Get your patient on Tamoxifen Citrate - Tamoxifen Citrate tablet, Film Coated (Tamoxifen Citrate)
Tamoxifen Citrate - Tamoxifen Citrate tablet, Film Coated prescribing information
WARNING - For Women with Ductal Carcinoma in Situ (DCIS) and Women at High Risk for Breast Cancer : Serious and life-threatening events associated with tamoxifen citrate in the risk reduction setting (women at high risk for cancer and women with DCIS) include uterine malignancies, stroke and pulmonary embolism. Incidence rates for these events were estimated from the NSABP P-1 trial (see CLINICAL PHARMACOLOGY-Clinical Studies – Reduction in Breast Cancer Incidence In High Risk Women ). Uterine malignancies consist of both endometrial adenocarcinoma (incidence rate per 1,000 women-years of 2.20 for tamoxifen citrate vs 0.71 for placebo) and uterine sarcoma (incidence rate per 1,000 women-years of 0.17 for tamoxifen citrate vs 0.4 for placebo)•. For stroke, the incidence rate per 1,000 women-years was 1.43 for tamoxifen citrate vs 1.00 for placebo••. For pulmonary embolism, the incidence rate per 1,000 women-years was 0.75 for tamoxifen citrate versus 0.25 for placebo••.
Some of the strokes, pulmonary emboli, and uterine malignancies were fatal.
Health care providers should discuss the potential benefits versus the potential risks of these serious events with women at high risk of breast cancer and women with DCIS considering tamoxifen citrate to reduce their risk of developing breast cancer.
The benefits of tamoxifen citrate outweigh its risks in women already diagnosed with breast cancer.
•Updated long-term follow-up data (median length of follow-up is 6.9 years) from NSABP P-1 study. See WARNINGS: Effects on the Uterus-Endometrial Cancer and Uterine Sarcoma .
••See Table 3 under CLINICAL PHARMACOLOGY-Clinical Studies .
INDICATIONS AND USAGE
Metastatic Breast Cancer:
Tamoxifen citrate tablets are effective in the treatment of metastatic breast cancer in women and men. In premenopausal women with metastatic breast cancer, tamoxifen citrate tablets are an alternative to oophorectomy or ovarian irradiation. Available evidence indicates that patients whose tumors are estrogen receptor positive are more likely to benefit from tamoxifen citrate tablets therapy.
Adjuvant Treatment of Breast Cancer:
Tamoxifen citrate tablets are indicated for the treatment of node-positive breast cancer in women following total mastectomy or segmental mastectomy, axillary dissection, and breast irradiation. In some tamoxifen citrate adjuvant studies, most of the benefit to date has been in the subgroup with four or more positive axillary nodes.
Tamoxifen citrate tablets are indicated for the treatment of axillary node-negative breast cancer in women following total mastectomy or segmental mastectomy, axillary dissection, and breast irradiation.
The estrogen and progesterone receptor values may help to predict whether adjuvant tamoxifen citrate tablets therapy are likely to be beneficial.
Tamoxifen citrate tablets reduces the occurrence of contralateral breast cancer in patients receiving adjuvant tamoxifen citrate tablets therapy for breast cancer.
Ductal Carcinoma in Situ (DCIS):
In women with DCIS, following breast surgery and radiation, tamoxifen citrate tablets are indicated to reduce the risk of invasive breast cancer (see BOXED WARNING at the beginning of the label). The decision regarding therapy with tamoxifen citrate tablets for the reduction in breast cancer incidence should be based upon an individual assessment of the benefits and risks of tamoxifen citrate tablets therapy.
Current data from clinical trials support five years of adjuvant tamoxifen citrate tablets therapy for patients with breast cancer.
Reduction in Breast Cancer Incidence in High Risk Women:
Tamoxifen citrate tablets are indicated to reduce the incidence of breast cancer in women at high risk for breast cancer. This effect was shown in a study of 5 years planned duration with a median follow-up of 4.2 years. Twenty-five percent of the participants received drug for 5 years. The longer-term effects are not known. In this study, there was no impact of tamoxifen on overall or breast cancer-related mortality (see BOXED WARNING at the beginning of the label).
Tamoxifen citrate tablets are indicated only for high-risk women. “High risk” is defined as women at least 35 years of age with a 5-year predicted risk of breast cancer ≥ 1.67%, as calculated by the Gail Model.
Examples of combinations of factors predicting a 5-year risk ≥ 1.67% are:
Age 35 or older and any of the following combination of factors:
- One first degree relative with a history of breast cancer, 2 or more benign biopsies, and a history of a breast biopsy showing atypical hyperplasia; or
- At least 2 first degree relatives with a history of breast cancer, and a personal history of at least one breast biopsy; or
- LCIS
Age 40 or older and any of the following combination of factors:
- One first degree relative with a history of breast cancer, 2 or more benign biopsies, age at first live birth 25 or older, and age at menarche 11 or younger; or
- At least 2 first degree relatives with a history of breast cancer, and age at first live birth 19 or younger; or
- One first degree relative with a history of breast cancer, and a personal history of a breast biopsy showing atypical hyperplasia.
Age 45 or older and any of the following combination of factors:
- At least 2 first degree relatives with a history of breast cancer and age at first live birth 24 or younger; or
- One first degree relative with a history of breast cancer with a personal history of a benign breast biopsy, age at menarche 11 or less and age at first live birth 20 or more.
Age 50 or older and any of the following combination of factors:
- At least 2 first degree relatives with a history of breast cancer; or
- History of one breast biopsy showing atypical hyperplasia, and age at first live birth 30 or older and age at menarche 11 or less; or
- History of at least two breast biopsies with a history of atypical hyperplasia, and age at first live birth 30 or more.
Age 55 or older and any of the following combination of factors:
- One first degree relative with a history of breast cancer with a personal history of a benign breast biopsy, and age at menarche 11 or less; or
- History of at least 2 breast biopsies with a history of atypical hyperplasia, and age at first live birth 20 or older.
Age 60 or older and:
- 5-year predicted risk of breast cancer ≥ 1.67%, as calculated by the Gail Model.
For women whose risk factors are not described in the above examples, the Gail Model is necessary to estimate absolute breast cancer risk. Health Care Professionals can obtain a Gail Model Risk Assessment Tool by dialing 1-866-850-2876.
There are insufficient data available regarding the effect of tamoxifen citrate tablets on breast cancer incidence in women with inherited mutations (BRCA1, BRCA2) to be able to make specific recommendations on the effectiveness of tamoxifen citrate in these patients.
After an assessment of the risk of developing breast cancer, the decision regarding therapy with tamoxifen citrate tablets for the reduction in breast cancer incidence should be based upon an individual assessment of the benefits and risks of tamoxifen citrate tablets therapy. In the NSABP P-1 trial, tamoxifen citrate tablets treatment lowered the risk of developing breast cancer during the follow-up period of the trial, but did not eliminate breast cancer risk (See Table 3 in CLINICAL PHARMACOLOGY ).
DOSAGE AND ADMINISTRATION
For patients with breast cancer, the recommended daily dose is 20 to 40 mg. Dosages greater than 20 mg per day should be given in divided doses (morning and evening).
In three single agent adjuvant studies in women, one 10 mg tamoxifen citrate tablet was administered two (ECOG and NATO) or three (Toronto) times a day for two years. In the NSABP B-14 adjuvant study in women with node-negative breast cancer, one 10 mg tamoxifen citrate tablet was given twice a day for at least 5 years. Results of the B-14 study suggest that continuation of therapy beyond five years does not provide additional benefit (see CLINICAL PHARMACOLOGY ). In the EBCTCG 1995 overview, the reduction in recurrence and mortality was greater in those studies that used tamoxifen for about 5 years than in those that used tamoxifen for a shorter period of therapy. There was no indication that doses greater than 20 mg per day were more effective. Current data from clinical trials support 5 years of adjuvant tamoxifen citrate therapy for patients with breast cancer.
Ductal Carcinoma in Situ (DCIS):
The recommended dose is tamoxifen citrate 20 mg daily for 5 years.
Reduction in Breast Cancer Incidence in High Risk Women:
The recommended dose is tamoxifen citrate 20 mg daily for 5 years. There are no data to support the use of tamoxifen citrate other than for 5 years (See CLINICAL PHARMACOLOGY-Clinical Studies - Reduction in Breast Cancer Incidence in High Risk Women ).
CONTRAINDICATIONS
Tamoxifen citrate tablets are contraindicated in patients with known hypersensitivity to the drug or any of its ingredients.
Reduction in Breast Cancer Incidence in High Risk Women and Women with DCIS:
Tamoxifen citrate tablets are contraindicated in women who require concomitant coumarin-type anticoagulant therapy or in women with a history of deep vein thrombosis or pulmonary embolus.
ADVERSE REACTIONS
Adverse reactions to tamoxifen citrate are relatively mild and rarely severe enough to require discontinuation of treatment in breast cancer patients.
Continued clinical studies have resulted in further information which better indicates the incidence of adverse reactions with tamoxifen citrate as compared to placebo.
