Chlorpromazine Hydrochloride - Chlorpromazine Hydrochloride tablet, Film Coated prescribing information
Warning
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Chlorpromazine hydrochloride is not approved for the treatment of patients with dementia-related psychosis [see WARNINGS ].
INDICATIONS AND USAGE
For the management of manifestations of psychotic disorders.
For the treatment of schizophrenia.
To control nausea and vomiting.
For relief of restlessness and apprehension before surgery.
For acute intermittent porphyria.
As an adjunct in the treatment of tetanus.
To control the manifestations of the manic type of manic-depressive illness.
For relief of intractable hiccups.
For the treatment of severe behavioral problems in children (1 to 12 years of age) marked by combativeness and/or explosive hyperexcitable behavior (out of proportion to immediate provocations), and in the short-term treatment of hyperactive children who show excessive motor activity with accompanying conduct disorders consisting of some or all of the following symptoms: impulsivity, difficulty sustaining attention, aggressivity, mood lability and poor frustration tolerance.
DOSAGE AND ADMINISTRATION – ADULTS
Adjust dosage to individual and the severity of his condition, recognizing that the milligram for milligram potency relationship among all dosage forms has not been precisely established clinically. It is important to increase dosage until symptoms are controlled. Dosage should be increased more gradually in debilitated or emaciated patients. In continued therapy, gradually reduce dosage to the lowest effective maintenance level, after symptoms have been controlled for a reasonable period.
The 100 mg and 200 mg tablets are for use in severe neuropsychiatric conditions.
Elderly Patients
In general, dosages in the lower range are sufficient for most elderly patients. Since they appear to be more susceptible to hypotension and neuromuscular reactions, such patients should be observed closely. Dosage should be tailored to the individual, response carefully monitored, and dosage adjusted accordingly. Dosage should be increased more gradually in elderly patients.
Psychotic Disorders
Increase dosage gradually until symptoms are controlled. Maximum improvement may not be seen for weeks or even months. Continue optimum dosage for 2 weeks; then gradually reduce dosage to the lowest effective maintenance level. Daily dosage of 200 mg is not unusual. Some patients require higher dosages (e.g., 800 mg daily is not uncommon in discharged mental patients).
Hospitalized Patients
Acute Schizophrenic or Manic States
It is recommended that initial treatment be with chlorpromazine HCl injection until patient is controlled. Usually patient becomes quiet and co-operative within 24 to 48 hours and oral doses may be substituted and increased until the patient is calm. 500 mg a day is generally sufficient. While gradual increases to 2,000 mg a day or more may be necessary, there is usually little therapeutic gain to be achieved by exceeding 1,000 mg a day for extended periods. In general, dosage levels should be lower in the elderly, the emaciated and the debilitated.
Less Acutely Disturbed
25 mg t.i.d. Increase gradually until effective dose is reached – usually 400 mg daily.
Outpatients
10 mg t.i.d. or q.i.d., or 25 mg b.i.d. or t.i.d.
More Severe Cases
25 mg t.i.d. After 1 or 2 days, daily dosage may be increased by 20 to 50 mg at semi-weekly intervals until patient becomes calm and cooperative.
Prompt Control of Severe Symptoms
Initial treatment should be with intramuscular chlorpromazine. Subsequent doses should be oral, 25 to 50 mg t.i.d.
Nausea and Vomiting
10 to 25 mg q4 to 6h, p.r.n., increased, if necessary.
Presurgical Apprehension
25 to 50 mg, 2 to 3 hours before the operation.
Intractable Hiccups
25 to 50 mg t.i.d. or q.i.d. If symptoms persist for 2 to 3 days, parenteral therapy is indicated.
Acute Intermittent Porphyria
25 to 50 mg t.i.d. or q.i.d. Can usually be discontinued after several weeks, but maintenance therapy may be necessary for some patients.
CONTRAINDICATIONS
Do not use in patients with known hypersensitivity to phenothiazines.
Do not use in comatose states or in the presence of large amounts of central nervous system depressants (alcohol, barbiturates, narcotics, etc.).
