Baclofen - Baclofen injection, Solution prescribing information
Abrupt discontinuation of intrathecal baclofen, regardless of the cause, has resulted in sequelae that include high fever, altered mental status, exaggerated rebound spasticity, and muscle rigidity, that in rare cases has advanced to rhabdomyolysis, multiple organ-system failure and death.
Prevention of abrupt discontinuation of intrathecal baclofen requires careful attention to programming and monitoring of the infusion system, refill scheduling and procedures, and pump alarms. Patients and caregivers should be advised of the importance of keeping scheduled refill visits and should be educated on the early symptoms of baclofen withdrawal. Special attention should be given to patients at apparent risk (e.g. spinal cord injuries at T-6 or above, communication difficulties, history of withdrawal symptoms from oral or intrathecal baclofen). Consult the technical manual of the implantable infusion system for additional postimplant clinician and patient information (see WARNINGS ).
INDICATIONS AND USAGE
Baclofen injection (intrathecal) is indicated for use in the management of severe spasticity. Patients should first respond to a screening dose of intrathecal baclofen prior to consideration for long term infusion via an implantable pump. For spasticity of spinal cord origin, chronic infusion of baclofen injection (intrathecal) via an implantable pump should be reserved for patients unresponsive to oral baclofen therapy, or those who experience intolerable CNS side effects at effective doses. Patients with spasticity due to traumatic brain injury should wait at least one year after the injury before consideration of long term intrathecal baclofen therapy. Baclofen injection (intrathecal) is intended for use by the intrathecal route in single bolus test doses (via spinal catheter or lumbar puncture) and, for chronic use, only in implantable pumps approved by the FDA specifically for the administration of baclofen injection (intrathecal) into the intrathecal space.
Spasticity of Spinal Cord Origin: Evidence supporting the efficacy of baclofen injection (intrathecal) was obtained in randomized, controlled investigations that compared the effects of either a single intrathecal dose or a three day intrathecal infusion of baclofen injection (intrathecal) to placebo in patients with severe spasticity and spasms due to either spinal cord trauma or multiple sclerosis. Baclofen injection (intrathecal) was superior to placebo on both principal outcome measures employed: change from baseline in the Ashworth rating of spasticity and the frequency of spasms.
Spasticity of Cerebral Origin: The efficacy of baclofen injection (intrathecal) was investigated in three controlled clinical trials; two enrolled patients with cerebral palsy and one enrolled patients with spasticity due to previous brain injury. The first study, a randomized controlled cross-over trial of 51 patients with cerebral palsy, provided strong, statistically significant results; baclofen injection (intrathecal) was superior to placebo in reducing spasticity as measured by the Ashworth Scale. A second cross-over study was conducted in 11 patients with spasticity arising from brain injury. Despite the small sample size, the study yielded a nearly significant test statistic (p= 0.066) and provided directionally favorable results. The last study, however, did not provide data that could be reliably analyzed.
Baclofen injection (intrathecal) therapy may be considered an alternative to destructive neurosurgical procedures. Prior to implantation of a device for chronic intrathecal infusion of baclofen injection (intrathecal), patients must show a response to baclofen injection (intrathecal) in a screening trial (see Dosage and Administration ).
DOSAGE AND ADMINISTRATION
Refer to the manufacturer's manual for the implantable pump approved for intrathecal infusion for specific instructions and precautions for programming the pump and/or refilling the reservoir. There are various pumps with varying reservoir volumes and there are various refill kits available. It is important to be familiar with all of these products in order to select the appropriate refill kit for the particular pump in use.
Screening Phase: Prior to pump implantation and initiation of chronic infusion of baclofen injection (intrathecal), patients must demonstrate a positive clinical response to a baclofen injection (intrathecal) bolus dose administered intrathecally in a screening trial. The screening trial employs baclofen injection (intrathecal) at a concentration of 50 mcg per mL. A 1 mL ampule (50 mcg per mL) is available for use in the screening trial. The screening procedure is as follows. An initial bolus containing 50 micrograms in a volume of 1 milliliter is administered into the intrathecal space by barbotage over a period of not less than one minute. The patient is observed over the ensuing 4 to 8 hours. A positive response consists of a significant decrease in muscle tone and/or frequency and/or severity of spasms. If the initial response is less than desired, a second bolus injection may be administered 24 hours after the first. The second screening bolus dose consists of 75 micrograms in 1.5 milliliters. Again, the patient should be observed for an interval of 4 to 8 hours. If the response is still inadequate, a final bolus screening dose of 100 micrograms in 2 milliliters may be administered 24 hours later.
