Get your patient on Zovia 1/35 - Ethynodiol Diacetate And Ethinyl Estradiol Tablets (Ethynodiol Diacetate And Ethinyl Estradiol Tablets)
Zovia 1/35 - Ethynodiol Diacetate And Ethinyl Estradiol Tablets prescribing information
INDICATIONS AND USAGE
Zovia 1/35 is indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception.
Oral contraceptives are highly effective. Table 1 lists the typical accidental pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization and progestogen implants and injections, depends upon the reliability with which they are used. Correct and consistent use of methods can result in lower failure rates.
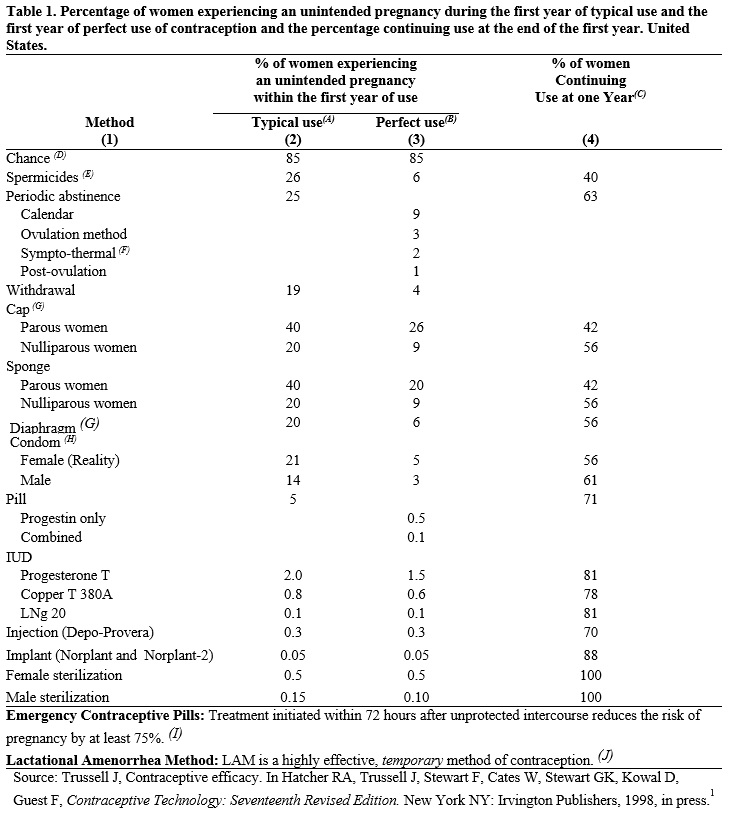
(A) Among typical couples who initiate use of a method (not necessarily for the first time), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
(B) Among couples who initiate use of a method (not necessarily for the first time) and who use it perfectly (both consistently and correctly), the percentage who experience an accidental pregnancy during the first year if they do not stop use for any other reason.
(C) Among couples attempting to avoid pregnancy, the percentage who continue to use a method for one year.
(D) The percents becoming pregnant in columns (2) and (3) are based on data from populations where contraception is not used and from women who cease using contraception in order to become pregnant. Among such populations, about 89% become pregnant within one year. This estimate was lowered slightly (to 85%) to represent the percent who would become pregnant within one year among women now relying on reversible methods of contraception if they abandoned contraception altogether.
(E) Foams, creams, gels, vaginal suppositories, and vaginal film.
(F) Cervical mucus (ovulation) method supplemented by calendar in the pre-ovulatory and basal body temperature in the post-ovulatory phases.
(G) With spermicidal cream or jelly.
(H) Without spermicides.
(I) The treatment schedule is one dose within 72 hours after unprotected intercourse and a second dose 12 hours after the first dose. The Food and Drug Administration has declared the following brands of oral contraceptives to be safe and effective for emergency contraception: Ovral (1 dose is 2 white pills), Alesse (1 dose is 5 pink pills), Nordette or Levlen (1 dose is 2 light-orange pills), Lo/Ovral (1 dose is 4 white pills), Triphasil or Tri-Levlen (1 dose is 4 yellow pills).
(J) However, to maintain effective protection against pregnancy, another method of contraception must be used as soon as menstruation resumes, the frequency or duration of breastfeeds is reduced, bottle feeds are introduced, or the baby reaches six months of age.
DOSAGE AND ADMINISTRATION
To achieve maximum contraceptive effectiveness, oral contraceptives must be taken exactly as directed and at intervals of 24 hours.