ADVERSE REACTIONS
Note: Some adverse effects of chlorpromazine may be more likely to occur, or occur with greater intensity, in patients with special medical problems, e.g., patients with mitral insufficiency or pheochromocytoma have experienced severe hypotension following recommended doses.
Drowsiness
Usually mild to moderate, may occur, particularly during the first or second week, after which it generally disappears. If troublesome, dosage may be lowered.
Jaundice
Overall incidence has been low, regardless of indication or dosage. Most investigators conclude it is a sensitivity reaction. Most cases occur between the second and fourth weeks of therapy. The clinical picture resembles infectious hepatitis, with laboratory features of obstructive jaundice, rather than those of parenchymal damage. It is usually promptly reversible on withdrawal of the medication; however, chronic jaundice has been reported.
There is no conclusive evidence that preexisting liver disease makes patients more susceptible to jaundice. Alcoholics with cirrhosis have been successfully treated with chlorpromazine without complications. Nevertheless, the medication should be used cautiously in patients with liver disease. Patients who have experienced jaundice with a phenothiazine should not, if possible, be reexposed to chlorpromazine or other phenothiazines.
If fever with grippe-like symptoms occurs, appropriate liver studies should be conducted. If tests indicate an abnormality, stop treatment.
Liver function tests in jaundice induced by the drug may mimic extrahepatic obstruction; withhold exploratory laparotomy until extrahepatic obstruction is confirmed.
Hematological Disorders
including agranulocytosis, eosinophilia, leukopenia, hemolytic anemia, aplastic anemia, thrombocytopenic purpura and pancytopenia have been reported.
Agranulocytosis
Warn patients to report the sudden appearance of sore throat or other signs of infection. If white blood cell and differential counts indicate cellular depression, stop treatment and start antibiotic and other suitable therapy.
Most cases have occurred between the fourth and tenth weeks of therapy; patients should be watched closely during that period.
Moderate suppression of white blood cells is not an indication for stopping treatment unless accompanied by the symptoms described above.
Cardiovascular
Hypotensive Effects
Postural hypotension, simple tachycardia, momentary fainting and dizziness may occur rarely, after the first oral dose. Usually recovery is spontaneous and symptoms disappear within ½ to 2 hours. Occasionally, these effects may be more severe and prolonged, producing a shock-like condition.
To control hypotension, place patient in head-low position with legs raised. If a vasoconstrictor is required, norepinephrine and phenylephrine are the most suitable. Other pressor agents, including epinephrine, should not be used as they may cause a paradoxical further lowering of blood pressure.
EKG Changes
particularly nonspecific, usually reversible Q and T wave distortions - have been observed in some patients receiving phenothiazine tranquilizers, including chlorpromazine.
Note: Sudden death, apparently due to cardiac arrest, has been reported.
CNS Reactions
Extrapyramidal Symptoms
Neuromuscular reactions include dystonias, motor restlessness, pseudo-parkinsonism and tardive dyskinesia, and appear to be dose-related. They are discussed in the following paragraphs:
Dystonia
Class effect
Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first-generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Motor Restlessness
Symptoms may include agitation or jitteriness and sometimes insomnia. These symptoms often disappear spontaneously. At times these symptoms may be similar to the original neurotic or psychotic symptoms. Dosage should not be increased until these side effects have subsided.
If these symptoms become too troublesome, they can usually be controlled by a reduction of dosage or change of drug. Treatment with anti-parkinsonian agents, benzodiazepines or propranolol may be helpful.
Pseudo-parkinsonism
Symptoms may include: mask-like facies, drooling, tremors, pill rolling motion, cogwheel rigidity and shuffling gait. In most cases these symptoms are readily controlled when an anti-parkinsonism agent is administered concomitantly. Anti-parkinsonism agents should be used only when required. Generally, therapy of a few weeks to 2 or 3 months will suffice. After this time patients should be evaluated to determine their need for continued treatment. ( Note : Levodopa has not been found effective in antipsychotic-induced pseudo-parkinsonism.) Occasionally, it is necessary to lower the dosage of chlorpromazine or to discontinue the drug.
