Get your patient on Ursodiol - Ursodiol capsule (Ursodiol)
Ursodiol - Ursodiol capsule prescribing information
INDICATIONS AND USAGE
- Ursodiol is indicated for patients with radiolucent, noncalcified gallbladder stones < 20 mm in greatest diameter in whom elective cholecystectomy would be undertaken except for the presence of increased surgical risk due to systemic disease, advanced age, idiosyncratic reaction to general anesthesia, or for those patients who refuse surgery. Safety of use of ursodiol beyond 24 months is not established.
- Ursodiol is indicated for the prevention of gallstone formation in obese patients experiencing rapid weight loss.
DOSAGE AND ADMINISTRATION
Gallstone Dissolution
The recommended dose for ursodiol treatment of radiolucent gallbladder stones is 8 - 10 mg/kg/day given in 2 or 3 divided doses. Ultrasound images of the gallbladder should be obtained at 6-month intervals for the first year of ursodiol therapy to monitor gallstone response. If gallstones appear to have dissolved, ursodiol therapy should be continued and dissolution confirmed on a repeat ultrasound examination within 1 to 3 months. Most patients who eventually achieve complete stone dissolution will show partial or complete dissolution at the first on-treatment reevaluation. If partial stone dissolution is not seen by 12 months of ursodiol therapy, the likelihood of success is greatly reduced.
Gallstone Prevention
The recommended dosage of ursodiol for gallstone prevention in patients undergoing rapid weight loss is 600 mg/day (300 mg b.i.d.).
CONTRAINDICATIONS
- Ursodiol will not dissolve calcified cholesterol stones, radiopaque stones, or radiolucent bile pigment stones. Hence, patients with such stones are not candidates for ursodiol therapy.
- Patients with compelling reasons for cholecystectomy including unremitting acute cholecystitis, cholangitis, biliary obstruction, gallstone pancreatitis, or biliary-gastrointestinal fistula are not candidates for ursodiol therapy.
- Allergy to bile acids.
ADVERSE REACTIONS
The nature and frequency of adverse experiences were similar across all groups.
The following tables provide comprehensive listings of the adverse experiences reported that occurred with a 5% incidence level:
| Gallstone Dissolution In Ursodiol and Placebo Patients | ||||
GALLSTONE DISSOLUTION | ||||
Ursodiol 8 - 10 mg/kg/day (N=155) | Placebo (N=159) | |||
N | (%) | N | (%) | |
Body as a Whole | ||||
Allergy | 8 | (5.2) | 7 | (4.4) |
Chest Pain | 5 | (3.2) | 10 | (6.3) |
Fatigue | 7 | (4.5) | 8 | (5.0) |
Infection Viral | 30 | (19.4) | 41 | (25.8) |
Digestive System | ||||
Abdominal Pain | 67 | (43.2) | 70 | (44.0) |
Cholecystitis | 8 | (5.2) | 7 | (4.4) |
Constipation | 15 | (9.7) | 14 | (8.8) |
Diarrhea | 42 | (27.1) | 34 | (21.4) |
Dyspepsia | 26 | (16.8) | 18 | (11.3) |
Flatulence | 12 | (7.7) | 12 | (7.5) |
Gastrointestinal Disorder | 6 | (3.9) | 8 | (5.0) |
Nausea | 22 | (14.2) | 27 | (17.0) |
Vomiting | 15 | (9.7) | 11 | (6.9) |
Musculoskeletal System | ||||
Arthralgia | 12 | (7.7) | 24 | (15.1) |
Arthritis | 9 | (5.8) | 4 | (2.5) |
Back Pain | 11 | (7.1) | 18 | (11.3) |
Myalgia | 9 | (5.8) | 9 | (5.7) |
Nervous System | ||||
Headache | 28 | (18.1) | 34 | (21.4) |
Insomnia | 3 | (1.9) | 8 | (5.0) |
Respiratory System | ||||
Bronchitis | 10 | (6.5) | 6 | (3.8) |
Coughing | 11 | (7.1) | 7 | (4.4) |
Pharyngitis | 13 | (8.4) | 5 | (3.1) |
Rhinitis | 8 | (5.2) | 11 | (6.9) |
Sinusitis | 17 | (11.0) | 18 | (11.3) |
Upper Respiratory Tract Infection | 24 | (15.5) | 21 | (13.2) |
Urogenital System | ||||
Urinary Tract Infection | 10 | (6.5) | 7 | (4.4) |
| Gallstone Prevention in Ursodiol and Placebo-Treated Patients | ||||
| Postmarketing Experience | ||||
| The following adverse reactions, presented by system organ class in alphabetical order, have been identified during post-approval use of ursodiol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a casual relationship to drug exposure. | ||||
| Gastrointestinal disorders : enteroliths (bezoars). | ||||
GALLSTONE PREVENTION | ||||
Ursodiol 600 mg (N=322) | Placebo (N=325) | |||
N | (%) | N | (%) | |
Body as a Whole | ||||
Fatigue | 25 | (7.8) | 33 | (10.2) |
Infection Viral | 29 | (9.0) | 29 | (8.9) |
Influenza-like Symptoms | 21 | (6.5) | 19 | (5.8) |
Digestive System | ||||
Abdominal Pain | 20 | (6.2) | 39 | (12.0) |
Constipation | 85 | (26.4) | 72 | (22.2) |
Diarrhea | 81 | (25.2) | 68 | (20.9) |
Flatulence | 15 | (4.7) | 24 | (7.4) |
Nausea | 56 | (17.4) | 43 | (13.2) |
Vomiting | 44 | (13.7) | 44 | (13.5) |
Musculoskeletal System | ||||
Back Pain | 38 | (11.8) | 21 | (6.5) |
Musculoskeletal Pain | 19 | (5.9) | 15 | (4.6) |
Nervous System | ||||
Dizziness | 53 | (16.5) | 42 | (12.9) |
Headache | 80 | (24.8) | 78 | (24.0) |
Respiratory System | ||||
Pharyngitis | 10 | (3.1) | 19 | (5.8) |
Sinusitis | 17 | (5.3) | 18 | (5.5) |
Upper Respiratory Tract Infection | 40 | (12.4) | 35 | (10.8) |
Skin and Appendages | ||||
Alopecia | 17 | (5.3) | 8 | (2.5) |
Urogenital System | ||||
Dysmenorrhea | 18 | (5.6) | 19 | (5.8) |
DESCRIPTION
Ursodiol is a bile acid available as 300 mg capsules suitable for oral administration.
