Get your patient on Lidocaine Hydrochloride And Dextrose - Lidocaine Hydrochloride Anhydrous And Dextrose Monohydrate injection, Solution (Lidocaine Hydrochloride Anhydrous And Dextrose Monohydrate)
Lidocaine Hydrochloride And Dextrose - Lidocaine Hydrochloride Anhydrous And Dextrose Monohydrate injection, Solution prescribing information
INDICATIONS AND USAGE
Lidocaine hydrochloride administered intravenously is specifically indicated in the acute management of (1) ventricular arrhythmias occurring during cardiac manipulations, such as cardiac surgery and (2) life-threatening arrhythmias which are ventricular in origin, such as occur during acute myocardial infarction.
DOSAGE AND ADMINISTRATION
Therapy of ventricular arrhythmias is often initiated with a single IV bolus of 1 mg/kg of Lidocaine Hydrochloride Injection USP. Following acute treatment by bolus in patients in whom arrhythmias tend to recur and who are incapable of receiving oral antiarrhythmic agents, intravenous infusion of Lidocaine Hydrochloride and 5% Dextrose Injection USP is administered continuously.
Rate of Administration
Adults (20 to 50 mcg/kg/min):
| Average 70 kg adult | |||
|---|---|---|---|
| mg/min | mL/hr | mL/min | |
| 0.4% Lidocaine Hydrochloride and 5% Dextrose Injection USP (4 mg lidocaine hydrochloride/mL) | 1–4 | 15–60 | 0.25–1.0 |
| 0.8% Lidocaine Hydrochloride and 5% Dextrose Injection USP (8 mg lidocaine hydrochloride/mL) | 1–4 | 7.5–30 | 0.12–0.5 |
Pediatric Patients (30 mcg/kg/min). Standards and Guidelines for Cardiopulmonary Resuscitation (CPR) and Emergency Cardiac Care (ECC). American Heart Association, JAMA 244 (5):453–509, 1980.
Pharmacokinetic data indicate reduced elimination of lidocaine after prolonged infusion (24 hours) with resultant prolongation of the half-life to approximately three times that seen following a single administration. Failure to adjust the rate of infusion in keeping with this altered ability to eliminate lidocaine may result in toxic accumulation of the drug in the patient's serum. LeLorier J, Grenon D, Latour Y, et al.: Pharmacokinetics of lidocaine after prolonged intravenous infusions in uncomplicated myocardial infarction. Ann Int Med 87:700–702, 1977.
Intravenous infusions of lidocaine hydrochloride must be administered under constant ECG monitoring to avoid potential overdosage and toxicity. Intravenous infusion should be terminated as soon as the patient's basic cardiac rhythm appears to be stable or at the earliest signs of toxicity. It should rarely be necessary to continue intravenous infusions beyond 24 hours. As soon as possible and when indicated, patients should be changed to an oral antiarrhythmic agent for maintenance therapy.
Caution: Concentrated solutions of lidocaine hydrochloride (greater than 0.2%) should be administered by carefully calibrated infusion devices.
Pediatric Use
Therapy should be initiated with a single IV bolus of 1 mg/kg of Lidocaine Hydrochloride Injection USP. A maintenance intravenous infusion of Lidocaine Hydrochloride and 5% Dextrose Injection USP administered at a recommended infusion rate of 30 mcg/kg/min may be given.
Geriatric Use
Patients with reduced hepatic function or diminished hepatic blood flow (as in heart failure and after cardiac surgery), or those over 70 years of age should receive half the usual loading dose and also should be given lower maintenance levels of intravenous lidocaine. Patients over 65 years may benefit from dosing based upon body weight (see CLINICAL PHARMACOLOGY and PRECAUTIONS, Geriatric Use ).
Lidocaine hydrochloride should not be added to blood transfusion assemblies.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
Lidocaine hydrochloride is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type.
Lidocaine should not be used in patients with Stokes-Adams syndrome, Wolff-Parkinson-White syndrome, or with severe degrees of sinoatrial, atrioventricular, or intraventricular block.
Solutions containing dextrose may be contraindicated in patients with known allergy to corn or corn products.
ADVERSE REACTIONS
Systemic reactions of the following types have been reported:
- Central Nervous System: Light-headedness; drowsiness; dizziness; apprehension; euphoria; tinnitus; blurred or double vision; nausea and vomiting; sensation of heat, cold or numbness; twitching; tremors; convulsions; unconsciousness; respiratory depression and arrest.
- Cardiovascular System: Hypotension; cardiovascular arrest; and bradycardia which may lead to cardiac arrest.
- Hematologic Effects: methemoglobinemia
- Allergic reactions, including anaphylactic reactions, may occur but are infrequent. There have been no reports of cross-sensitivity between lidocaine hydrochloride and procainamide or between lidocaine hydrochloride and quinidine.
