Get your patient on Baqsimi (Glucagon)
Baqsimi prior authorization resources
Most recent state uniform prior authorization forms
Baqsimi patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
- BAQSIMI is for intranasal use only. (2.1 )
- The recommended dose of BAQSIMI is 3 mg administered as one actuation of the intranasal device into one nostril. (2.2 )
- Administer BAQSIMI according to the printed instructions on the shrink-wrapped tube label and the Instructions for Use. (2.1 )
- Administer the dose by inserting the tip into one nostril and pressing the device plunger all the way in until the green line is no longer showing. The dose does not need to be inhaled. (2.1 )
- Call for emergency assistance immediately after administering the dose. (2.1 )
- When the patient responds to treatment, give oral carbohydrates. (2.1 )
- Do not attempt to reuse BAQSIMI. Each BAQSIMI device contains one dose of glucagon and cannot be reused. Discard any unused portion. (2.1 )
- If there has been no response after 15 minutes, an additional 3 mg dose may be administered while waiting for emergency assistance. (2.2 )
Important Administration Instructions
BAQSIMI is for intranasal use only.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires help of others to recover, instruct the patient to inform those around them about BAQSIMI and its Instructions for Use. Administer BAQSIMI as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for BAQSIMI. Emphasize the following instructions to the patient or caregiver:
- Do not push the plunger or test the device prior to administration.
- Administer BAQSIMI according to the printed instructions on the shrink-wrapped tube label and the Instructions for Use.
- Administer the dose by inserting the tip into one nostril and pressing the device plunger all the way in until the green line is no longer showing. The dose does not need to be inhaled.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, an additional dose of BAQSIMI may be administered while waiting for emergency assistance.
- When the patient responds to treatment, give oral carbohydrates to restore the liver glycogen and prevent recurrence of hypoglycemia.
- Do not attempt to reuse BAQSIMI. Each BAQSIMI device contains one dose of glucagon and cannot be reused. Discard any unused portion.
Dosage in Adults and Pediatric Patients Aged 1 Year and Older
The recommended dose of BAQSIMI is 3 mg administered as one actuation of the intranasal device into one nostril.
If there has been no response after 15 minutes, an additional 3 mg dose of BAQSIMI from a new device may be administered while waiting for emergency assistance.

Baqsimi prescribing information
INDICATIONS AND USAGE
BAQSIMI™ is indicated for the treatment of severe hypoglycemia in adults and pediatric patients aged 1 year and older with diabetes.
DOSAGE AND ADMINISTRATION
- BAQSIMI is for intranasal use only. (2.1 )
- The recommended dose of BAQSIMI is 3 mg administered as one actuation of the intranasal device into one nostril. (2.2 )
- Administer BAQSIMI according to the printed instructions on the shrink-wrapped tube label and the Instructions for Use. (2.1 )
- Administer the dose by inserting the tip into one nostril and pressing the device plunger all the way in until the green line is no longer showing. The dose does not need to be inhaled. (2.1 )
- Call for emergency assistance immediately after administering the dose. (2.1 )
- When the patient responds to treatment, give oral carbohydrates. (2.1 )
- Do not attempt to reuse BAQSIMI. Each BAQSIMI device contains one dose of glucagon and cannot be reused. Discard any unused portion. (2.1 )
- If there has been no response after 15 minutes, an additional 3 mg dose may be administered while waiting for emergency assistance. (2.2 )
Important Administration Instructions
BAQSIMI is for intranasal use only.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires help of others to recover, instruct the patient to inform those around them about BAQSIMI and its Instructions for Use. Administer BAQSIMI as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions for Use at the time they receive a prescription for BAQSIMI. Emphasize the following instructions to the patient or caregiver:
- Do not push the plunger or test the device prior to administration.
- Administer BAQSIMI according to the printed instructions on the shrink-wrapped tube label and the Instructions for Use.
- Administer the dose by inserting the tip into one nostril and pressing the device plunger all the way in until the green line is no longer showing. The dose does not need to be inhaled.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, an additional dose of BAQSIMI may be administered while waiting for emergency assistance.
- When the patient responds to treatment, give oral carbohydrates to restore the liver glycogen and prevent recurrence of hypoglycemia.
- Do not attempt to reuse BAQSIMI. Each BAQSIMI device contains one dose of glucagon and cannot be reused. Discard any unused portion.