Pediatric Patients: The starting screening dose for pediatric patients is the same as in adult patients, i.e., 50 mcg. However, for very small patients, a screening dose of 25 mcg may be tried first. Patients who do not respond to a 100 mcg intrathecal bolus should not be considered candidates for an implanted pump for chronic infusion .
Post-Implant Dose Titration Period: To determine the initial total daily dose of baclofen injection (intrathecal) following implant, the screening dose that gave a positive effect should be doubled and administered over a 24-hour period, unless the efficacy of the bolus dose was maintained for more than 8 hours, in which case the starting daily dose should be the screening dose delivered over a 24-hour period. No dose increases should be given in the first 24 hours (i.e., until the steady state is achieved).
Adult Patients with Spasticity of Spinal Cord Origin: After the first 24 hours, for adult patients, the daily dosage should be increased slowly by 10 to 30% increments and only once every 24 hours, until the desired clinical effect is achieved.
Adult Patients with Spasticity of Cerebral Origin: After the first 24 hours, the daily dose should be increased slowly by 5 to 15% only once every 24 hours, until the desired clinical effect is achieved.
Pediatric Patients: After the first 24 hours, the daily dose should be increased slowly by 5 to 15% only once every 24 hours, until the desired clinical effect is achieved. If there is not a substantive clinical response to increases in the daily dose, check for proper pump function and catheter patency. Patients must be monitored closely in a fully equipped and staffed environment during the screening phase and dose-titration period immediately following implant. Resuscitative equipment should be immediately available for use in case of life-threatening or intolerable side effects.
Maintenance Therapy:
Spasticity of Spinal Cord Origin Patients: The clinical goal is to maintain muscle tone as close to normal as possible, and to minimize the frequency and severity of spasms to the extent possible, without inducing intolerable side effects. Very often, the maintenance dose needs to be adjusted during the first few months of therapy while patients adjust to changes in life style due to the alleviation of spasticity. During periodic refills of the pump, the daily dose may be increased by 10 to 40%, but no more than 40%, to maintain adequate symptom control. The daily dose may be reduced by 10 to 20% if patients experience side effects. Most patients require gradual increases in dose over time to maintain optimal response during chronic therapy. A sudden large requirement for dose escalation suggests a catheter complication (i.e., catheter kink or dislodgement).
Maintenance dosage for long term continuous infusion of baclofen injection (intrathecal) has ranged from 12 mcg/day to 2,003 mcg/day, with most patients adequately maintained on 300 micrograms to 800 micrograms per day. There is limited experience with daily doses greater than 1,000 mcg/day. Determination of the optimal baclofen injection (intrathecal) dose requires individual titration. The lowest dose with an optimal response should be used.
Spasticity of Cerebral Origin Patients: The clinical goal is to maintain muscle tone as close to normal as possible and to minimize the frequency and severity of spasms to the extent possible, without inducing intolerable side effects, or to titrate the dose to the desired degree of muscle tone for optimal functions. Very often the maintenance dose needs to be adjusted during the first few months of therapy while patients adjust to changes in life style due to the alleviation of spasticity. During periodic refills of the pump, the daily dose may be increased by 5 to 20%, but no more than 20%, to maintain adequate symptom control. The daily dose may be reduced by 10 to 20% if patients experience side effects. Many patients require gradual increases in dose over time to maintain optimal response during chronic therapy. A sudden large requirement for dose escalation suggests a catheter complication (i.e., catheter kink or dislodgement) .
Maintenance dosage for long term continuous infusion of baclofen injection (intrathecal) has ranged from 22 mcg/day to 1,400 mcg/day, with most patients adequately maintained on 90 micrograms to 703 micrograms per day. In clinical trials, only 3 of 150 patients required daily doses greater than 1,000 mcg/day.
Pediatric Patients: Use same dosing recommendations for patients with spasticity of cerebral origin. Pediatric patients under 12 years seemed to require a lower daily dose in clinical trials. Average daily dose for patients under 12 years was 274 mcg/day, with a range of 24 to 1,199 mcg/day. Dosage requirement for pediatric patients over 12 years does not seem to be different from that of adult patients. Determination of the optimal baclofen injection (intrathecal) dose requires individual titration. The lowest dose with an optimal response should be used.