Metastatic Breast Cancer:
Increased bone and tumor pain and, also, local disease flare have occurred, which are sometimes associated with a good tumor response. Patients with increased bone pain may require additional analgesics. Patients with soft tissue disease may have sudden increases in the size of pre-existing lesions, sometimes associated with marked erythema within and surrounding the lesions and/or the development of new lesions. When they occur, the bone pain or disease flare are seen shortly after starting tamoxifen citrate and generally subside rapidly.
In patients treated with tamoxifen citrate for metastatic breast cancer, the most frequent adverse reaction to tamoxifen citrate is hot flashes.
Other adverse reactions which are seen infrequently are hypercalcemia, peripheral edema, distaste for food, pruritus vulvae, depression, dizziness, light-headedness, headache, hair thinning and/or partial hair loss, and vaginal dryness.
Premenopausal Women:
The following table summarizes the incidence of adverse reactions reported at a frequency of 2% or greater from clinical trials (Ingle, Pritchard, Buchanan) which compared tamoxifen citrate therapy to ovarian ablation in premenopausal patients with metastatic breast cancer.
| •Some women had more than one adverse reaction. | ||
| TAMOXIFEN CITRATE All Effects % of Women | OVARIAN ABLATION All Effects % of Women | |
| Adverse Reactions• | n=104 | n=100 |
| Flush | 33 | 46 |
| Amenorrhea | 16 | 69 |
| Altered Menses | 13 | 5 |
| Oligomenorrhea | 9 | 1 |
| Bone Pain | 6 | 6 |
| Menstrual Disorder | 6 | 4 |
| Nausea | 5 | 4 |
| Cough/Coughing | 4 | 1 |
| Edema | 4 | 1 |
| Fatigue | 4 | 1 |
| Musculoskeletal Pain | 3 | 0 |
| Pain | 3 | 4 |
| Ovarian Cyst(s) | 3 | 2 |
| Depression | 2 | 2 |
| Abdominal Cramps | 1 | 2 |
| Anorexia | 1 | 2 |
Male Breast Cancer:
Tamoxifen citrate is well tolerated in males with breast cancer. Reports from the literature and case reports suggest that the safety profile of tamoxifen citrate in males is similar to that seen in women. Loss of libido and impotence have resulted in discontinuation of tamoxifen therapy in male patients. Also, in oligospermic males treated with tamoxifen, LH, FSH, testosterone and estrogen levels were elevated. No significant clinical changes were reported.
Adjuvant Breast Cancer:
In the NSABP B-14 study, women with axillary node-negative breast cancer were randomized to 5 years of tamoxifen citrate 20 mg/day or placebo following primary surgery. The reported adverse effects are tabulated below (mean follow-up of approximately 6.8 years) showing adverse events more common on tamoxifen citrate than on placebo. The incidence of hot flashes (64% vs. 48%), vaginal discharge (30% vs. 15%), and irregular menses (25% vs. 19%) were higher with tamoxifen citrate compared with placebo. All other adverse effects occurred with similar frequency in the 2 treatment groups, with the exception of thrombotic events; a higher incidence was seen in tamoxifen citrate-treated patients (through 5 years, 1.7% vs. 0.4%). Two of the patients treated with tamoxifen citrate who had thrombotic events died.
| • Defined as a platelet count of <100,000/mm 3 | ||
| NSABP B-14 Study | ||
| Adverse Effect | % of Women | |
| Tamoxifen Citrate (n=1422) | Placebo (n=1437) | |
| Hot Flashes | 64 | 48 |
| Fluid Retention | 32 | 30 |
| Vaginal Discharge | 30 | 15 |
| Nausea | 26 | 24 |
| Irregular Menses | 25 | 19 |
| Weight Loss (>5%) | 23 | 18 |
| Skin Changes | 19 | 15 |
| Increased SGOT | 5 | 3 |
| Increased Bilirubin | 2 | 1 |
| Increased Creatinine | 2 | 1 |
| Thrombocytopenia• | 2 | 1 |
| Thrombotic Events | ||
| Deep Vein Thrombosis | 0.8 | 0.2 |
| Pulmonary Embolism | 0.5 | 0.2 |
| Superficial Phlebitis | 0.4 | 0.0 |
In the Eastern Cooperative Oncology Group (ECOG) adjuvant breast cancer trial, tamoxifen citrate or placebo was administered for 2 years to women following mastectomy. When compared to placebo, tamoxifen citrate showed a significantly higher incidence of hot flashes (19% vs. 8% for placebo). The incidence of all other adverse reactions was similar in the 2 treatment groups with the exception of thrombocytopenia where the incidence for tamoxifen citrate was 10% vs. 3% for placebo, an observation of borderline statistical significance.
In other adjuvant studies, Toronto and Tamoxifen Citrate Adjuvant Trial Organization (NATO), women received either tamoxifen citrate or no therapy. In the Toronto study, hot flashes were observed in 29% of patients for tamoxifen citrate vs. 1% in the untreated group. In the NATO trial, hot flashes and vaginal bleeding were reported in 2.8% and 2.0% of women, respectively, for tamoxifen citrate vs. 0.2% for each in the untreated group.
Anastrozole Adjuvant Trial - Study of Anastrozole compared to Tamoxifen Citrate for Adjuvant Treatment of Early Breast Cancer (see CLINICAL PHARMACOLOGY - Clinical Studies ). At a median follow-up of 33 months, the combination of anastrozole and tamoxifen citrate did not demonstrate any efficacy benefit when compared to tamoxifen citrate therapy given alone in all patients as well as in the hormone receptor positive subpopulation. This treatment arm was discontinued from the trial. The median duration of adjuvant treatment for safety evaluation was 59.8 months and 59.6 months for patients receiving anastrozole 1 mg and tamoxifen citrate 20 mg, respectively.
Adverse events occurring with an incidence of at least 5% in either treatment group during treatment or within 14 days of the end of treatment are presented in the following table.
Adverse events occurring with an incidence of at least 5% in either treatment group during treatment, or within 14 days of the end of treatment
| COSTART Coding Symbols for Thesaurus of Adverse Reaction Terms. N = Number of patients receiving the treatment. • A patient may have had more than 1 adverse event, including more than 1 adverse event in the same body system. † Vaginal Hemorrhage without further diagnosis. •• The combination arm was discontinued due to lack of efficacy benefit at 33 months of follow-up. | ||
| Body system and adverse event by COSTART-preferred term• | Anastrozole 1 mg (N = 3092) | Tamoxifen Citrate 20 mg (N = 3094) |
| Body as a whole | ||
| Asthenia | 575 (19) | 544 (18) |
| Pain | 533 (17) | 485 (16) |
| Back pain | 321 (10) | 309 (10) |
| Headache | 314 (10) | 249 (8) |
| Abdominal pain | 271 (9) | 276 (9) |
| Infection | 285 (9) | 276 (9) |
| Accidental injury | 311 (10) | 303 (10) |
| Flu syndrome | 175 (6) | 195 (6) |
| Chest pain | 200 (7) | 150 (5) |
| Neoplasm | 162 (5) | 144 (5) |
| Cyst | 138 (5) | 162 (5) |
| Cardiovascular | ||
| Vasodilatation | 1104 (36) | 1264 (41) |
| Hypertension | 402 (13) | 349 (11) |
| Digestive | ||
| Nausea | 343 (11) | 335 (11) |
| Constipation | 249 (8) | 252 (8) |
| Diarrhea | 265 (9) | 216 (7) |
| Dyspepsia | 206 (7) | 169 (6) |
| Gastrointestinal disorder | 210 (7) | 158 (5) |
| Hemic and lymphatic | ||
| Lymphoedema | 304 (10) | 341 (11) |
| Anemia | 113 (4) | 159 (5) |
| Metabolic and nutritional | ||
| Peripheral edema | 311 (10) | 343 (11) |
| Weight gain | 285 (9) | 274 (9) |
| Hypercholesterolemia | 278 (9) | 108 (3.5) |
| Musculoskeletal | ||
| Arthritis | 512 (17) | 445 (14) |
| Arthralgia | 467 (15) | 344 (11) |
| Osteoporosis | 325 (11) | 226 (7) |
| Fracture | 315 (10) | 209 (7) |
| Bone pain | 201 (7) | 185 (6) |
| Arthrosis | 207 (7) | 156 (5) |
| Joint Disorder | 184 (6) | 160 (5) |
| Myalgia | 179 (6) | 160 (5) |
| Nervous system | ||
| Depression | 413 (13) | 382 (12) |
| Insomnia | 309 (10) | 281 (9) |
| Dizziness | 236 (8) | 234 (8) |
| Anxiety | 195 (6) | 180 (6) |
| Paraesthesia | 215 (7) | 145 (5) |
| Respiratory | ||
| Pharyngitis | 443 (14) | 422 (14) |
| Cough increased | 261 (8) | 287 (9) |
| Dyspnea | 234 (8) | 237 (8) |
| Sinusitis | 184 (6) | 159 (5) |
| Bronchitis | 167 (5) | 153 (5) |
| Skin and appendages | ||
| Rash | 333 (11) | 387 (13) |
| Sweating | 145 (5) | 177 (6) |
| Special Senses | ||
| Cataract Specified | 182 (6) | 213 (7) |
| Urogenital | ||
| Leukorrhea | 86 (3) | 286 (9) |
| Urinary tract infection | 244 (8) | 313 (10) |
| Breast pain | 251 (8) | 169 (6) |
| Breast Neoplasm | 164 (5) | 139 (5) |
| Vulvovaginitis | 194 (6) | 150 (5) |
| Vaginal Hemorrhage † | 122 (4) | 180 (6) |
| Vaginitis | 125 (4) | 158 (5) |
Certain adverse events and combinations of adverse events were prospectively specified for analysis, based on the known pharmacologic properties and side effect profiles of the two drugs (see the following table).