Tardive Dyskinesia
As with all antipsychotic agents, tardive dyskinesia may appear in some patients on long-term therapy or may appear after drug therapy has been discontinued. The syndrome can also develop, although much less frequently, after relatively brief treatment periods at low doses. This syndrome appears in all age groups. Although its prevalence appears to be highest among elderly patients, especially elderly women, it is impossible to rely upon prevalence estimates to predict at the inception of antipsychotic treatment which patients are likely to develop the syndrome. The symptoms are persistent and, in some patients, appear to be irreversible. The syndrome is characterized by rhythmical involuntary movements of the tongue, face, mouth or jaw (e.g., protrusion of tongue, puffing of cheeks, puckering of mouth, chewing movements). Sometimes these may be accompanied by involuntary movements of extremities. In rare instances, these involuntary movements of the extremities are the only manifestations of tardive dyskinesia. A variant of tardive dyskinesia, tardive dystonia, has also been described.
There is no known effective treatment for tardive dyskinesia; anti-parkinsonism agents do not alleviate the symptoms of this syndrome. If clinically feasible, it is suggested that all antipsychotic agents be discontinued if these symptoms appear. Should it be necessary to reinstitute treatment, or increase the dosage of the agent, or switch to a different antipsychotic agent, the syndrome may be masked.
It has been reported that fine vermicular movements of the tongue may be an early sign of the syndrome and, if the medication is stopped at that time, the syndrome may not develop.
Adverse Behavioral Effects
Psychotic symptoms and catatonic-like states have been reported rarely.
Other CNS Effects
Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs [See WARNINGS ] .
Cerebral edema has been reported.
Convulsive seizures (petit mal and grand mal) have been reported, particularly in patients with EEG abnormalities or history of such disorders.
Abnormality of the cerebrospinal fluid proteins has also been reported.
Allergic Reactions of a mild urticarial type of photosensitivity are seen. Avoid undue exposure to sun. More severe reactions, including exfoliative dermatitis and toxic epidermal necrolysis (TEN), have been reported occasionally.
Contact dermatitis has been reported in nursing personnel; accordingly, the use of rubber gloves when administering chlorpromazine liquid or injectable is recommended.
In addition, asthma, laryngeal edema, angioneurotic edema and anaphylactoid reactions have been reported.
Endocrine Disorders
Lactation and moderate breast engorgement may occur in females on large doses. If persistent, lower dosage or withdraw drug. False-positive pregnancy tests have been reported but are less likely to occur when a serum test is used. Amenorrhea and gynecomastia have also been reported. Hyperglycemia, hypoglycemia and glycosuria have been reported.
Autonomic Reactions
Occasional dry mouth; nasal congestion; nausea; obstipation; constipation; adynamic ileus; urinary retention, priapism; miosis and mydriasis, atonic colon, ejaculatory disorders/impotence.
Special Considerations In Long-Term Therapy
Skin pigmentation and ocular changes have occurred in some patients taking substantial doses of chlorpromazine for prolonged periods.
Skin Pigmentation
Rare instances of skin pigmentation have been observed in hospitalized mental patients, primarily females who have received the drug usually for 3 years or more in dosages ranging from 500 mg to 1,500 mg daily.
The pigmentary changes, restricted to exposed areas of the body, range from an almost imperceptible darkening of the skin to a slate gray color, sometimes with a violet hue. Histological examination reveals a pigment, chiefly in the dermis, which is probably a melanin-like complex. This pigmentation may fade following discontinuance of the drug.
Ocular Changes
Ocular changes have occurred more frequently than skin pigmentation and have been observed both in pigmented and nonpigmented patients receiving chlorpromazine usually for 2 years or more in dosages of 300 mg daily and higher. Eye changes are characterized by deposition of fine particulate matter in the lens and cornea. In more advanced cases, star-shaped opacities have also been observed in the anterior portion of the lens. The nature of the eye deposits has not yet been determined. A small number of patients with more severe ocular changes have had some visual impairment. In addition to these corneal and lenticular changes, epithelial keratopathy and pigmentary retinopathy have been reported. Reports suggest that the eye lesions may regress after withdrawal of the drug.
Since the occurrence of eye changes seems to be related to dosage levels and/or duration of therapy, it is suggested that long-term patients on moderate to high dosage levels have periodic ocular examinations.
