Get your patient on Drospirenone And Ethinyl Estradiol - Drospirenone And Ethinyl Estradiol (Drospirenone And Ethinyl Estradiol)
Drospirenone And Ethinyl Estradiol - Drospirenone And Ethinyl Estradiol prescribing information
WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS
WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS
See full prescribing information for complete boxed warning.
- Women over 35 years old who smoke should not use drospirenone and ethinyl estradiol tablets. (4)
- Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive (COC) use. (4)
Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptives (COC) use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, COCs should not be used by women who are over 35 years of age and smoke [see CONTRAINDICATIONS (4)].
RECENT MAJOR CHANGES
Dosage and Administration (2.3) 5/2023
Contraindications, Pregnancy (4) Removed 5/2023
Warnings and Precautions, (5.11) Removed 5/2023
INDICATIONS AND USAGE
Drospirenone and Ethinyl Estradiol Tablets USP is a combination of drospirenone, a progestin, and ethinyl estradiol, an estrogen, indicated for use by females of reproductive potential to prevent pregnancy.(1)
Drospirenone and Ethinyl Estradiol Tablets USP are indicated for use by females of reproductive potential to prevent pregnancy
DOSAGE AND ADMINISTRATION
- Take one tablet daily by mouth at the same time every day. (2.1)
- Tablets must be taken in the order directed on the blister pack. (2.1)
How to Take Drospirenone and Ethinyl Estradiol Tablets
Take one tablet by mouth at the same time every day. The failure rate may increase when pills are missed or taken incorrectly.
To achieve maximum contraceptive effectiveness, drospirenone and ethinyl estradiol tablets must be taken as directed, in the order directed on the blister pack. Single missed pills should be taken as soon as remembered.
How to Start Drospirenone and Ethinyl Estradiol Tablets
Instruct the patient to begin taking drospirenone and ethinyl estradiol tablets either on the first day of her menstrual period (Day 1 Start) or on the first Sunday after the onset of her menstrual period (Sunday Start).
Day 1 Start
During the first cycle of drospirenone and ethinyl estradiol tablets use, instruct the patient to take one yellow drospirenone and ethinyl estradiol tablets daily, beginning on Day 1 of her menstrual cycle. (The first day of menstruation is Day 1.) She should take one yellow drospirenone and ethinyl estradiol tablet daily for 21 consecutive days, followed by one white tablet daily on Days 22 through 28. Drospirenone and ethinyl estradiol tablets should be taken in the order directed on the package at the same time each day, preferably after the evening meal or at bedtime with some liquid, as needed. Drospirenone and ethinyl estradiol tablets can be taken without regard to meals. If Drospirenone and ethinyl estradiol tablets are first taken later than the first day of the menstrual cycle, drospirenone and ethinyl estradiol tablets should not be considered effective as a contraceptive until after the first 7 consecutive days of product administration. Instruct the patient to use a non-hormonal contraceptive as back-up during the first 7 days. The possibility of ovulation and conception prior to initiation of medication should be considered.
Sunday Start
During the first cycle of drospirenone and ethinyl estradiol tablets use, instruct the patient to take one yellow drospirenone and ethinyl estradiol tablet daily, beginning on the first Sunday after the onset of her menstrual period. She should take one yellow drospirenone and ethinyl estradiol tablet daily for 21 consecutive days, followed by one white tablet daily on Days 22 through 28. Drospirenone and ethinyl estradiol tablets should be taken in the order directed on the package at the same time each day, preferably after the evening meal or at bedtime with some liquid, as needed. Drospirenone and ethinyl estradiol tablets can be taken without regard to meals. Drospirenone and ethinyl estradiol tablets should not be considered effective as a contraceptive until after the first 7 consecutive days of product administration. Instruct the patient to use a non-hormonal contraceptive as back-up during the first 7 days. The possibility of ovulation and conception prior to initiation of medication should be considered.
The patient should begin her next and all subsequent 28-day regimens of drospirenone and ethinyl estradiol tablets on the same day of the week that she began her first regimen, following the same schedule. She should begin taking her yellow tablets on the next day after ingestion of the last white tablet, regardless of whether or not a menstrual period has occurred or is still in progress. Anytime a subsequent cycle of drospirenone and ethinyl estradiol tablets is started later than the day following administration of the last white to off-white tablet, the patient should use another method of contraception until she has taken a yellow drospirenone and ethinyl estradiol tablets daily for seven consecutive days.
When Switching from a Different Birth Control Pill
When switching from another birth control pill, drospirenone and ethinyl estradiol tablets should be started on the same day that a new pack of the previous oral contraceptive would have been started.
When Switching from a Method Other Than a Birth Control Pill
When switching from a transdermal patch or vaginal ring, drospirenone and ethinyl estradiol tablets should be started when the next application would have been due. When switching from an injection, drospirenone and ethinyl estradiol tablets should be started when the next dose would have been due. When switching from an intrauterine contraceptive or an implant, drospirenone and ethinyl estradiol tablets should be started on the day of removal.
Withdrawal bleeding usually occurs within 3 days following the last yellow tablet. If spotting or breakthrough bleeding occurs while taking drospirenone and ethinyl estradiol tablets, instruct the patient to continue taking drospirenone and ethinyl estradiol tablets by the regimen described above. Counsel her that this type of bleeding is usually transient and without significance; however, advise her that if the bleeding is persistent or prolonged, she should consult her healthcare provider.
Although the occurrence of pregnancy is low if drospirenone and ethinyl estradiol tablets are taken according to directions, if withdrawal bleeding does not occur, consider the possibility of pregnancy. If the patient has not adhered to the prescribed dosing schedule (missed one or more active tablets or started taking them on a day later than she should have), consider the possibility of pregnancy at the time of the first missed period and take appropriate diagnostic measures. If the patient has adhered to the prescribed regimen and misses two consecutive periods, rule out pregnancy. Discontinue drospirenone and ethinyl estradiol tablets if pregnancy is confirmed.
The risk of pregnancy increases with each active yellow tablet missed.If breakthrough bleeding occurs following missed tablets, it will usually be transient and of no consequence. If the patient misses one or more white to off-white tablets, she should still be protected against pregnancy provided she begins taking a new cycle of yellow tablets on the proper day.
For postpartum women who do not breastfeed or after a second trimester abortion, start drospirenone and ethinyl estradiol tablets no earlier than 4 weeks postpartum due to the increased risk of thromboembolism. If the patient starts drospirenone and ethinyl estradiol tablets postpartum and has not yet had a period, evaluate for possible pregnancy, and instruct her to use an additional method of contraception until she has taken drospirenone and ethinyl estradiol tablets for 7 consecutive days.