| 1 Patients with multiple events in the same category are counted only once in that category. 2 Refers to joint symptoms, including joint disorder, arthritis, arthrosis and arthralgia. 3 Percentages calculated based upon the numbers of patients with an intact uterus at baseline. 4 The odds ratios < 1.00 favor Anastrozole and those > 1.00 favor Tamoxifen Citrate | ||||
| Anastrozole N=3092 (%) | Tamoxifen Citrate N=3094 (%) | Odds- ratio 4 | 95% CI 4 | |
| Hot Flashes | 1104 (36) | 1264 (41) | 0.80 | 0.73 to 0.89 |
| Musculoskeletal Events 2 | 1100 (36) | 911 (29) | 1.32 | 1.19 to 1.47 |
| Fatigue/Asthenia | 575 (19) | 544 (18) | 1.07 | 0.94 to 1.22 |
| Mood Disturbances | 597 (19) | 554 (18) | 1.10 | 0.97 to 1.25 |
| Nausea and Vomiting | 393 (13) | 384 (12) | 1.03 | 0.88 to 1.19 |
| All Fractures | 315 (10) | 209 (7) | 1.57 | 1.30 to 1.88 |
| Fractures of Spine, Hip, or Wrist | 133 (4) | 91 (3) | 1.48 | 1.13 to 1.95 |
| Wrist/Colles’ fractures | 67 (2) | 50 (2) | ||
| Spine fractures | 43 (1) | 22 (1) | ||
| Hip fractures | 28 (1) | 26 (1) | ||
| Cataracts | 182 (6) | 213 (7) | 0.85 | 0.69 to 1.04 |
| Vaginal Bleeding | 167 (5) | 317 (10) | 0.50 | 0.41 to 0.61 |
| Ischemic Cardiovascular Disease | 127 (4) | 104 (3) | 1.23 | 0.95 to 1.60 |
| Vaginal Discharge | 109 (4) | 408 (13) | 0.24 | 0.19 to 0.30 |
| Venous Thromboembolic events | 87 (3) | 140 (5) | 0.61 | 0.47 to 0.80 |
| Deep Venous Thromboembolic Events | 48 (2) | 74 (2) | 0.64 | 0.45 to 0.93 |
| Ischemic Cerebrovascular Event | 62 (2) | 88 (3) | 0.70 | 0.50 to 0.97 |
| Endometrial Cancer 3 | 4 (0.2) | 13 (0.6) | 0.31 | 0.10 to 0.94 |
Patients receiving anastrozole had an increase in joint disorders (including arthritis, arthrosis and arthralgia) compared with patients receiving tamoxifen citrate. Patients receiving anastrozole had an increase in the incidence of all fractures (specifically fractures of spine, hip and wrist) [315 (10%)] compared with patients receiving tamoxifen citrate [209 (7%)]. Patients receiving anastrozole had a decrease in hot flashes, vaginal bleeding, vaginal discharge, endometrial cancer, venous thromboembolic events and ischemic cerebrovascular events compared with patients receiving tamoxifen citrate.
Patients receiving tamoxifen citrate had a decrease in hypercholesterolemia (108 [3.5%]) compared to patients receiving anastrozole (278 [9%]). Angina pectoris was reported in 71 [2.3%] patients in the anastrozole arm and 51 [1.6%] patients in the tamoxifen citrate arm; myocardial infarction was reported in 37 [1.2%] patients in the anastrozole arm and in 34 [1.1%] patients in the tamoxifen citrate arm.
Results from the adjuvant trial bone substudy, at 12 and 24 months demonstrated that patients receiving anastrozole had a mean decrease in both lumbar spine and total hip bone mineral density (BMD) compared to baseline. Patients receiving tamoxifen citrate had a mean increase in both lumbar spine and total hip BMD compared to baseline.
Ductal Carcinoma in Situ (DCIS):
The type and frequency of adverse events in the NSABP B-24 trial were consistent with those observed in the other adjuvant trials conducted with tamoxifen citrate.
Reduction in Breast Cancer Incidence in High Risk Women:
In the NSABP P-1 Trial, there was an increase in five serious adverse effects in the tamoxifen citrate group: endometrial cancer (33 cases in the tamoxifen citrate group vs. 14 in the placebo group); pulmonary embolism (18 cases in the tamoxifen citrate group vs. 6 in the placebo group); deep vein thrombosis (30 cases in the tamoxifen citrate group vs. 19 in the placebo group); stroke (34 cases in the tamoxifen citrate group vs. 24 in the placebo group); cataract formation (540 cases in the tamoxifen citrate group vs. 483 in the placebo group) and cataract surgery (101 cases in the tamoxifen citrate group vs. 63 in the placebo group) (See WARNINGS and Table 3 in CLINICAL PHARMACOLOGY ).
The following table presents the adverse events observed in NSABP P-1 by treatment arm. Only adverse events more common on tamoxifen citrate than placebo are shown.
| 1 Number with Quality of Life Questionnaires 2 Number with Treatment Follow-up Forms 3 Number with Adverse Drug Reaction Forms | ||
| NSABP P-1 Trial: All Adverse Events % of Women | ||
| TAMOXIFEN CITRATE N=6681 | PLACEBO N=6707 | |
| Self Reported Symptoms | N=6441 1 | N=6469 1 |
| Hot Flashes | 80 | 68 |
| Vaginal Discharges | 55 | 35 |
| Vaginal Bleeding | 23 | 22 |
| Laboratory Abnormalities | N=6520 2 | N=6535 2 |
| Platelets decreased | 0.7 | 0.3 |
| Adverse Effects | N=6492 3 | N=6484 3 |
| Other Toxicities | ||
| Mood | 11.6 | 10.8 |
| Infection/Sepsis | 6.0 | 5.1 |
| Constipation | 4.4 | 3.2 |
| Alopecia | 5.2 | 4.4 |
| Skin | 5.6 | 4.7 |
| Allergy | 2.5 | 2.1 |
In the NSABP P-1 trial, 15.0% and 9.7% of participants receiving tamoxifen citrate and placebo therapy, respectively withdrew from the trial for medical reasons. The following are the medical reasons for withdrawing from tamoxifen citrate and placebo therapy, respectively: Hot flashes (3.1% vs. 1.5%) and Vaginal Discharge (0.5% vs. 0.1%).
In the NSABP P-1 trial, 8.7% and 9.6% of participants receiving tamoxifen citrate and placebo therapy, respectively withdrew for non-medical reasons.
On the NSABP P-1 trial, hot flashes of any severity occurred in 68% of women on placebo and in 80% of women on tamoxifen citrate. Severe hot flashes occurred in 28% of women on placebo and 45% of women on tamoxifen citrate. Vaginal discharge occurred in 35% and 55% of women on placebo and tamoxifen citrate respectively; and was severe in 4.5% and 12.3% respectively.
There was no difference in the incidence of vaginal bleeding between treatment arms.
Pediatric Patients - McCune-Albright Syndrome:
Mean uterine volume increased after 6 months of treatment and doubled at the end of the one- year study. A causal relationship has not been established; however, as an increase in the incidence of endometrial adenocarcinoma and uterine sarcoma has been noted in adults treated with tamoxifen citrate (see BOXED WARNING ), continued monitoring of McCune-Albright patients treated with tamoxifen citrate for long-term effects is recommended. The safety and efficacy of tamoxifen citrate for girls aged two to 10 years with McCune-Albright Syndrome and precocious puberty have not been studied beyond one year of treatment. The long-term effects of tamoxifen citrate therapy in girls have not been established .
Postmarketing experience:
Less frequently reported adverse reactions are vaginal bleeding, vaginal discharge, menstrual irregularities, skin rash and headaches. Usually these have not been of sufficient severity to require dosage reduction or discontinuation of treatment. Very rare reports of erythema multiforme, Stevens-Johnson syndrome, bullous pemphigoid, interstitial pneumonitis, and rare reports of hypersensitivity reactions including angioedema have been reported with tamoxifen citrate therapy. In some of these cases, the time to onset was more than one year. Rarely, elevation of serum triglyceride levels, in some cases with pancreatitis, may be associated with the use of tamoxifen citrate (see PRECAUTIONS- Drug/Laboratory Testing Interactions section). To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions:
When tamoxifen citrate is used in combination with coumarin-type anticoagulants, a significant increase in anticoagulant effect may occur. Where such coadministration exists, careful monitoring of the patient’s prothrombin time is recommended.
