Get your patient on Carbidopa And Levodopa - Carbidopa And Levodopa tablet, Extended Release (Carbidopa And Levodopa)
Carbidopa And Levodopa - Carbidopa And Levodopa tablet, Extended Release prescribing information
INDICATIONS AND USAGE
Carbidopa and levodopa extended-release tablets, USP are indicated in the treatment of Parkinson’s disease, post-encephalitic parkinsonism, and symptomatic parkinsonism that may follow carbon monoxide intoxication or manganese intoxication.
DOSAGE AND ADMINISTRATION
Carbidopa and levodopa extended-release tablet contains carbidopa and levodopa in a 1:4 ratio as either the 25 mg/100 mg tablet or the 50 mg/200 mg tablet. The daily dosage of carbidopa and levodopa extended-release tablets must be determined by careful titration. Patients should be monitored closely during the dose adjustment period, particularly with regard to appearance or worsening of involuntary movements, dyskinesias or nausea. Carbidopa and levodopa extended-release tablets should not be chewed or crushed.
Standard drugs for Parkinson’s disease, other than levodopa without a decarboxylase inhibitor, may be used concomitantly while carbidopa and levodopa extended-release tablet is being administered, although their dosage may have to be adjusted.
Since carbidopa prevents the reversal of levodopa effects caused by pyridoxine, carbidopa and levodopa extended-release tablets can be given to patients receiving supplemental pyridoxine (vitamin B 6 ).
Management of Vitamin B 6 Levels
Evaluate vitamin B 6 levels prior to initiating carbidopa/levodopa therapies including carbidopa and levodopa extended-release tablets, periodically during treatment, and as clinically indicated (see WARNINGS, Vitamin B 6 Deficiency and Seizures). If vitamin B 6 levels are low, supplement to sufficient levels per standard of care. Patients may initiate and continue treatment with carbidopa and levodopa extended-release tablets while supplementing vitamin B 6 .
Initial Dosage
Patients currently treated with conventional carbidopa levodopa preparations: Studies show that peripheral dopa-decarboxylase is saturated by the bioavailable carbidopa at doses of 70 mg a day and greater. Because the bioavailabilities of carbidopa and levodopa in carbidopa and levodopa tablets and carbidopa and levodopa extended-release tablets are different, appropriate adjustments should be made, as shown in Table 2.
Table 2: Approximate Bioavailabilities at Steady State •
| Tablet | Amount of Levodopa (mg) in Each Tablet | Approximate Bioavailability | Approximate Amount of Bioavailable Levodopa (mg) in Each Tablet |
| Carbidopa and levodopa extended-release tablets 50 mg/200 mg | 200 | 0.7 to 0.75 † | 140 to 150 |
| Carbidopa and levodopa tablets 25 mg/100 mg | 100 | 0.99 ‡ | 99 |
• This table is only a guide to bioavailabilities since other factors such as food, drugs, and inter-patient variabilities may affect the bioavailability of carbidopa and levodopa.
† The extent of availability of levodopa from carbidopa and levodopa extended-release tablets was about 70 to 75% relative to intravenous levodopa or standard carbidopa and levodopa tablets in the elderly.
‡ The extent of availability of levodopa from carbidopa and levodopa tablets was 99% relative to intravenous levodopa in the healthy elderly.
Dosage with carbidopa and levodopa extended-release tablets should be substituted at an amount that provides approximately 10% more levodopa per day, although this may need to be increased to a dosage that provides up to 30% more levodopa per day depending on clinical response (see DOSAGE AND ADMINISTRATION, Titration with carbidopa and levodopa extended-release tablets ). The interval between doses of carbidopa and levodopa extended-release tablets should be 4 to 8 hours during the waking day. (See CLINICAL PHARMACOLOGY, Pharmacodynamics .)
A guideline for initiation of carbidopa and levodopa extended-release tablet is shown in Table 3. Table 3: Guidelines for Initial Conversion from Carbidopa and Levodopa Tablets to Carbidopa and Levodopa Extended-Release Tablets
| Carbidopa and Levodopa Tablets | Carbidopa and Levodopa Extended-Release Tablets |
| Total Daily Dose • | Suggested |
| Levodopa (mg) | Dosage Regimen |
| 300 to 400 | 200 mg b.i.d. |
| 500 to 600 | 300 mg b.i.d. or 200 mg t.i.d. |
| 700 to 800 | A total of 800 mg in 3 or more divided doses (e.g., 300 mg a.m., 300 mg early p.m., and 200 mg later p.m.) |
| 900 to 1000 | A total of 1000 mg in 3 or more divided doses (e.g., 400 mg a.m., 400 mg early p.m., and 200 mg later p.m.) |
• For dosing ranges not shown in the table see DOSAGE AND ADMINISTRATION, Initial Dosage — Patients currently treated with conventional carbidopa levodopa preparations.