Missed Dose
Table 1: Instructions for Drospirenone and Ethinyl Estradiol Tablets Missed Doses
| If one yellow active tablet is missed | Take it as soon as possible. Take the next tablet at the regular time. This means two tablets may be taken in one day. A back-up birth control method is not required if the patient has sex |
| If two yellow active tablets in a row are missed in Week 1 or Week 2 | Take two tablets as soon as possible and two tablets the next day. Then take one tablet a day until the pack is finished. Additional nonhormonal contraception (such as condoms and spermicide) should be used as back-up if the patient has sex within 7 days after missing tablets. |
| If two yellow active tablets in a row are missed in Week 3 or Week 4 | Day 1 Start: Throw out the rest of the pack and start a new pack that same day. Sunday Start: Keep taking one tablet every day until Sunday. On Sunday, throw out the rest of the pack and start a new pack that same day. Additional nonhormonal contraception (such as condoms and spermicide) should be used as back-up if the patient has sex within 7 days after missing tablets. The patient may not have their period this month but this is expected. However, if they miss their period two months in a row, they should call their healthcare provider because they might be pregnant. |
| If three or more yellow active tablets in a row are missed during any week | Day 1 Start: Throw out the rest of the pack and start a new pack that same day. Sunday Start: Keep taking one tablet every day until Sunday. On Sunday, throw out the rest of the pack and start a new pack that same day. Additional nonhormonal contraception (such as condoms and spermicide) should be used as back-up if the patient has sex within 7 days after missing tablets. The patient may not have their period this month but this is expected. However, if they miss their period two months in a row, they should call their healthcare provider because they might be pregnant. |
| If any of the seven white inactive tablets are missed in Week 4 | Throw away the tablets that were missed. Keep taking one tablet each day until the pack is empty. They do not need a back-up method. |
| Finally, if they are still not sure what to do about the tablets they have missed | Use nonhormonal contraception (such as condoms and spermicides) anytime they have sex. Contact their healthcare provider and continue taking one active yellow tablet each day until otherwise directed. |
Advice in Case of Gastrointestinal Disturbances
In case of severe vomiting or diarrhea, absorption may not be complete and additional contraceptive measures should be taken. If vomiting occurs within 3-4 hours after tablet-taking, this can be regarded as a missed tablet.
DOSAGE FORMS AND STRENGTHS
Drospirenone and ethinyl estradiol tablets consists of 28, biconvex tablets in the following order (3):
- 21 yellow film-coated tablets, each containing 3 mg drospirenone (DRSP) and 0.03 mg ethinyl estradiol (EE),
- 7 inert white tablets
Drospirenone and ethinyl estradiol tablets, 3 mg and 0.03 mg are available in blister packs.
Each blister pack contains 28, round, bi-convex tablets in the following order:
- 21 yellow tablets each containing 3 mg drospirenone (DRSP) and 0.03 mg ethinyl estradiol (EE) debossed with a “EV1” on one side and plain on other side
- 7 inert white tablets debossed with a “EV2” on one side and plain on other side
USE IN SPECIFIC POPULATIONS
Lactation: Can reduce milk production in breast-feeding females (8.2)
PREGNANCY
Risk Summary
There is no use for contraception in pregnancy; therefore, Drospirenone and Ethinyl Estradiol Tablets should be discontinued during pregnancy. Epidemiologic studies and meta-analyses have not found an increased risk of genital or non-genital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to CHCs before conception or during early pregnancy. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4 percent and 15 to 20 percent, respectively.
Data
Human Data
A retrospective database study of women in Norway, that included 44,734 pregnancies of which 368 were women who inadvertently took drospirenone/ethinyl estradiol during the first trimester of a pregnancy, found there were no adverse effects on pre-term birth, small for gestational age, or birth weight Z-scores.
Post-marketing adverse event data on the use of Drospirenone and Ethinyl Estradiol Tablets USP in pregnant women suggest that frequencies of miscarriage and congenital anomalies were not higher than the estimated background risk in the general population.
Lactation
Risk Summary DRSP is present in human milk. After a single oral administration of 3 mg DRSP/0.03 mg EE tablets, DRSP concentration in breast milk over the 24-h period ranged from 1.4 to 7.0 ng/mL, with a mean ± standard deviation value of 3.7 ± 1.9 ng/mL. The estimated mean infant dose was 0.003 mg/day, which is about 0.1% of maternal dose (see Data). There is limited information on the effects of Drospirenone and Ethinyl Estradiol Tablets on the breast-fed infant. CHCs can reduce milk production in breast-feeding females. This reduction can occur at any time but is less likely to occur once breast-feeding is well-established. When possible, advise the nursing female to use other methods of contraception until she discontinues breastfeeding. [See also Dosage and Administration (2.2)]. The developmental and health benefits of breast-feeding should be considered along with the mother’s clinical need for Drospirenone and Ethinyl Estradiol Tablets and any potential adverse effects on the breast-fed child from Drospirenone and Ethinyl Estradiol Tablets or from the underlying maternal condition. Data Human Data An open-label study evaluated the degree of DRSP transfer into milk within 72 hours following a single oral administration of 3 mg DRSP/0.03 mg EE tablets to 6 healthy lactating women who were 1 week to 3 months postpartum. DRSP was present in breast milk with a mean Cmax of 13.5 ng/mL, while the mean Cmax in serum of lactating women was 30.8 ng/mL. The DRSP concentration in breast milk over the 24-hour period following dosing ranged from 1.4 to 7.0 ng/mL, with a mean ± standard deviation value of 3.7 ± 1.9 ng/mL. Based on single dose data, the maximal daily infant dose of DRSP was calculated to be 0.003 mg/day, which represented a mean of 0.1% of the maternal dose.
PEDIATRIC USE
Safety and efficacy of drospirenone and ethinyl estradiol tablets have been established in women of reproductive age. Efficacy is expected to be the same for postpubertal adolescents under the age of 18 and for users 18 years and older. Use of this product before menarche is not indicated.
GERIATRIC USE
Drospirenone and ethinyl estradiol tablets have not been studied in postmenopausal women and are not indicated in this population.
Patients with Renal Impairment
Drospirenone and ethinyl estradiol tablets are contraindicated in patients with renal impairment [see CONTRAINDICATIONS (4) and WARNINGS AND PRECAUTIONS (5.2) ].
In subjects with creatinine clearance (CLcr) of 50 to 79 mL/min, serum DRSP concentrations were comparable to those in a control group with CLcr ≥ 80 mL/min. In subjects with CLcr of 30 to 49 mL/min, serum DRSP concentrations were on average 37% higher than those in the control group. In addition, there is a potential to develop hyperkalemia in subjects with renal impairment whose serum potassium is in the upper reference range, and who are concomitantly using potassium sparing drugs [see CLINICAL PHARMACOLOGY (12.3) ].
Patients with Hepatic Impairment
Drospirenone and ethinyl estradiol tablets are contraindicated in patients with hepatic disease [see CONTRAINDICATIONS (4) and WARNINGS AND PRECAUTIONS (5.4) ]. The mean exposure to DRSP in women with moderate liver impairment is approximately three times higher than the exposure in women with normal liver function. Drospirenone and ethinyl estradiol tablets have not been studied in women with severe hepatic impairment.
Race
No clinically significant difference was observed between the pharmacokinetics of DRSP or EE in Japanese versus Caucasian women [see CLINICAL PHARMACOLOGY (12.3) ].
CONTRAINDICATIONS
- Renal impairment (4 )
- Adrenal insufficiency (4 )
- A high risk of arterial or venous thrombotic diseases (4 )
- Undiagnosed abnormal uterine bleeding (4 )
- Breast cancer (4 )
- Liver tumors or liver disease (4 )
- Co-administration with Hepatitis C drug combinations containing ombitasvir, paritaprevir/ritonavir, with or without dasabuvir (4 )
Drospirenone and ethinyl estradiol tablet is contraindicated in females who are known to have or develop the following conditions:
• Renal impairment
• Adrenal insufficiency
• A high risk of arterial or venous thrombotic diseases. Examples include women who are known to:
○ Smoke, if over age 35 [see Boxed Warning and Warnings and Precautions (5.1 ) ]
○ Have deep vein thrombosis or pulmonary embolism, now or in the past [see Warnings and Precautions (5.1 ) ]
○ Have cerebrovascular disease [see Warnings and Precautions (5.1 ) ]
○ Have coronary artery disease [ see Warnings and Precautions (5.1 ) ]
○ Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see Warnings AND Precautions (5.1 ) ]
○ Have inherited or acquired hypercoagulopathies [see Warnings and Precautions (5.1 ) ]
○ Have uncontrolled hypertension [see Warnings and Precautions (5.6 ) ]
○ Have diabetes mellitus with vascular disease [see Warnings and Precautions (5.8 ) ]
○ Have headaches with focal neurological symptoms or have migraine headaches with or without aura if over age 35 [see Warnings And Precautions (5.9 ) ]
- Undiagnosed abnormal uterine bleeding [see Warnings and Precautions (5.10 ) ]
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive [see Warnings And Precautions (5.3 ) ]
- Liver tumor (benign or malignant) or liver disease [see Warnings And Precautions (5.4 ) and Use In Specific Populations (8.7 ) ]
- Use of Hepatitis C drug combinations containing ombitasvir, paritaprevir/ritonavir, with or without dasabuvir due to the potential for ALT elevations [see Warnings And Precautions (5.5 ) and Drug Interactions (7.2 ) ].