Potential need for dose adjustments in chronic use: During long term treatment, approximately 5% (28/627) of patients become refractory to increasing doses. There is not sufficient experience to make firm recommendations for tolerance treatment; however, this “tolerance” has been treated on occasion, in hospital, by a “drug holiday” consisting of the gradual reduction of baclofen injection (intrathecal) over a 2 to 4 week period and switching to alternative methods of spasticity management. After the “drug holiday,” baclofen injection (intrathecal) may be restarted at the initial continuous infusion dose.
Stability
Parenteral drug products should be inspected for particulate matter and discoloration prior to administration, whenever solution and container permit.
Delivery Specifications
The specific concentration that should be used depends upon the total daily dose required as well as the delivery rate of the pump. Baclofen injection (intrathecal) may require dilution when used with certain implantable pumps. Please consult manufacturer's manual for specific recommendations.
Preparation Instruction:
Screening
Use the 1 mL screening ampule only (50 mcg per mL) for bolus injection into the subarachnoid space. For a 50 mcg bolus dose, use 1 mL of the screening ampule. Use 1.5 mL of 50 mcg per mL baclofen injection (intrathecal) for a 75 mcg bolus dose. For the maximum screening dose of 100 mcg, use 2 mL of 50 mcg per mL baclofen injection (intrathecal) (2 screening ampules).
Maintenance
For patients who require concentrations other than 500 mcg per mL or 2,000 mcg per mL, baclofen injection (intrathecal) must be diluted.
Baclofen injection (intrathecal) must be diluted with sterile preservative free Sodium Chloride for Injection, U.S.P.
Delivery Regimen:
Baclofen injection (intrathecal) is most often administered in a continuous infusion mode immediately following implant. For those patients implanted with programmable pumps who have achieved relatively satisfactory control on continuous infusion, further benefit may be attained using more complex schedules of baclofen injection (intrathecal) delivery. For example, patients who have increased spasms at night may require a 20% increase in their hourly infusion rate. Changes in flow rate should be programmed to start two hours before the time of desired clinical effect.
CONTRAINDICATIONS
Hypersensitivity to baclofen. Baclofen injection (intrathecal) is not recommended for intravenous, intramuscular, subcutaneous or epidural administration.
ADVERSE REACTIONS
Spasticity of Spinal Cord Origin – Clinical Studies:
Commonly Observed in Patients with Spasticity of Spinal Origin — In pre- and post- marketing clinical trials, the most commonly observed adverse events associated with use of baclofen injection (intrathecal) which were not seen at an equivalent incidence among placebo-treated patients were: somnolence, dizziness, nausea, hypotension, headache, convulsions and hypotonia.
Associated with Discontinuation of Treatment — 8/474 patients with spasticity of spinal cord origin receiving long term infusion of baclofen injection (intrathecal) in pre- and post- marketing clinical studies in the U.S. discontinued treatment due to adverse events. These include: pump pocket infections (3), meningitis (2), wound dehiscence (1), gynecological fibroids (1) and pump overpressurization (1) with unknown, if any, sequela. Eleven patients who developed coma secondary to overdose had their treatment temporarily suspended, but all were subsequently re-started and were not, therefore, considered to be true discontinuations.
Fatalities — See Warnings .
Incidence in Controlled Trials — Experience with baclofen injection (intrathecal) obtained in parallel, placebo-controlled, randomized studies provides only a limited basis for estimating the incidence of adverse events because the studies were of very brief duration (up to three days of infusion) and involved only a total of 63 patients. The following events occurred among the 31 patients receiving baclofen injection (intrathecal) in two randomized, placebo-controlled trials: hypotension (2), dizziness (2), headache (2), dyspnea (1). No adverse events were reported among the 32 patients receiving placebo in these studies.
Events Observed during the Pre- and Post-marketing Evaluation of Baclofen Injection (Intrathecal) — Adverse events associated with the use of baclofen injection (intrathecal) reflect experience gained with 576 patients followed prospectively in the United States. They received baclofen injection (intrathecal) for periods of one day (screening) (N = 576) to over eight years (maintenance) (N = 10). The usual screening bolus dose administered prior to pump implantation in these studies was typically 50 mcg. The maintenance dose ranged from 12 mcg to 2003 mcg per day. Because of the open, uncontrolled nature of the experience, a causal linkage between events observed and the administration of baclofen injection (intrathecal) cannot be reliably assessed in many cases and many of the adverse events reported are known to occur in association with the underlying conditions being treated. Nonetheless, many of the more commonly reported reactions— hypotonia, somnolence, dizziness, paresthesia, nausea/vomiting and headache— appear clearly drug-related.
