Get your patient on Sevenfact - Coagulation Factor Viia Recombinant Human (Coagulation Factor Viia Recombinant Human)
Sevenfact - Coagulation Factor Viia Recombinant Human prescribing information
WARNING: THROMBOSIS
● Serious arterial and venous thrombotic events may occur following administration of SEVENFACT ® . [See Warnings and Precautions (5.1 )]
● Discuss the risks and explain the signs and symptoms of thrombotic and thromboembolic events to patients who will receive SEVENFACT ® .
● Monitor patients for signs or symptoms of activation of the coagulation system and for thrombosis.
1 INDICATIONS AND USAGE
SEVENFACT is indicated for the treatment and control of bleeding episodes occurring in adults and adolescents 12 years of age and older with hemophilia A or B with inhibitors.
Limitation s of Use:
SEVENFACT is not indicated for the treatment of patients with congenital Factor VII deficiency.
2 DOSAGE AND ADMINISTRATION
For i ntravenous u se after reconstitution only .
| Type of Bleeding | Dosing Regimen Recommendation |
| For Mild or Moderate bleeds | 75 mcg/kg repeated every 3 hours until hemostasis is achieved or Initial dose of 225 mcg/kg. If hemostasis is not achieved within 9 hours, additional 75 mcg/kg doses may be administered every 3 hours as needed to achieve hemostasis |
| For Severe bleeds | 225 mcg/kg, followed if necessary 6 hours later with 75 mcg/kg every 2 hours |
Consider alternative treatments if successful control of bleeding does not occur within 24 hours of the first administration of SEVENFACT.
Dose
For intravenous use after reconstitution only.
- Dose and duration of SEVENFACT depend on the location and severity of the bleeding, need for urgent hemostasis, frequency of administration, and known patient responsiveness to FVIIa-containing bypassing agents during prior bleeding events. Treatment with SEVENFACT should be initiated as soon as a bleeding event occurs.
- The dose, frequency, and duration of SEVENFACT therapy should be based on the patient’s clinical response and hemostasis evaluation.
- The use of laboratory assessment(s) of coagulation (PT/INR, aPTT, FVII:C) does not necessarily correlate with or predict the hemostatic effectiveness of SEVENFACT.
- Dose adjustment may be required if the patient has received other procoagulant therapies prior to treatment with SEVENFACT.
Based on the clinical trial program for SEVENFACT, the recommended initial dose should be adjusted based on the criteria provided in Table 1.
| Type of Bleeding | Dosing Regimen Recommendation | Duration of Therapy |
| Mild and Moderate Joint, superficial muscle, soft tissue and mucous membranes. | 75 mcg/kg repeated every 3 hours until hemostasis is achieved or Initial dose of 225 mcg/kg. If hemostasis is not achieved within 9 hours, additional 75 mcg/kg doses may be administered every 3 hours as needed to achieve hemostasis. Consider alternative treatments if successful control of bleeding does not occur within 24 hours of the first administration of SEVENFACT. | Continue therapy to support healing and prevent recurrent hemorrhage after hemostasis to maintain the hemostatic plug. The site and severity of bleeding should determine therapy duration. |
| Severe Life or limb threatening hemorrhage, iliopsoas and deep muscle with neurovascular injury, retroperitoneum, intracranial, or gastrointestinal. Patients should seek immediate medical care if signs or symptoms of severe bleeding occur in the home setting. | 225 mcg/kg initially, followed if necessary 6 hours later with 75 mcg/kg every 2 hours until hemostasis is achieved. Subsequent Dosing: After achieving hemostasis, base the decision for dosing on clinical assessment and the type of bleeding. Consider the risk of thrombosis with subsequent dosing after achieving hemostatic efficacy. | Continue therapy to support healing and prevent recurrent hemorrhage. The site and severity of bleeding and the use of other procoagulant therapies should determine therapy duration. |
Reconstitution
- Follow the procedures below for reconstitution of SEVENFACT.
- Calculate the amount of SEVENFACT required and select the appropriate SEVENFACT packages containing the matching pre-filled syringe of sterile Water for Injection, and the vial adapters.
- Reconstitute each vial with the pre-filled syringe provided with each vial of SEVENFACT.
Overview of SEVENFACT P ackage :
Figure 1 Vial with SEVENFACT L yophilized P owder
Lyophilized Powder Drug Vial

Figure 2 Syringe P lunger R od and P re-filled S yringe with Water for Injection D iluent
Syringe Plunger Rod Pre-filled S yringe with Diluent
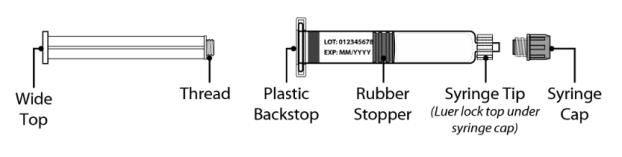
Figure 3 SEVENFACT 1 mg and 2 mg V ial A dapter and SEVENFACT 5 mg V ial A dapter
Vial Adapters• and Packaging
1 mg and 2 mg V ial A dapter 5 mg V ial A dapter
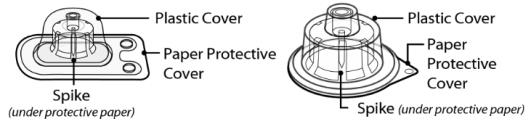
• N ote : Each SEVENFACT kit will contain only one vial adapter.