WARNINGS AND PRECAUTIONS
- Vascular risks : Stop drospirenone and ethinyl estradiol tablets if a thrombotic event occurs. Stop at least 4 weeks before and through 2 weeks after major surgery. Start no earlier than 4 weeks after delivery, in women who are not breastfeeding. (5.1) COCs containing DRSP may be associated with a higher risk of venous thromboembolism (VTE) than COCs containing levonorgestrel or some other progestins. Before initiating drospirenone and ethinyl estradiol tablets in a new COC user or a woman who is switching from a contraceptive that does not contain DRSP, consider the risks and benefits of a DRSP-containing COC in light of her risk of a VTE. (5.1)
- Hyperkalemia : DRSP has anti-mineralocorticoid activity. Do not use in patients predisposed to hyperkalemia. Check serum potassium concentration during the first treatment cycle in women on long-term treatment with medications that may increase serum potassium concentration. (5.2, 7.1, 7.2)
- Liver disease : Discontinue drospirenone and ethinyl estradiol tablets if jaundice occurs. (5.4)
- High blood pressure : Do not prescribe drospirenone and ethinyl estradiol tablets for women with uncontrolled hypertension or hypertension with vascular disease. (5.6)
- Carbohydrate and lipid metabolic effects : Monitor prediabetic and diabetic women taking drospirenone and ethinyl estradiol tablets. Consider an alternate contraceptive method for women with uncontrolled dyslipidemia. (5.8)
- Headache : Evaluate significant change in headaches and discontinue drospirenone and ethinyl estradiol tablets if indicated. (5.9)
- Uterine bleeding : Evaluate irregular bleeding or amenorrhea. (5.10)
Hyperkalemia
Drospirenone and ethinyl estradiol tablets contain 3 mg of the progestin DRSP, which has anti-mineralocorticoid activity, including the potential for hyperkalemia in high-risk patients, comparable to a 25 mg dose of spironolactone. Drospirenone and ethinyl estradiol tablet is contraindicated in patients with conditions that predispose to hyperkalemia (that is, renal impairment, hepatic impairment, and adrenal insufficiency). Women receiving daily, long-term treatment for chronic conditions or diseases with medications that may increase serum potassium concentration should have their serum potassium concentration checked during the first treatment cycle. Medications that may increase serum potassium concentration include ACE inhibitors, angiotensin–II receptor antagonists, potassium-sparing diuretics, potassium supplementation, heparin, aldosterone antagonists, and NSAIDs.
Consider monitoring serum potassium concentration in high-risk patients who take a strong CYP3A4 inhibitor long-term and concomitantly. Strong CYP3A4 inhibitors include azole antifungals (e.g. ketoconazole, itraconazole, voriconazole), HIV/HCV protease inhibitors (e.g., indinavir, boceprevir), and clarithromycin [see Clinical Pharmacology (12.3 ) ]
Malignant Neoplasms
Breast Cancer
Drospirenone and ethinyl estradiol tablet is contraindicated in females who currently have or have had breast cancer because breast cancer may be hormonally sensitive [see Contraindications (4 ) ].
Epidemiology studies have not found a consistent association between use of combined oral contraceptives (COCs) and breast cancer risk. Studies do not show an association between ever (current or past) use of COCs and risk of breast cancer. However, some studies report a small increase in the risk of breast cancer among current or recent users (<6 months since last use) and current users with longer duration of COC use [see Adverse Reactions (6.2 ) ].
Cervical Cancer
Some studies suggest that COCs are associated with an increase in the risk of cervical cancer or intraepithelial neoplasia. However, there is controversy about the extent to which these findings may be due to differences in sexual behavior and other factors.
Liver Disease
Discontinue drospirenone and ethinyl estradiol tablets if jaundice develops. Steroid hormones may be poorly metabolized in patients with impaired liver function. Acute or chronic disturbances of liver function may necessitate the discontinuation of COC use until markers of liver function return to normal and COC causation has been excluded.
Hepatic adenomas are associated with COC use. An estimate of the attributable risk is 3.3 cases/100,000 COC users. Rupture of hepatic adenomas may cause death through intra-abdominal hemorrhage.
Studies have shown an increased risk of developing hepatocellular carcinoma in long-term (> 8 years) COC users. However, the attributable risk of liver cancers in COC users is less than one case per million users.
Oral contraceptive-related cholestasis may occur in women with a history of pregnancy-related cholestasis. Women with a history of COC-related cholestasis may have the condition recur with subsequent COC use.
Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in women using ethinyl estradiol-containing medications, such as COCs. Discontinue drospirenone and ethinyl estradiol tablets prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir [see CONTRAINDICATIONS (4)]. Drospirenone and ethinyl estradiol tablets can be restarted approximately 2 weeks following completion of treatment with the Hepatitis C combination drug regimen.
High Blood Pressure
For women with well-controlled hypertension, monitor blood pressure and stop drospirenone and ethinyl estradiol tablets if blood pressure rises significantly. Women with uncontrolled hypertension or hypertension with vascular disease should not use COCs.
An increase in blood pressure has been reported in women taking COCs, and this increase is more likely in older women and with extended duration of use. The incidence of hypertension increases with increasing concentration of progestin.
Gallbladder Disease
Studies suggest a small increased relative risk of developing gallbladder disease among COC users.
Carbohydrate and Lipid Metabolic Effects
Carefully monitor prediabetic and diabetic women who are taking drospirenone and ethinyl estradiol tablets. COCs may decrease glucose tolerance in a dose-related fashion.
Consider alternative contraception for women with uncontrolled dyslipidemia. A small proportion of women will have adverse lipid changes while on COCs.
Women with hypertriglyceridemia, or a family history thereof, may be at an increased risk of pancreatitis when using COCs.
Headache
If a woman taking drospirenone and ethinyl estradiol tablets develops new headaches that are recurrent, persistent, or severe, evaluate the cause and discontinue drospirenone and ethinyl estradiol tablets if indicated.
An increase in frequency or severity of migraine during COC use (which may be prodromal of a cerebrovascular event) may be a reason for immediate discontinuation of the COC.
Bleeding Irregularities
Unscheduled (breakthrough or intracyclic) bleeding and spotting sometimes occur in patients on COCs, especially during the first three months of use. If bleeding persists or occurs after previously regular cycles, check for causes such as pregnancy or malignancy. If pathology and pregnancy are excluded, bleeding irregularities may resolve over time or with a change to a different COC.