Adverse experiences reported during all U.S. studies (both controlled and uncontrolled) are shown in the following table. Eight of 474 patients who received chronic infusion via implanted pumps had adverse experiences which led to a discontinuation of long term treatment in the pre- and post-marketing studies.
a Following administration of test bolus | |||
b Two month period following implant | |||
c Beyond two months following implant | |||
N= total number of patients entering each period | |||
%=% of patients evaluated | |||
| INCIDENCE OF MOST FREQUENT (≥1%) ADVERSE EVENTS IN PATIENTS WITH SPASTICITY OF SPINAL ORIGIN IN PROSPECTIVELY MONITORED CLINICAL TRIALS | |||
| Percent of Patients Reporting Events | |||
| Adverse Event | N = 576 Screening a Percent | N = 474 Titration b Percent | N = 430 Maintenance c Percent |
| Hypotonia | 5.4 | 13.5 | 25.3 |
| Somnolence | 5.7 | 5.9 | 20.9 |
| Dizziness | 1.7 | 1.9 | 7.9 |
| Paresthesia | 2.4 | 2.1 | 6.7 |
| Nausea and Vomiting | 1.6 | 2.3 | 5.6 |
| Headache | 1.6 | 2.5 | 5.1 |
| Constipation | 0.2 | 1.5 | 5.1 |
| Convulsion | 0.5 | 1.3 | 4.7 |
| Urinary Retention | 0.7 | 1.7 | 1.9 |
| Dry Mouth | 0.2 | 0.4 | 3.3 |
| Accidental Injury | 0.0 | 0.2 | 3.5 |
| Asthenia | 0.7 | 1.3 | 1.4 |
| Confusion | 0.5 | 0.6 | 2.3 |
| Death | 0.2 | 0.4 | 3.0 |
| Pain | 0.0 | 0.6 | 3.0 |
| Speech Disorder | 0.0 | 0.2 | 3.5 |
| Hypotension | 1.0 | 0.2 | 1.9 |
| Ambylopia | 0.5 | 0.2 | 2.3 |
| Diarrhea | 0.0 | 0.8 | 2.3 |
| Hypoventilation | 0.2 | 0.8 | 2.1 |
| Coma | 0.0 | 1.5 | 0.9 |
| Impotence | 0.2 | 0.4 | 1.6 |
| Peripheral Edema | 0.0 | 0.0 | 2.3 |
| Urinary Incontinence | 0.0 | 0.8 | 1.4 |
| Insomnia | 0.0 | 0.4 | 1.6 |
| Anxiety | 0.2 | 0.4 | 0.9 |
| Depression | 0.0 | 0.0 | 1.6 |
| Dyspnea | 0.3 | 0.0 | 1.2 |
| Fever | 0.5 | 0.2 | 0.7 |
| Pneumonia | 0.2 | 0.2 | 1.2 |
| Urinary Frequency | 0.0 | 0.6 | 0.9 |
| Urticaria | 0.2 | 0.2 | 1.2 |
| Anorexia | 0.0 | 0.4 | 0.9 |
| Diplopia | 0.0 | 0.4 | 0.9 |
| Dysautonomia | 0.2 | 0.2 | 0.9 |
| Hallucinations | 0.3 | 0.4 | 0.5 |
| Hypertension | 0.2 | 0.6 | 0.5 |
In addition to the more common (1% or more) adverse events reported in the prospectively followed 576 domestic patients in pre- and post-marketing studies, experience from an additional 194 patients exposed to baclofen injection (intrathecal) from foreign studies has been reported. The following adverse events, not described in the table, and arranged in decreasing order of frequency, and classified by body system, were reported:
Nervous System: Abnormal gait, thinking abnormal, tremor, amnesia, twitching, vasodilitation, cerebrovascular accident, nystagmus, personality disorder, psychotic depression, cerebral ischemia, emotional lability, euphoria, hypertonia, ileus, drug dependence, incoordination, paranoid reaction and ptosis.
Digestive System: Flatulence, dysphagia, dyspepsia and gastroenteritis.
Cardiovascular: Postural hypotension, bradycardia, palpitations, syncope, arrhythmia ventricular, deep thrombophlebitis, pallor and tachycardia.