Patients currently treated with levodopa without a decarboxylase inhibitor: Levodopa must be discontinued at least twelve hours before therapy with carbidopa and levodopa extended-release tablet is started. Carbidopa and levodopa extended-release tablets should be substituted at a dosage that will provide approximately 25% of the previous levodopa dosage. In patients with mild to moderate disease, the initial dose is usually 1 tablet of carbidopa and levodopa extended-release tablet 50 mg/200 mg b.i.d.
Patients not receiving levodopa: In patients with mild to moderate disease, the initial recommended dose is 1 tablet of carbidopa and levodopa extended-release tablet 50 mg/200 mg b.i.d. Initial dosage should not be given at intervals of less than 6 hours.
Titration with Carbidopa and Levodopa Extended-Release Tablets
Following initiation of therapy, doses and dosing intervals may be increased or decreased depending upon therapeutic response. Most patients have been adequately treated with doses of carbidopa and levodopa extended-release tablets that provide 400 to 1600 mg of levodopa per day, administered as divided doses at intervals ranging from 4 to 8 hours during the waking day. Higher doses of carbidopa and levodopa extended-release tablets (2400 mg or more of levodopa per day) and shorter intervals (less than 4 hours) have been used, but are not usually recommended.
When doses of carbidopa and levodopa extended-release tablets are given at intervals of less than 4 hours, and/or if the divided doses are not equal, it is recommended that the smaller doses be given at the end of the day.
An interval of at least 3 days between dosage adjustments is recommended.
Maintenance
Because Parkinson’s disease is progressive, periodic clinical evaluations are recommended; adjustment of the dosage regimen of carbidopa and levodopa extended-release tablets may be required.
Addition of Other Antiparkinson Medications
Anticholinergic agents, dopamine agonists, and amantadine can be given with carbidopa and levodopa extended-release tablets. Dosage adjustment of carbidopa and levodopa extended-release tablets may be necessary when these agents are added.
A dose of carbidopa levodopa immediate release 25 mg/100 mg or 10 mg/100 mg (one half or a whole tablet) can be added to the dosage regimen of carbidopa and levodopa extended-release tablets in selected patients with advanced disease who need additional immediate-release levodopa for a brief time during daytime hours. Interruption of Therapy
Sporadic cases of hyperpyrexia and confusion have been associated with dose reductions and withdrawal of carbidopa and levodopa tablets or carbidopa and levodopa extended-release tablets.
Patients should be observed carefully if abrupt reduction or discontinuation of carbidopa and levodopa extended-release tablet is required, especially if the patient is receiving neuroleptics. (See WARNINGS.)
If general anesthesia is required, carbidopa and levodopa extended-release tablets may be continued as long as the patient is permitted to take oral medication. If therapy is interrupted temporarily, the patient should be observed for symptoms resembling NMS, and the usual dosage should be administered as soon as the patient is able to take oral medication.
CONTRAINDICATIONS
Nonselective monoamine oxidase (MAO) inhibitors are contraindicated for use with carbidopa and levodopa extended-release tablets. These inhibitors must be discontinued at least two weeks prior to initiating therapy with carbidopa and levodopa extended-release tablets. Carbidopa and levodopa extended-release tablets may be administered concomitantly with the manufacturer’s recommended dose of an MAO inhibitor with selectivity for MAO type B (e.g., selegiline HCl) (see PRECAUTIONS, Drug Interactions ).
Carbidopa and levodopa extended-release tablets are contraindicated in patients with known hypersensitivity to any component of this drug, and in patients with narrow-angle glaucoma.
ADVERSE REACTIONS
In controlled clinical trials, patients predominantly with moderate to severe motor fluctuations while on carbidopa and levodopa tablets were randomized to therapy with either carbidopa and levodopa tablets or carbidopa and levodopa extended-release tablets. The adverse experience frequency profile of carbidopa and levodopa extended-release tablets did not differ substantially from that of carbidopa and levodopa, as shown in Table 1.