Dosage in Adults and Pediatric Patients Aged 1 Year and Older
The recommended dose of BAQSIMI is 3 mg administered as one actuation of the intranasal device into one nostril.
If there has been no response after 15 minutes, an additional 3 mg dose of BAQSIMI from a new device may be administered while waiting for emergency assistance.
DOSAGE FORMS AND STRENGTHS
Nasal Powder:
- 3 mg glucagon: as a white powder in an intranasal device containing one dose of glucagon
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Available data from case reports and a small number of observational studies with glucagon use in pregnant women over decades of use have not identified a drug associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Multiple small studies have demonstrated a lack of transfer of pancreatic glucagon across the human placental barrier during early gestation. In a rat reproduction study, no embryofetal toxicity was observed with glucagon administered by injection during the period of organogenesis at doses representing up to 40 times the human dose, based on body surface area (mg/m 2 ) (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data
In pregnant rats given animal sourced glucagon twice-daily by injection at doses up to 2 mg/kg (up to 40 times the human dose based on body surface area extrapolation, mg/m 2 ) during the period of organogenesis, there was no evidence of increased malformations or embryofetal lethality.
Lactation
Risk Summary
There is no information available on the presence of glucagon in human or animal milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. However, glucagon is a peptide and would be expected to be broken down to its constituent amino acids in the infant's digestive tract and is therefore, unlikely to cause harm to an exposed infant.
Pediatric Use
The safety and effectiveness of BAQSIMI for the treatment of severe hypoglycemia in patients with diabetes have been established in pediatric patients aged 1 year and older. Use of BAQSIMI for this indication is supported by evidence from an adequate and well-controlled study in adults with type 1 diabetes mellitus [see Clinical Studies (14.1 )] , a study in 48 pediatric patients aged 4 to less than 17 years with type 1 diabetes mellitus [see Clinical Studies (14.2 )] , and additional pharmacokinetic and safety data from a study of seven pediatric patients aged 1 to less than 4 years with type 1 diabetes mellitus [see Adverse Reactions (6.1 ),Clinical Pharmacology (12.2 , 12.3 ), and Clinical Studies (14.2 )] .
The safety and effectiveness of BAQSIMI have not been established in pediatric patients younger than 1 year of age.
Geriatric Use
Clinical studies of BAQSIMI did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger adult patients.
CONTRAINDICATIONS
BAQSIMI is contraindicated in patients with:
- Pheochromocytoma because of the risk of substantial increase in blood pressure [see Warnings and Precautions (5.1 )]
- Insulinoma because of the risk of hypoglycemia [see Warnings and Precautions (5.2 )]
- Prior hypersensitivity reaction to glucagon or to any of the excipients in BAQSIMI. Allergic reactions have been reported with glucagon and include anaphylactic shock with breathing difficulties and hypotension [see Warnings and Precautions (5.3 )]
WARNINGS AND PRECAUTIONS
- Substantial Increase in Blood Pressure in Patients with Pheochromocytoma : Contraindicated in patients with pheochromocytoma because BAQSIMI may stimulate the release of catecholamines from the tumor. (5.1 )
- Hypoglycemia in Patients with Insulinoma : In patients with insulinoma, administration may produce an initial increase in blood glucose; however, BAQSIMI may stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. If a patient develops symptoms of hypoglycemia after a dose of BAQSIMI, give glucose orally or intravenously. (5.2 )
- Serious Hypersensitivity Reactions : Serious hypersensitivity reactions have been reported and include generalized rash, and in some cases anaphylactic shock with breathing difficulties, and hypotension. (5.3 )
- Lack of Efficacy in Patients with Decreased Hepatic Glycogen : BAQSIMI is effective in treating hypoglycemia only if sufficient hepatic glycogen is present. Patients in states of starvation, with adrenal insufficiency or chronic hypoglycemia may not have adequate levels of hepatic glycogen for BAQSIMI to be effective. Patients with these conditions should be treated with glucose. (5.4 )
Substantial Increase in Blood Pressure in Patients with Pheochromocytoma
BAQSIMI is contraindicated in patients with pheochromocytoma because glucagon may stimulate release of catecholamines from the tumor [see Contraindications (4 )]. If the patient develops a substantial increase in blood pressure and a previously undiagnosed pheochromocytoma is suspected, 5 to 10 mg of phentolamine mesylate, administered intravenously, has been shown to be effective in lowering blood pressure.