Data from ten contraceptive efficacy clinical trials (N=2,467) show that the percent of women who took drospirenone and ethinyl estradiol tablets and experienced unscheduled bleeding decreased over time from 12% at cycle 2 to 6% (cycle 13). A total of 24 subjects out of 2,837 in the drospirenone and ethinyl estradiol tablets trials (<1%) discontinued due to bleeding complaints. These are described as metrorrhagia, vaginal hemorrhage, menorrhagia, abnormal withdrawal bleeding, and menometrorrhagia.
The average duration of scheduled bleeding episodes in the majority of subjects (86% to 88%) was 4 to 7 days. Women who use drospirenone and ethinyl estradiol tablets may experience absence of withdrawal bleeding, even if they are not pregnant. Based on subject diaries from contraceptive efficacy trials, during cycles 2 to 13, 1 to 11% of women per cycle experienced no withdrawal bleeding. Some women may encounter post-pill amenorrhea or oligomenorrhea, especially when such a condition was pre-existent.
If withdrawal bleeding does not occur, consider the possibility of pregnancy. If the patient has not adhered to the prescribed dosing schedule (missed one or more active tablets or started taking them on a day later than she should have), consider the possibility of pregnancy at the time of the first missed period and take appropriate diagnostic measures. If the patient has adhered to the prescribed regimen and misses two consecutive periods, rule out pregnancy.
Depression
Women with a history of depression should be carefully observed and drospirenone and ethinyl estradiol tablets discontinued if depression recurs to a serious degree .
Interference with Laboratory Tests
The use of COCs may change the results of some laboratory tests, such as coagulation factors, lipids, glucose tolerance, and binding proteins. Women on thyroid hormone replacement therapy may need increased doses of thyroid hormone because serum concentrations of thyroid-binding globulin increase with use of COCs [see DRUG INTERACTIONS (7.2) ] .
DRSP causes an increase in plasma renin activity and plasma aldosterone induced by its mild anti-mineralocorticoid activity.
Monitoring
A woman who is taking COCs should have a yearly visit with her healthcare provider for a blood pressure check and for other indicated healthcare.
Other Conditions
In women with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema. Chloasma may occasionally occur, especially in women with a history of chloasma gravidarum. Women with a tendency to chloasma should avoid exposure to the sun or ultraviolet radiation while taking COCs.
Thromboembolic Disorders and Other Vascular Problems
Stop Drospirenone and Ethinyl Estradiol Tablets if an arterial or venous thrombotic (VTE) event occurs.
Based on presently available information on Drospirenone and Ethinyl Estradiol Tablets, DRSP-containing COCs may be associated with a higher risk of venous thromboembolism (VTE) than COCs containing the progestin levonorgestrel or some other progestins. Epidemiologic studies that compared the risk of VTE reported that the risk ranged from no increase to a three-fold increase. Before initiating use of Drospirenone and Ethinyl Estradiol Tablets in a new COC user or a woman who is switching from a contraceptive that does not contain DRSP, consider the risks and benefits of a DRSP-containing COC in light of her risk of a VTE. Known risk factors for VTE include smoking, obesity, and family history of VTE, in addition to other factors that contraindicate use of COCs [see Contraindications (4)].
A number of studies have compared the risk of VTE for users of Drospirenone and Ethinyl Estradiol Tablets to the risk for users of other COCs, including COCs containing levonorgestrel. Those that were required or sponsored by regulatory agencies are summarized in Table 2.
| Table 2: Estimates (Hazard Ratios) of Venous Thromboembolism Risk in Current Users of Drospirenone and Ethinyl Estradiol Tablets Compared to Users of Oral Contraceptives that Contain Other Progestins | ||
| Epidemiologic Study (Author, Year of Publication) Population Studied | Comparator Product (all are low-dose COCs; with ≤ 0.04 mg of EE) | Hazard Ratio (HR) (95% CI) |
i3 Ingenix (Seeger 2007) Initiators, including new users a | All COCs available in the US during the conduct of the study b | HR: 0.9 (0.5 - 1.6) |
EURAS (Dinger 2007) Initiators, including new users a | All COCs available in Europe during the conduct of the study c Levonorgestrel/EE | HR: 0.9 (0.6 - 1.4) HR: 1.0 (0.6 - 1.8) |
“FDA-funded study” (2011) New users a All users (i.e., initiation and continuing use of study combination hormonal contraception) | Other COCs available during the course of the study d Levonorgestrel/0.03 mg EE Other COCs available during the course of the study d Levonorgestrel/0.03 mg EE | HR: 1.8 (1.3 - 2.4) HR: 1.6 (1.1 - 2.2) HR: 1.7 (1.4 - 2.1) HR: 1.5 (1.2 - 1.8) |
“New users” - no use of combination hormonal contraception for at least the prior 6 months Includes low-dose COCs containing the following progestins: norgestimate, norethindrone, levonorgestrel, desogestrel, norgestrel, medroxyprogesterone, or ethynodiol diacetate Includes low-dose COCs containing the following progestins: levonorgestrel, desogestrel, dienogest, chlormadinone acetate, gestodene, cyproterone acetate, norgestimate, or norethindrone Includes low-dose COCs containing the following progestins: norgestimate, norethindrone, or levonorgestrel
In addition to these “regulatory studies,” other studies of various designs have been conducted. Overall, there are two prospective cohort studies (see Table 2): the US post-approval safety study Ingenix [Seeger 2007], the European post-approval safety study EURAS (European Active Surveillance Study) [Dinger 2007]. An extension of the EURAS study, the Long-Term Active Surveillance Study (LASS), did not enroll additional subjects, but continued to assess VTE risk. There are three retrospective cohort studies: one study in the US funded by the FDA (see Table 2), and two from Denmark [Lidegaard 2009, Lidegaard 2011]. There are two case-control studies: the Dutch MEGA study analysis [van Hylckama Vlieg 2009] and the German case-control study [Dinger 2010]. There are two nested case-control studies that evaluated the risk of non-fatal idiopathic VTE: the PharMetrics study [Jick 2011] and the GPRD study [Parkin 2011]. The results of all of these studies are presented in Figure 1.
Figure 1: VTE Risk with Drospirenone and Ethinyl Estradiol Tablets Relative to LNG-Containing COCs (adjusted risk#)
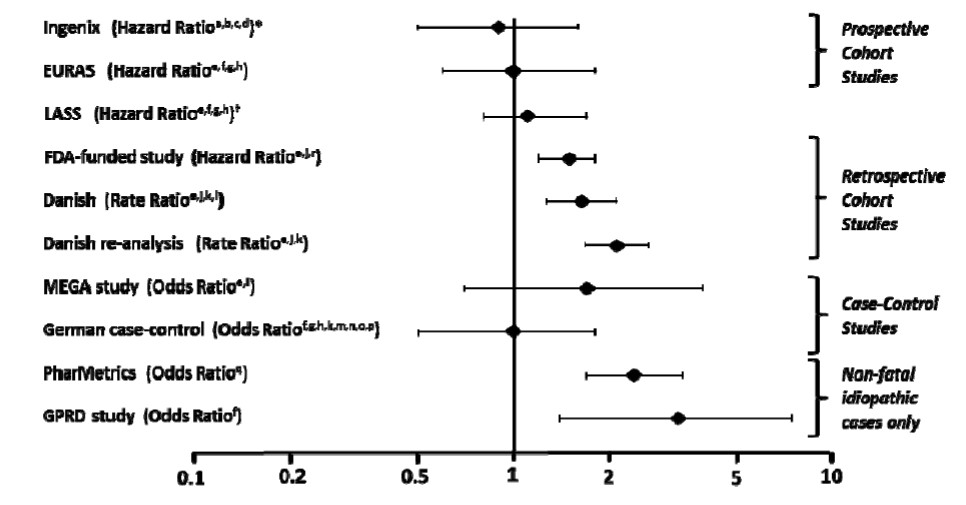
Risk ratios displayed on logarithmic scale; risk ratio < 1 indicates a lower risk of VTE for DRSP, > 1 indicates an increased risk of VTE for DRSP.