In the NSABP P-1 trial, women who required coumarin-type anticoagulants for any reason were ineligible for participation in the trial (See CONTRAINDICATIONS ).
There is an increased risk of thromboembolic events occurring when cytotoxic agents are used in combination with tamoxifen citrate.
Tamoxifen reduced letrozole plasma concentrations by 37%. The effect of tamoxifen on metabolism and excretion of other antineoplastic drugs, such as cyclophosphamide and other drugs that require mixed function oxidases for activation, is not known. Tamoxifen and N-desmethyl tamoxifen plasma concentrations have been shown to be reduced when coadministered with rifampin or aminoglutethimide. Induction of CYP3A4-mediated metabolism is considered to be the mechanism by which these reductions occur; other CYP3A4 inducing agents have not been studied to confirm this effect.
One patient receiving tamoxifen citrate with concomitant phenobarbital exhibited a steady state serum level of tamoxifen lower than that observed for other patients (i.e., 26 ng/mL vs. mean value of 122 ng/mL). However, the clinical significance of this finding is not known. Rifampin induced the metabolism of tamoxifen and significantly reduced the plasma concentrations of tamoxifen in 10 patients. Aminoglutethimide reduces tamoxifen and N-desmethyl tamoxifen plasma concentrations. Medroxyprogesterone reduces plasma concentrations of N-desmethyl, but not tamoxifen.
Concomitant bromocriptine therapy has been shown to elevate serum tamoxifen and N-desmethyl tamoxifen.
Based on clinical and pharmacokinetic results from the anastrozole adjuvant trial, tamoxifen citrate should not be administered with anastrozole (see CLINICAL PHARMACOLOGY – Drug- Drug Interactions section).
DESCRIPTION
Tamoxifen citrate tablets, USP a nonsteroidal antiestrogen, are for oral administration. Tamoxifen citrate tablets are available as:
10 mg Tablets:
Each tablet contains 15.2 mg of tamoxifen citrate USP equivalent to 10 mg of tamoxifen.
20 mg Tablets:
Each tablet contains 30.4 mg of tamoxifen citrate USP equivalent to 20 mg of tamoxifen.
Inactive Ingredients: corn starch (maize), croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, polyethylene glycol, povidone and titanium dioxide.
Chemically, tamoxifen citrate is the trans-isomer of a triphenylethylene derivative. The chemical name is (Z)2-[4-(1,2-diphenyl-1-butenyl) phenoxy]-N, N-dimethylethanamine 2 hydroxy-1,2,3- propanetricarboxylate (1:1). The structural and molecular formulas are:
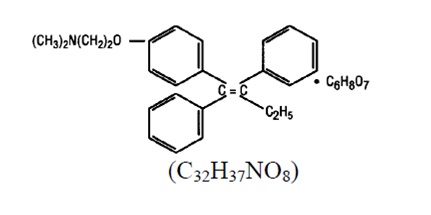
Tamoxifen citrate has a molecular weight of 563.62, the pKa' is 8.85, the equilibrium solubility in water at 37°C is 0.3159 mg/mL and in 0.02 N HCl at 37°C, it is 0.4045 mg/mL.
CLINICAL PHARMACOLOGY
Tamoxifen citrate is a nonsteroidal agent that has demonstrated potent antiestrogenic properties in animal test systems. The antiestrogenic effects may be related to its ability to compete with estrogen for binding sites in target tissues such as breast. Tamoxifen inhibits the induction of rat mammary carcinoma induced by dimethylbenzanthracene (DMBA) and causes the regression of already established DMBA-induced tumors. In this rat model, tamoxifen appears to exert its antitumor effects by binding the estrogen receptors.
In cytosols derived from human breast adenocarcinomas, tamoxifen competes with estradiol for estrogen receptor protein.
Absorption and Distribution:
Following a single oral dose of 20 mg tamoxifen, an average peak plasma concentration of 40 ng/mL (range 35 to 45 ng/mL) occurred approximately 5 hours after dosing. The decline in plasma concentrations of tamoxifen is biphasic with a terminal elimination half-life of about 5 to 7 days. The average peak plasma concentration of N-desmethyl tamoxifen is 15 ng/mL (range 10 to 20 ng/mL). Chronic administration of 10 mg tamoxifen given twice daily for 3 months to patients results in average steady-state plasma concentrations of 120 ng/mL (range 67 to 183 ng/mL) for tamoxifen and 336 ng/mL (range 148 to 654 ng/mL) for N-desmethyl tamoxifen. The average steady-state plasma concentrations of tamoxifen and N-desmethyl tamoxifen after administration of 20 mg tamoxifen once daily for 3 months are 122 ng/mL (range 71 to 183 ng/mL) and 353 ng/mL (range 152 to 706 ng/mL), respectively. After initiation of therapy, steady state concentrations for tamoxifen are achieved in about 4 weeks and steady-state concentrations for N-desmethyl tamoxifen are achieved in about 8 weeks, suggesting a half-life of approximately 14 days for this metabolite. In a steady-state, crossover study of 10 mg tamoxifen citrate tablets given twice a day vs. a 20 mg tamoxifen citrate tablet given once daily, the 20 mg tamoxifen citrate tablet was bioequivalent to the 10 mg tamoxifen citrate tablets.
Metabolism:
Tamoxifen is extensively metabolized after oral administration. N-desmethyl tamoxifen is the major metabolite found in patients’ plasma. The biological activity of N-desmethyl tamoxifen appears to be similar to that of tamoxifen. 4-Hydroxytamoxifen and a side chain primary alcohol derivative of tamoxifen have been identified as minor metabolites in plasma. Tamoxifen is a substrate of cytochrome P-450 3A, 2C9 and 2D6, and an inhibitor of P-glycoprotein.
Excretion:
Studies in women receiving 20 mg of 14 C tamoxifen have shown that approximately 65% of the administered dose was excreted from the body over a period of 2 weeks with fecal excretion as the primary route of elimination. The drug is excreted mainly as polar conjugates, with unchanged drug and unconjugated metabolites accounting for less than 30% of the total fecal radioactivity.
Special Populations:
The effects of age, gender and race on the pharmacokinetics of tamoxifen have not been determined. The effects of reduced liver function on the metabolism and pharmacokinetics of tamoxifen have not been determined.
Pediatric Patients:
The pharmacokinetics of tamoxifen and N-desmethyl tamoxifen were characterized using a population pharmacokinetic analysis with sparse samples per patient obtained from 27 female pediatric patients aged 2 to 10 years enrolled in a study designed to evaluate the safety, efficacy, and pharmacokinetics of tamoxifen citrate in treating McCune-Albright Syndrome. Rich data from two tamoxifen citrate pharmacokinetic trials in which 59 postmenopausal women with breast cancer completed the studies were included in the analysis to determine the structural pharmacokinetic model for tamoxifen. A one-compartment model provided the best fit to the data.
In pediatric patients, an average steady state peak plasma concentration (C ss , max ) and AUC were of 187 ng/mL and 4110 ng hr/mL, respectively, and C ss , max occurred approximately 8 hours after dosing. Clearance (CL/F) as body weight adjusted in female pediatric patients was approximately 2.3-fold higher than in female breast cancer patients. In the youngest cohort of female pediatric patients (2 to 6 year olds), CL/F was 2.6-fold higher; in the oldest cohort (7 to 10.9 year olds) CL/F was approximately 1.9-fold higher. Exposure to N-desmethyl tamoxifen was comparable between the pediatric and adult patients. The safety and efficacy of tamoxifen citrate for girls aged two to 10 years with McCune-Albright Syndrome and precocious puberty have not been studied beyond one year of treatment. The long-term effects of tamoxifen citrate therapy in girls have not been established. In adults treated with tamoxifen citrate an increase in incidence of uterine malignancies, stroke and pulmonary embolism has been noted (see BOXED WARNING ).
Drug-Drug Interactions:
In vitro studies showed that erythromycin, cyclosporin, nifedipine and diltiazem competitively inhibited formation of N-desmethyl tamoxifen with apparent K 1 of 20, 1, 45 and 30 μM, respectively. The clinical significance of these in vitro studies is unknown.
Tamoxifen reduced the plasma concentration of letrozole by 37% when these drugs were coadministered. Rifampin, a cytochrome P-450 3A4 inducer reduced tamoxifen AUC and C max by 86% and 55%, respectively. Aminoglutethimide reduces tamoxifen and N-desmethyl tamoxifen plasma concentrations. Medroxyprogesterone reduces plasma concentrations of N-desmethyl, but not tamoxifen.
In the anastrozole adjuvant trial, co-administration of anastrozole and tamoxifen citrate in breast cancer patients reduced anastrozole plasma concentration by 27% compared to those achieved with anastrozole alone; however, the coadministration did not affect the pharmacokinetics of tamoxifen or N-desmethyltamoxifen (see PRECAUTIONS -Drug Interactions ). Tamoxifen citrate should not be co-administered with anastrozole.
