyperlipidemia), and such patients would be expected to benefit from more aggressive treatment to a lower blood pressure goal.
Some antihypertensive drugs have smaller blood pressure effects (as monotherapy) in black patients, and many antihypertensive drugs have additional approved indications and effects (e.g., on angina, heart failure, or diabetic kidney disease). These considerations may guide selection of therapy.
Prazosin Hydrochloride Capsules, USP can be used alone or in combination with other antihypertensive drugs such as diuretics or beta-adrenergic blocking agents.
DOSAGE & ADMINISTRATION
The dose of prazosin hydrochloride capsules should be adjusted according to the patient’s individual blood pressure response. The following is a guide to its administration:
Initial Dose
1 mg two or three times a day (see WARNINGS).
Maintenance Dose
Dosage may be slowly increased to a total daily dose of 20 mg given in divided doses. The therapeutic dosages most commonly employed have ranged from 6 mg to 15 mg daily given in divided doses. Doses higher than 20 mg usually do not increase efficacy, however a few patients may benefit from further increases up to a daily dose of 40 mg given in divided doses. After initial titration some patients can be maintained adequately on a twice daily dosage regimen.
Use With Other Drugs
When adding a diuretic or other antihypertensive agent, the dose of prazosin hydrochloride capsules should be reduced to 1 mg or 2 mg three times a day and retitration then carried out.
Concomitant administration of prazosin hydrochloride capsules with a PDE-5 inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension; therefore, PDE-5 inhibitor therapy should be initiated at the lowest dose in patients taking prazosin hydrochloride capsules.
CONTRAINDICATIONS
Prazosin Hydrochloride Capsules are contraindicated in patients with known sensitivity to quinazolines, prazosin, or any of the inert ingredients.
ADVERSE REACTIONS
Clinical trials were conducted on more than 900 patients. During these trials and subsequent marketing experience, the most frequent reactions associated with prazosin hydrochloride therapy are: dizziness 10.3%, headache 7.8%, drowsiness 7.6%, lack of energy 6.9%, weakness 6.5%, palpitations 5.3%, and nausea 4.9%. In most instances, side effects have disappeared with continued therapy or have been tolerated with no decrease in dose of drug.
Less frequent adverse reactions which are reported to occur in 1% to 4% of patients are:
Gastrointestinal: vomiting, diarrhea, constipation.
Cardiovascular: edema, orthostatic hypotension, dyspnea, syncope.
Central Nervous System: vertigo, depression, nervousness.
Dermatologic: rash.
Genitourinary: urinary frequency.
EENT: blurred vision, reddened sclera, epistaxis, dry mouth, nasal congestion.
In addition, fewer than 1% of patients have reported the following (in some instances, exact causal relationships have not been established):
Gastrointestinal: abdominal discomfort and/or pain, liver function abnormalities, pancreatitis.
Cardiovascular: tachycardia.
Central Nervous System: paresthesia, hallucinations.
Dermatologic: pruritus, alopecia, lichen planus.
Genitourinary: incontinence, impotence, priapism.
EENT: tinnitus.
Other: diaphoresis, fever, positive ANA titer, arthralgia.
Single reports of pigmentary mottling and serous retinopathy, and a few reports of cataract development or disappearance have been reported. In these instances, the exact causal relationship has not been established because the baseline observations were frequently inadequate.
In more specific slit-lamp and funduscopic studies, which included adequate baseline examinations, no drug-related abnormal ophthalmological findings have been reported.
Literature reports exist associating prazosin hydrochloride therapy with a worsening of preexisting narcolepsy. A causal relationship is uncertain in these cases.
In post-marketing experience, the following adverse events have been reported:
Autonomic Nervous System: flushing.
Body As A Whole: allergic reaction, asthenia, malaise, pain.
Cardiovascular, General: angina pectoris, hypotension.
Endocrine: gynecomastia.
Heart Rate/Rhythm: bradycardia.
Psychiatric: insomnia.
Skin/Appendages: urticaria.
Vascular (Extracardiac): vasculitis.
Vision: eye pain.
Special Senses: During cataract surgery, a variant of small pupil syndrome known as Intraoperative Floppy Iris Syndrome (IFIS) has been reported in association with alpha-1 blocker therapy (see PRECAUTIONS).
DRUG INTERACTIONS
Prazosin hydrochloride has been administered without any adverse drug interaction in limited clinical experience to date with the following: (1) cardiac glycosides-digitalis and digoxin; (2) hypoglycemics-insulin, chlorpropamide, phenformin, tolazamide, and tolbutamide; (3) tranquilizers and sedatives-chlordiazepoxide, diazepam, and phenobarbital; (4) antigout-allopurinol, colchicine, and probenecid; (5) antiarrhythmics-procainamide, propranolol (see WARNINGS however), and quinidine; and (6) analgesics, antipyretics and anti-inflammatories-propoxyphene, aspirin, indomethacin, and phenylbutazone.
Addition of a diuretic or other antihypertensive agent to prazosin hydrochloride has been shown to cause an additive hypotensive effect. This effect can be minimized by reducing the prazosin hydrochloride dose to 1 mg to 2 mg three times a day, by introducing additional antihypertensive drugs cautiously, and then by retitrating prazosin hydrochloride based on clinical response.
Concomitant administration of prazosin hydrochloride with a phosphodiesterase-5 (PDE-5) inhibitor can result in additive blood pressure lowering effects and symptomatic hypotension (see DOSAGE AND ADMINISTRATION).