•Comparator “Other COCs”, including LNG-containing COCs
† LASS is an extension of the EURAS study
#Some adjustment factors are indicated by superscript letters: a) Current heavy smoking, b) hypertension, c) obesity, d) family history, e) age, f) BMI, g) duration of use, h) VTE history, i) period of inclusion, j) calendar year, k) education, l) length of use, m) parity, n) chronic disease, o) concomitant medication, p) smoking, q) duration of exposure, r) site
(References: Ingenix [Seeger 2007]1, EURAS (European Active Surveillance Study) [Dinger 2007]2, LASS (Long-Term Active Surveillance Study) [Dinger, unpublished document on file], FDA-funded study [Sidney 2011]3, Danish [Lidegaard 2009]4, Danish re- analysis [ Lidegaard 2011]5, MEGA study [van Hylckama Vlieg 2009]6, German Case-Control study [Dinger 2010]7, PharMetrics [Jick 2011]8, GPRD study [Parkin 2011]9)
Although the absolute VTE rates are increased for users of hormonal contraceptives compared to non-users, the rates during pregnancy are even greater, especially during the post-partum period (see Figure 2). The risk of VTE in women using COCs has been estimated to be 3 to 9 per 10,000 woman-years. The risk of VTE is highest during the first year of use. Data from a large, prospective cohort safety study of various COCs suggest that this increased risk, as compared to that in non-COC users, is greatest during the first 6 months of COC use. Data from this safety study indicate that the greatest risk of VTE is present after initially starting a COC or restarting (following a 4 week or greater pill-free interval) the same or a different COC.
The risk of thromboembolic disease due to oral contraceptives gradually disappears after COC use is discontinued.
Figure 2 shows the risk of developing a VTE for women who are not pregnant and do not use oral contraceptives, for women who use oral contraceptives, for pregnant women, and for women in the postpartum period. To put the risk of developing a VTE into perspective: If 10,000 women who are not pregnant and do not use oral contraceptives are followed for one year, between 1 and 5 of these women will develop a VTE.
Figure 2: Likelihood of Developing a VTE
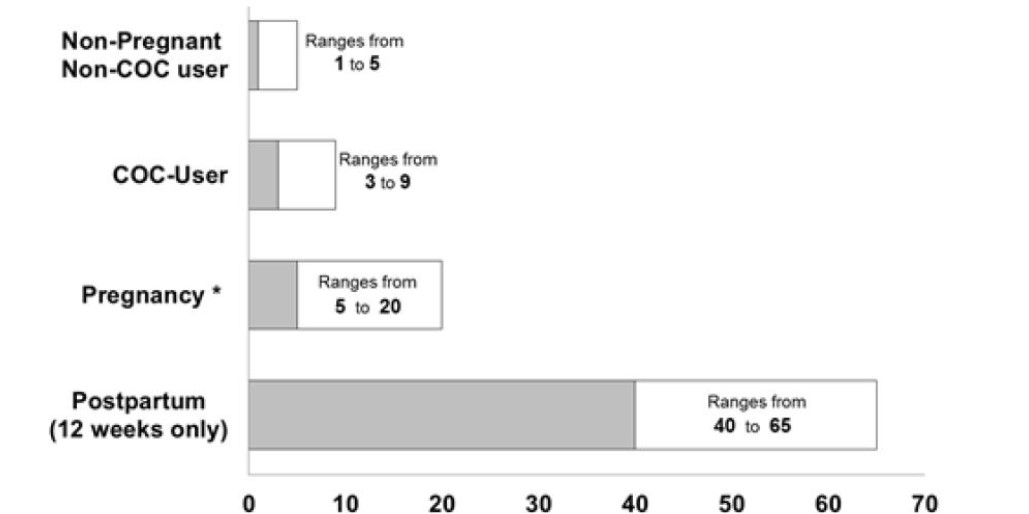
Number of Women with a Blood Clot
Out of 10,000 Women Years (WY)
• Pregnancy data based on actual duration of pregnancy in the reference studies. Based on a model assumption that pregnancy duration is nine months, the rate is 7 to 27 per 10,000 WY
If feasible, stop Drospirenone and Ethinyl Estradiol Tablets at least 4 weeks before and through 2 weeks after major surgery or other surgeries known to have an elevated risk of thromboembolism.
Start Drospirenone and Ethinyl Estradiol Tablets no earlier than 4 weeks after delivery, in women who are not breastfeeding. The risk of postpartum thromboembolism decreases after the third postpartum week, whereas the risk of ovulation increases after the third postpartum week.
Use of COCs also increases the risk of arterial thromboses such as strokes and myocardial infarctions, especially in women with other risk factors for these events.
COCs have been shown to increase both the relative and attributable risks of cerebrovascular events (thrombotic and hemorrhagic strokes), although, in general, the risk is greatest among older (>35 years of age), hypertensive women who also smoke. COCs also increase the risk for stroke in women with other underlying risk factors.
Oral contraceptives must be used with caution in women with cardiovascular disease risk factors.
Stop Drospirenone and Ethinyl Estradiol Tablets if there is unexplained loss of vision, proptosis, diplopia, papilledema, or retinal vascular lesions. Evaluate for retinal vein thrombosis immediately. [See Adverse Reactions (6).]
ADVERSE REACTIONS
The following serious adverse reactions with the use of COCs are discussed elsewhere in the labeling: • Serious cardiovascular events and stroke [see Boxed Warning and Warnings and Precautions (5.1)] • Vascular events [see Warnings and Precautions (5.1)] • Liver disease [see Warnings and Precautions (5.4)]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other clinical trials and may not reflect the rates observed in practice. The data provided reflect the experience with the use of drospirenone and ethinyl estradiol tablets (3 mg DRSP/0.03 mg EE) in the adequate and well-controlled studies for contraception (N=2,837). The US pivotal clinical study (N=326) was a multicenter, open-label trial in healthy women aged 18 to 35 who were treated for up to 13 cycles. The second pivotal study (N=442)was a multicenter, randomized, open-label comparative European study of drospirenone and ethinyl estradiol tablets vs. 0.150 mg desogestrel/0.03 mg EE conducted in healthy women aged 17 to 40 who were treated for up to 26 cycles. The most common adverse reactions (≥ 2% of users) were: premenstrual syndrome (13.2%), headache/migraine (10.7%), breast pain/tenderness/discomfort (8.3%), nausea/vomiting (4.5%) abdominal pain/discomfort/tenderness (2.3%) and mood changes (depression, depressed mood, irritability, mood swings, mood altered and affect lability (2.3%). Adverse Reactions (≥ 1%) Leading to Study Discontinuation: Of 2,837 women, 6.7% discontinued from the clinical trials due to an adverse reaction; the most frequent adverse reaction leading to discontinuation was headache/migraine (1.5%). Serious Adverse Reactions:
Depression, pulmonary embolism, toxic skin eruption, and uterine leiomyoma.