Clinical Studies
Metastatic Breast Cancer:
Premenopausal Women (Tamoxifen Citrate vs. Ablation):
Three prospective, randomized studies (Ingle, Pritchard, Buchanan) compared tamoxifen citrate to ovarian ablation (oophorectomy or ovarian irradiation) in premenopausal women with advanced breast cancer. Although the objective response rate, time to treatment failure, and survival were similar with both treatments, the limited patient accrual prevented a demonstration of equivalence. In an overview analysis of survival data from the 3 studies, the hazard ratio for death (tamoxifen citrate/ovarian ablation) was 1.00 with two-sided 95% confidence intervals of 0.73 to 1.37. Elevated serum and plasma estrogens have been observed in premenopausal women receiving tamoxifen citrate, but the data from the randomized studies do not suggest an adverse effect of this increase. A limited number of premenopausal patients with disease progression during tamoxifen citrate therapy responded to subsequent ovarian ablation.
Male Breast Cancer:
Published results from 122 patients (119 evaluable) and case reports in 16 patients (13 evaluable) treated with tamoxifen citrate have shown that tamoxifen citrate is effective for the palliative treatment of male breast cancer. Sixty-six of these 132 evaluable patients responded to tamoxifen citrate which constitutes a 50% objective response rate.
Adjuvant Breast Cancer:
Overview:
The Early Breast Cancer Trialists’ Collaborative Group (EBCTCG) conducted worldwide overviews of systemic adjuvant therapy for early breast cancer in 1985, 1990, and again in 1995. In 1998, 10-year outcome data were reported for 36,689 women in 55 randomized trials of adjuvant tamoxifen citrate using doses of 20 to 40 mg/day for 1 to 5+ years. Twenty-five percent of patients received 1 year or less of trial treatment, 52% received 2 years, and 23% received about 5 years. Forty-eight percent of tumors were estrogen receptor (ER) positive (> 10 fmol/mg), 21% were ER poor (< 10 fmol/l), and 31% were ER unknown. Among 29,441 patients with ER positive or unknown breast cancer, 58% were entered into trials comparing tamoxifen citrate to no adjuvant therapy and 42% were entered into trials comparing tamoxifen citrate in combination with chemotherapy vs. the same chemotherapy alone. Among these patients, 54% had node positive disease and 46% had node negative disease.
Among women with ER positive or unknown breast cancer and positive nodes who received about 5 years of treatment, overall survival at 10 years was 61.4% for tamoxifen citrate vs. 50.5% for control (logrank 2p < 0.00001). The recurrence-free rate at 10 years was 59.7% for tamoxifen citrate vs. 44.5% for control (logrank 2p < 0.00001). Among women with ER positive or unknown breast cancer and negative nodes who received about 5 years of treatment, overall survival at 10 years was 78.9% for tamoxifen citrate vs. 73.3% for control (logrank 2p < 0.00001). The recurrence-free rate at 10 years was 79.2% for tamoxifen citrate versus 64.3% for control (logrank 2p < 0.00001).
The effect of the scheduled duration of tamoxifen may be described as follows. In women with ER positive or unknown breast cancer receiving 1 year or less, 2 years or about 5 years of tamoxifen citrate, the proportional reductions in mortality were 12%, 17% and 26%, respectively (trend significant at 2p < 0.003). The corresponding reductions in breast cancer recurrence were 21%, 29% and 47% (trend significant at 2p < 0.00001).
Benefit is less clear for women with ER poor breast cancer in whom the proportional reduction in recurrence was 10% (2p = 0.007) for all durations taken together, or 9% (2p = 0.02) if contralateral breast cancers are excluded. The corresponding reduction in mortality was 6% (NS). The effects of about 5 years of tamoxifen citrate on recurrence and mortality were similar regardless of age and concurrent chemotherapy. There was no indication that doses greater than 20 mg per day were more effective.
Anastrozole Adjuvant ATAC Trial - Study of Anastrozole compared to Tamoxifen Citrate for Adjuvant Treatment of Early Breast Cancer – An anastrozole adjuvant trial was conducted in 9366 postmenopausal women with operable breast cancer who were randomized to receive adjuvant treatment with either anastrozole 1 mg daily, tamoxifen citrate 20 mg daily, or a combination of these two treatments for five years or until recurrence of the disease. At a median follow-up of 33 months, the combination of anastrozole and tamoxifen citrate did not demonstrate any efficacy benefit when compared with tamoxifen citrate therapy alone in all patients as well as in the hormone receptor-positive subpopulation. This treatment arm was discontinued from the trial. Please refer to CLINICAL PHARMACOLOGY-Special Populations-Drug-Drug Interactions , PRECAUTIONS-Laboratory Tests , PRECAUTIONS-Drug Interactions and ADVERSE REACTIONS sections for safety information from this trial. Please refer to the full prescribing information for ARIMIDEX ® (anastrozole) 1 mg Tablets for additional information on this trial.
Patients in the two monotherapy arms of the ATAC trial were treated for a median of 60 months (5 years) and followed for a median of 68 months. Disease-free survival in the intent-to-treat population was statistically significantly improved [Hazard Ratio (HR) = 0.87, 95% CI: 0.78, 0.97, p=0.0127] in the anastrozole arm compared to the tamoxifen citrate arm.
Node Positive - Individual Studies:
Two studies (Hubay and NSABP B-09) demonstrated an improved disease-free survival following radical or modified radical mastectomy in postmenopausal women or women 50 years of age or older with surgically curable breast cancer with positive axillary nodes when tamoxifen citrate was added to adjuvant cytotoxic chemotherapy. In the Hubay study, tamoxifen citrate was added to “low-dose” CMF (cyclophosphamide, methotrexate and fluorouracil). In the NSABP B-09 study, tamoxifen citrate was added to melphalan [L-phenylalanine mustard (P)] and fluorouracil (F).
In the Hubay study, patients with a positive (more than 3 fmol) estrogen receptor were more likely to benefit. In the NSABP B-09 study in women age 50 to 59 years, only women with both estrogen and progesterone receptor levels 10 fmol or greater clearly benefited, while there was a nonstatistically significant trend toward adverse effect in women with both estrogen and progesterone receptor levels less than 10 fmol. In women age 60 to 70 years, there was a trend toward a beneficial effect of tamoxifen citrate without any clear relationship to estrogen or progesterone receptor status.
Three prospective studies (ECOG-1178, Toronto, NATO) using tamoxifen citrate adjuvantly as a single agent demonstrated an improved disease-free survival following total mastectomy and axillary dissection for postmenopausal women with positive axillary nodes compared to placebo/no treatment controls. The NATO study also demonstrated an overall survival benefit.
Node Negative - Individual Studies:
NSABP B-14, a prospective, double-blind, randomized study, compared tamoxifen citrate to placebo in women with axillary node-negative, estrogen-receptor positive (≥10 fmol/mg cytosol protein) breast cancer (as adjuvant therapy, following total mastectomy and axillary dissection, or segmental resection, axillary dissection, and breast radiation). After five years of treatment, there was a significant improvement in disease-free survival in women receiving tamoxifen citrate. This benefit was apparent both in women under age 50 and in women at or beyond age 50.
One additional randomized study (NATO) demonstrated improved disease-free survival for tamoxifen citrate compared to no adjuvant therapy following total mastectomy and axillary dissection in postmenopausal women with axillary node-negative breast cancer. In this study, the benefits of tamoxifen citrate appeared to be independent of estrogen receptor status.
Duration of Therapy:
In the EBCTCG 1995 overview, the reduction in recurrence and mortality was greater in those studies that used tamoxifen for about 5 years than in those that used tamoxifen for a shorter period of therapy.
In the NSABP B-14 trial, in which patients were randomized to tamoxifen citrate 20 mg/day for 5 years vs. placebo and were disease-free at the end of this 5-year period were offered rerandomization to an additional 5 years of tamoxifen citrate or placebo. With 4 years of follow-up after this rerandomization, 92% of the women that received 5 years of tamoxifen citrate were alive and disease-free, compared to 86% of the women scheduled to receive 10 years of tamoxifen citrate (p=0.003). Overall survivals were 96% and 94%, respectively (p=0.08). Results of the B-14 study suggest that continuation of therapy beyond 5 years does not provide additional benefit.
A Scottish trial of 5 years of tamoxifen vs. indefinite treatment found a disease-free survival of 70% in the five-year group and 61% in the indefinite group, with 6.2 years median follow-up (HR=1.27, 95% CI 0.87 to 1.85).
In a large randomized trial conducted by the Swedish Breast Cancer Cooperative Group of adjuvant tamoxifen citrate 40 mg/day for 2 or 5 years, overall survival at 10 years was estimated to be 80% in the patients in the 5-year tamoxifen group, compared with 74% among corresponding patients in the 2-year treatment group (p=0.03). Disease-free survival at 10 years was 73% in the 5-year group and 67% in the 2-year group (p=0.009). Compared with 2 years of tamoxifen treatment, 5 years of treatment resulted in a slightly greater reduction in the incidence of contralateral breast cancer at 10 years, but this difference was not statistically significant.