IMPORTANT: If the Sunday start schedule is selected, the patient should be instructed to use an additional method of protection until after the first week of administration in the initial cycle.
The possibility of ovulation and conception prior to initiation of use should be considered.
Zovia 1/35
Dosage Schedules
The Zovia 1/35 tablet dispensers contain 21 pale pink active tablets arranged in three numbered rows of 7 tablets each, followed by a fourth row of 7 white placebo (inactive) tablets .
Days of the week are embossed on the plastic compact just above the tablets, starting with Sunday on the left.
28-Day Schedule: For a DAY 1 START, count the first day of menstrual flow as Day 1 and the first tablet (pale pink) is then taken on Day 1. For a SUNDAY START when menstrual flow begins on or before Sunday, the first tablet (pale pink) is taken on that day. With either a DAY 1 START or SUNDAY START, 1 tablet (pale pink) is taken each day at the same time for 21 days. Then the white tablets are taken for 7 days, whether bleeding has stopped or not. After all 28 tablets have been taken, whether bleeding has stopped or not, the same dosage schedule is repeated beginning on the following day.
Special notes
Spotting, breakthrough bleeding, or nausea. If spotting (bleeding insufficient to require a pad), breakthrough bleeding (heavier bleeding similar to a menstrual flow), or nausea occurs the patient should continue taking her tablets as directed. The incidence of spotting, breakthrough bleeding or nausea is minimal, most frequently occurring in the first cycle. Ordinarily spotting or breakthrough bleeding will stop within a week. Usually the patient will begin to cycle regularly within two or three courses of tablet-taking. In the event of spotting or breakthrough bleeding organic causes should be borne in mind. (See WARNING No. 12 .)
Missed menstrual periods. Withdrawal flow will normally occur 2 or 3 days after the last active tablet is taken. Failure of withdrawal bleeding ordinarily does not mean that the patient is pregnant, providing the dosage schedule has been correctly followed. (See WARNING No. 7. )
If the patient has not adhered to the prescribed dosage regimen, the possibility of pregnancy should be considered after the first missed period, and oral contraceptives should be withheld until pregnancy has been ruled out.
If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out before continuing the contraceptive regimen.
The first intermenstrual interval after discontinuing the tablets is usually prolonged; consequently, a patient for whom a 28-day cycle is usual might not begin to menstruate for 35 days or longer. Ovulation in such prolonged cycles will occur correspondingly later in the cycle. Post-treatment cycles after the first one, however, are usually typical for the individual woman prior to taking tablets. (See WARNING No. 12 .)
Missed tablets. If a woman misses taking one active tablet, the missed tablet should be taken as soon as it is remembered. In addition, the next tablet should be taken at the usual time. If two consecutive active tablets are missed in week 1 or week 2 of the dispenser, the dosage should be doubled for the next 2 days. The regular schedule should then be resumed, but an additional method of protection must be used as backup for the next 7 days if she has sex during that time or she may become pregnant.
If two consecutive active tablets are missed in week 3 of the dispenser or three consecutive active tablets are missed during any of the first 3 weeks of the dispenser, direct the patient to do one of the following: Day 1 Starters should discard the rest of the dispenser and begin a new dispenser that same day; Sunday Starters should continue to take 1 tablet daily until Sunday, discard the rest of the dispenser and begin a new dispenser that same day. The patient may not have a period this month; however, if she has missed two consecutive periods, pregnancy should be ruled out. An additional method of protection must be used as a backup for the next 7 days after the tablets are missed if she has sex during that time or she may become pregnant.
While there is little likelihood of ovulation if only one active tablet is missed, the possibility of spotting or breakthrough bleeding is increased and should be expected if two or more successive active tablets are missed. However, the possibility of ovulation increases with each successive day that scheduled active tablets are missed.
If one or more placebo tablets of Zovia 1/35 are missed, the schedule should be resumed on the eighth day after the last pale pink colored tablet was taken. Omission of placebo tablets in the 28-tablet courses does not increase the possibility of conception provided that this schedule is followed.