Table 1: Clinical Adverse Experiences Occurring in 1% or Greater of Patients
| Carbidopa and Levodopa Extended-Release Tablets n=491 | Carbidopa and Levodopa Tablets n=524 | |
| Adverse Experience | % | % |
| Dyskinesia | 16.5 | 12.2 |
| Nausea | 5.5 | 5.7 |
| Hallucinations | 3.9 | 3.2 |
| Confusion | 3.7 | 2.3 |
| Dizziness | 2.9 | 2.3 |
| Depression | 2.2 | 1.3 |
| Urinary tract infection | 2.2 | 2.3 |
| Headache | 2 | 1.9 |
| Dream abnormalities | 1.8 | 0.8 |
| Dystonia | 1.8 | 0.8 |
| Vomiting | 1.8 | 1.9 |
| Upper respiratory infection | 1.8 | 1 |
| Dyspnea | 1.6 | 0.4 |
| ‘On-Off’ phenomena | 1.6 | 1.1 |
| Back pain | 1.6 | 0.6 |
| Dry mouth | 1.4 | 1.1 |
| Anorexia | 1.2 | 1.1 |
| Diarrhea | 1.2 | 0.6 |
| Insomnia | 1.2 | 1 |
| Orthostatic hypotension | 1 | 1.1 |
| Shoulder pain | 1 | 0.6 |
| Chest pain | 1 | 0.8 |
| Muscle cramps | 0.8 | 1 |
| Paresthesia | 0.8 | 1.1 |
| Urinary frequency | 0.8 | 1.1 |
| Dyspepsia | 0.6 | 1.1 |
| Constipation | 0.2 | 1.5 |
Abnormal laboratory findings occurring at a frequency of 1% or greater in approximately 443 patients who received carbidopa and levodopa extended-release tablets and 475 who received carbidopa and levodopa tablets during controlled clinical trials included: decreased hemoglobin and hematocrit; elevated serum glucose; white blood cells, bacteria and blood in the urine.
The adverse experiences observed in patients in uncontrolled studies were similar to those seen in controlled clinical studies.
Other adverse experiences reported overall in clinical trials in 748 patients treated with carbidopa and levodopa extended-release tablets, listed by body system in order of decreasing frequency, include:
Body as a Whole
Asthenia, fatigue, abdominal pain, orthostatic effects.
Cardiovascular
Palpitation, hypertension, hypotension, myocardial infarction.
Gastrointestinal
Gastrointestinal pain, dysphagia, heartburn.
Metabolic
Weight loss.
Musculoskeletal
Leg pain.
Nervous System/Psychiatric
Chorea, somnolence, falling, anxiety, disorientation, decreased mental acuity, gait abnormalities, extrapyramidal disorder, agitation, nervousness, sleep disorders, memory impairment.
Respiratory
Cough, pharyngeal pain, common cold.
Skin
Rash.
Special Senses
Blurred vision.
Urogenital
Urinary incontinence.
Laboratory Tests
Decreased white blood cell count and serum potassium; increased BUN, serum creatinine and serum LDH; protein and glucose in the urine.
The following adverse experiences have been reported in postmarketing experience with carbidopa and levodopa extended-release tablets:
Cardiovascular
Cardiac irregularities, syncope.
Gastrointestinal
Taste alterations, dark saliva.
Hypersensitivity
Angioedema, urticaria, pruritus, bullous lesions (including pemphigus-like reactions).
Nervous System/Psychiatric
Increased tremor, peripheral neuropathy, psychotic episodes including delusions and paranoid ideation, pathological gambling, increased libido including hypersexuality, impulse control symptoms, seizures (including convulsions).
Skin
Alopecia, flushing, dark sweat.
Urogenital
Dark urine.
Other adverse reactions that have been reported with levodopa alone and with various carbidopa levodopa formulations and may occur with carbidopa and levodopa extended-release tablets are:
Cardiovascular
Phlebitis.
Gastrointestinal
Gastrointestinal bleeding, development of duodenal ulcer, sialorrhea, bruxism, hiccups, flatulence, burning sensation of tongue.
Hematologic
Hemolytic and non-hemolytic anemia, thrombocytopenia, leukopenia, agranulocytosis.
Hypersensitivity
Henoch-Schönlein purpura.
Metabolic
Weight gain, edema.
Nervous System/Psychiatric
Ataxia, depression with suicidal tendencies, dementia, euphoria, bradykinetic episodes, numbness, muscle twitching, blepharospasm (which may be taken as an early sign of excess dosage; consideration of dosage reduction may be made at this time), trismus, activation of latent Horner’s syndrome, nightmares.
Skin
Malignant melanoma, increased sweating.
