Get your patient on Progesterone - Progesterone injection, Solution (Progesterone)
Progesterone - Progesterone injection, Solution prescribing information
INDICATIONS AND USAGE
This drug is indicated in amenorrhea and abnormal uterine bleeding due to hormonal imbalance in the absence of organic pathology, such as submucous fibroids or uterine cancer.
DOSAGE AND ADMINISTRATION
Progesterone is administered by intramuscular injection. It differs from other commonly used steroids in that it is irritating at the place of injection.
Amenorrhea: Five to 10 mg are given for six to eight consecutive days. If there has been sufficient ovarian activity to produce a proliferative endometrium, one can expect withdrawal bleeding forty-eight to seventy-two hours after the last injection. This may be followed by spontaneous normal cycles.
Functional Uterine Bleeding: Five to 10 mg are given daily for six doses. Bleeding may be expected to cease within six days. When estrogen is given as well, the administration of progesterone is begun after two weeks of estrogen therapy. If menstrual flow begins during the course of injections of progesterone, they are discontinued.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever the solution and container permit.
CONTRAINDICATIONS
- Current or past history of thrombophlebitis, thromboembolic disorders, or cerebral apoplexy.
- Liver dysfunction or disease.
- Known or suspected malignancy of breast or genital organs.
- Undiagnosed vaginal bleeding.
- Missed abortion.
- Known sensitivity to progesterone injection.
- Known sensitivity to sesame oil/seeds.
ADVERSE REACTIONS
Breakthrough bleeding; spotting; change in menstrual flow; amenorrhea; edema; change in weight (increase or decrease); changes in cervical erosion and cervical secretions; cholestatic jaundice; breast tenderness and galactorrhea; pain, irritation, and/or redness at the injection area; skin sensitivity reactions consisting of urticaria, pruritus, edema and generalized rash; acne, alopecia and hirsutism; rash (allergic) with and without pruritus; anaphylactoid reactions (including eosinophilic pneumonia); mental depression; pyrexia; insomnia; nausea; and somnolence.
A statistically significant association has been demonstrated between use of estrogen-progestin combination drugs and pulmonary embolism and cerebral thrombosis and embolism. For this reason patients on progestin therapy should be carefully observed. There is also evidence suggestive of an association with neuro-ocular lesions, e.g., retinal thrombosis and optic neuritis.
The following adverse reactions have been observed in patients receiving estrogen-progestin combination drugs: rise in blood pressure in susceptible individual, premenstrual syndrome, changes in libido, changes in appetite, cystitis-like syndrome, headache, nervousness, fatigue, backache, hirsutism, loss of scalp hair, erythema multiforme, erythema nodosum, hemorrhagic eruption, itching, and dizziness.
The following laboratory results may be altered by the use of estrogen-progestin combination drugs: increased sulfobromophthalein retention and other hepatic function tests; coagulation tests: increase in prothrombin factors VII, VIII, IX, and X; metyrapone test; pregnanediol determinations; thyroid function: increase in PBI and butanol extractable protein bound iodine, and decrease in T 3 uptake values.
DESCRIPTION
Progesterone injection, a progestin, is a sterile solution of progesterone in a suitable vegetable oil available for intramuscular use.
Progesterone occurs as a white or creamy white, crystalline powder. It is odorless and is stable in air. Practically insoluble in water, it is soluble in alcohol, acetone, and dioxane and sparingly soluble in vegetable oils.
It has the following structural formula:
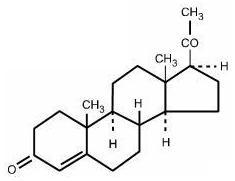
C 21 H 30 O 2 M.W. 314.47
Pregn-4-ene-3, 20-dione
Each mL contains: Progesterone 50 mg, Benzyl Alcohol 10% as preservative in Sesame Oil q.s.
CLINICAL PHARMACOLOGY
Transforms proliferative endometrium into secretory endometrium.
Inhibits (at the usual dose range) the secretion of pituitary gonadotropins, which in turn prevents follicular maturation and ovulation.
Pharmacokinetics and Metabolism:
Absorption: After intramuscular administration of 10 mg of progesterone in oil, maximum plasma concentrations (geometric mean of 7 ng/mL) were reached within approximately 8 hours after injection and plasma concentrations remained above baseline for about 24 hours after injection. Injection of 10, 25, and 50 mg resulted in geometric mean values for maximum plasma concentration (C MAX ) of 7, 28, and 50 ng/mL, respectively.
Distribution: Progesterone is extensively bound to plasma proteins, primarily albumin (50-54%) and cortisol-binding protein (43-48%).
Metabolism: Progesterone is metabolized primarily in the liver by reduction to pregnanediol, pregnanetriol, and pregnanolone. Subsequent conjugation results in the formation of glucuronide and sulfate metabolites. The mean plasma metabolic clearance rate in cycling women is 2510 ± 135 (SEM) L/day.
Excretion: The glucuronide and sulfate conjugates of pregnanediol and pregnanolone are excreted in the urine and bile. Progesterone metabolites which are excreted in the bile may undergo enterohepatic recycling or may be excreted in the feces.
The pharmacokinetic data was determined in a small number of patients, limiting the precision in which population values may be estimated.
Special Populations:
Renal Insufficiency: The safety and effectiveness in patients with renal insufficiency have not been established. Since progesterone metabolites are excreted mainly by the kidneys, progesterone should be administered with caution and careful monitoring in this patient population (see PRECAUTIONS ).
Hepatic Insufficiency: The safety and effectiveness in patients with hepatic insufficiency have not been established. Since progesterone is metabolized by the liver, use in patients with liver dysfunction or disease is contraindicated (see CONTRAINDICATIONS ).
Drug Interactions:
The metabolism of progesterone by human liver microsomes was inhibited by ketoconazole (IC 50 < 01 μM). Ketoconazole is a known inhibitor of cytochrome P450 3A4 and these data suggest that ketoconazole or other known inhibitors of this enzyme may increase the bioavailability of progesterone. The clinical relevance of the in vitro findings is unknown.
HOW SUPPLIED
Progesterone Injection USP, 500 mg/10 mL (50 mg/mL) is available in 10 mL multiple dose vials, individually boxed.
(NDC 0591-3128-79)
Store at 20°-25°C (68°-77°F). [See USP controlled room temperature.]
Rx only
For all medical inquiries contact: ACTAVIS Medical Communications Parsippany, NJ 07054 800-272-5525 Distributed By: Actavis Pharma, Inc. Parsippany, NJ 07054 USA
Manufactured By: Hikma Farmacêutica Fervença 2705-906 Terrugem SNT Portugal
Revised:August 2018