Etiology
Etiology of both of these reactions is not clear, but exposure to light, along with dosage/duration of therapy, appears to be the most significant factor. If either of these reactions is observed, the physician should weigh the benefits of continued therapy against the possible risks and, on the merits of the individual case, determine whether or not to continue present therapy, lower the dosage, or withdraw the drug.
Other Adverse Reactions
Mild fever may occur after large I.M. doses. Hyperpyrexia has been reported. Increases in appetite and weight sometimes occur. Peripheral edema and a systemic lupus erythematosus-like syndrome have been reported.
Note: There have been occasional reports of sudden death in patients receiving phenothiazines. In some cases, the cause appeared to be cardiac arrest or asphyxia due to failure of the cough reflex.
DESCRIPTION
Chlorpromazine hydrochloride, a dimethylamine derivative of phenothiazine, has a chemical formula of 2-chloro-10-[3-(dimethylamino) propyl] phenothiazine monohydrochloride. It is available in tablets for oral administration. It has the following structural formula:
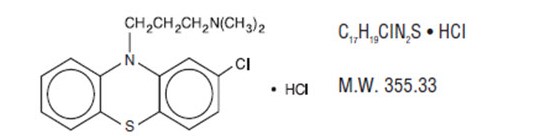

Chlorpromazine hydrochloride occurs as white or slightly creamy white, odorless, crystalline powder which darkens on prolonged exposure to light.
Each tablet for oral administration contains 10 mg, 25 mg, 50 mg, 100 mg, or 200 mg of chlorpromazine hydrochloride, USP.
Inactive ingredients: silicon dioxide, lactose monohydrate, magnesium stearate, povidone, hypromellose, polyethylene glycol and titanium dioxide.
CLINICAL PHARMACOLOGY
The precise mechanism whereby the therapeutic effects of chlorpromazine are produced is not known. The principal pharmacological actions are psychotropic. It also exerts sedative and antiemetic activity. Chlorpromazine has actions at all levels of the central nervous system –primarily at subcortical levels – as well as on multiple organ systems. Chlorpromazine has strong antiadrenergic and weaker peripheral anticholinergic activity; ganglionic blocking action is relatively slight. It also possesses slight antihistaminic and anti-serotonin activity.
HOW SUPPLIED
Chlorpromazine Hydrochloride Tablets, USP, 10 mg are round, white film coated tablets, imprinted on one side with "832" above "10" and no print on the reverse side. They are supplied as follows:
Bottles of 100 - NDC 0832-6017-00
Chlorpromazine Hydrochloride Tablets, USP, 25 mg are round, white film coated tablets, imprinted on one side with "832" above "25" and no print on the reverse side. They are supplied as follows:
Bottles of 100 - NDC 0832-6018-00
Bottles of 1,000 - NDC 0832-6018-10
Chlorpromazine Hydrochloride Tablets, USP, 50 mg are round, white film coated tablets, imprinted on one side with "832" above "50" and no print on the reverse side. They are supplied as follows:
Bottles of 100 - NDC 0832-6019-00
Bottles of 1,000 - NDC 0832-6019-10
Unit-Dose cartons of 100 - NDC 0832-6019-01
THE TABLET STRENGTHS LISTED BELOW ARE FOR USE ONLY IN SEVERE NEUROPSYCHIATRIC CONDITIONS.
Chlorpromazine Hydrochloride Tablets, USP, 100 mg are round, white film coated tablets, imprinted on one side with "832" above "100" and no print on the reverse side. They are supplied as follows:
Bottles of 100 - NDC 0832-6020-00
Bottles of 1,000 - NDC 0832-6020-10
Unit-Dose cartons of 100 - NDC 0832-6020-01
Chlorpromazine Hydrochloride Tablets, USP, 200 mg are round, white film coated tablets, imprinted on one side with "832" above "200" and no print on the reverse side. They are supplied as follows:
Bottles of 100 - NDC 0832-6021-00
Bottles of 1,000 - NDC 0832-6021-10
Unit-Dose cartons of 100 - NDC 0832-6021-01
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light and moisture.
This package is not for household dispensing. If dispensed for outpatient use, a well closed, light-resistant, child-resistant container should be utilized.
Keep out of reach of children.