The instructions below serve as a general guideline for reconstitution of SEVENFACT.
Reconstitution :
- Based on the prescribed dose, take out the number of SEVENFACT kits (each kit containing one vial of SEVENFACT powder and one pre-filled Water for Injection diluent syringe with one vial adapter for needleless reconstitution), an infusion set (not supplied in the kit) and an alcohol swab (not supplied in the kit). Check the expiration date on the side of the box(es) for the SEVENFACT kit(s).
- Always use aseptic technique. Wash your hands with soap and water and dry them using a clean towel or air dry.
- Take out the contents of one kit and one alcohol swab. Place items on a clean surface.
- Inspect all contents of the kit. Make sure each vial has a matching colored syringe.
- Bring SEVENFACT (lyophilized powder) and the specified pre-filled syringe (diluent) to room temperature. The specified volume of diluent corresponding to the amount of SEVENFACT is as follows: 1 mg (1000 micrograms) vial + 1.1 mL Water for Injection diluent in pre-filled syringe 2 mg (2000 micrograms) vial + 2.2 mL Water for Injection diluent in pre-filled syringe 5 mg (5000 micrograms) vial + 5.2 mL Water for Injection diluent in pre-filled syringe
- Remove the plastic cap from the SEVENFACT vials to expose the central portion of the rubber stopper. Cleanse the rubber stoppers with an alcohol swab and allow to dry prior to use.
- Peel back the protective paper from the vial adapter. Do not remove the vial adapter from the package.
- Place the SEVENFACT vial on a flat surface. While holding the vial adapter package, place the vial adapter over the SEVENFACT vial and press down firmly on the package until the vial adapter spike breaks through the rubber stopper.
- Lightly squeeze the plastic cover and lift up to remove it from the vial adapter. Note: the 5 mg vial adapter may not sit flat against the vial, but it is fully functional.
- Remove the syringe cap from the pre-filled syringe by holding the syringe body with one hand to unscrew the syringe cap (turn to the left).
- While holding the edges of the vial adapter, screw on the pre-filled syringe (turn to the right) a few turns until it starts to tighten.
- Insert the plunger rod into the syringe, then screw a few turns (turn to the right) so that the plunger rod is attached to the gray rubber stopper in the syringe.
- Push the plunger rod to slowly inject all the diluent into the vial. Keep the plunger rod pressed down and swirl the vial gently until the powder is dissolved.
- The reconstituted solution is clear to slightly opaque. All powder must be mixed with no particles floating in the liquid.
- Without withdrawing any drug back into the syringe, unscrew the syringe from the vial adapter (turn to the left) until it is completely detached.
- Withdraw the liquid drug from the vial(s), using an infusion syringe provided by the pharmacy; the syringe should be large enough to hold the prescribed dose.
- The reconstituted solution should be stored in the vial at room temperature, but can be stored between 36 o F to 86 o F (2 o C to 30 o C) for up to 4 hours after reconstitution. After reconstitution with the specified volume of diluent, each vial contains approximately 1 mg per mL SEVENFACT (1000 micrograms per mL).
Administration
For Intravenous Use Only .
- Visually inspect the reconstituted solution for particulate matter and discoloration prior to administration. Do not use if particulate matter or discoloration is observed.
- Do not freeze reconstituted solution or store it in a syringe.
- SEVENFACT must be infused within 4 hours after reconstitution.
- SEVENFACT should be infused over 2 minutes or less as a bolus intravenous infusion.
- Do not mix with other infusion solutions.
- Any unused solution should be discarded 4 hours after reconstitution.
3 DOSAGE FORMS AND STRENGTHS
SEVENFACT is a white to off-white lyophilized powder for reconstitution in a colorless solution for injection. It is supplied in single-dose vial sizes containing 1 mg, 2 mg or 5 mg of coagulation factor VIIa (recombinant)-jncw.
The diluent for reconstitution of SEVENFACT is supplied in single-dose prefilled glass syringes containing 1.1 mL, 2.2 mL or 5.2 mL sterile Water for Injection. It is a clear colorless solution.
After reconstitution with the appropriate volume of Water for Injection diluent, each mL of SEVENFACT contains 1 mg per mL of coagulation factor VIIa (recombinant)-jncw (1,000 micrograms per mL).
8 USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no adequate and well-controlled studies using SEVENFACT in pregnant women to determine whether there is a drug-associated risk. Animal studies evaluating the embryo-fetal teratogenic potential of SEVENFACT have not been conducted. It is unknown whether SEVENFACT can cause fetal harm when administered to a pregnant woman or can affect fertility.
