Get your patient on Dextrose - Dextrose Monohydrate injection, Solution (Dextrose Monohydrate)
Dextrose - Dextrose Monohydrate injection, Solution prescribing information
INDICATIONS AND USAGE:
Intravenous solutions containing dextrose are indicated for parenteral replenishment of fluid and minimal carbohydrate calories as required by the clinical condition of the patient.
DOSAGE AND ADMINISTRATION:
The dose is dependent upon the age, weight and clinical condition of the patient.
As reported in the literature, the dosage and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low birth weight infants, because of the increased risk of hyperglycemia/hypoglycemia.
Drug Interactions
Additives may be incompatible. Consult with pharmacist, if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit (see PRECAUTIONS ).
CONTRAINDICATIONS:
5% Dextrose Injection, USP without electrolytes should not be administered simultaneously with blood through the same infusion set because of the possibility that pseudoagglutination of red cells may occur.
ADVERSE REACTIONS:
Reactions which may occur because of the solution or the technique of administration include febrile response, infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary.
To report SUSPECTED ADVERSE REACTIONS, contact Fresenius Kabi USA, LLC at 1-800-551-7176 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Additives may be incompatible. Consult with pharmacist, if available. When introducing additives, use aseptic technique, mix thoroughly and do not store.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit (see PRECAUTIONS ).
DESCRIPTION:
5% Dextrose Injection, USP solution is sterile and nonpyrogenic. It is a parenteral solution containing dextrose in water for injection intended for intravenous administration.
Each 100 mL of 5% Dextrose Injection, USP, contains dextrose monohydrate, 5 g in water for injection. The caloric value is 170 kcal/L. The osmolarity is 252 mOsmol/L (calc.), which is slightly hypotonic.
The solution pH is 4.3 (3.2 to 6.5).
This solution contains no bacteriostat, antimicrobial agent or added buffer and is intended only as a single-dose injection. When smaller doses are required the unused portion should be discarded.
5% Dextrose Injection, USP is a parenteral fluid and nutrient replenisher.
Dextrose, USP is chemically designated D-glucose monohydrate (C 6 H 12 O 6 • H 2 O), a hexose sugar freely soluble in water. It has the following structural formula:
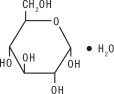
Water for Injection, USP is chemically designated H 2 O.
The flexible container is fabricated from a specially formulated non-plasticized film containing polypropylene and thermoplastic elastomers ( free flex ® bag). The free flex ® + bag has a needle-free injection port and can accept standard luer lock syringes to add medication. The amount of water that can permeate from the container into the overwrap is insufficient to affect the solution significantly. Solutions in contact with the flexible container can leach out certain of the container's chemical components in very small amounts within the expiration period. The suitability of the container material has been confirmed by tests in animals according to USP biological tests for plastic containers.
CLINICAL PHARMACOLOGY:
When administered intravenously, these solutions provide a source of water and carbohydrate.
Isotonic and hypertonic concentrations of dextrose are suitable for parenteral maintenance of water requirements when salt is not needed or should be avoided.
Solutions containing carbohydrate in the form of dextrose restore blood glucose levels and provide calories. Carbohydrate in the form of dextrose may aid in minimizing liver glycogen depletion and exerts a protein- sparing action. Dextrose injected parenterally undergoes oxidation to carbon dioxide and water.
Water is an essential constituent of all body tissues and accounts for approximately 70% of total body weight. Average normal adult daily requirements range from two to three liters (1 to 1.5 liters each for insensible water loss by perspiration and urine production).
Water balance is maintained by various regulatory mechanisms. Water distribution depends primarily on the concentration of electrolytes in the body compartments and sodium (Na + ) plays a major role in maintaining physiologic equilibrium.
HOW SUPPLIED:
5% Dextrose Injection, USP is supplied in single dose flexible plastic containers as follows:
| Product Code | Unit of Sale | Strength | Each |
| 252300 | NDC 65219-234-10 Package of 60 free flex ® + bags | 2.5 grams per 50 mL (50 mg per mL) | NDC 65219-234-01 50 mL in a 100 mL free flex ® + bag |
| 262300 | NDC 65219-236-10 Package of 50 free flex ® + bags | 5 grams per 100 mL (50 mg per mL) | NDC 65219-236-01 100 mL free flex ® + bag |
| 262325 | NDC 65219-238-25 Package of 30 free flex ® + bags | 12.5 grams per 250 mL (50 mg per mL) | NDC 65219-238-01 250 mL free flex ® + bag |
| 262450 | NDC 65219-240-50 Package of 20 free flex ® + bags | 25 grams per 500 mL (50 mg per mL) | NDC 65219-240-01 500 mL free flex ® + bag |
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing.
The container closure is not made with natural rubber latex. Non-PVC, Non-DEHP, Sterile.

Lake Zurich, IL 60047
www.fresenius-kabi.com/us
Revised: August 2024
INSTRUCTIONS FOR USE:
Check flexible container solution composition, lot number, and expiry date.
Do not remove solution container from its overwrap until immediately before use.
Use sterile equipment and aseptic technique.
To Open
- Turn solution container over so that the text is face down. Using the pre-cut corner tabs, peel open the overwrap and remove solution container.
- Check the solution container for leaks by squeezing firmly. If leaks are found, or if the seal is not intact, discard the solution.
- Do not use if the solution is cloudy or a precipitate is present.
To Add Medication
The Additive port of the free flex ® + container accepts standard luer lock syringes. Do not use a needle for additions.
- Identify LIGHT BLUE Additive Port with arrow pointing toward solution container.
- Immediately before injecting additives, break off LIGHT BLUE Additive Port Cap with the arrow pointing toward solution container.
- Hold base of LIGHT BLUE Additive Port.
- Attach Luer Lock syringe to the threaded LIGHT BLUE Additive Port. Inject additive.
- Mix solution container contents thoroughly.
Preparation for Administration
- Immediately before inserting the infusion set, break off BLUE Infusion Port Cap with the arrow pointing away from container.
- Use a non-vented infusion set or close the air-inlet on a vented set.
- Close the roller clamp of the infusion set.
- Hold the base of BLUE Infusion Port.
- Insert spike through BLUE Infusion Port by rotating wrist slightly until the spike is inserted.
NOTE: See full directions accompanying administration set.
WARNING: Do not use flexible container in series connections.