Contralateral Breast Cancer:
The incidence of contralateral breast cancer is reduced in breast cancer patients (premenopausal and postmenopausal) receiving tamoxifen citrate compared to placebo. Data on contralateral breast cancer are available from 32,422 out of 36,689 patients in the 1995 overview analysis of the Early Breast Cancer Trialists Collaborative Group (EBCTCG). In clinical trials with tamoxifen citrate of 1 year or less, 2 years, and about 5 years duration, the proportional reductions in the incidence rate of contralateral breast cancer among women receiving tamoxifen citrate were 13% (NS), 26% (2p = 0.004) and 47% (2p < 0.00001), with a significant trend favoring longer tamoxifen duration (2p = 0.008). The proportional reductions in the incidence of contralateral breast cancer were independent of age and ER status of the primary tumor. Treatment with about 5 years of tamoxifen citrate reduced the annual incidence rate of contralateral breast cancer from 7.6 per 1,000 patients in the control group compared with 3.9 per 1,000 patients in the tamoxifen group.
In a large randomized trial in Sweden (the Stockholm Trial) of adjuvant tamoxifen citrate 40 mg/day for 2 to 5 years, the incidence of second primary breast tumors was reduced 40% (p <0.008) on tamoxifen compared to control. In the NSABP B-14 trial in which patients were randomized to tamoxifen citrate 20 mg/day for 5 years vs. placebo, the incidence of second primary breast cancers was also significantly reduced (p < 0.01). In NSABP B-14, the annual rate of contralateral breast cancer was 8.0 per 1000 patients in the placebo group compared with 5.0 per 1,000 patients in the tamoxifen group, at 10 years after first randomization.
Ductal Carcinoma in Situ:
NSABP B-24, a double-blind, randomized trial included women with ductal carcinoma in situ (DCIS). This trial compared the addition of tamoxifen citrate or placebo to treatment with lumpectomy and radiation therapy for women with DCIS. The primary objective was to determine whether 5 years of tamoxifen citrate therapy (20 mg/day) would reduce the incidence of invasive breast cancer in the ipsilateral (the same) or contralateral (the opposite) breast.
In this trial 1,804 women were randomized to receive either tamoxifen citrate or placebo for 5 years: 902 women were randomized to tamoxifen citrate 10 mg tablets twice a day and 902 women were randomized to placebo. As of December 31, 1998, follow-up data were available for 1,798 women and the median duration of follow-up was 74 months.
The tamoxifen citrate and placebo groups were well balanced for baseline demographic and prognostic factors. Over 80% of the tumors were less than or equal to 1 cm in their maximum dimension, were not palpable, and were detected by mammography alone. Over 60% of the study population was postmenopausal. In 16% of patients, the margin of the resected specimen was reported as being positive after surgery. Approximately half of the tumors were reported to contain comedo necrosis.
For the primary endpoint, the incidence of invasive breast cancer was reduced by 43% among women assigned to tamoxifen citrate (44 cases - tamoxifen citrate, 74 cases - placebo; p=0.004; relative risk (RR)=0.57, 95% CI: 0.39 to 0.84). No data are available regarding the ER status of the invasive cancers. The stage distribution of the invasive cancers at diagnosis was similar to that reported annually in the SEER data base.
Results are shown in Table 1. For each endpoint the following results are presented: the number of events and rate per 1,000 women per year for the placebo and tamoxifen citrate groups; and the relative risk (RR) and its associated 95% confidence interval (CI) between tamoxifen citrate and placebo. Relative risks less than 1.0 indicate a benefit of tamoxifen citrate therapy. The limits of the confidence intervals can be used to assess the statistical significance of the benefits of tamoxifen citrate therapy. If the upper limit of the CI is less than 1.0, then a statistically significant benefit exists.
Table 1. Major Outcomes of the NSABP B-24 Trial
| 1 Updated follow-up data (median 8.1 years) | ||||||
| Type of Event | Lumpectomy, radiotherapy, and placebo | Lumpectomy, radiotherapy, and tamoxifen citrate | RR | 95% CI Limits | ||
| No. of events | Rate per 1000 women per year | No. of events | Rate per 1000 women per year | |||
| Invasive breast cancer (Primary endpoint) | 74 | 16.73 | 44 | 9.60 | 0.57 | 0.39 to 0.84 |
| -Ipsilateral | 47 | 10.61 | 27 | 5.90 | 0.56 | 0.33 to 0.91 |
| -Contralateral | 25 | 5.64 | 17 | 3.71 | 0.66 | 0.33 to 1.27 |
| -Side undertermined | 2 | -- | 0 | -- | -- | |
| Secondary Endpoints | ||||||
| DCIS | 56 | 12.66 | 41 | 8.95 | 0.71 | 0.46 to 1.08 |
| -Ipsilateral | 46 | 10.40 | 38 | 8.29 | 0.88 | 0.51 to 1.25 |
| -Contralateral | 10 | 2.26 | 3 | 0.65 | 0.29 | 0.05 to 1.13 |
| All Breast Cancer Events | 129 | 29.16 | 84 | 18.34 | 0.63 | 0.47 to 0.83 |
| -All ipsilateral events | 96 | 21.70 | 65 | 14.19 | 0.65 | 0.47 to 0.91 |
| -All contralateral events | 37 | 8.36 | 20 | 4.37 | 0.52 | 0.29 to 0.92 |
| Deaths | 32 | 28 | ||||
| Uterine Malignancies 1 | 4 | 9 | ||||
| Endometrial Adenocarcinoma 1 | 4 | 0.57 | 8 | 1.15 | ||
| Uterine Sarcoma 1 | 0 | 0.0 | 1 | 0.14 | ||
| Second primary malignancies (other than endometrial and breast) | 30 | 29 | ||||
| Stroke | 2 | 7 | ||||
| Thromboembolic events (DVT, PE) | 5 | 15 | ||||
Survival was similar in the placebo and tamoxifen citrate groups. At 5 years from study entry, survival was 97% for both groups.
Reduction in Breast Cancer Incidence in High Risk Women:
The Breast Cancer Prevention Trial (BCPT, NSABP P-1) was a double-blind, randomized, placebo-controlled trial with a primary objective to determine whether 5 years of tamoxifen citrate therapy (20 mg/day) would reduce the incidence of invasive breast cancer in women at high risk for the disease (See INDICATIONS AND USAGE ). Secondary objectives included an evaluation of the incidence of ischemic heart disease; the effects on the incidence of bone fractures; and other events that might be associated with the use of tamoxifen citrate, including: endometrial cancer, pulmonary embolus, deep vein thrombosis, stroke, and cataract formation and surgery (See WARNINGS ).
The Gail Model was used to calculate predicted breast cancer risk for women who were less than 60 years of age and did not have lobular carcinoma in situ (LCIS). The following risk factors were used: age; number of first-degree female relatives with breast cancer; previous breast biopsies; presence or absence of atypical hyperplasia; nulliparity; age at first live birth; and age at menarche. A 5-year predicted risk of breast cancer of ≥ 1.67% was required for entry into the trial.
In this trial, 13,388 women of at least 35 years of age were randomized to receive either tamoxifen citrate or placebo for five years. The median duration of treatment was 3.5 years. As of January 31, 1998, follow-up data is available for 13,114 women. Twenty-seven percent of women randomized to placebo (1,782) and 24% of women randomized to tamoxifen citrate (1,596) completed 5 years of therapy. The demographic characteristics of women on the trial with follow-up data are shown in Table 2.