In the U.S. general population, the estimated background risks of major birth defect and miscarriage in clinically recognized pregnancies are 2-4% and 15-20%, respectively.
Lactation
Risk Summary
There is no information regarding the presence of SEVENFACT in human milk, the effect on the breastfed infant, and the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for SEVENFACT and any potential adverse effects on the breastfed infant from SEVENFACT or from the underlying maternal condition.
Females and Males of Reproductive Potential
Risk Summary
In Study 1, male patients cautioned to avoid sexual activity without condoms received SEVENFACT for the treatment of bleeding episodes. No pregnancies from sexual partners were reported. The relative benefits of SEVENFACT should be weighed against any potential risk arising from exposure in sexually active patients.
All clinical studies of SEVENFACT were performed on males, as males are predominantly affected with the congenital form of hemophilia. No adverse effects on the mating index, fertility, or conception rate were observed following administration of SEVENFACT at dose levels up to 13-fold higher than the highest recommended human dose in healthy male rats prior to and during cohabitation with healthy untreated female rats [ See Carcinogenesis, Mutagenesis, Impairment of Fertility (13.1 ) ].
Pediatric Use
The safety and effectiveness of SEVENFACT have been established for pediatric patients ≥ 12 years of age for the treatment and control of bleeding episodes. Limited clinical data for SEVENFACT in adolescents (≥12 to <18 years) were collected in an adult and adolescent study (Study 1). A total of five patients were dosed with SEVENFACT. These five patients were treated for a total of 79 bleeding episodes (all mild or moderate) that occurred while patients were still under 18 years of age. Hemostatic efficacy in this subgroup (n=5) was comparable to efficacy observed in the overall population [ See Clinical Studies (14 ) ].
The safety and effectiveness of SEVENFACT for the treatment and control of bleeding episodes have not been established in children < 12 years of age. Effectiveness was not demonstrated in a trial of 25 pediatric patients 6 months to < 12 years of age. The safety and effectiveness of SEVENFACT in infants less than 6 months of age have not been evaluated.
Geriatric Use
Safety and effectiveness of SEVENFACT in patients >65 years of age have not been evaluated in clinical trials (Study 1 and Study 2). The presence of age-related comorbidities and the attendant risks associated with thrombotic and thromboembolic events should be considered when administering SEVENFACT to patients older than 50 years of age.
4 CONTRAINDICATIONS
SEVENFACT is contraindicated in patients with:
- known allergy to rabbits or rabbit proteins. Exposure to SEVENFACT in these patients can result in severe hypersensitivity reaction.
- severe hypersensitivity reaction to SEVENFACT or any of its components. Exposure to SEVENFACT in these patients can result in severe hypersensitivity reaction.
5 WARNINGS AND PRECAUTIONS
- Patients with hemophilia A or B with inhibitors who have other risk factors for thrombosis may be at increased risk of serious arterial and venous thrombotic events (5.1 ).
- Hypersensitivity reactions, including anaphylaxis, are possible with SEVENFACT. Should symptoms occur, patients should discontinue SEVENFACT and seek appropriate medical intervention (5.2 ).
Thrombosis
Serious arterial and venous thrombosis can occur with coagulation factor VIIa containing products including SEVENFACT.
The following patients may have increased risk of thrombosis with use of SEVENFACT:
- History of congenital or acquired hemophilia receiving concomitant treatment with aPCC/PCC (activated or non-activated prothrombin complex) or other hemostatic agents
- History of atherosclerotic disease, coronary artery disease, cerebrovascular disease, crush injury, septicemia, or thromboembolism.
- Monitor patients who receive SEVENFACT for the development of signs and symptoms of activation of the coagulation system or thrombosis. When there is laboratory confirmation of intravascular coagulation or presence of clinical thrombosis, reduce the dose of SEVENFACT or stop treatment, depending on the patient’s condition.
Hypersensitivity and Infusion-Related Reactions
Hypersensitivity and infusion-related reactions including anaphylaxis can occur with coagulation factor VIIa containing products including SEVENFACT. Sign and symptoms may include hives, itching, rash, difficulty breathing, swelling around the mouth and throat, tightness of the chest, wheezing, dizziness or fainting, and low blood pressure. Patients with known IgE-based hypersensitivity to casein may be at higher risk of hypersensitivity reactions. In the event of hypersensitivity or infusion-related reactions, discontinue SEVENFACT and manage according to clinical practice guideline.
Neutralizing Antibodies
Neutralizing antibodies may occur with the use of SEVENFACT. If treatment with SEVENFACT does not result in adequate hemostasis, then suspect development of neutralizing antibody as the possible cause and perform testing as clinically indicated.
Neutralizing antibodies to other Factor VIIa-containing products have been observed in congenital Factor VII-deficient patients. SEVENFACT has not been studied in this patient population. [ See limitation of use statement under Indications and Usage (1 )] .
Laboratory Tests
Laboratory coagulation parameters (PT/INR, aPTT, FVII:C) do not correlate with clinical response to SEVENFACT treatment.