Special Senses
Oculogyric crises, mydriasis, diplopia.
Urogenital
Urinary retention, priapism.
Miscellaneous
Faintness, hoarseness, malaise, hot flashes, sense of stimulation, bizarre breathing patterns.
Laboratory Tests
Abnormalities in alkaline phosphatase, SGOT (AST), SGPT (ALT), bilirubin, Coombs test, uric acid, vitamin B 6 levels.
To report SUSPECTED ADVERSE REACTIONS, contact Alembic Pharmaceuticals Limited at 1-866-210-9797 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Caution should be exercised when the following drugs are administered concomitantly with carbidopa and levodopa extended-release tablets .
Symptomatic postural hypotension has occurred when carbidopa levodopa preparations were added to the treatment of patients receiving some antihypertensive drugs. Therefore, when therapy with carbidopa and levodopa extended-release tablet is started, dosage adjustment of the antihypertensive drug may be required.
For patients receiving MAO inhibitors (Type A or B), see CONTRAINDICATIONS. Concomitant therapy with selegiline and carbidopa levodopa may be associated with severe orthostatic hypotension not attributable to carbidopa levodopa alone (see CONTRAINDICATIONS).
There have been rare reports of adverse reactions, including hypertension and dyskinesia, resulting from the concomitant use of tricyclic antidepressants and carbidopa levodopa preparations.
Dopamine D 2 receptor antagonists (e.g., phenothiazines, butyrophenones, risperidone) and isoniazid may reduce the therapeutic effects of levodopa. In addition, the beneficial effects of levodopa in Parkinson’s disease have been reported to be reversed by phenytoin and papaverine. Patients taking these drugs with carbidopa and levodopa extended-release tablets should be carefully observed for loss of therapeutic response.
Use of carbidopa and levodopa extended-release tablets with dopamine-depleting agents (e.g., reserpine and tetrabenazine) or other drugs known to deplete monoamine stores is not recommended.
Carbidopa and levodopa extended-release tablets and iron salts or multivitamins containing iron salts should be coadministered with caution. Iron salts can form chelates with levodopa and carbidopa and consequently reduce the bioavailability of carbidopa and levodopa.
Although metoclopramide may increase the bioavailability of levodopa by increasing gastric emptying, metoclopramide may also adversely affect disease control by its dopamine receptor antagonistic properties.
DESCRIPTION
Carbidopa and levodopa extended-release tablets, USP are an extended-release combination of carbidopa and levodopa for the treatment of Parkinson’s disease and syndrome.
Carbidopa, USP, an inhibitor of aromatic amino acid decarboxylation, is a white to creamy white, odorless or practically odorless powder, freely soluble in 3N hydrochloric acid, slightly soluble in water and in methanol, practically insoluble in alcohol, in acetone, in chloroform and in ether, with a molecular weight of 244.24. It is designated chemically as (-)-L-αhydrazino-α-methyl-β-(3,4-dihydroxybenzene) propanoic acid monohydrate. Its empirical formula is C 10 H 14 N 2 O 4 •H 2 O, and its structural formula is:
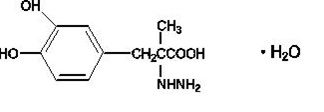
Tablet content is expressed in terms of anhydrous carbidopa, which has a molecular weight of 226.24. Levodopa, USP, an aromatic amino acid, is a white to off-white, odorless, crystalline powder, slightly soluble in water, freely soluble in 3N hydrochloric acid and insoluble in alcohol with a molecular weight of 197.19. It is designated chemically as (-)-L-α-amino-β-(3,4-dihydroxybenzene) propanoic acid. Its empirical formula is C 9 H 11 NO 4 , and its structural formula is:
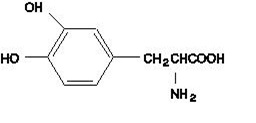
Carbidopa and levodopa extended-release tablets, USP are supplied as extended-release tablets containing either 25 mg of carbidopa and 100 mg of levodopa, or 50 mg of carbidopa and 200 mg of levodopa. Inactive ingredients are hydroxypropyl cellulose, hypromellose and magnesium stearate. Carbidopa and levodopa extended-release tablets USP, 25 mg/100 mg and carbidopa and levodopa extended-release tablets USP, 50 mg/200 mg also contain FD&C Blue #2 Aluminium Lake and FD&C Red #40 Aluminium Lake.
