Get your patient on Fluocinonide - Fluocinonide solution (Fluocinonide)
Fluocinonide - Fluocinonide solution prescribing information
INDICATIONS AND USAGE
Fluocinonide topical solution USP, 0.05% is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
DOSAGE AND ADMINISTRATION
Fluocinonide topical solution USP, 0.05% is generally applied to the affected area as a thin film from two to four times daily depending on the severity of the condition.
Occlusive dressings may be used for the management of psoriasis or recalcitrant conditions.
If an infection develops, the use of occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted.
CONTRAINDICATIONS
Topical corticosteroids are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
ADVERSE REACTIONS:
The following local adverse reactions are reported infrequently with topical corticosteroids, but may occur more frequently with the use of occlusive dressings. These reactions are listed in an approximate decreasing order of occurrence:
Burning Itching Irritation Dryness Folliculitis Hypertrichosis Acneiform eruptions Hypopigmentation Perioral dermatitis Allergic contact dermatitis Maceration of the skin Secondary infection Skin atrophy Striae Miliaria
To report SUSPECTED ADVERSE REACTIONS, contact Quagen Pharmaceuticals LLC. at 1-888-344-9603 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch for voluntary reporting of adverse reactions.
DESCRIPTION
Fluocinonide topical solution USP, 0.05% is intended for topical administration. The active component is the corticosteroid fluocinonide, which is the 21-acetate ester of fluocinolone acetonide and has the chemical name pregna-1,4-diene-3,20-dione,21-(acetyloxy)-6,9-difluoro-11-hydroxy-16,17-[(1-methylethylidene)bis(oxy)]-,(6α,11β,16α)-. It has the following chemical structure:
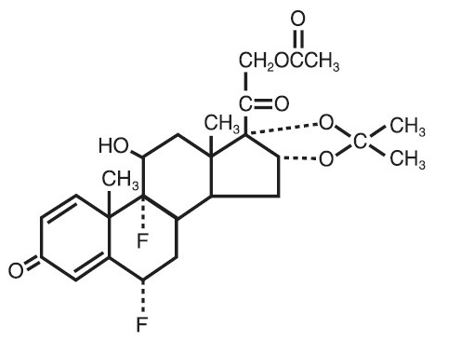
Fluocinonide topical solution USP, 0.05% contains fluocinonide 0.5 mg/mL in a clear, colorless to pale yellow solution of alcohol (35%), citric acid anhydrous, diisopropyl adipate, and propylene glycol.
CLINICAL PHARMACOLOGY
Topical corticosteroids share anti-inflammatory, anti-pruritic and vasoconstrictive actions. The mechanism of anti-inflammatory activity of the topical corticosteroids is unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. A significantly greater amount of fluocinonide is absorbed from the solution than from the cream or gel formulations.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. Thus, occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses. (See DOSAGE AND ADMINISTRATION .)
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
HOW SUPPLIED
Fluocinonide Topical Solution USP, 0.05% is a clear, colorless to pale yellow solution supplied in plastic squeeze bottle:
60 mL - NDC 70752-154-05
Tamper Evident - Do not use if shrink seal is broken or missing
Store at 20°-25°C (68°-77°F); excursions permitted to 15°-30°C (59°-86°F). See USP Controlled Room Temperature. Avoid excess heat, above 40°C (104°F).
Manufactured by: Quagen Pharmaceuticals LLC West Caldwell, NJ 07006
52037 Rev. 04/25