Postmarketing Experience
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 3). Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 3). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 - 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
Figure 3: Relative Studies of Risk of Breast Cancer with Combined Oral Contraceptives
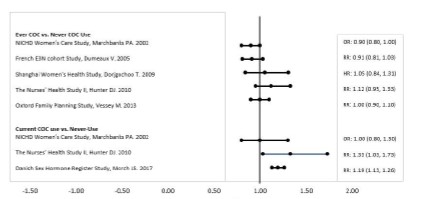
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ever COC” are females with current or past COC use; “never COC use” are females that never used COCs. The following adverse reactions have been identified during post-approval use of drospirenone and ethinyl estradiol tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Adverse reactions, including fatalities, are grouped into System Organ Classes and ordered by frequency. Vascular disorders: Venous and arterial thromboembolic events (including pulmonary emboli, deep vein thrombosis, intracardiac thrombosis, intracranial venous sinus thrombosis, sagittal sinus thrombosis, retinal vein occlusion, myocardial infarction and stroke), hypertension Hepatobiliary disorders: Gallbladder disease
Immune system disorders: Hypersensitivity
Metabolism and nutrition disorders: Hyperkalemia Skin and subcutaneous tissue disorders: Chloasma
DRUG INTERACTIONS
Drugs or herbal products that induce certain enzymes (for example, CYP3A4) may decrease the effectiveness of COCs or increase breakthrough bleeding.
Counsel patients to use a back-up or alternative method of contraception when enzyme inducers are used with COCs. (7.1)
Consult the labeling of all concurrently-used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
Effects of Other Drugs on Combined Oral Contraceptives
Substances Diminishing the Efficacy of COCs
Drugs or herbal products that induce certain enzymes, including cytochrome P450 3A4 (CYP3A4), may decrease the effectiveness of COCs or increase breakthrough bleeding. Some drugs or herbal products that may decrease the effectiveness of hormonal contraceptives include phenytoin, barbiturates, carbamazepine, bosentan, felbamate, griseofulvin, oxcarbazepine, rifampin, topiramate and products containing St. John's wort. Interactions between oral contraceptives and other drugs may lead to breakthrough bleeding and/or contraceptive failure. Counsel women to use an alternative method of contraception or a back-up method when enzyme inducers are used with COCs, and to continue back-up contraception for 28 days after discontinuing the enzyme inducer to ensure contraceptive reliability.
Substances Increasing the Plasma Concentrations of COCs
Co-administration of atorvastatin and certain COCs containing EE increase AUC values for EE by approximately 20%. Ascorbic acid and acetaminophen may increase plasma EE concentrations, possibly by inhibition of conjugation.
Concomitant administration of moderate or strong CYP3A4 inhibitors such as azole antifungals (e.g., ketoconazole, itraconazole, voriconazole, fluconazole), verapamil, macrolides (e.g., clarithromycin, erythromycin), diltiazem, and grapefruit juice can increase the plasma concentrations of the estrogen or the progestin or both. In a clinical drug-drug interaction study conducted in premenopausal women, once daily co-administration of DRSP 3 mg/EE 0.02 mg containing tablets with strong CYP3A4 inhibitor, ketoconazole 200 mg twice daily for 10 days resulted in a moderate increase of DRSP systemic exposure. The exposure of EE was increased mildly [see WARNINGS AND PRECAUTIONS (5.2) and CLINICAL PHARMACOLOGY (12.3) ].
Human Immunodeficiency Virus (HIV)/Hepatitis C Virus (HCV) Protease Inhibitors and Non-nucleoside Reverse Transcriptase Inhibitors
Significant changes (increase or decrease) in the plasma concentrations of estrogen and progestin have been noted in some cases of co-administration with HIV/HCV protease inhibitors or with non-nucleoside reverse transcriptase inhibitors.
Antibiotics
There have been reports of pregnancy while taking hormonal contraceptives and antibiotics, but clinical pharmacokinetic studies have not shown consistent effects of antibiotics on plasma concentrations of synthetic steroids.
Effects of Combined Oral Contraceptives on Other Drugs
COCs containing EE may inhibit the metabolism of other compounds. COCs have been shown to significantly decrease plasma concentrations of lamotrigine, likely due to induction of lamotrigine glucuronidation. This may reduce seizure control; therefore, dosage adjustments of lamotrigine may be necessary. Consult the labeling of the concurrently-used drug to obtain further information about interactions with COCs or the potential for enzyme alterations.
COCs Increasing the Plasma Concentrations of CYP450 Enzymes
In clinical studies, administration of a hormonal contraceptive containing EE did not lead to any increase or only to a weak increase in plasma concentrations of CYP3A4 substrates (e.g., midazolam) while plasma concentrations of CYP2C19 substrates (e.g., omeprazole and voriconazole) and CYP1A2 substrates (e.g., theophylline and tizanidine) can have a weak or moderate increase.
Clinical studies did not indicate an inhibitory potential of DRSP towards human CYP enzymes at clinically relevant concentrations [see CLINICAL PHARMACOLOGY (12.3) ].
Women on thyroid hormone replacement therapy may need increased doses of thyroid hormone because serum concentration of thyroid-binding globulin increases with use of COCs.
Potential to Increase Serum Potassium Concentration
There is a potential for an increase in serum potassium concentration in women taking drospirenone and ethinyl estradiol tablets with other drugs that may increase serum potassium concentration [see WARNINGS AND PRECAUTIONS (5.2) and CLINICAL PHARMACOLOGY (12.3) ].
Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation
Do not co-administer drospirenone and ethinyl estradiol tablets with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations [see WARNINGS AND PRECAUTIONS (5.5)].
Interference with Laboratory Tests
The use of contraceptive steroids may influence the results of certain laboratory tests, such as coagulation factors, lipids, glucose tolerance, and binding proteins. DRSP causes an increase in plasma renin activity and plasma aldosterone induced by its mild anti-mineralocorticoid activity [see Warnings And Precautions (5.13 ) and Drug Interactions (7.2 ) ].
DESCRIPTION
Drospirenone and ethinyl estradiol tablets USP, 3 mg and 0.03 mg, provide an oral contraceptive regimen consisting of 28 tablets that contain the ingredients specified for each tablet below:
- 21 Yellow, round, biconvex film coated tablets, debossed with ‘EV1’ on one side and plain on other side containing 3 mg drospirenone and 0.03 mg ethinyl estradiol.
- 7 inert white to off white, round, biconvex, film coated tablets, debossed with ‘EV2’ on one side and plain on other side.
The inactive ingredients in the yellow tablets are lactose monohydrate, corn starch, pregelatinized starch (maize), povidone, magnesium stearate, hypromellose, macrogol, titanium dioxide, talc, and ferric oxide pigment, yellow. The white inert film coated tablets contain lactose monohydrate, corn starch, pregelatinized starch (maize), povidone, magnesium stearate, hypromellose, talc, and triacetin.
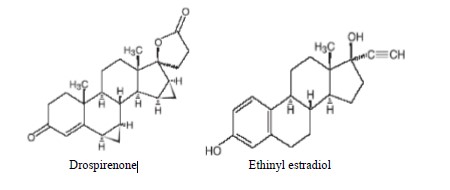
Drospirenone (6R,7R,8R,9S,10R,13S,14S,15S,16S,17S)-1,3',4',6,6a,7,8,9,10,11,12,13,14,15, 15a,16-hexadecahydro-10,13-dimethylspiro-[17H-dicyclopropa- [6,7:15,16]cyclopenta[a] phenanthrene-17,2'(5H)-furan]-3,5'(2H)-dione) is a synthetic progestational compound and has a molecular weight of 366.5 and a molecular formula of C 24 H 30 O 3 .
Ethinyl estradiol (19-nor-17α-pregna 1,3,5(10)-triene-20-yne-3,17-diol) is a synthetic estrogenic compound and has a molecular weight of 296.4 and a molecular formula of C 20 H 24 O 2 .
The structural formulas are as follows:
CLINICAL PHARMACOLOGY
MECHANISM OF ACTION
COCs lower the risk of becoming pregnant primarily by suppressing ovulation.