Respiratory: Respiratory disorder, aspiration pneumonia, hyperventilation, pulmonary embolus and rhinitis.
Urogenital: Hematuria and kidney failure.
Skin and Appendages: Alopecia and sweating.
Metabolic and Nutritional Disorders: Weight loss, albuminuria, dehydration and hyperglycemia.
Special Senses: Abnormal vision, abnormality of accommodation, photophobia, taste loss and tinnitus.
Body as a Whole: Suicide, lack of drug effect, abdominal pain, hypothermia, neck rigidity, chest pain, chills, face edema, flu syndrome and overdose.
Hemic and Lymphatic System: Anemia.
Spasticity of Cerebral Origin – Clinical Studies:
Commonly Observed — In pre-marketing clinical trials, the most commonly observed adverse events associated with use of baclofen injection (intrathecal) which were not seen at an equivalent incidence among placebo-treated patients included: agitation, constipation, somnolence, leukocytosis, chills, urinary retention and hypotonia.
Associated with Discontinuation of Treatment — Nine of 211 patients receiving baclofen injection (intrathecal) in pre-marketing clinical studies in the U.S. discontinued long term infusion due to adverse events associated with intrathecal therapy.
The nine adverse events leading to discontinuation were: infection (3), CSF leaks (2), meningitis (2), drainage (1), and unmanageable trunk control (1).
Fatalities — Three deaths, none of which were attributed to baclofen injection (intrathecal), were reported in patients in clinical trials involving patients with spasticity of cerebral origin. See Warnings on other deaths reported in spinal spasticity patients.
Incidence in Controlled Trials — Experience with baclofen injection (intrathecal) obtained in parallel, placebo-controlled, randomized studies provides only a limited basis for estimating the incidence of adverse events because the studies involved a total of 62 patients exposed to a single 50 mcg intrathecal bolus. The following events occurred among the 62 patients receiving baclofen injection (intrathecal) in two randomized, placebo-controlled trials involving cerebral palsy and head injury patients, respectively: agitation, constipation, somnolence, leukocytosis, nausea, vomiting, nystagmus, chills, urinary retention, and hypotonia.
Events Observed during the Pre-marketing Evaluation of Baclofen Injection (Intrathecal) — Adverse events associated with the use of baclofen injection (intrathecal) reflect experience gained with a total of 211 U.S. patients with spasticity of cerebral origin, of whom 112 were pediatric patients (under age 16 at enrollment). They received baclofen injection (intrathecal) for periods of one day (screening) (N= 211) to 84 months (maintenance) (N= 1). The usual screening bolus dose administered prior to pump implantation in these studies was 50 to 75 mcg. The maintenance dose ranged from 22 mcg to 1,400 mcg per day. Doses used in this patient population for long term infusion are generally lower than those required for patients with spasticity of spinal cord origin.
Because of the open, uncontrolled nature of the experience, a causal linkage between events observed and the administration of baclofen injection (intrathecal) cannot be reliably assessed in many cases. Nonetheless, many of the more commonly reported reactions— somnolence, dizziness, headache, nausea, hypotension, hypotonia and coma— appear clearly drug-related.
The most frequent (≥1%) adverse events reported during all clinical trials are shown in the following table. Nine patients discontinued long term treatment due to adverse events.