6 ADVERSE REACTIONS
The most common adverse reactions (incidence ≥1%) were headache, dizziness, infusion-site discomfort, infusion-site hematoma, infusion-related reaction, and fever (6 ).
To report SUSPECTED ADVERSE REACTIONS, contact HEMA Biologics at 855-718-HEMA (4362) or FDA at 1-800-FDA-1088 or https://www.fda.gov/medwatch.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety database described in this section reflect exposure to SEVENFACT in two clinical studies, Study 1 and Study 2. A total of 42 patients with Hemophilia A or B with or without inhibitors received SEVENFACT: 27 patients in Study 1 at doses 75 mcg/kg and 225 mcg/kg and 15 patients in Study 2 at three escalating dose levels 25 mcg/kg, 75 mcg/kg and 225 mcg/kg [see Clinical Studies (14 )] .
The most common adverse reactions (incidence ≥1%) reported in clinical trials for SEVENFACT were headache, dizziness, infusion-site discomfort, infusion-site hematoma, infusion-related reaction, and fever.
Adverse reactions reported in the two clinical studies are shown in Table 2.
| Preferred Terms | Number of Patient Adverse Reactions (% Incidence Rate) Study 2 (N=15) | Number of A dverse R eactions • Study 2 | Number of Patient Adverse Reactions (% Incidence Rate) Study 1 (N=27) | Number of A dverse R eactions • Study 1 |
| Infusion site discomfort | - | - | 1 (4) | 4 |
| Infusion site hematoma | - | - | 1 (4) | 2 |
| Dizziness | 1 (7) | 2 | - | - |
| Headache | 1 (7) | 1 | - | - |
| Body temperature increase | - | - | 1 (4) | 1 |
| Infusion related reaction•• | 1 (7) | 1 | - | - |
• Three patients experienced adverse reactions in Study 2 and two patients experienced adverse reactions in Study 1.
•• Symptoms resolved without any intervention and did not recur with subsequent administration.
7 DRUG INTERACTIONS
Clinical experience with pharmacologic use of FVIIa-containing products indicates an elevated risk of serious thrombotic events when used simultaneously with activated prothrombin complex concentrates.
11 DESCRIPTION
SEVENFACT [coagulation factor VIIa (recombinant)-jncw] is a sterile, white to off-white lyophilized powder in a single-dose vial containing either 1 mg, 2 mg or 5 mg of coagulation factor VIIa (recombinant)-jncw as the active ingredient. SEVENFACT is to be reconstituted with Sterile Water for Injection in a pre-filled syringe supplied with the product. The reconstituted product is a clear to slightly opaque solution of coagulation factor VIIa (recombinant)-jncw at a concentration of 1 mg of protein per mL with a pH of approximately 6.0. SEVENFACT is formulated with arginine, isoleucine, citrate, glycine, lysine and polysorbate 80. It does not contain any antimicrobial preservatives nor human or bovine plasma-derived proteins.
The active ingredient in SEVENFACT, activated coagulation Factor VII, is a glycoprotein of 406 amino acids with a molecular weight of approximately 50 kilodaltons. The amino acid sequence of activated coagulation Factor VII is identical to that of human plasma-derived Factor VIIa. It is >99% pure with a nominal specific activity of 45,000 IU/mg of protein when tested against the World Health Organization international standard for human Factor VIIa activity.
SEVENFACT is produced by recombinant DNA technology using genetically engineered rabbits into which the DNA coding sequence for human Factor VII has been introduced. Human Factor VII is expressed in the rabbit mammary gland and secreted into the milk. During purification and processing, Factor VII is enzymatically converted to activated Factor VII. The manufacturing process of SEVENFACT includes specific steps to reduce impurities. SEVENFACT may contain trace amounts of rabbit proteins. The purification process also includes steps that are validated to inactivate or remove viruses, such as Xenotropic murine leukemia virus (X-MuLV), bovine viral diarrhea virus (BVDV), Pseudorabies virus (PRV), Feline Calicivirus (FCV), and Porcine Parvovirus (PPV).
12 CLINICAL PHARMACOLOGY
Mechanism of Action
The active ingredient in SEVENFACT is a recombinant analog of human Factor VIIa, a vitamin K-dependent coagulation factor. In the presence of both calcium and phospholipids, Factor VIIa in a complex with tissue factor (TF) activates Factor X to Factor Xa, directly bypassing the reactions that require Factor VIII or Factor IX. Activation of Factor X to Factor Xa initiates the common pathway of the coagulation cascade in which prothrombin is activated to thrombin, which then converts fibrinogen to fibrin to form a hemostatic plug, thereby achieving clot formation at the site of hemorrhage (hemostasis). This process may also occur in the absence of TF on the surface of activated platelets.
Pharmacodynamics
Laboratory assessment of coagulation does not necessarily correlate with or predict the hemostatic effectiveness of SEVENFACT.