Hypoglycemia in Patients with Insulinoma
In patients with insulinoma, administration of glucagon may produce an initial increase in blood glucose; however, BAQSIMI administration may directly or indirectly (through an initial rise in blood glucose) stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. BAQSIMI is contraindicated in patients with insulinoma [see Contraindications (4 )]. If a patient develops symptoms of hypoglycemia after a dose of BAQSIMI, give glucose orally or intravenously.
Serious Hypersensitivity Reactions
Serious hypersensitivity reactions have been reported with glucagon products, including generalized rash, and in some cases anaphylactic shock with breathing difficulties and hypotension. Discontinue BAQSIMI if symptoms of serious hypersensitivity reactions occur. Advise patients and/or caregivers to seek immediate medical attention if the patient experiences any symptoms of serious hypersensitivity reactions. BAQSIMI is contraindicated in patients with a prior hypersensitivity reaction [see Contraindications (4 )].
Lack of Efficacy in Patients with Decreased Hepatic Glycogen
Patients with insufficient hepatic stores of glycogen may not respond to BAQSIMI for the treatment of severe hypoglycemia [see Clinical Pharmacology (12.2 )]. Insufficient hepatic stores of glycogen may be present in conditions such as states of starvation or in patients with adrenal insufficiency or chronic hypoglycemia.
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in labeling:
- Substantial Increase in Blood Pressure in Patients with Pheochromocytoma [see Warnings and Precautions (5.1 )] .
- Hypoglycemia in Patients with Insulinoma [see Warnings and Precautions (5.2 )] .
- Serious Hypersensitivity Reactions [see Warnings and Precautions (5.3 )] .
- Lack of Efficacy in Patients with Decreased Hepatic Glycogen [see Warnings and Precautions (5.4 )] .
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of BAQSIMI cannot be directly compared with rates in clinical trials of other drugs and may not reflect the rates observed in practice.
Adverse Reactions in Adult Patients
Two similarly designed comparator-controlled trials, Study 1 and Study 2, evaluated the safety of a single intranasal dose of BAQSIMI compared to a 1 mg dose of intra-muscular glucagon (IMG) in adult patients with diabetes [see Clinical Studies (14.1 )].
Table 1 presents adverse reactions that occurred with BAQSIMI at an incidence of ≥2% in a pool of Study 1 and Study 2.
a Upper Respiratory Tract Irritation: rhinorrhea, nasal discomfort, nasal congestion, cough, and epistaxis. | |
| Adverse Reaction | BAQSIMI 3 mg (N=153) % |
| Nausea | 26 |
| Headache | 18 |
| Vomiting | 15 |
| Upper Respiratory Tract Irritation a | 12 |
Nasal and ocular symptoms with BAQSIMI were solicited through a patient questionnaire in Study 1 and 2 and these adverse reactions are presented in Table 2 .
a Patients were asked to report whether they have the symptom, as well as severity (mild, moderate, severe) at baseline, and after glucagon administration. | |
| Adverse Reaction a | BAQSIMI 3 mg (n=153) % |
| Any increase in symptom severity a | |
| Watery eyes | 59 |
| Nasal congestion | 43 |
| Nasal itching | 39 |
| Runny nose | 35 |
| Redness of eyes | 25 |
| Itchy eyes | 22 |
| Sneezing | 20 |
| Itching of throat | 12 |
| Itching of ears | 3 |
Adverse Reactions in Pediatric Patients Aged 1 Year and Above
A single dose of BAQSIMI was compared to weight-based doses of 0.5 mg or 1 mg of IMG in pediatric patients aged 4 to less than 17 years with type 1 diabetes in Study 3 [see Clinical Studies (14.2 )].