a Following administration of test bolus | |||
b Two month period following implant | |||
c Beyond two months following implant | |||
N= Total number of patients entering each period. 211 patients received drug; (1 of 212) received placebo only. | |||
| INCIDENCE OF MOST FREQUENT (≥ 1%) ADVERSE EVENTS IN PATIENTS WITH SPASTICITY OF CEREBRAL ORIGIN IN PROSPECTIVELY MONITORED CLINICAL TRIALS | |||
| Percent of Patients Reporting Events | |||
| N = 211 | N = 153 | N = 150 | |
| Adverse Event | Screening a Percent | Titration b Percent | Maintenance c Percent |
| Hypotonia | 2.4 | 14.4 | 34.7 |
| Somnolence | 7.6 | 10.5 | 18.7 |
| Headache | 6.6 | 7.8 | 10.7 |
| Nausea and Vomiting | 6.6 | 10.5 | 4.0 |
| Vomiting | 6.2 | 8.5 | 4.0 |
| Urinary Retention | 0.9 | 6.5 | 8.0 |
| Convulsion | 0.9 | 3.3 | 10.0 |
| Dizziness | 2.4 | 2.6 | 8.0 |
| Nausea | 1.4 | 3.3 | 7.3 |
| Hypoventilation | 1.4 | 1.3 | 4.0 |
| Hypertonia | 0.0 | 0.7 | 6.0 |
| Paresthesia | 1.9 | 0.7 | 3.3 |
| Hypotension | 1.9 | 0.7 | 2.0 |
| Increased Salivation | 0.0 | 2.6 | 2.7 |
| Back Pain | 0.9 | 0.7 | 2.0 |
| Constipation | 0.5 | 1.3 | 2.0 |
| Pain | 0.0 | 0.0 | 4.0 |
| Pruritus | 0.0 | 0.0 | 4.0 |
| Diarrhea | 0.5 | 0.7 | 2.0 |
| Peripheral Edema | 0.0 | 0.0 | 3.3 |
| Thinking Abnormal | 0.5 | 1.3 | 0.7 |
| Agitation | 0.5 | 0.0 | 1.3 |
| Asthenia | 0.0 | 0.0 | 2.0 |
| Chills | 0.5 | 0.0 | 1.3 |
| Coma | 0.5 | 0.0 | 1.3 |
| Dry Mouth | 0.5 | 0.0 | 1.3 |
| Pneumonia | 0.0 | 0.0 | 2.0 |
| Speech Disorder | 0.5 | 0.7 | 0.7 |
| Tremor | 0.5 | 0.0 | 1.3 |
| Urinary Incontinence | 0.0 | 0.0 | 2.0 |
| Urination Impaired | 0.0 | 0.0 | 2.0 |
The more common (1% or more) adverse events reported in the prospectively followed 211 patients exposed to baclofen injection (intrathecal) have been reported. In the total cohort, the following adverse events, not described in the table, and arranged in decreasing order of frequency, and classified by body system, were reported:
Nervous System: Akathisia, ataxia, confusion, depression, opisthotonos, amnesia, anxiety, hallucinations, hysteria, insomnia, nystagmus, personality disorder, reflexes decreased, and vasodilitation.
Digestive System: Dysphagia, fecal incontinence, gastrointestinal hemorrhage and tongue disorder.
Cardiovascular: Bradycardia.
Respiratory: Apnea, dyspnea and hyperventilation.
Urogenital: Abnormal ejaculation, kidney calculus, oliguria and vaginitis.
Skin and Appendages: Rash, sweating, alopecia, contact dermatitis and skin ulcer.
Special Senses: Abnormality of accommodation.
Body as a Whole: Death, fever, abdominal pain, carcinoma, malaise and hypothermia.
Hemic and Lymphatic System: Leukocytosis and petechial rash.
Postmarketing Experience:
The following adverse events have been reported during post-approval use of baclofen injection (intrathecal). Because these events are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency.
Musculoskeletal: The onset of scoliosis or worsening of a pre-existing scoliosis has been reported.
Urogenital: Sexual dysfunction in men and women, including decreased libido and orgasm dysfunction, have been reported. Erectile dysfunction in men has also been reported. Priapism has been reported following baclofen withdrawal.
To report SUSPECTED ADVERSE REACTIONS, contact Sagent Pharmaceuticals, Inc. at 1-866-625-1618 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DRUG INTERACTIONS
There is inadequate systematic experience with the use of baclofen injection (intrathecal) in combination with other medications to predict specific drug-drug interactions. Interactions attributed to the combined use of baclofen injection (intrathecal) and epidural morphine include hypotension and dyspnea.
DESCRIPTION
Baclofen injection (intrathecal), USP• is a muscle relaxant and antispastic. Its chemical name is 4-amino-3- (4-chlorophenyl) butanoic acid, and its structural formula is:
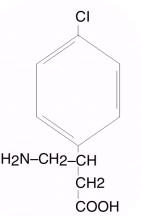
Baclofen is a white to off-white, odorless or practically odorless crystalline powder, with a molecular weight of 213.66. It is slightly soluble in water, very slightly soluble in methanol, and insoluble in chloroform.
Baclofen injection (intrathecal), USP• is a sterile, nonpyrogenic, isotonic solution free of antioxidants, preservatives or other potentially neurotoxic additives indicated only for intrathecal administration. The drug is stable in solution at 37° C and compatible with CSF. Each milliliter of baclofen injection (intrathecal) contains baclofen U. S. P., 500 mcg or 2,000 mcg and sodium chloride 9 mg in Water for Injection; pH range is 5.0 to 7.0. Each vial is intended for SINGLE USE ONLY. Discard any unused portion. DO NOT AUTOCLAVE .