Table 2. Demographic Characteristics of Women in the NSABP P-1 Trial
| Characteristic | Placebo | Tamoxifen | ||
| # | % | # | % | |
| Age (yrs.) | ||||
| 35 to 39 | 184 | 3 | 158 | 2 |
| 40 to 49 | 2,394 | 36 | 2,411 | 37 |
| 50 to 59 | 2,011 | 31 | 2,019 | 31 |
| 60 to 69 | 1,588 | 24 | 1,563 | 24 |
| ≥70 | 393 | 6 | 393 | 6 |
| Age at first live birth (yrs.) Nulliparous | 1,202 | 18 | 1,205 | 18 |
| 12 to 19 | 915 | 14 | 946 | 15 |
| 20 to 24 | 2,448 | 37 | 2,449 | 37 |
| 25 to 29 | 1,399 | 21 | 1,367 | 21 |
| ≥30 | 606 | 9 | 577 | 9 |
| Race | ||||
| White | 6,333 | 96 | 6,323 | 96 |
| Black | 109 | 2 | 103 | 2 |
| Other | 128 | 2 | 118 | 2 |
| Age at menarche | ||||
| ≥14 | 1,243 | 19 | 1,170 | 18 |
| 12 to 13 | 3,610 | 55 | 3,610 | 55 |
| ≤11 | 1,717 | 26 | 1,764 | 27 |
| # of first degree relatives with breast cancer | ||||
| 0 | 1,584 | 24 | 1,525 | 23 |
| 1 | 3,714 | 57 | 3,744 | 57 |
| 2+ | 1,272 | 19 | 1,275 | 20 |
| Prior Hysterectomy | ||||
| No | 4,173 | 63.5 | 4,018 | 62.4 |
| Yes | 2,397 | 36.5 | 2,464 | 37.7 |
| # of previous breast biopsies | ||||
| 0 | 2,935 | 45 | 2,923 | 45 |
| 1 | 1,833 | 28 | 1,850 | 28 |
| ≥2 | 1,802 | 27 | 1,771 | 27 |
| History of atypical hyperplasia in the breast | ||||
| No | 5,958 | 91 | 5,969 | 91 |
| Yes | 612 | 9 | 575 | 9 |
| History of LCIS at entry | ||||
| No | 6,165 | 94 | 6,135 | 94 |
| Yes | 405 | 6 | 409 | 6 |
| 5-year predicted breast cancer risk (%) | ||||
| ≤2.00 | 1,646 | 25 | 1,626 | 25 |
| 2.01 to 3.00 | 2,028 | 31 | 2,057 | 31 |
| 3.01 to 5.00 | 1,787 | 27 | 1,707 | 26 |
| ≥5.01 | 1,109 | 17 | 1,162 | 18 |
| Total | 6,570 | 100.0 | 6,544 | 100.0 |
Results are shown in Table 3. After a median follow-up of 4.2 years, the incidence of invasive breast cancer was reduced by 44% among women assigned to tamoxifen citrate (86 cases- tamoxifen citrate, 156 cases-placebo; p<0.00001; relative risk (RR)=0.56, 95% CI: 0.43 to 0.72). A reduction in the incidence of breast cancer was seen in each prospectively specified age group (≤ 49, 50 to 59, ≥ 60), in women with or without LCIS, and in each of the absolute risk levels specified in Table 3. A non-significant decrease in the incidence of ductal carcinoma in situ (DCIS) was seen (23- tamoxifen citrate, 35-placebo; RR=0.66; 95% CI: 0.39 to 1.11).
There was no statistically significant difference in the number of myocardial infarctions, severe angina, or acute ischemic cardiac events between the two groups (61- tamoxifen citrate, 59-placebo; RR=1.04, 95% CI: 0.73 to 1.49).
No overall difference in mortality (53 deaths in tamoxifen citrate group vs. 65 deaths in placebo group) was present. No difference in breast cancer-related mortality was observed (4 deaths in tamoxifen citrate group vs. 5 deaths in placebo group).
Although there was a non-significant reduction in the number of hip fractures (9 on tamoxifen citrate, 20 on placebo) in the tamoxifen citrate group, the number of wrist fractures was similar in the two treatment groups (69 on tamoxifen citrate, 74 on placebo). A subgroup analysis of the P-1 trial, suggests a difference in effect in bone mineral density (BMD) related to menopausal status in patients receiving tamoxifen citrate. In postmenopausal women there was no evidence of bone loss of the lumbar spine and hip. Conversely, tamoxifen citrate was associated with significant bone loss of the lumbar spine and hip in premenopausal women.
The risks of tamoxifen citrate therapy include endometrial cancer, DVT, PE, stroke, cataract formation and cataract surgery (See Table 3). In the NSABP P-1 trial, 33 cases of endometrial cancer were observed in the tamoxifen citrate group vs. 14 in the placebo group (RR=2.48, 95% CI: 1.27 to 4.92). Deep vein thrombosis was observed in 30 women receiving tamoxifen citrate vs. 19 in women receiving placebo (RR=1.59, 95% CI: 0.86 to 2.98). Eighteen cases of pulmonary embolism were observed in the tamoxifen citrate group vs. 6 in the placebo group (RR=3.01, 95% CI: 1.15 to 9.27). There were 34 strokes on the tamoxifen citrate arm and 24 on the placebo arm (RR=1.42; 95% CI: 0.82 to 2.51). Cataract formation in women without cataracts at baseline was observed in 540 women taking tamoxifen citrate vs. 483 women receiving placebo (RR=1.13, 95% CI: 1.00 to 1.28). Cataract surgery (with or without cataracts at baseline) was performed in 201 women taking tamoxifen citrate vs. 129 women receiving placebo (RR=1.51, 95% CI: 1.21 to 1.89) (See WARNINGS ).
Table 3 summarizes the major outcomes of the NSABP P-1 trial. For each endpoint, the following results are presented: the number of events and rate per 1000 women per year for the placebo and tamoxifen citrate groups; and the relative risk (RR) and its associated 95% confidence interval (CI) between tamoxifen citrate and placebo. Relative risks less than 1.0 indicate a benefit of tamoxifen citrate therapy. The limits of the confidence intervals can be used to assess the statistical significance of the benefits or risks of tamoxifen citrate therapy. If the upper limit of the CI is less than 1.0, then a statistically significant benefit exists.
For most participants, multiple risk factors would have been required for eligibility. This table considers risk factors individually, regardless of other co-existing risk factors, for women who developed breast cancer. The 5-year predicted absolute breast cancer risk accounts for multiple risk factors in an individual and should provide the best estimate of individual benefit (See INDICATIONS AND USAGE ).
Table 3. Major Outcomes of the NSABP P-1 Trial
| TYPE OF EVENT | # OF EVENTS | RATE/1000 WOMEN/YEAR | 95% CI | |||
| PLACEBO | TAMOXIFEN CITRATE | PLACEBO | TAMOXIFEN CITRATE | RR | LIMITS | |
| Invasive Breast Cancer | 156 | 86 | 6.49 | 3.58 | 0.56 | 0.43 to 0.72 |
| Age ≤49 | 59 | 38 | 6.34 | 4.11 | 0.65 | 0.43 to 0.98 |
| Age 50 to 59 | 46 | 25 | 6.31 | 3.53 | 0.56 | 0.35 to 0.91 |
| Age ≥60 | 51 | 23 | 7.17 | 3.22 | 0.45 | 0.27 to 0.74 |
| Risk Factors for Breast Cancer History, LCIS | ||||||
| No | 140 | 78 | 6.23 | 3.51 | 0.56 | 0.43 to 0.74 |
| Yes | 16 | 8 | 12.73 | 6.33 | 0.50 | 0.21 to 1.17 |
| History, Atypical Hyperplasia No | 138 | 84 | 6.37 | 3.89 | 0.61 | 0.47 to 0.80 |
| Yes | 18 | 2 | 8.69 | 1.05 | 0.12 | 0.03 to 0.52 |
| No. First Degree Relatives 0 | 32 | 17 | 5.97 | 3.26 | 0.55 | 0.30 to 0.98 |
| 1 | 80 | 45 | 5.81 | 3.31 | 0.57 | 0.40 to 0.82 |
| 2 | 35 | 18 | 8.92 | 4.67 | 0.52 | 0.30 to 0.92 |
| ≥3 | 9 | 6 | 13.33 | 7.58 | 0.57 | 0.20 to 1.59 |
| 5-Year Predicted Breast Cancer Risk (as calculated by the Gail Model) | ||||||
| ≤2.00% | 31 | 13 | 5.36 | 2.26 | 0.42 | 0.22 to 0.81 |
| 2.01 to 3.00% | 39 | 28 | 5.25 | 3.83 | 0.73 | 0.45 to 1.18 |
| 3.01 to 5.00% | 36 | 26 | 5.37 | 4.06 | 0.76 | 0.46 to 1.26 |
| ≥5.00% | 50 | 19 | 13.15 | 4.71 | 0.36 | 0.21 to 0.61 |
| DCIS | 35 | 23 | 1.47 | 0.97 | 0.66 | 0.39 to 1.11 |
| Fractures (protocol-specified sites) | 92 1 | 76 1 | 3.87 | 3.20 | 0.61 | 0.83 to 1.12 |
| Hip | 20 | 9 | 0.84 | 0.38 | 0.45 | 0.18 to 1.04 |
| Wrist 2 | 74 | 69 | 3.11 | 2.91 | 0.93 | 0.67 to 1.29 |
| Total Ischemic Events | 59 | 61 | 2.47 | 2.57 | 1.04 | 0.71 to 1.51 |
| Myocardial Infarction | 27 | 27 | 1.13 | 1.13 | 1.00 | 0.57 to 1.78 |
| Fatal | 8 | 7 | 0.33 | 0.29 | 0.88 | 0.27 to 2.77 |
| Nonfatal | 19 | 20 | 0.79 | 0.84 | 1.06 | 0.54 to 2.09 |
| Angina 3 | 12 | 12 | 0.50 | 0.50 | 1.00 | 0.41 to 2.44 |
| Acute Ischemic Syndrome 4 | 20 | 22 | 0.84 | 0.92 | 1.11 | 0.58 to 2.13 |
| Uterine Malignancies (among women with an intact uterus) 10 | 17 | 57 | ||||
| Endometrial Adenocarcinoma 10 | 17 | 53 | 0.71 | 2.20 | ||
| Uterine Sarcoma 10 | 0 | 4 | 0.0 | 0.17 | ||
| Stroke 5 | 24 | 34 | 1.00 | 1.43 | 1.42 | 0.82 to 2.51 |
| Transient Ischemic Attack | 21 | 18 | 0.88 | 0.75 | 0.86 | 0.43 to 1.70 |
| Pulmonary Emboli 6 | 6 | 18 | 0.25 | 0.75 | 3.01 | 1.15 to 9.27 |
| Deep-Vein Thrombosis 7 | 19 | 30 | 0.79 | 1.26 | 1.59 | 0.86 to 2.98 |
| Cataracts Developing on Study 8 | 483 | 540 | 22.51 | 25.41 | 1.13 | 1.00 to 1.28 |
| Underwent Cataract Surgery 8 | 63 | 101 | 2.83 | 4.57 | 1.62 | 1.18 to 2.22 |
| Underwent Cataract Surgery 9 | 129 | 201 | 5.44 | 8.56 | 1.58 | 1.26 to 1.97 |
| 1 Two women had hip and wrist fractures 2 Includes Colles’ and other lower radius fractures 3 Requiring angioplasty or CABG 4 New Q-wave on ECG; no angina or elevation of serum enzymes; or angina requiring hospitalization without surgery 5 Seven cases were fatal; three in the placebo group and four in the tamoxifen citrate group 6 Three cases in the tamoxifen citrate group were fatal 7 All but three cases in each group required hospitalization 8 Based on women without cataracts at baseline (6,230-Placebo, 6,199-Tamoxifen Citrate) 9 All women (6,707-Placebo, 6,681-Tamoxifen Citrate) 10 Updated long-term follow-up data (median 6.9 years) from NSABP P-1 study added after cut-off for the other information in this table. | ||||||
Table 4 describes the characteristics of the breast cancers in the NSABP P-1 trial and includes tumor size, nodal status, ER status. Tamoxifen citrate decreased the incidence of small estrogen receptor positive tumors, but did not alter the incidence of estrogen receptor negative tumors or larger tumors.