CONTRAINDICATIONS
Oral contraceptives should not be used in women who have the following conditions:
● Thrombophlebitis or thromboembolic disorders
● A past history of deep vein thrombophlebitis or thromboembolic disorders
● Cerebral vascular disease, myocardial infarction, or coronary artery disease, or a past history of these conditions
● Known or suspected carcinoma of the breast, or a history of this condition
● Known or suspected carcinoma of the female reproductive organs or suspected estrogen-dependent neoplasia, or a history of these conditions
● Undiagnosed abnormal genital bleeding
● History of cholestatic jaundice of pregnancy or jaundice with prior oral contraceptive use
● Past or present, benign or malignant liver tumors
● Known or suspected pregnancy
● Are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations (see Warnings , RISK OF LIVER ENZYME ELEVATIONS WITH CONCOMITANT HEPATITIS C TREATMENT )
WARNING SIGNALS
If any of these adverse effects occur while you are taking oral contraceptives, call your doctor immediately:
● Sharp chest pain, coughing up blood, or sudden shortness of breath (indicating a possible blood clot in the lung).
● Pain in the calf (indicating a possible blood clot in the leg).
● Crushing chest pain or heaviness in the chest (indicating a possible heart attack).
● Sudden severe headache or vomiting, dizziness or fainting, disturbances of vision or speech, or numbness in an arm or leg (indicating a possible stroke).
● Sudden partial or complete loss of vision (indicating a possible blood clot in the blood vessels of the eye).
● Breast lumps (indicating possible breast cancer or fibrocystic disease of the breast). Ask your doctor or health care provider to show you how to examine your own breasts.
● Severe pain or tenderness or a mass in the stomach area (indicating a possible ruptured liver tumor).
● Difficulty in sleeping, weakness, lack of energy, fatigue, or change in mood (possibly indicating severe depression).
● Jaundice or a yellowing of the skin or eyeballs, accompanied frequently by fever, fatigue, loss of appetite, dark-colored urine, or light-colored bowel movements (indicating possible liver problems).
● Unusual swelling.
● Other unusual conditions.
SIDE EFFECTS OF ORAL CONTRACEPTIVES
1. Vaginal bleeding.
Spotting. This is a slight staining between your menstrual periods that may not even require a pad. Some women spot even though they take their pills exactly as directed. Many women spot although they have never taken the pills. Spotting does not mean that your ovaries are releasing an egg. Spotting may be the result of irregular pill-taking. Getting back on schedule will usually stop it.
If you should spot while taking the pills, you should not be alarmed, because spotting usually stops by itself within a few days. It seldom occurs after the first pill cycle. Consult your doctor if spotting persists for more than a few days or if it occurs after the second cycle.
Unexpected (breakthrough) bleeding. Unexpected (breakthrough) bleeding does not mean that your ovaries have released an egg. It seldom occurs, but when it does happen it is most common in the first pill cycle. It is a flow much like a regular period, requiring the use of a pad or tampon.
If you experience breakthrough bleeding use a pad or tampon and continue with your schedule. Usually your periods will become regular within a few cycles. Breakthrough bleeding will seldom bother you again.
Consult your doctor or health care provider if breakthrough bleeding is heavy, does not stop within a week, or if it occurs after the second cycle.
2. Contact lenses. If you wear contact lenses and notice a change in vision or an inability to wear your lenses, contact your doctor or health care provider.
3. Fluid retention or raised blood pressure. Oral contraceptives may cause edema (fluid retention), with swelling of the fingers or ankles. If you experience fluid retention, contact your doctor or health care provider. Some women develop high blood pressure while on the pill, which ordinarily, but not always, returns to the original levels when the pill is stopped. High blood pressure predisposes one to strokes, heart attacks, kidney disease, and other diseases of the blood vessels.
4. Melasma. A spotty darkening of the skin is possible, particularly of the face. This may persist after the pill is discontinued.
5. Other side effects. Other side effects may include nausea and vomiting, change in appetite, headache, nervousness, depression, dizziness, loss of scalp hair, rash, and vaginal infections.
If any of these, or other, side effects occur, call your doctor or health care provider.