Table 3 presents adverse reactions that occurred with BAQSIMI in pediatric patients at an incidence of ≥2% in Study 3.
a Upper Respiratory Tract Irritation: nasal discomfort, nasal congestion, sneezing. | |
| Adverse Reaction | BAQSIMI 3 mg (n=36) % |
| Vomiting | 31 |
| Headache | 25 |
| Nausea | 17 |
| Upper Respiratory Tract Irritation a | 17 |
Nasal and ocular symptoms with BAQSIMI were solicited through a patient questionnaire in pediatric patients in Study 3 and these adverse reactions are presented in Table 4 .
a Subjects were asked to report whether they have the symptom, as well as severity (mild, moderate, severe) at baseline, and after glucagon administration. | |
| Adverse Reaction a | BAQSIMI 3 mg (n=36) % |
| Any increase in symptom severity a | |
| Watery eyes | 47 |
| Nasal congestion | 42 |
| Nasal itching | 28 |
| Runny nose | 25 |
| Sneezing | 19 |
| Itchy eyes | 17 |
| Redness of eyes | 14 |
| Itching of throat | 3 |
| Itching of ears | 3 |
The safety of a single 3 mg intranasal dose of BAQSIMI was assessed in an open-label study of 7 pediatric patients aged 1 to less than 4 years with type 1 diabetes mellitus [see Clinical Studies (14.2 )] . The safety profile observed in this trial in pediatric patients was comparable to that observed in adults and pediatric patients aged 4 to less than 17 years [see Clinical Pharmacology (12.2 , 12.3 )] .
Other Adverse Reactions in Adult and Pediatric Patients
Other observed adverse reactions with BAQSIMI-treated patients across clinical trials were, dysgeusia, pruritus, tachycardia, hypertension, and additional upper respiratory tract irritation events (nasal pruritus, throat irritation, and parosmia).
DRUG INTERACTIONS
- Beta-blockers : Patients taking beta-blockers may have a transient increase in pulse and blood pressure. (7.1 )
- Indomethacin : In patients taking indomethacin BAQSIMI may lose its ability to raise glucose or may produce hypoglycemia. (7.2 )
- Warfarin : BAQSIMI may increase the anticoagulant effect of warfarin. (7.3 )
Beta-blockers
Patients taking beta-blockers may have a transient increase in pulse and blood pressure when given BAQSIMI.
Indomethacin
In patients taking indomethacin, BAQSIMI may lose its ability to raise blood glucose or may even produce hypoglycemia.
Warfarin
BAQSIMI may increase the anticoagulant effect of warfarin.
DESCRIPTION
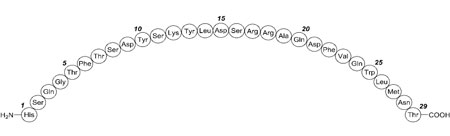
BAQSIMI contains glucagon, an antihypoglycemic agent used to treat severe hypoglycemia. Glucagon is a single-chain polypeptide containing 29 amino acid residues and has a molecular weight of 3483, and is identical to human glucagon.
Its molecular formula is C 153 H 225 N 43 O 49 S, with the following molecular structure:
BAQSIMI is a preservative-free, white powder for intranasal administration in an intranasal device containing one dose of 3 mg glucagon. BAQSIMI contains glucagon as the active ingredient and betadex, and dodecylphosphocholine as the excipients.
CLINICAL PHARMACOLOGY
Mechanism of Action
Glucagon increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for glucagon to produce an antihypoglycemic effect.
Pharmacodynamics
After administration of BAQSIMI in adult patients with diabetes, the mean maximum glucose increase from baseline was 140 mg/dL (Figure 1 ).
In pediatric patients aged 1 to less than 17 years with type 1 diabetes, the mean maximum glucose increase from baseline was 132 mg/dL (1 to less than 4 years), 138 mg/dL (4 to less than 8 years), 133 mg/dL (8 to less than 12 years), and 102 mg/dL (12 to less than 17 years) (Figure 2 ).
Sex and body weight had no clinically meaningful effects on the pharmacodynamics of BAQSIMI.
Common cold with nasal congestion tested with or without use of decongestant did not impact pharmacodynamics of BAQSIMI.