Drug Interactions
Lidocaine should be used with caution in patients with digitalis toxicity accompanied by atrioventricular block (see CONTRAINDICATIONS ).
Coadministration of propranolol or cimetidine with lidocaine has been reported to reduce clearance from the plasma and may result in toxic accumulation of the drug (see CLINICAL PHARMACOLOGY ).
When lidocaine is administered with other antiarrhythmic drugs such as phenytoin, procainamide, propranolol, amiodarone, or quinidine, the cardiac effects may be additive or antagonistic and toxic effects may be additive. Phenytoin may stimulate the hepatic metabolism of lidocaine, but the clinical significance of this effect is not known.
DESCRIPTION
Lidocaine Hydrochloride and 5% Dextrose Injection USP is a sterile, nonpyrogenic solution prepared from lidocaine hydrochloride and dextrose in water for injection.
Lidocaine hydrochloride is designated chemically as 2-(Diethylamino)-2',6'-acetoxylidide monohydrochloride. The solution serves as a cardiac antiarrhythmic agent intended for intravenous use.
| Solution | Lidocaine Hydrochloride Anhydrous USP | Hydrous Dextrose USP | pH | Calculated Osmolarity mOsmol/liter |
|---|---|---|---|---|
| 0.4% Lidocaine HCl and 5% Dextrose Injection USP | 0.4 g | 5 g | 4.4 (3.0–7.0) | 280 |
| 0.8% Lidocaine HCl and 5% Dextrose Injection USP | 0.8 g | 5 g | 4.2 (3.0–7.0) | 305 |
| Water for Injection USP qs |
The formulas of the active ingredients are:
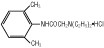 | 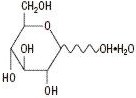 |
| Lidocaine Hydrochloride Anhydrous USP (M.W. 270.80) | Hydrous Dextrose USP (M.W. 198.17) |
Not made with natural rubber latex, PVC or DEHP.
The plastic container is made from a multilayered film specifically developed for parenteral drugs. It contains no plasticizers and exhibits virtually no leachables. The solution contact layer is a rubberized copolymer of ethylene and propylene. The container is nontoxic and biologically inert. The container-solution unit is a closed system and is not dependent upon entry of external air during administration. The container is overwrapped to provide protection from the physical environment and to provide an additional moisture barrier when necessary.
The closure system has two ports; the one for the administration set has a tamper evident plastic protector. Refer to the Directions for Use of the container.
CLINICAL PHARMACOLOGY
Lidocaine hydrochloride exerts an antiarrhythmic effect by increasing the electric stimulation threshold of the ventricle during diastole. In usual therapeutic doses, lidocaine hydrochloride produces no change in myocardial contractility, in systemic arterial pressure, or in absolute refractory period.
About 90% of an administered dose of the drug is metabolized in the liver. The remaining 10% is excreted unchanged via the kidneys.
Lidocaine toxicity is related to systemic blood levels. The decreased clearance and longer half-life of lidocaine should be taken into consideration with prolonged (24 hour) infusions. Constant rate of infusion may result in toxic accumulation of lidocaine. Infusion should be reduced to approximately one-half to compensate for decreased rate of clearance and concomitant or prior administration of propranolol may further increase blood concentrations by as much as 30% in patients without cardiac or hepatic failure. In clinical studies, patients over 65 years showed decreased lidocaine clearance. This was partly due to the tendency of elderly patients to have lower body weight and the increased risk of cardiac failure in these patients.
This solution provides approximately 170 calories per liter.
HOW SUPPLIED
Lidocaine Hydrochloride and 5% Dextrose Injection USP is supplied sterile and nonpyrogenic in Full Fill 500 mL and 250 mL EXCEL® Containers packaged 24 per case.
| Fill | NDC | REF | Solution |
|---|---|---|---|
| 2 g Lidocaine Hydrochloride: | |||
| 500 mL | 0264-9594-10 | P5941 | 0.4% Lidocaine Hydrochloride |
| and 5% Dextrose Injection USP | |||
| 250 mL | 0264-9598-20 | P5982 | 0.8% Lidocaine Hydrochloride |
| and 5% Dextrose Injection USP | |||
| 1 g Lidocaine Hydrochloride: | |||
| 250 mL | 0264-9594-20 | P5942 | 0.4% Lidocaine Hydrochloride |
| and 5% Dextrose Injection USP | |||
Exposure of pharmaceutical products to heat should be minimized. Avoid excessive heat. Protect from freezing. It is recommended that the product be stored at room temperature (25°C); however, brief exposure up to 40°C does not adversely affect the product.
Storage in automated dispensing machines: Brief exposure up to 2 weeks to ultraviolet or fluorescent light does not adversely affect the product labeling legibility; prolonged exposure can cause fading of the red label. Rotate stock frequently.