SEVENFACT demonstrated a dose and concentration-dependent pharmacodynamics effect on the coagulation system, including shortening of the activated partial thromboplastin time (aPTT) and the prothrombin time (PT), and increasing the thrombin generation with platelets (TGT) and the maximum clot firmness (ROTEM-FIBTEM test).
Pharmacokinetics
Pharmacokinetic (PK) evaluation was conducted in an open-label, randomized, dose-comparison study to evaluate safety and pharmacokinetic of SEVENFACT in 28 adult patients (ages 18-75 yrs) with hemophilia A with or without inhibitors who were not actively bleeding. Patients received a single intravenous administration of either 75 mcg/kg or 225 mcg/kg dose of SEVENFACT. The PK parameters evaluated were maximum plasma concentration (C max ) of FVIIa (ng/mL), clearance (L/h), volume of distribution (Vss (L)), area under the plasma concentration-time curve from Time 0 (dosing) to infinity (AUC 0-inf ) (ng•h/mL), and half-life time (t 1/2 (h)), calculated from non-compartmental analysis (NCA) PK analyses using a validated activity assay.
| PK Parameter ( Arithmetic Mean [ CV% ] ) | C max ( ng/mL ) | Clearance (L/h) | Vss (L) | AUC 0-inf (ng•h/mL) | t 1/2 (h) |
| 75 mcg/kg (n=14) | 938 (37) | 5.1 (37) | 8.2 (37) | 1108 (447) | 2.3 (16) |
| 225 mcg/kg (n=14) | 3211 (23) | 4.5 (20) | 7.0 (22) | 3571 (26) | 2.0 (8) |
Five minutes after infusion, plasma SEVENFACT levels were 938 ng/mL and 3211 ng/mL for 75 mcg/kg and 225 mcg/kg dose groups respectively. Observed plasma concentration-time profiles show a biexponential decay from the maximal concentration to return to baseline approximately 8 hours post-administration. Exposure PK parameters C max and AUC 0-inf suggested dose dependence over the ranges studied. The clearance of SEVENFACT was approximately 5.1 L/h and 4.5 L/h for 75 mcg/kg and 225 mcg/kg dose levels respectively. Half-life time was approximately 2 hours at both levels. Results were based on Non-Compartmental Analysis. (Table 3)
Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of SEVENFACT or of other Factor VIIa containing products.
In Study 1, two out of 27 patients had a positive screening assay for anti-SEVENFACT antibody at baseline, prior to exposure to SEVENFACT, and at follow-up visits. One of these two patients had a transient SEVENFACT antibody with an additional confirmatory test for anti-SEVENFACT antibody, which was confirmed as non-neutralizing.
In Study 2, five of 15 patients tested positive for anti-SEVENFACT antibody using a screening assay. The confirmatory assay was negative for all patients at all time points.
No patient developed anti-rabbit milk protein antibodies during treatment with SEVENFACT.
There was no identified clinically significant effect of anti-drug antibodies on pharmacokinetics, pharmacodynamics, safety, or effectiveness of SEVENFACT over the study duration.
13 NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in animals to evaluate the potential effect of SEVENFACT on carcinogenesis or mutagenesis were not conducted. Male rats intravenously infused with SEVENFACT at 0.1, 0.3, 1 and 3 mg/kg/day (up to 13-fold higher than the highest recommended human dose of 225 mcg/kg), beginning 28 days before cohabitation and during cohabitation did not display adverse effects for the mating index, fertility, or conception rate. Comparing animals administered vehicle to animals administered SEVENFACT, there were no differences in sperm motility, morphology, or concentration.
Animal Toxicology and/or Pharmacology
The no observed adverse effect level (NOAEL) in 28-day repeat-dose toxicity studies in male Sprague-Dawley rats and Cynomolgus monkeys was 1 mg/kg/day, which is 4-fold higher than the highest recommended human dose. Anti-drug antibody formation was observed by Day 29 in both animal species. The NOAEL in a 13-week repeat-dose toxicity study in male and female Cynomolgus monkeys was 1 mg/kg/day. Anti-drug antibody formation was observed in all animals by Day 28. Antibody presence was not associated with any adverse effects in either animal species.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety database described in this section reflect exposure to SEVENFACT in two clinical studies, Study 1 and Study 2. A total of 42 patients with Hemophilia A or B with or without inhibitors received SEVENFACT: 27 patients in Study 1 at doses 75 mcg/kg and 225 mcg/kg and 15 patients in Study 2 at three escalating dose levels 25 mcg/kg, 75 mcg/kg and 225 mcg/kg [see Clinical Studies (14 )] .
The most common adverse reactions (incidence ≥1%) reported in clinical trials for SEVENFACT were headache, dizziness, infusion-site discomfort, infusion-site hematoma, infusion-related reaction, and fever.