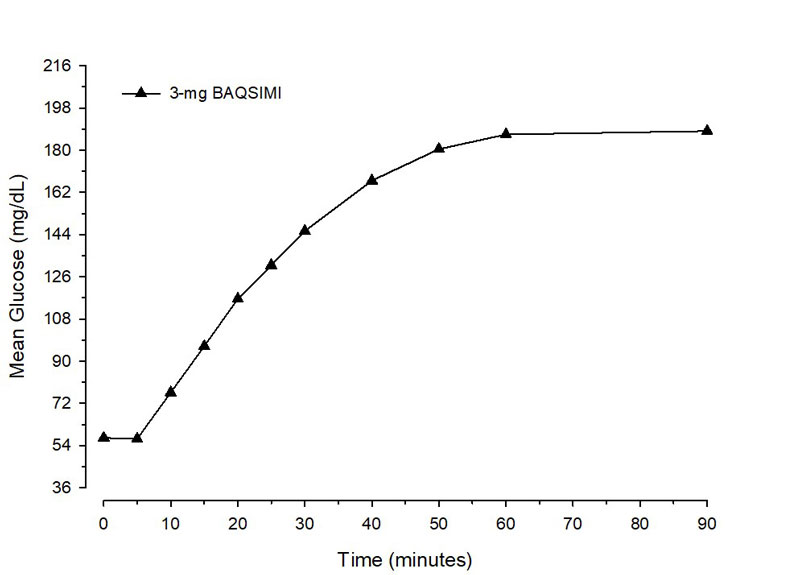
Figure 1 Mean glucose concentration over time after glucagon dose in adult Type 1 Diabetes patients with insulin-induced hypoglycemia.
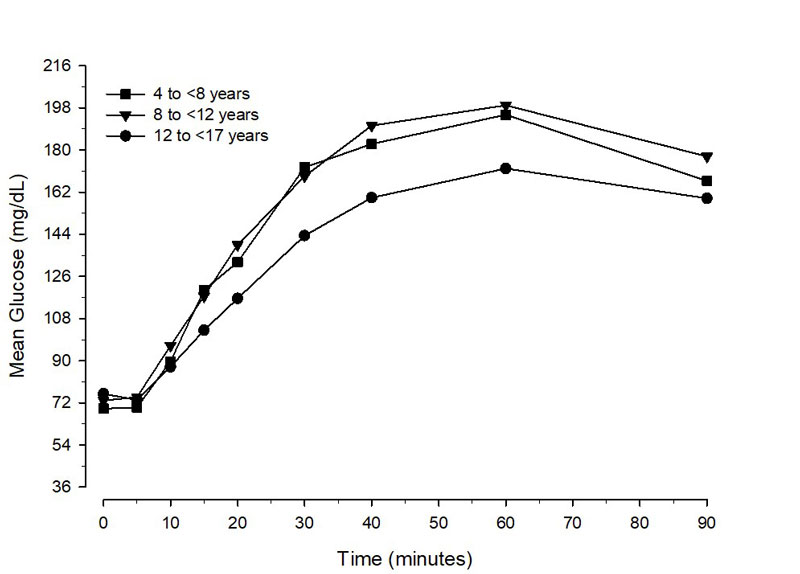
Fi gure 2 Mean glucose concentration over time in pediatric Type 1 Diabetes patients administered BAQSIMI
Pharmacokinetics
Absorption
Glucagon absorption via the intranasal route, achieved mean peak plasma levels of 6130 pg/mL at around 15 minutes.
Distribution
The apparent volume of distribution was approximately 885 L.
Elimination
The median half-life was approximately 35 minutes.
Metabolism
Glucagon is known to be degraded in the liver, kidneys, and plasma.
Specific Populations
Pediatrics
In pediatric patients (aged 1 to less than 17 years), glucagon via the intranasal route, achieved mean peak plasma levels between 10 and 20 minutes. The median half-life was 21 to 31 minutes.
Patients with Colds
Common cold with nasal congestion did not impact the pharmacokinetics of BAQSIMI.
Drug Interaction Studies
Common cold with use of decongestant did not impact the pharmacokinetics of BAQSIMI.
Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of anti-drug antibodies in other studies, including those of glucagon or of other glucagon products.
In 3 clinical trials, 3/124 (2%) of BAQSIMI-treated patients had treatment-emergent ADAs as detected by an affinity capture elution (ACE) ligand-binding immunogenicity assay. No neutralizing antibodies were detected. Because of the low occurrence of ADA, the effect of these antibodies on the pharmacokinetics, pharmacodynamics, and/or effectiveness of BAQSIMI is unknown.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long term studies in animals to evaluate carcinogenic potential have not been performed. Recombinant glucagon was positive in the bacterial Ames assay. It was determined that an increase in colony counts was related to technical difficulties in running this assay with peptides. Studies in rats have shown that glucagon does not cause impaired fertility.