Ursodiol, USP (ursodeoxycholic acid), is a naturally occurring bile acid found in small quantities in normal human bile and in the biles of certain other mammals. It is a bitter-tasting, white powder freely soluble in ethanol, methanol, and glacial acetic acid; sparingly soluble in chloroform; slightly soluble in ether; and insoluble in water. The chemical name for ursodiol is 3α, 7β-Dihydroxy-5β-cholan-24-oic acid (C 24 H 40 O 4 ). Ursodiol, USP has a molecular weight of 392.57. Its structure is shown below:
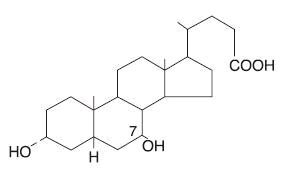
Inactive Ingredients : Corn starch, magnesium stearate, silicon dioxide and the capsule shell contain the following ingredients, gelatin, titanium dioxide, D&C Red #28, FD&C Blue #1 and FD&C Red #40.
The imprinting ink contains the following: black iron oxide, D&C Yellow #10 Aluminum Lake, FD&C Blue #1 Aluminum Lake, FD&C Blue #2 Aluminum Lake, FD&C Red #40 Aluminum Lake, propylene glycol and shellac glaze.
CLINICAL PHARMACOLOGY
About 90% of a therapeutic dose of ursodiol is absorbed in the small bowel after oral administration. After absorption, ursodiol enters the portal vein and undergoes efficient extraction from portal blood by the liver (i.e., there is a large “first-pass” effect) where it is conjugated with either glycine or taurine and is then secreted into the hepatic bile ducts. Ursodiol in bile is concentrated in the gallbladder and expelled into the duodenum in gallbladder bile via the cystic and common ducts by gallbladder contractions provoked by physiologic responses to eating. Only small quantities of ursodiol appear in the systemic circulation and very small amounts are excreted into urine. The sites of the drug’s therapeutic actions are in the liver, bile, and gut lumen.
Beyond conjugation, ursodiol is not altered or catabolized appreciably by the liver or intestinal mucosa. A small proportion of orally administered drug undergoes bacterial degradation with each cycle of enterohepatic circulation. Ursodiol can be both oxidized and reduced at the 7-carbon, yielding either 7-keto-lithocholic acid or lithocholic acid, respectively. Further, there is some bacterially catalyzed deconjugation of glyco- and tauro-ursodeoxycholic acid in the small bowel. Free ursodiol, 7-keto-lithocholic acid, and lithocholic acid are relatively insoluble in aqueous media and larger proportions of these compounds are lost from the distal gut into the feces. Reabsorbed free ursodiol is reconjugated by the liver. Eighty percent of lithocholic acid formed in the small bowel is excreted in the feces, but the 20% that is absorbed is sulfated at the 3-hydroxyl group in the liver to relatively insoluble lithocholyl conjugates which are excreted into bile and lost in feces. Absorbed 7-keto-lithocholic acid is stereospecifically reduced in the liver to chenodiol.
Lithocholic acid causes cholestatic liver injury and can cause death from liver failure in certain species unable to form sulfate conjugates. Lithocholic acid is formed by 7-dehydroxylation of the dihydroxy bile acids (ursodiol and chenodiol) in the gut lumen. The 7-dehydroxylation reaction appears to be alpha-specific, i.e., chenodiol is more efficiently 7-dehydroxylated than ursodiol and, for equimolar doses of ursodiol and chenodiol, levels of lithocholic acid appearing in bile are lower with the former. Man has the capacity to sulfate lithocholic acid. Although liver injury has not been associated with ursodiol therapy, a reduced capacity to sulfate may exist in some individuals, but such a deficiency has not yet been clearly demonstrated.
HOW SUPPLIED
Ursodiol Capsules USP, 300 mg are #0 capsules with a pink opaque cap, white opaque body, imprinted “Є503” in black ink on cap and body, filled with white powder.
They are supplied:
NDC 24658-780-01 Bottles of 100 Capsules.
Store at 20° - 25°C (68° - 77°F) [see USP Controlled Room Temperature]. Dispense contents in a tight, light-resistant container as defined in the USP.
Keep out of reach of children.
Distributed by:
PuraCap Laboratories, LLC
DBA Blu Pharmaceuticals
Greenvale, NY 11548
Manufactured in USA
Rev. 08-2024-00
MF780REV08/24B
OE2631