Adverse reactions reported in the two clinical studies are shown in Table 2.
| Preferred Terms | Number of Patient Adverse Reactions (% Incidence Rate) Study 2 (N=15) | Number of A dverse R eactions • Study 2 | Number of Patient Adverse Reactions (% Incidence Rate) Study 1 (N=27) | Number of A dverse R eactions • Study 1 |
| Infusion site discomfort | - | - | 1 (4) | 4 |
| Infusion site hematoma | - | - | 1 (4) | 2 |
| Dizziness | 1 (7) | 2 | - | - |
| Headache | 1 (7) | 1 | - | - |
| Body temperature increase | - | - | 1 (4) | 1 |
| Infusion related reaction•• | 1 (7) | 1 | - | - |
• Three patients experienced adverse reactions in Study 2 and two patients experienced adverse reactions in Study 1.
•• Symptoms resolved without any intervention and did not recur with subsequent administration.
1 6 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
- SEVENFACT [coagulation factor VIIa (recombinant)-jncw], is supplied as a room temperature stable, white to off-white, lyophilized powder in single-dose vials, one vial per carton. The diluent for reconstitution of SEVENFACT is Water for Injection supplied as a clear colorless solution in a pre-filled syringe.
- Single 1 mg, 2 mg or 5 mg vials of SEVENFACT are available in packages as indicated in Table 6.
| Presentation | Cap Color Indication | NDC Number | Contents |
| 1 mg per vial | Yellow | NDC 71127-1000-1 |
|
| 2 mg per vial | Green | NDC 71127-2000-1 |
|
| 5 mg per vial | Purple | NDC 71127-5000-1 |
|
- The SEVENFACT vials are made of glass, closed with a bromobutyl rubber stopper (not made with natural rubber latex), and sealed with an aluminum cap.
- The pre-filled diluent syringes are made of glass, with a siliconized bromobutyl rubber plunger (not made with natural rubber latex).
Storage and Handling
- Prior to reconstitution, the SEVENFACT kit should be stored at room temperature but can be stored between 36°F to 86°F (2°C to 30°C), protected from light in the product package. Do not freeze.
- After reconstitution, SEVENFACT should be stored at room temperature but can be stored between 36°F to 86°F (2°C to 30°C), for up to 4 hours. Do not freeze or store in syringes.
INSTRUCTIONS FOR USE:
READ BEFORE YOU START USING SEVENFACT ®
Your healthcare provider should show you and/or your caregiver how to reconstitute and administer SEVENFACT ® the first time it is used. Use aseptic techniques.
Your SEVENFACT ® kit contains:

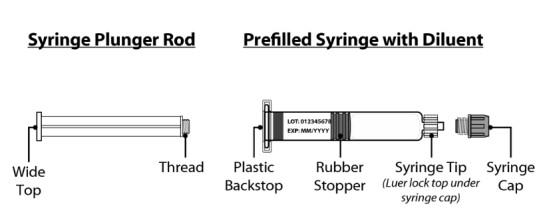
Vial Adapters• and Packaging
1 mg and 2 mg V ial A dapter 5 mg V ial A dapter

•N ote : Each SEVENFACT kit will contain only one vial adapter.
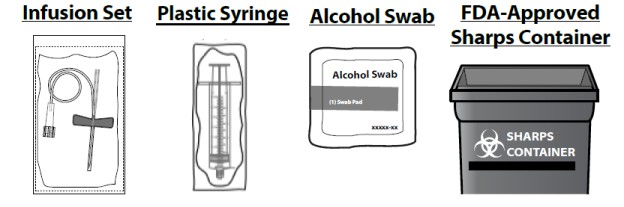
You will also need:
1 Collect Supplies and Prepare Vial
- Take out the number of SEVENFACT ® kits you need to fulfill your prescribed dose, an infusion set (not supplied) and an alcohol swab (not supplied). Do not use the kit if the tamper seal has been broken or you suspect the kit is contaminated. Use a new kit instead.
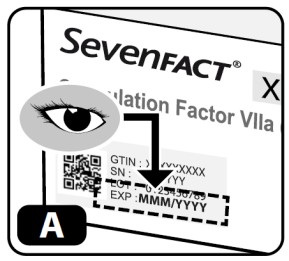
- Check the expiration date on the side of the kit (Fig ure A) . Do not use if expired.
- Clean a flat surface before starting the steps for reconstituting SEVENFACT ® .
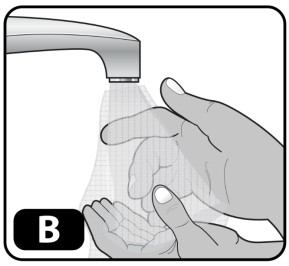
- Wash your hands with soap and water and dry using a clean towel or air dry (Fig ure B) .
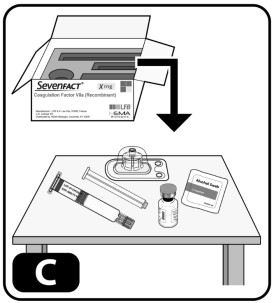
- Take out the contents of one kit and one alcohol swab. Place items on the clean surface (Fig ure C) .
- Inspect all contents of the kit. Make sure each vial has a matching colored syringe. Do not use the contents if they have been dropped or if they are damaged. Use a new kit instead.