PHARMACODYNAMICS
Drospirenone is a spironolactone analogue with anti-mineralocorticoid activity. The estrogen in drospirenone and ethinyl estradiol tablets is ethinyl estradiol (EE).
No specific pharmacodynamic studies were conducted with drospirenone and ethinyl estradiol tablets.
Pharmacokinetics
Absorption
The absolute bioavailability of DRSP from a single entity tablet is about 76%. The absolute bioavailability of EE is approximately 40% as a result of presystemic conjugation and first-pass metabolism. The absolute bioavailability of Drospirenone and Ethinyl Estradiol Tablets, which is a combination tablet of DRSP and EE, has not been evaluated. Serum concentrations of DRSP and EE reached peak levels within 1 to 2 hours after administration of Drospirenone and Ethinyl Estradiol Tablets.
The pharmacokinetics of DRSP are dose proportional following single doses ranging from 1 to 10 mg. Following daily dosing of Drospirenone and Ethinyl Estradiol Tablets, steady state DRSP concentrations were observed after 8 days. There was about 2 to 3 fold accumulation in serum Cmax and AUC (0-24h) values of DRSP following multiple dose administration of Drospirenone and Ethinyl Estradiol Tablets (see Table 3).
For EE, steady-state conditions are reported during the second half of a treatment cycle. Following daily administration of Drospirenone and Ethinyl Estradiol Tablets serum Cmax and AUC (0-24h) values of EE accumulate by a factor of about 1.5 to 2 (see Table 3).
Table 3: Mean Pharmacokinetic Parameters of Drospirenone and Ethinyl Estradiol Tablets (DRSP 3 mg and EE 0.03 mg )
DRSP Mean (%CV) Values | |||||
Cycle / Day | No. of Subjects | Cmax (ng/mL) | Tmax (h) | AUC(0-24h) (ng•h/mL) | t1/2 (h) |
| 1/1 | 12 | 36.9 (13) | 1.7 (47) | 288 (25) | NA |
| 1/21 | 12 | 87.5 (59) | 1.7 (20) | 827 (23) | 30.9 (44) |
| 6/21 | 12 | 84.2 (19) | 1.8 (19) | 930 (19) | 32.5 (38) |
| 9/21 | 12 | 81.3 (19) | 1.6 (38) | 957 (23) | 31.4 (39) |
| 13/21 | 12 | 78.7 (18) | 1.6 (26) | 968 (24) | 31.1 (36) |
EE Mean (%CV) Values | |||||
| Cycle / Day | No. of Subjects | Cmax (pg/mL) | Tmax (h) | AUC(0-24h) (pg•h/mL) | t1/2 (h) |
| 1/1 | 11 | 53.5 (43) | 1.9 (45) | 280 (87) | NA |
| 1/21 | 11 | 92.1 (35) | 1.5 (40) | 461 (94) | NA |
| 6/21 | 11 | 99.1 (45) | 1.5 (47) | 346 (74) | NA |
9/21 | 11 | 87 (43) | 1.5 (42) | 485 (92) | NA |
| 13/21 | 10 | 90.5 (45) | 1.6 (38) | 469 (83) | NA |
NA – Not available
Food Effect
The rate of absorption of DRSP and EE following single administration of a formulation similar to Drospirenone and Ethinyl Estradiol Tablets was slower under fed (high fat meal) conditions with the serum Cmax being reduced about 40% for both components. The extent of absorption of DRSP, however, remained unchanged. In contrast, the extent of absorption of EE was reduced by about 20% under fed conditions.
Distribution
DRSP and EE serum concentrations decline in two phases. The apparent volume of distribution of DRSP is approximately 4 L/kg and that of EE is reported to be approximately 4 to 5 L/kg.
DRSP does not bind to sex hormone binding globulin (SHBG) or corticosteroid binding globulin (CBG) but binds about 97% to other serum proteins. Multiple dosing over 3 cycles resulted in no change in the free fraction (as measured at trough concentrations). EE is reported to be highly but non-specifically bound to serum albumin (approximately 98.5 %) and induces an increase in the serum concentrations of both SHBG and CBG. EE induced effects on SHBG and CBG were not affected by variation of the DRSP dosage in the range of 2 to 3 mg.
Metabolism
The two main metabolites of DRSP found in human plasma were identified to be the acid form of DRSP generated by opening of the lactone ring and the 4,5-dihydrodrospirenone-3-sulfate, formed by reduction and subsequent sulfation. These metabolites were shown not to be pharmacologically active. Drospirenone is also subject to oxidative metabolism catalyzed by CYP3A4.
EE has been reported to be subject to significant gut and hepatic first-pass metabolism. Metabolism of EE and its oxidative metabolites occur primarily by conjugation with glucuronide or sulfate. CYP3A4 in the liver is responsible for the 2-hydroxylation which is the major oxidative reaction. The 2-hydroxy metabolite is further transformed by methylation and glucuronidation prior to urinary and fecal excretion.
Excretion
DRSP serum concentrations are characterized by a terminal disposition phase half-life of approximately 30 hours after both single and multiple dose regimens. Excretion of DRSP was nearly complete after ten days and amounts excreted were slightly higher in feces compared to urine. DRSP was extensively metabolized and only trace amounts of unchanged DRSP were excreted in urine and feces. At least 20 different metabolites were observed in urine and feces. About 38 to 47% of the metabolites in urine were glucuronide and sulfate conjugates. In feces, about 17 to 20% of the metabolites were excreted as glucuronides and sulfates.
For EE the terminal disposition phase half-life has been reported to be approximately 24 hours. EE is not excreted unchanged. EE is excreted in the urine and feces as glucuronide and sulfate conjugates and undergoes enterohepatic circulation.
Use in Specific Populations
Pediatric Use: Safety and efficacy of Drospirenone and Ethinyl Estradiol Tablets have been established in women of reproductive age. Efficacy is expected to be the same for postpubertal adolescents under the age of 18 and for users 18 years and older. Use of this product before menarche is not indicated.
Geriatric Use: Drospirenone and Ethinyl Estradiol Tablets have not been studied in postmenopausal women and is not indicated in this population.
Race: No clinically significant difference was observed between the pharmacokinetics of DRSP or EE in Japanese versus Caucasian women (age 25 to 35) when 3 mg DRSP/0.02 mg EE was administered daily for 21 days. Other ethnic groups have not been specifically studied.
Renal Impairment: Drospirenone and Ethinyl Estradiol Tablets is contraindicated in patients with renal impairment.
The effect of renal impairment on the pharmacokinetics of DRSP (3 mg daily for 14 days) and the effect of DRSP on serum potassium concentrations were investigated in three separate groups of female subjects (n=28, age 30 to 65). All subjects were on a low potassium diet. During the study, 7 subjects continued the use of potassium-sparing drugs for the treatment of their underlying illness. On the 14th day (steady-state) of DRSP treatment, the serum DRSP concentrations in the group with CLcr of 50 to 79 mL/min were comparable to those in the control group with CLcr ≥ 80 mL/min. The serum DRSP concentrations were on average 37% higher in the group with CLcr of 30 to 49 mL/min compared to those in the control group. DRSP treatment did not show any clinically significant effect on serum potassium concentration. Although hyperkalemia was not observed in the study, in five of the seven subjects who continued use of potassium sparing drugs during the study, mean serum potassium concentrations increased by up to 0.33 mEq/L. [See Contraindications (4) and Warnings and Precautions (5.2).]
Hepatic Impairment: Drospirenone and Ethinyl Estradiol Tablets is contraindicated in patients with hepatic disease.