Table 4. Characteristics of Breast Cancer in NSABP P-1 Trial
| Staging Parameter | Placebo N=156 | Tamoxifen N=86 | Total N=242 |
| Tumor Size: T1 | 117 | 60 | 177 |
| T2 | 28 | 20 | 48 |
| T3 | 7 | 3 | 10 |
| T4 | 1 | 2 | 3 |
| Unknown | 3 | 1 | 4 |
| Nodal status: Negative | 103 | 56 | 159 |
| 1 to 3 positive nodes | 29 | 14 | 43 |
| ≥4 positive nodes | 10 | 12 | 22 |
| Unknown | 14 | 4 | 18 |
| Stage: I | 88 | 47 | 135 |
| II: node negative | 15 | 9 | 24 |
| II: node positive | 33 | 22 | 55 |
| III | 6 | 4 | 10 |
| IV | 2 1 | 1 | 3 |
| Unknown Estrogen receptor: Positive | 12 115 | 3 38 | 15 153 |
| Negative | 27 | 36 | 63 |
| Unknown | 14 | 12 | 26 |
| 1 One participant presented with a suspicious bone scan but did not have documented metastases. She subsequently died of metastatic breast cancer. | |||
Interim results from 2 trials in addition to the NSABP P-1 trial examining the effects of tamoxifen in reducing breast cancer incidence have been reported.
The first was the Italian Tamoxifen Prevention trial. In this trial women between the ages of 35 and 70, who had had a total hysterectomy, were randomized to receive 20 mg tamoxifen or matching placebo for 5 years. The primary endpoints were occurrence of, and death from, invasive breast cancer. Women without any specific risk factors for breast cancer were to be entered. Between 1992 and 1997, 5408 women were randomized. Hormone Replacement Therapy (HRT) was used in 14% of participants. The trial closed in 1997 due to the large number of dropouts during the first year of treatment (26%). After 46 months of follow-up there were 22 breast cancers in women on placebo and 19 in women on tamoxifen. Although no decrease in breast cancer incidence was observed, there was a trend for a reduction in breast cancer among women receiving protocol therapy for at least 1 year (19-placebo, 11- tamoxifen). The small numbers of participants along with the low level of risk in this otherwise healthy group precluded an adequate assessment of the effect of tamoxifen in reducing the incidence of breast cancer.
The second trial, the Royal Marsden Trial (RMT) was reported as an interim analysis. The RMT was begun in 1986 as a feasibility study of whether larger scale trials could be mounted. The trial was subsequently extended to a pilot trial to accrue additional participants to further assess the safety of tamoxifen. Twenty-four hundred and seventy-one women were entered between 1986 and 1996; they were selected on the basis of a family history of breast cancer. HRT was used in 40% of participants. In this trial, with a 70 month median follow-up, 34 and 36 breast cancers (8 noninvasive, 4 on each arm) were observed among women on tamoxifen and placebo, respectively. Patients in this trial were younger than those in the NSABP P-1 trial and may have been more likely to develop ER (-) tumors, which are unlikely to be reduced in number by tamoxifen therapy. Although women were selected on the basis of family history and were thought to have a high risk of breast cancer, few events occurred, reducing the statistical power of the study. These factors are potential reasons why the RMT may not have provided an adequate assessment of the effectiveness of tamoxifen in reducing the incidence of breast cancer.
In these trials, an increased number of cases of deep vein thrombosis, pulmonary embolus, stroke, and endometrial cancer were observed on the tamoxifen arm compared to the placebo arm. The frequency of events was consistent with the safety data observed in the NSABP P-1 trial.
McCune-Albright Syndrome:
A single, uncontrolled multicenter trial of tamoxifen citrate 20 mg once a day was conducted in a heterogenous group of girls with McCune-Albright Syndrome and precocious puberty manifested by physical signs of pubertal development, episodes of vaginal bleeding and/or advanced bone age (bone age of at least 12 months beyond chronological age). Twenty-eight female pediatric patients, aged 2 to 10 years, were treated for up to 12 months. Effect of treatment on frequency of vaginal bleeding, bone age advancement, and linear growth rate was assessed relative to prestudy baseline. Tamoxifen citrate treatment was associated with a 50% reduction in frequency of vaginal bleeding episodes by patient or family report (mean annualized frequency of 3.56 episodes at baseline and 1.73 episodes on-treatment). Among the patients who reported vaginal bleeding during the pre-study period, 62% (13 out of 21 patients) reported no bleeding for a 6-month period and 33% (7 out of 21 patients) reported no vaginal bleeding for the duration of the trial. Not all patients improved on treatment and a few patients not reporting vaginal bleeding in the 6 months prior to enrollment reported menses on treatment. Tamoxifen citrate therapy was associated with a reduction in mean rate of increase of bone age. Individual responses with regard to bone age advancement were highly heterogeneous. Linear growth rate was reduced during the course of tamoxifen citrate treatment in a majority of patients (mean change of 1.68 cm/year relative to baseline; change from 7.47 cm/year at baseline to 5.79 cm/year on study). This change was not uniformly seen across all stages of bone maturity; all recorded response failures occurred in patients with bone ages less than 7 years at screening.
Mean uterine volume increased after 6 months of treatment and doubled at the end of the one- year study. A causal relationship has not been established; however, as an increase in the incidence of endometrial adenocarcinoma and uterine sarcoma has been noted in adults treated with tamoxifen citrate (see BOXED WARNING ), continued monitoring of McCune-Albright patients treated with tamoxifen citrate for long-term uterine effects is recommended. The safety and efficacy of tamoxifen citrate for girls aged two to 10 years with McCune-Albright Syndrome and precocious puberty have not been studied beyond one year of treatment. The long-term effects of tamoxifen citrate therapy in girls have not been established.
HOW SUPPLIED
Tamoxifen Citrate Tablets, USP 10 mg contains 15.2 mg of tamoxifen citrate equivalent to 10 mg of tamoxifen. The tablets are white to off-white, round, biconvex, film-coated, unscored tablets debossed with T10 on one side and plain on the other side.
Bottles of 60 NDC 59651-299-60
Tamoxifen Citrate Tablets, USP 20 mg contains 30.4 mg of tamoxifen citrate equivalent to 20 mg of tamoxifen. The tablets are white to off-white, round, biconvex, film-coated, unscored tablets debossed with T20 on one side and plain on the other side.
Bottles of 30 NDC 59651-300-30 Bottles of 90 NDC 59651-300-90
Store at 20° to 25°C (68° to 77°F) [see USP controlled room temperature]. Dispense in a well-closed, light-resistant container. The brands listed are the trademarks of their respective owners and are not trademarks of the Aurobindo Pharma Limited.
Dispense with Medication Guide available at: www.aurobindousa.com/product-medication-guides.
Distributed by: Aurobindo Pharma USA, Inc. 279 Princeton-Hightstown Road East Windsor, NJ 08520 Manufactured by: Eugia Pharma Specialities Limited Medchal-Malkajgiri District- 500101 India Issued: August 2019