- If not already at room temperature, bring the vial and the pre-filled syringe to room temperature. You can do this by holding them in your hands until they feel as warm as your hands.
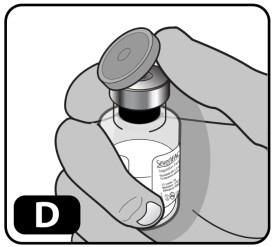
- Remove the plastic cap from the vial (Fig ure D). If the plastic cap is loose or missing, do not use the vial.
- Wipe the rubber stopper with an alcohol swab (Fig ure E) and allow it to air dry for a few seconds to ensure that it is as germ free as possible.
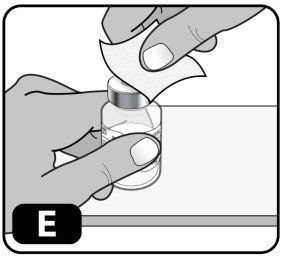
- After cleaning with the swab, do not touch the rubber stopper with your fingers and do not allow it to touch any other object until you attach the vial adapter, as this can transfer germs.
2 Attach Vial Adapter
- Peel off the paper protective cover from the vial adapter package (Fig ure F) .
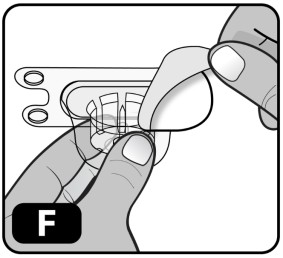
- Hold the vial on the clean flat surface with one hand, using your other hand to hold the plastic cover (with the vial adapter inside) directly over the drug vial. The spike of the adapter should line up with the middle of the gray rubber stopper.
- Firmly press down so the vial adapter spike breaks through the rubber stopper (you may hear/see it "snap" into place) (Fig ure G).
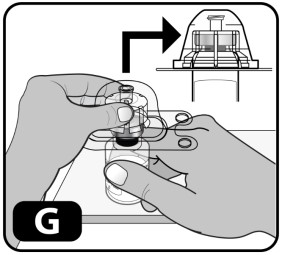
- Lightly squeeze the plastic cover and lift up to remove it from the vial adapter (Fig ure H) .
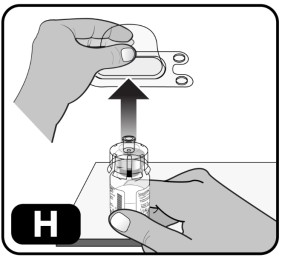 Do not touch the top of the vial adapter once the plastic cover is removed to avoid transferring germs from your fingers. NOTE: The 5 mg vial adapter may not sit flat against the vial, but it is fully functional.
Do not touch the top of the vial adapter once the plastic cover is removed to avoid transferring germs from your fingers. NOTE: The 5 mg vial adapter may not sit flat against the vial, but it is fully functional.
3 Attach Pre-filled Syringe and Install the Plunger Rod
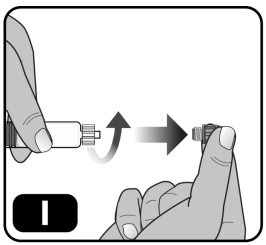
- Remove the syringe cap from the pre-filled syringe by holding the syringe body with one hand and using the other hand to unscrew the syringe cap (turn to the left) (Fig ure I) . Do not touch the syringe tip under the syringe cap to avoid transferring germs from your fingers. Do not use the pre-filled syringe if the syringe cap is loose or missing.
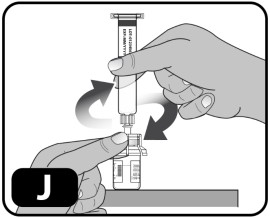
- While holding the edges of the vial adapter, screw on the pre-filled syringe (turn to the right) a few turns until it starts to tighten (Fig ure J) . Be careful not to overtighten as you will need to remove the syringe later.
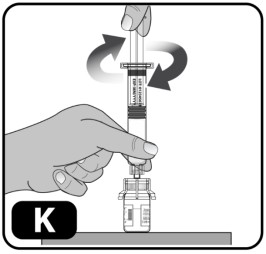
- Hold the plunger rod by the wide top end in one hand and the syringe body using your other hand.
- Insert the plunger rod into the syringe, then screw a few turns (turn to the right) so that the plunger rod is attached to the gray rubber stopper in the syringe (Fig ure K) .
4 Mix Drug in Vial
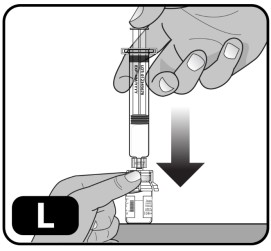
- Very slowly push down on the plunger rod to the bottom of the syringe, to transfer all of the liquid from the syringe into the drug vial (Fig ure L) . Do not push too quickly as it can result in excess foam and air in the vial.
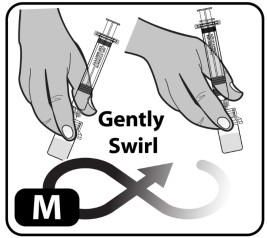
- Swirl the vial gently or roll between hands until all powder is dissolved (Fig ure M) . Do not shake the vial as this creates foam and air.