CLINICAL STUDIES
Adult Patients with Type 1 or Type 2 Diabetes Mellitus
Study 1 (NCT03339453 ) was a randomized, multicenter, open-label, 2-period, crossover study in adult patients with type 1 diabetes. The efficacy of a single 3 mg dose of BAQSIMI was compared to a 1 mg dose of intra-muscular glucagon (IMG). Insulin was used to reduce blood glucose levels to <60 mg/dL. Seventy patients were enrolled, with a mean age of 41.7 years and a mean diabetes duration of 20.1 years. Twenty-seven (39%) were female.
The primary efficacy outcome measure was the proportion of patients achieving treatment success, which was defined as either an increase in blood glucose to ≥70 mg/dL or an increase of ≥20 mg/dL from glucose nadir within 30 minutes after receiving study glucagon, without receiving additional actions to increase the blood glucose level. Glucose nadir was defined as the minimum glucose measurement at the time of, or within 10 minutes, following glucagon administration.
The mean nadir blood glucose was 54.5 mg/dL for BAQSIMI and 55.8 mg/dL for IMG. BAQSIMI demonstrated non-inferiority to IMG in reversing insulin-induced hypoglycemia with 100% of BAQSIMI-treated patients and 100% of IMG-treated patients achieving treatment success. The mean time to treatment success was 11.6 and 9.9 minutes in the BAQSIMI and IMG 1 mg treatment groups, respectively.
Table 5 : Adult Patients with Type 1 Diabetes Meeting Treatment Success and Other Glucose Criteria in Study 1
a The Efficacy Analysis Population consisted of all patients who received both doses of the Study Drug with evaluable primary outcome. | ||
b Difference calculated as (percentage with success in BAQSIMI) – (percentage with success in IMG). | ||
c 2-sided 95% confidence interval (CI) of paired differences using a Wald-Min correction; non-inferiority margin = -10%. | ||
| Type 1 Diabetes (N=66) a | ||
| BAQSIMI 3 mg | IMG 1 mg | |
| Treatment Success – n (%) | 66 (100%) | 66 (100%) |
| Treatment Difference (2-sided 95% confidence limit) b, c | 0% (-2.9%, 2.9%) | |
| Glucose criterion met – n (%) | ||
| (i) ≥70 mg/dL (ii) Increase by ≥20 mg/dL from nadir Both (i) and (ii) | 66 (100%) 66 (100%) 66 (100%) | 66 (100%) 66 (100%) 66 (100%) |
Study 2 (NCT01994746 ) was a randomized, multicenter, open-label, 2-period, crossover study in adult patients with type 1 diabetes or type 2 diabetes. The efficacy of a single 3 mg dose of BAQSIMI was compared to a 1 mg dose of intra-muscular glucagon (IMG). Insulin was used to reduce blood glucose levels to the hypoglycemic range with a target blood glucose nadir of <50 mg/dL.
Study 2 enrolled 83 patients 18 to <65 years of age. The mean age of patients with type 1 diabetes (N=77) was 32.9 years and a mean diabetes duration of 18.1 years, and 45 (58%) patients were female. The mean age of patients with type 2 diabetes (N=6) was 47.8 years, with a mean diabetes duration of 18.8 years, and 4 (67%) patients were female.
The mean nadir blood glucose was 44.2 mg/dL for BAQSIMI and 47.2 mg/dL for IMG. BAQSIMI demonstrated non-inferiority to IMG in reversing insulin-induced hypoglycemia with 98.8% of BAQSIMI-treated patients and 100% of IMG-treated patients achieving treatment success within 30 minutes.
The mean time to treatment success was 15.9 and 12.1 minutes in the BAQSIMI and IMG 1 mg treatment groups, respectively.
a The Efficacy Analysis Population consisted of all patients who received both doses of the Study Drug with evaluable primary outcome. | ||
b Difference calculated as (percentage with success in BAQSIMI) – (percentage with success in IMG). | ||
c 2-sided 95% confidence interval (CI) of paired differences using a Wald-Min correction; non-inferiority margin = -10%. | ||
d Percentage based on number of patients. | ||
| Type 1 and Type 2 Diabetes (N=80) a | ||
| BAQSIMI 3 mg | IMG 1 mg | |
| Treatment Success – n (%) | 79 (98.8%) | 80 (100%) |
| Treatment Difference (2-sided 95% confidence limit) b,c | -1.3% (-4.6%, 2.2%) | |
| Glucose criterion met – n (%) d | ||
| (i) ≥70 mg/dL | 77 (96%) | 79 (99%) |
| (ii) Increase by ≥20 mg/dL from nadir | 79 (99%) | 80 (100%) |
| Both (i) and (ii) | 77 (96%) | 79 (99%) |
Pediatric Patients Aged 1 to less than 17 Years with Type 1 Diabetes Mellitus
Study 3 (NCT01997411 ) was a randomized, multicenter, clinical study that assessed BAQSIMI compared to intra-muscular glucagon (IMG) in pediatric patients aged 4 to less than 17 years with type 1 diabetes. Insulin was used to reduce blood glucose levels, and glucagon was administered after glucose reached <80 mg/dL. Efficacy was assessed based on percentage of patients with a glucose increase of ≥20 mg/dL from glucose nadir within 30 minutes following BAQSIMI administration.