The 25 mg/100 mg tablet is supplied as an oval shaped mottled tablet that is dappled-purple in color and is debossed with “L519” on one side and plain on the other. The 50 mg/200 mg tablet is supplied as an oval shaped mottled tablet that is dappled-purple in color and is debossed with “L520” on one side and plain on the other. Carbidopa and levodopa extended-release tablets, USP are polymeric-based drug delivery system that controls the release of carbidopa and levodopa as it slowly erodes. Carbidopa and levodopa extended-release tablet 25 mg/100 mg is available to facilitate titration when 100 mg steps are required. FDA approved dissolution specifications differs from the USP dissolution specifications.
CLINICAL PHARMACOLOGY
Mechanism of Action Parkinson’s disease is a progressive, neurodegenerative disorder of the extrapyramidal nervous system affecting the mobility and control of the skeletal muscular system. Its characteristic features include resting tremor, rigidity, and bradykinetic movements. Symptomatic treatments, such as levodopa therapies, may permit the patient better mobility.
Current evidence indicates that symptoms of Parkinson's disease are related to depletion of dopamine in the corpus striatum. Administration of dopamine is ineffective in the treatment of Parkinson’s disease apparently because it does not cross the blood-brain barrier. However, levodopa, the metabolic precursor of dopamine, does cross the blood-brain barrier, and presumably is converted to dopamine in the brain. This is thought to be the mechanism whereby levodopa relieves symptoms of Parkinson’s disease.
Pharmacodynamics When levodopa is administered orally, it is rapidly decarboxylated to dopamine in extracerebral tissues so that only a small portion of a given dose is transported unchanged to the central nervous system. For this reason, large doses of levodopa are required for adequate therapeutic effect, and these may often be accompanied by nausea and other adverse reactions, some of which are attributable to dopamine formed in extracerebral tissues.
Since levodopa competes with certain amino acids for transport across the gut wall, the absorption of levodopa may be impaired in some patients on a high protein diet.
Carbidopa inhibits decarboxylation of peripheral levodopa. It does not cross the blood-brain barrier and does not affect the metabolism of levodopa within the central nervous system.
Since its decarboxylase inhibiting activity is limited to extracerebral tissues, administration of carbidopa with levodopa makes more levodopa available for transport to the brain.
Patients treated with levodopa therapy for Parkinson’s disease may develop motor fluctuations characterized by end-of-dose failure, peak dose dyskinesia, and akinesia. The advanced form of motor fluctuations (‘on-off’ phenomenon) is characterized by unpredictable swings from mobility to immobility. Although the causes of the motor fluctuations are not completely understood, in some patients they may be attenuated by treatment regimens that produce steady plasma levels of levodopa.
Carbidopa and levodopa extended-release tablets contain either 25 mg of carbidopa and 100 mg of levodopa, or 50 mg of carbidopa and 200 mg of levodopa in a extended-release dosage form designed to release these ingredients over a 4- to 6-hour period. With carbidopa and levodopa extended-release tablets there is less variation in plasma levodopa levels than with carbidopa and levodopa tablets, the conventional formulation. However, carbidopa and levodopa extended-release tablets are less systemically bioavailable than carbidopa and levodopa tablets and may require increased daily doses to achieve the same level of symptomatic relief as provided by carbidopa and levodopa tablets.
In clinical trials, patients with moderate to severe motor fluctuations who received carbidopa and levodopa extended-release tablets did not experience quantitatively significant reductions in ‘off’ time when compared to carbidopa and levodopa tablets. However, global ratings of improvement as assessed by both patient and physician were better during therapy with carbidopa and levodopa extended-release tablets than with carbidopa and levodopa tablets. In patients without motor fluctuations, carbidopa and levodopa extended-release tablets, under controlled conditions, provided the same therapeutic benefit with less frequent dosing when compared to carbidopa and levodopa tablets.
Pharmacokinetics Carbidopa reduces the amount of levodopa required to produce a given response by about 75% and, when administered with levodopa, increases both plasma levels and the plasma half-life of levodopa, and decreases plasma and urinary dopamine and homovanillic acid.
Elimination half-life of levodopa in the presence of carbidopa is about 1.5 hours. Following carbidopa and levodopa extended-release tablets, the apparent half-life of levodopa may be prolonged because of continuous absorption.