CLINICAL PHARMACOLOGY
The precise mechanism of action of baclofen as a muscle relaxant and antispasticity agent is not fully understood. Baclofen inhibits both monosynaptic and polysynaptic reflexes at the spinal level, possibly by decreasing excitatory neurotransmitter release from primary afferent terminals, although actions at supraspinal sites may also occur and contribute to its clinical effect. Baclofen is a structural analog of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA), and may exert its effects by stimulation of the GABA B receptor subtype.
Baclofen injection (intrathecal) when introduced directly into the intrathecal space permits effective CSF concentrations to be achieved with resultant plasma concentrations 100 times less than those occurring with oral administration.
In people, as well as in animals, baclofen has been shown to have general CNS depressant properties as indicated by the production of sedation with tolerance, somnolence, ataxia, and respiratory and cardiovascular depression.
Pharmacodynamics of Baclofen Injection (Intrathecal):
Intrathecal Bolus:
Adult Patients: The onset of action is generally one-half hour to one hour after an intrathecal bolus. Peak spasmolytic effect is seen at approximately four hours after dosing and effects may last four to eight hours. Onset, peak response, and duration of action may vary with individual patients depending on the dose and severity of symptoms.
Pediatric Patients: The onset, peak response and duration of action is similar to those seen in adult patients.
Continuous Infusion:
Baclofen injection (intrathecal)'s antispastic action is first seen at 6 to 8 hours after initiation of continuous infusion. Maximum activity is observed in 24 to 48 hours.
Continuous Infusion: No additional information is available for pediatric patients.
Pharmacokinetics of Baclofen Injection (Intrathecal):
The pharmacokinetics of CSF clearance of baclofen injection (intrathecal) calculated from intrathecal bolus or continuous infusion studies approximates CSF turnover, suggesting elimination is by bulk-flow removal of CSF.
Intrathecal Bolus: After a bolus lumbar injection of 50 or 100 mcg baclofen injection (intrathecal) in seven patients, the average CSF elimination half-life was 1.51 hours over the first four hours and the average CSF clearance was approximately 30 mL/hour.
Continuous Infusion: The mean CSF clearance for baclofen injection (intrathecal) was approximately 30 mL/hour in a study involving ten patients on continuous intrathecal infusion.
Concurrent plasma concentrations of baclofen during intrathecal administration are expected to be low (0 to 5 ng/mL).
Limited pharmacokinetic data suggest that a lumbar-cisternal concentration gradient of about 4:1 is established along the neuroaxis during baclofen infusion. This is based upon simultaneous CSF sampling via cisternal and lumbar tap in 5 patients receiving continuous baclofen infusion at the lumbar level at doses associated with therapeutic efficacy; the interpatient variability was great. The gradient was not altered by position.
Six pediatric patients (age 8 to 18 years) receiving continuous intrathecal baclofen infusion at doses of 77 to 400 mcg/day had plasma baclofen levels near or below 10 ng/mL.
HOW SUPPLIED
Baclofen injection (intrathecal), USP• is a clear, colorless, isotonic solution consisting of the active ingredient, Baclofen USP, and the excipients Sodium Chloride USP and Water for Injection USP and is supplied in single-dose clear glass vials as follows:
| NDC | Baclofen Injection (Intrathecal), USP• ( 500 mcg per mL) | Package Factor |
| 25021-678-20 | 10 mg per 20 mL Single-Dose Vial | 1 vial per carton |
| NDC | Baclofen Injection (Intrathecal), USP• ( 2,000 mcg per mL) | Package Factor |
| 25021-679-20 | 40 mg per 20 mL Single-Dose Vial | 1 vial per carton |
Sterile, Nonpyrogenic, Preservative-free. The container closure is not made with natural rubber latex.
Storage Conditions:
Does not require refrigeration.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F). [See USP Controlled Room Temperature.]
Do not freeze.
Do not heat sterilize
•The USP osmolality specification is pending
SAGENT ® Mfd. for SAGENT Pharmaceuticals Schaumburg, IL 60195 (USA) Made in India ©2021 Sagent Pharmaceuticals, Inc.
June 2021
SAGENT Pharmaceuticals ®