Forty-eight patients were enrolled and received at least one dose of study drug. The mean age in the Young Children cohort (4 to <8 years) was 6.5 years. In the Children cohort (8 to <12 years), mean age was 11.1 years and in the Adolescents cohort (12 to <17 years) mean age was 14.6 years. In all age cohorts, the population was predominantly male and white.
Across all age groups, all (100%) patients in both treatment arms achieved an increase in glucose ≥20 mg/dL from glucose nadir within 20 minutes of glucagon administration. The mean time to reach a glucose increase of ≥20 mg/dL for BAQSIMI and IMG for all age groups is shown in Table 7 .
| Increase from Nadir | Mean Time Post-Glucagon Administration (minutes) | |||||
|---|---|---|---|---|---|---|
| Young Children (4 to <8 years old) | Children (8 to <12 years old) | Adolescents (12 to <17 years old) | ||||
| IMG a N=6 | BAQSIMI 3 mg N=12 | IMG a N=6 | BAQSIMI 3 mg N=12 | IMG a N=12 | BAQSIMI 3 mg N=12 | |
a 0.5 mg or 1 mg of IMG (based upon body weight) | ||||||
| ≥20 mg/dL | 10.8 | 10.8 | 12.5 | 11.3 | 12.5 | 14.2 |
Study 4 (NCT04992312 ) was a phase 1, open-label, multi-center study with a primary objective of assessing the safety and tolerability of a single 3 mg dose of BAQSIMI in pediatric participants aged 1 to less than 4 years with type 1 diabetes mellitus. Patients were recommended to fast overnight before the dosing visit on Day 1, to achieve the target range glucose of 70 to 140 mg/dL (3.9 to 7.8 mmol/L) at baseline. Efficacy was assessed based on the percentage of patients with a glucose increase of ≥20 mg/dL from baseline within 30 minutes following BAQSIMI administration.
Seven patients were enrolled in the study, all received the planned 3 mg dose of BAQSIMI and completed the study. The mean age of the patients enrolled in the study was 2.98 years, with ages ranging from 1.8 to 4 years old. There were 4 males and 3 females enrolled in the study, all who were white.
All (100%) patients achieved an increase in glucose ≥20 mg/dL from baseline within 30 minutes of BAQSIMI administration.
HOW SUPPLIED/STORAGE AND HANDLING
BAQSIMI is supplied as an intranasal device containing one 3 mg dose of glucagon as a preservative free, white powder.
- BAQSIMI One Pack carton contains 1 intranasal device (NDC 0548-8351-01)
- BAQSIMI Two Pack carton contains 2 intranasal devices (NDC 0548-8352-02)
- Store at temperatures up to 86°F (30°C) in the shrink wrapped tube provided.
- Keep BAQSIMI in the shrink wrapped tube until ready to use. If the tube has been opened, BAQSIMI may have been exposed to moisture and may not work as expected.
- Discard BAQSIMI and tube after use.
INSTRUCTIONS FOR USE
BAQSIMI ®
(glucagon) nasal powder
3 mg
Read the Instructions for Use for BAQSIMI before using it. BAQSIMI is used to treat very low blood sugar (severe hypoglycemia) that will cause you to need help from others. You should make sure you show your caregivers, family and friends where you keep BAQSIMI and explain how to use it by sharing these instructions. They need to know how to use BAQSIMI before an emergency happens.