In healthy elderly subjects (56 to 67 years old) the mean time-to-peak concentration of levodopa after a single dose of carbidopa and levodopa extended-release tablets 50 mg/200 mg was about 2 hours as compared to 0.5 hours after standard carbidopa and levodopa tablets. The maximum concentration of levodopa after a single dose of carbidopa and levodopa extended-release tablets was about 35% of the standard carbidopa and levodopa tablets (1151 vs. 3256 ng/mL). The extent of availability of levodopa from carbidopa and levodopa extended-release tablets was about 70 to 75% relative to intravenous levodopa or standard carbidopa and levodopa tablets in the elderly. The absolute bioavailability of levodopa from carbidopa and levodopa extended-release tablets (relative to I.V.) in young subjects was shown to be only about 44%. The extent of availability and the peak concentrations of levodopa were comparable in the elderly after a single dose and at steady state after t.i.d. administration of carbidopa and levodopa extended-release tablets 50 mg/200 mg. In elderly subjects, the average trough levels of levodopa at steady state after the CR tablet were about 2 fold higher than after the standard carbidopa and levodopa tablets (163 vs. 74 ng/mL).
In these studies, using similar total daily doses of levodopa, plasma levodopa concentrations with carbidopa and levodopa extended-release tablets fluctuated in a narrower range than with carbidopa and levodopa tablets. Because the bioavailability of levodopa from carbidopa and levodopa extended-release tablets relative to carbidopa and levodopa tablets is approximately 70 to 75%, the daily dosage of levodopa necessary to produce a given clinical response with the sustained-release formulation will usually be higher.
The extent of availability and peak concentrations of levodopa after a single dose of carbidopa and levodopa extended-release tablets 50 mg/200 mg increased by about 50% and 25%, respectively, when administered with food.
At steady state, the bioavailability of carbidopa from carbidopa and levodopa tablet is approximately 99% relative to the concomitant administration of carbidopa and levodopa. At steady state, carbidopa bioavailability from carbidopa and levodopa extended-release tablets 50 mg/200 mg is approximately 58% relative to that from carbidopa and levodopa tablets.
Pyridoxine hydrochloride (vitamin B 6 ), in oral doses of 10 mg to 25 mg, may reverse the effects of levodopa by increasing the rate of aromatic amino acid decarboxylation. Carbidopa inhibits this action of pyridoxine.
Special Populations Geriatric: A study in eight young healthy subjects (21 to 22 yr) and eight elderly healthy subjects (69 to 76 yr) showed that the absolute bioavailability of levodopa was similar between young and elderly subjects following oral administration of levodopa and carbidopa. However, the systemic exposure (AUC) of levodopa was increased by 55% in elderly subjects compared to young subjects. Based on another study in forty patients with Parkinson’s disease, there was a correlation between age of patients and the increase of AUC of levodopa following administration of levodopa and an inhibitor of peripheral dopa decarboxylase. AUC of levodopa was increased by 28% in elderly patients (≥ 65 yr) compared to young patients (< 65 yr). Additionally, mean value of C max for levodopa was increased by 24% in elderly patients (≥ 65 yr) compared to young patients (< 65 yr) (see PRECAUTIONS, Geriatric Use ).
The AUC of carbidopa was increased in elderly subjects (n=10, 65 to 76 yr) by 29% compared to young subjects (n=24, 23 to 64 yr) following IV administration of 50 mg levodopa with carbidopa (50 mg). This increase is not considered a clinically significant impact.
HOW SUPPLIED
Carbidopa and levodopa extended-release tablets USP, 25 mg/100 mg containing 25 mg of carbidopa and 100 mg of levodopa, are dappled-purple in color, oval shaped mottled tablets debossed with “L519” on one side and plain on other side. They are supplied as follows:
NDC 62332-332-30 Bottle of 30 tablets
NDC 62332-332-31 Bottle of 100 tablets
NDC 62332-332-91 Bottle of 1000 tablets
Carbidopa and levodopa extended-release tablets USP, 50 mg/200 mg containing 50 mg of carbidopa and 200 mg of levodopa, are dappled-purple in color, oval shaped mottled tablets debossed with “L520” on one side and plain on other side. They are supplied as follows:
NDC 62332-333-30 Bottle of 30 tablets
NDC 62332-333-31 Bottle of 100 tablets
NDC 62332-333-91 Bottle of 1000 tablets
Storage and Handling
Store at 25°C (77°F), excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. Store in a tightly closed container, protected from light and moisture.
Dispense in a tightly closed, light-resistant container.
Manufactured by:
Alembic Pharmaceuticals Limited
(Formulation Division),
Panelav 389350, Gujarat, India
Manufactured for:
Alembic Pharmaceuticals, Inc.
Bedminster, NJ 07921, USA
Revised: 04/2026