The mean exposure to DRSP in women with moderate liver impairment is approximately three times higher than the exposure in women with normal liver function. Drospirenone and Ethinyl Estradiol Tablets have not been studied in women with severe hepatic impairment. [See Contraindications (4) and Warnings and Precautions (5.4).]
Drug Interactions
Consult the labeling of all concurrently used drugs to obtain further information about interactions with oral contraceptives or the potential for enzyme alterations.
Effects of Other Drugs on Combined Oral Contraceptives
Substances diminishing the efficacy of COCs: Drugs or herbal products that induce certain enzymes, including CYP3A4, may decrease the effectiveness of COCs or increase breakthrough bleeding.
Substances increasing the plasma concentrations of COCs: Co-administration of atorvastatin and certain COCs containing EE increase AUC values for EE by approximately 20%. Ascorbic acid and acetaminophen may increase plasma EE concentrations, possibly by inhibition of conjugation. In a clinical drug-drug interaction study conducted in 20 premenopausal women, co-administration of a DRSP (3 mg)/EE (0.02 mg) COC with the strong CYP3A4 inhibitor ketoconazole (200 mg twice daily) for 10 days increased the AUC(0-24h) of DRSP and EE by 2.68-fold (90% CI: 2.44, 2.95) and 1.40-fold (90% CI: 1.31, 1.49), respectively. The increases in Cmax were 1.97-fold (90% CI: 1.79, 2.17) and 1.39-fold (90% CI: 1.28, 1.52) for DRSP and EE, respectively. Although no clinically relevant effects on safety or laboratory parameters including serum potassium were observed, this study only assessed subjects for 10 days. The clinical impact for a patient taking a DRSP-containing COC concomitantly with chronic use of a CYP3A4/5 inhibitor is unknown [see Warnings and Precautions (5.2)].
HIV/HCV protease inhibitors and non-nucleoside reverse transcriptase inhibitors: Significant changes (increase or decrease) in the plasma concentrations of estrogen and progestin have been noted in some cases of co-administration with HIV/HCV protease inhibitors or with non-nucleoside reverse transcriptase inhibitors.
Antibiotics : There have been reports of pregnancy while taking hormonal contraceptives and antibiotics, but clinical pharmacokinetic studies have not shown consistent effects of antibiotics on plasma concentrations of synthetic steroids.
Effects of Combined Oral Contraceptives on Other Drugs
COCs containing EE may inhibit the metabolism of other compounds. COCs have been shown to significantly decrease plasma concentrations of lamotrigine, likely due to induction of lamotrigine glucuronidation. This may reduce seizure control; therefore, dosage adjustments of lamotrigine may be necessary. Consult the labeling of the concurrently-used drug to obtain further information about interactions with COCs or the potential for enzyme alterations.
In vitro, EE is a reversible inhibitor of CYP2C19, CYP1A1 and CYP1A2 as well as a mechanism-based inhibitor of CYP3A4/5, CYP2C8, and CYP2J2. Metabolism of DRSP and potential effects of DRSP on hepatic CYP enzymes have been investigated in in vitro and in vivo studies. In in vitro studies DRSP did not affect turnover of model substrates of CYP1A2 and CYP2D6, but had an inhibitory influence on the turnover of model substrates of CYP1A1, CYP2C9, CYP2C19, and CYP3A4, with CYP2C19 being the most sensitive enzyme. The potential effect of DRSP on CYP2C19 activity was investigated in a clinical pharmacokinetic study using omeprazole as a marker substrate. In the study with 24 postmenopausal women [including 12 women with homozygous (wild type) CYP2C19 genotype and 12 women with heterozygous CYP2C19 genotype] the daily oral administration of 3 mg DRSP for 14 days did not affect the oral clearance of omeprazole (40 mg, single oral dose) and the CYP2C19 product 5-hydroxy omeprazole. Furthermore, no significant effect of DRSP on the systemic clearance of the CYP3A4 product omeprazole sulfone was found. These results demonstrate that DRSP did not inhibit CYP2C19 and CYP3A4 in vivo.
Two additional clinical drug-drug interaction studies using simvastatin and midazolam as marker substrates for CYP3A4 were each performed in 24 healthy postmenopausal women. The results of these studies demonstrated that pharmacokinetics of the CYP3A4 substrates were not influenced by steady state DRSP concentrations achieved after administration of 3 mg DRSP/day.
Women on thyroid hormone replacement therapy may need increased doses of thyroid hormone because serum concentration of thyroid-binding globulin increases with use of COCs.
Interactions With Drugs That Have the Potential to Increase Serum Potassium Concentration: There is a potential for an increase in serum potassium concentration in women taking Drospirenone and Ethinyl Estradiol Tablets with other drugs that may increase serum potassium concentration [see Warnings and Precautions (5.2)].
A drug-drug interaction study of DRSP 3 mg/estradiol (E2) 1 mg versus placebo was performed in 24 mildly hypertensive postmenopausal women taking enalapril maleate 10 mg twice daily. Potassium concentrations were obtained every other day for a total of 2 weeks in all subjects. Mean serum potassium concentrations in the DRSP/E2 treatment group relative to baseline were 0.22 mEq/L higher than those in the placebo group. Serum potassium concentrations also were measured at multiple time points over 24 hours at baseline and on Day 14. On Day 14, the ratios for serum potassium Cmax and AUC in the DRSP/E2 group to those in the placebo group were 0.955 (90% CI: 0.914, 0.999) and 1.010 (90% CI: 0.944, 1.08), respectively. No patient in either treatment group developed hyperkalemia (serum potassium concentrations > 5.5 mEq/L).
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 24 month oral carcinogenicity study in mice dosed with 10 mg/kg/day DRSP alone or 1 + 0.01, 3 + 0.03 and 10 + 0.1 mg/kg/day of DRSP and EE, 0.1 to 2 times the exposure (AUC of DRSP) of women taking a contraceptive dose, there was an increase in carcinomas of the harderian gland in the group that received the high dose of DRSP alone. In a similar study in rats given 10 mg/kg/day DRSP alone or 0.3 + 0.003, 3 + 0.03 and 10 + 0.1 mg/kg/day DRSP and EE, 0.8 to 10 times the exposure of women taking a contraceptive dose, there was an increased incidence of benign and total (benign and malignant) adrenal gland pheochromocytomas in the group receiving the high dose of DRSP. Mutagenesis studies for DRSP were conducted in vivo and in vitro and no evidence of mutagenic activity was observed.[See Warnings and Precautions (5.3, 5.4)].
CLINICAL STUDIES
In the clinical efficacy studies of up to 2 years duration, 2,629 subjects completed 33,160 cycles of use without any other contraception. The mean age of the subjects was 25.5 ± 4.7 years. The age range was 16 to 37 years. The racial demographic was: 83% Caucasian, 1% Hispanic, 1% Black, <1% Asian, <1% other, <1% missing data, 14% not inquired and <1% unspecified. Pregnancy rates in the clinical trials were less than one per 100 woman-years of use.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Drospirenone and Ethinyl Estradiol Tablets, USP 3 mg/0.03 mg are available in packages of three blister packs (NDC 79929-019-05). Each blister pack contains 28 film coated tablets in the following order: • 21 Yellow, round, biconvex film coated tablets, debossed with ‘EV1’ on one side and plain on other side containing 3 mg drospirenone and 0.03 mg ethinyl estradiol • 7 White to off white, round, biconvex, film coated tablets, debossed with ‘EV2’ on one side and plain on other side.
Storage
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
MECHANISM OF ACTION
COCs lower the risk of becoming pregnant primarily by suppressing ovulation.