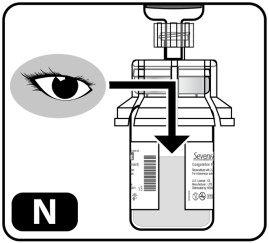
- Check the mixed solution (Fig ure N) . It should be clear to slightly opaque. All powder should be dissolved with no particles floating in the liquid. Do not use the drug if liquid has particles or is cloudy after mixing. Start over with a new kit.
5 Remove Empty Syringe from Vial Adapter
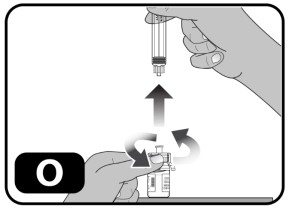
- Without withdrawing any drug back into the syringe, unscrew the syringe from the vial adapter (turn to the left) until it is completely detached (Fig ure O) .
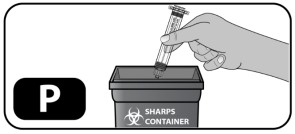
- Throw out empty syringe into an FDA-approved sharps container (Fig ure P) . Do not remove the vial adapter. Do not touch the luer lock top of the vial adapter. If you touch this top, germs from your fingers can be transferred.
6 Mix Additional Vial(s) and Infuse Dose
- If your dose requires more than one vial, repeat the above steps with additional kits until you have reached your required dose.
- Withdraw the liquid drug from the vial(s), using a syringe provided by your pharmacy that is large enough to hold your prescribed dose.
- SEVENFACT ® must be infused within 4 hours of reconstitution (Fig ure Q) . Do not use if more than 4 hours have passed since reconstitution.
- SEVENFACT ® can be infused in 2 minutes or less as an intravenous infusion per the instructions of your healthcare provider.
7 Throw Out Empty Drug Vial(s)
- After reconstitution and infusion, safely dispose of the vial with vial adapter attached, the infusion syringe, and any other waste materials into an FDA-approved sharps container (Fig ure R) . Do not throw out with the ordinary household trash. Do not disassemble the vial and vial adapter before disposal. Do not reuse any components of the kit.
- If you do not have an FDA-approved sharps container, you may use a container with the following:
- Made of heavy duty plastic,
- Can be closed, puncture resistant and prevents sharps from coming out,
- Upright and stable,
- Leak resistant,
- Properly labeled as hazardous
Follow local or state guidelines for proper disposal of sharps container. For more information about safe sharps disposal visit FDA’s website: http://www.fda.gov/safesharpsdisposal
Storage
SEVENFACT ® is supplied in a kit that should be stored at room temperature, but can be stored between 36°F to 86°F (2°C-30°C).
Do not open the kit contents until you are ready to use them.
Do not freeze or store in syringes reconstituted SEVENFACT ® solution.
Avoid exposure of the reconstituted SEVENFACT ® solution to direct light.
Important Information
SEVENFACT ® is for intravenous infusion only. Do not administer through any other route (subcutaneous or intramuscular).
Contact your healthcare provider or local hemophilia treatment center if you experience problems.
For Q uestions or C omplaints:
Call: 855.718.HEMA (4362)
or please contact your HEMA Biologics representative for assistance.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by LFB S.A. Puteaux, 92800, France
Distributed by HEMA Biologics, Louisville, KY 40241
Revised: November 13, 2025
Mechanism of Action
The active ingredient in SEVENFACT is a recombinant analog of human Factor VIIa, a vitamin K-dependent coagulation factor. In the presence of both calcium and phospholipids, Factor VIIa in a complex with tissue factor (TF) activates Factor X to Factor Xa, directly bypassing the reactions that require Factor VIII or Factor IX. Activation of Factor X to Factor Xa initiates the common pathway of the coagulation cascade in which prothrombin is activated to thrombin, which then converts fibrinogen to fibrin to form a hemostatic plug, thereby achieving clot formation at the site of hemorrhage (hemostasis). This process may also occur in the absence of TF on the surface of activated platelets.
Sevenfact - Coagulation Factor Viia Recombinant Human PubMed™ news
- Journal Article • 2026 MayCrosstalk between the monocytes and coagulation factor VⅡa aggravates the inflammation in patients with CAD.
- Journal Article • 2026 MayIntracranial hemorrhage in acquired hemophilia A: illustrative case.
- Journal Article • 2026 AprFactor X and Combined Factor VIIa/Factor X Augment Coagulation Potential in a Plasma Model of Tissue Factor Pathway Inhibitor-Reduced Haemophilia State.
- Journal Article • 2026 MarProbing Solution Dynamics of Tissue Factor Using Molecular Dynamics Simulations Guided by NMR Chemical Shifts.
- Journal Article • 2026 MarThe effects of recombinant human activated factor VII and tranexamic acid on traumatic bleeding and mortality in mice.