DESCRIPTION
Prazosin hydrochloride, USP a quinazoline derivative, is the first of a new chemical class of antihypertensives. It is the hydrochloride salt of 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-(2-furoyl) piperazine and its structural formula is: 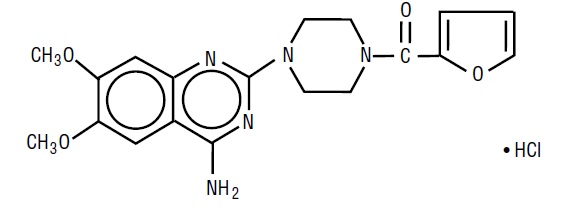 Molecular formula C 19 H 21 N 5 O 4 •HCl
Molecular formula C 19 H 21 N 5 O 4 •HCl
It is a white to tan powder, slightly soluble in water, practically insoluble in chloroform and acetone and has a molecular weight of 419.87. Each capsule, for oral administration, contains prazosin hydrochloride, USP equivalent (as the polyhydrate) to 1 mg, 2 mg or 5 mg of prazosin.
Inert ingredients in the formulations are: colloidal silicon dioxide, lactose monohydrate, lactose anhydrous, magnesium stearate and microcrystalline cellulose. The empty hard gelatin capsules contain black iron oxide, gelatin, red iron oxide, titanium dioxide and yellow iron oxide. In addition, the 1 mg empty gelatin capsules contain D&C Yellow No. 10 and FD&C Green No. 3; the 2 mg empty gelatin capsules contain D&C Red No. 28, D&C Yellow No. 10, FD&C Blue No. 1 and FD&C Red No. 40; and the 5 mg empty gelatin capsules contain FD&C Blue No. 1.
The capsules shells are imprinted in edible ink which contains concentrated ammonium solution, potassium hydroxide, propylene glycol, shellac and titanium dioxide.
CLINICAL PHARMACOLOGY
The exact mechanism of the hypotensive action of prazosin is unknown. Prazosin causes a decrease in total peripheral resistance and was originally thought to have a direct relaxant action on vascular smooth muscle. Recent animal studies, however, have suggested that the vasodilator effect of prazosin is also related to blockade of postsynaptic alpha- adrenoceptors. The results of dog forelimb experiments demonstrate that the peripheral vasodilator effect of prazosin is confined mainly to the level of the resistance vessels (arterioles). Unlike conventional alpha-blockers, the antihypertensive action of prazosin is usually not accompanied by a reflex tachycardia. Tolerance has not been observed to develop in long term therapy.
Hemodynamic studies have been carried out in man following acute single dose administration and during the course of long term maintenance therapy. The results confirm that the therapeutic effect is a fall in blood pressure unaccompanied by a clinically significant change in cardiac output, heart rate, renal blood flow and glomerular filtration rate. There is no measurable negative chronotropic effect.
In clinical studies to date, prazosin hydrochloride has not increased plasma renin activity.
In man, blood pressure is lowered in both the supine and standing positions. This effect is most pronounced on the diastolic blood pressure.
Following oral administration, human plasma concentrations reach a peak at about three hours with a plasma half-life of two to three hours. The drug is highly bound to plasma protein. Bioavailability studies have demonstrated that the total absorption relative to the drug in a 20% alcoholic solution is 90%, resulting in peak levels approximately 65% of that of the drug in solution. Animal studies indicate that prazosin hydrochloride is extensively metabolized, primarily by demethylation and conjugation, and excreted mainly via bile and feces. Less extensive human studies suggest similar metabolism and excretion in man.
In clinical studies in which lipid profiles were followed, there were generally no adverse changes noted between pre- and post-treatment lipid levels.
HOW SUPPLIED
Prazosin Hydrochloride Capsules, USP 1 mg for oral administration containing prazosin hydrochloride, USP equivalent to 1 mg of prazosin, are supplied as follows:
Hard gelatin capsule shell with dark green colored cap and light brown colored body, imprinted with white ink as 019 on cap and novitium on body, filled with white to off-white powder blend.
NDC 70954- 019 -50 Bottles of 90 capsules
NDC 70954- 019 -10 Bottles of 100 capsules
NDC 70954- 019 -20 Bottles of 1000 capsules
NDC 70954- 019 -40 Unit dose blister packages of 100 (10 cards of 10 capsules each)
Prazosin Hydrochloride Capsules, USP 2 mg for oral administration containing prazosin hydrochloride, USP equivalent to 2 mg of prazosin, are supplied as follows:
Hard gelatin capsule shell with brown colored cap and light brown colored body, imprinted with white ink as 020 on cap and novitium on body, filled with white to off-white powder blend.
NDC 70954- 020- 10 Bottles of 100 capsules
NDC 70954- 020 -20 Bottles of 1000 capsules
NDC 70954- 020 -40 Unit dose blister packages of 100 (10 cards of 10 capsules each)
Prazosin Hydrochloride Capsules, USP 5 mg for oral administration containing prazosin hydrochloride, USP equivalent to 5 mg of prazosin, are supplied as follows:
Hard gelatin capsule shell with light blue colored cap and light brown colored body, imprinted with white ink as 021 on cap and novitium on body, filled with white to off-white powder blend.
NDC 70954- 021 -10 Bottles of 100 capsules
NDC 70954- 021 -20 Bottles of 250 capsules
NDC 70954- 021 -40 Unit dose blister packages of 100 (10 cards of 10 capsules each)
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from moisture and light.
Dispense in a tight, light-resistant container as defined in the USP.
Manufactured by:
Esjay Pharma Private Limited
Sipcot Industrial Park, Kanchipuram,
Tamilnadu, India – 602117
Distributed by:
Novitium Pharma LLC
East Windsor, NJ 08520
P3319/00/25
Rev. 05/25