ADVERSE REACTIONS
An increased risk of the following serious adverse reactions has been associated with the use of oral contraceptives (see WARNINGS ):
● Thrombophlebitis and thrombosis
● Arterial thromboembolism
● Pulmonary embolism
● Myocardial infarction and coronary thrombosis
● Cerebral hemorrhage
● Cerebral thrombosis
● Hypertension
● Gallbladder disease
● Benign and malignant liver tumors, and other hepatic lesions
There is evidence of an association between the following conditions and the use of oral contraceptives, although additional confirmatory studies are needed:
● Mesenteric thrombosis
● Neuro-ocular lesions (e.g., retinal thrombosis and optic neuritis)
The following adverse reactions have been reported in patients receiving oral contraceptives and are believed to be drug-related:
● Nausea
● Vomiting
● Gastrointestinal symptoms (such as abdominal cramps and bloating)
● Breakthrough bleeding
● Spotting
● Change in menstrual flow
● Amenorrhea during or after use
● Temporary infertility after discontinuation of use
● Edema
● Chloasma or melasma, which may persist
● Breast changes: tenderness, enlargement, secretion
● Change in weight (increase or decrease)
● Change in cervical erosion or secretion
● Diminution in lactation when given immediately postpartum
● Cholestatic jaundice
● Migraine
● Rash (allergic)
● Mental depression
● Reduced tolerance to carbohydrates
● Vaginal candidiasis
● Change in corneal curvature (steepening)
● Intolerance to contact lenses
The following adverse reactions or conditions have been reported in users of oral contraceptives and the association has been neither confirmed nor refuted:
● Premenstrual syndrome
● Cataracts
● Changes in appetite
● Cystitis-like syndrome
● Headache
● Nervousness
● Dizziness
● Hirsutism
● Loss of scalp hair
● Erythema multiforme
● Erythema nodosum
● Hemorrhagic eruption
● Vaginitis
● Porphyria
● Impaired renal function
● Hemolytic uremic syndrome
● Acne
● Changes in libido
● Colitis
● Budd-Chiari syndrome
● Endocervical hyperplasia or ectropion
DESCRIPTION
Zovia 1/35: Each pale pink tablet contains 1 mg of ethynodiol diacetate and 35 mcg of ethinyl estradiol, and the inactive ingredients include lactose monohydrate, pregelatinized starch, vitamin e, magnesium stearate, microcrystalline cellulose, croscarmellose sodium, povidone, polyvinyl alcohol, titanium dioxide, talc, polyethylene glycol/macrogol, lecithin (soya), FD&C Red #40 aluminum lake, FD&C Blue #1 aluminum lake, and FD&C yellow #6 aluminum lake. Each white tablet in the Zovia 1/35 package is a placebo containing no active ingredients and the inactive ingredients include lactose monohydrate, magnesium stearate, pregelatinized starch, titanium dioxide, polydextrose, hypromellose, triacetin, and polyethylene glycol.
The chemical name for ethynodiol diacetate is 19-Nor-17α-pregn-4-en-20-yne-3β,17-diol diacetate, and for ethinyl estradiol it is 19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17-diol.
The structural formulas are as follows:
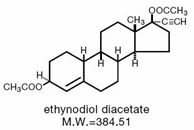
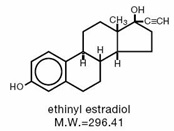
Therapeutic class: Oral contraceptive.
CLINICAL PHARMACOLOGY
Combination oral contraceptives act primarily by suppression of gonadotropins. Although the primary mechanism of this action is inhibition of ovulation, other alterations in the genital tract, including changes in the cervical mucus (which increase the difficulty of sperm entry into the uterus) and the endometrium (which may reduce the likelihood of implantation) may also contribute to contraceptive effectiveness.
NON-CONTRACEPTIVE HEALTH BENEFITS
The following non-contraceptive health benefits related to the use of oral contraceptives are supported by epidemiological studies that largely utilized oral contraceptive formulations containing estrogen doses exceeding 35 mcg of ethinyl estradiol or 50 mcg of mestranol. 148, 149
Effects on menses:
● Increased menstrual cycle regularity
● Decreased blood loss and decreased risk of iron-deficiency anemia
● Decreased frequency of dysmenorrhea
Effects related to inhibition of ovulation:
● Decreased risk of functional ovarian cysts
● Decreased risk of ectopic pregnancies
Effects from long-term use:
● Decreased risk of fibroadenomas and fibrocystic disease of the breast
● Decreased risk of acute pelvic inflammatory disease
● Decreased risk of endometrial cancer
● Decreased risk of ovarian cancer
● Decreased risk of uterine fibroids
HOW SUPPLIED
Zovia 1/35: Each pale pink Zovia 1/35 tablet is round, biconvex, unscored, debossed with X1 on one side and contains 1 mg of ethynodiol diacetate and 35 mcg of ethinyl estradiol.
Zovia 1/35 is packaged in cartons of three (NDC 51862-894-03) and six (NDC 51862-894-06) tablet compact dispensers. Each compact dispenser contains 21 pale pink tablets and 7 white placebo tablets. Placebo tablets are round, biconvex and have a debossed P on one side and N on the other side.
Store at 20° to 25°C (68° to 77°F). [See USP controlled room temperature.]