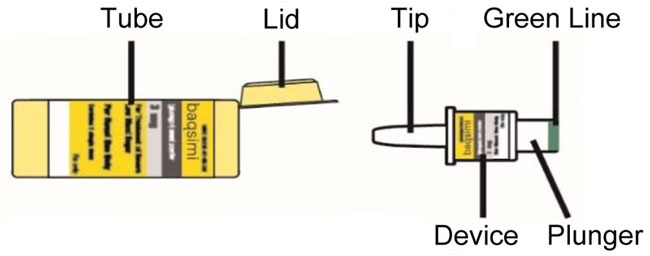
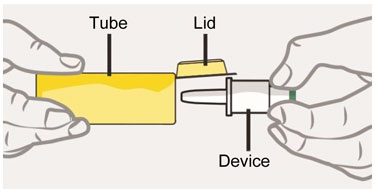
Tube and Device Parts
|
Important Information to Know
- Do not remove the Shrink Wrap or open the Tube until you are ready to use it.
- If the Tube has been opened, BAQSIMI could be exposed to moisture. This could cause BAQSIMI not to not work as expected.
- Do not push the plunger or test BAQSIMI before you are ready to use it.
- BAQSIMI contains 1 dose of glucagon nasal powder and cannot be reused.
- BAQSIMI is for nasal (nose) use only.
- BAQSIMI will work even if you have a cold or are taking cold medicine.
Preparing the Dose
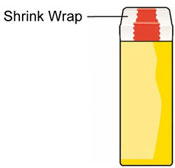 | Remove the Shrink Wrap by pulling on red stripe. |
 |
|
Giving the Dose
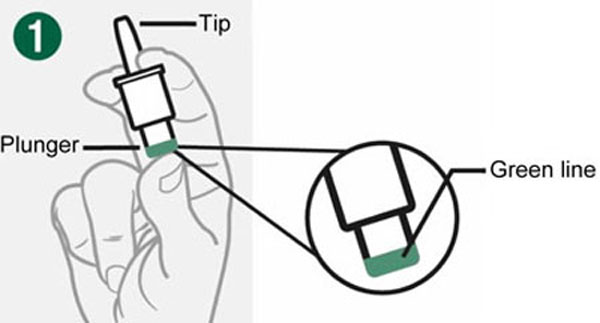 |
|
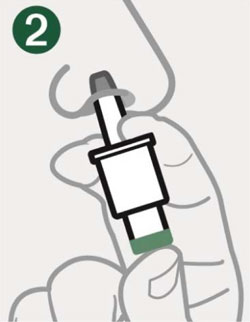 |
|
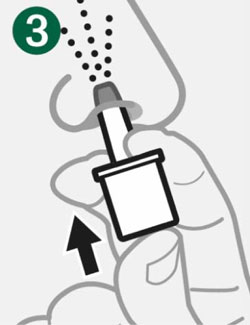 |
|
After giving BAQSIMI
- Call for emergency medical help right away.
- If the person is passed out (unconscious) turn the person on their side.
- Encourage the person to eat as soon as possible. When they can safely swallow, give the person a fast-acting source of sugar such as juice. Then encourage the person to eat a snack such as crackers with cheese or peanut butter.
- If the person does not respond after 15 minutes, another dose may be given, if available.
Storage and Handling
- Do not remove the Shrink Wrap or open the Tube until you are ready to use it.
- Store BAQSIMI in the shrink wrapped Tube at temperatures up to 86º F (30ºC ).
- Throw away (discard) BAQSIMI and Tube after use. Used BAQSIMI may be placed in household trash.
- Caution: Replace the used BAQSIMI right away so you will have a new BAQSIMI in case you need it.
- Replace BAQSIMI before the expiration date printed on the Tube or carton.
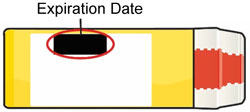
- Keep BAQSIMI and all medicines out of the reach of children.
For Questions or More Information about BAQSIMI
- Call your healthcare provider
- Call Amphastar Pharmaceuticals, Inc. at 1-800-423-4136
- Visit www.baqsimi.com
BAQSIMI is a registered trademark of Amphastar Pharmaceuticals, Inc.
Marketed by: Amphastar Pharmaceuticals, Inc. Rancho Cucamonga, CA 91730, U.S.A
Copyright © 2025, Amphastar Pharmaceuticals, Inc.
| BAQSIMI Device meets all applicable requirements defined in ISO 20072 |
This Instructions for Use has been approved by the U.S. Food and Drug Adminstration Revised: March, 2025
678351AMC/3-25
Mechanism of Action
Glucagon increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for glucagon to produce an antihypoglycemic effect.