Lorazepam - Lorazepam tablet prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of these drugs for patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS and PRECAUTIONS ).
- The use of benzodiazepines, including lorazepam, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing lorazepam and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (see WARNINGS ).
- The continued use of benzodiazepines, including lorazepam, may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of lorazepam after continued use may precipitate acute withdrawal reactions, which can be life-threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue lorazepam or reduce the dosage (DOSAGE AND ADMINISTRATION and WARNINGS ).
INDICATIONS AND USAGE
Lorazepam tablets areindicated for the management of anxietydisorders or for theshort-termrelief of the symptoms of anxiety or anxietyassociatedwithdepressivesymptoms.Anxiety or tensionassociatedwith the stress of everyday life usuallydoesnotrequiretreatmentwith an anxiolytic.
The effectiveness of lorazepam tablets in long-termuse,thatis,morethan 4 months,has not been assessed by systematicclinicalstudies. The physicianshouldperiodicallyreassess the usefulness of the drug for the individualpatient.
DOSAGE AND ADMINISTRATION
Lorazepam tablets are administeredorally. For optimalresults,dose,frequency of administration,andduration of therapyshould be individualizedaccording to patientresponse. To facilitatethis,0.5mg, 1 mg,and 2 mgtabletsareavailable.
The usualrange is 2 to 6 mg/daygivenindivideddoses, the largestdosebeingtakenbeforebedtime, but the dailydosagemayvaryfrom 1 to 10 mg/day.
For anxiety,mostpatientsrequireaninitialdose of 2 to 3 mg/daygiventwotimes a day or threetimes a day.
For insomnia due to anxiety or transientsituationalstress, a singledailydose of 2 to 4 mgmay be given,usuallyatbedtime.
For elderly or debilitatedpatients,aninitialdosage of 1 to 2 mg/day in divideddoses is recommended, to be adjustedasneededandtolerated.
The dosageoflorazepam tablets should be increasedgraduallywhenneeded to helpavoidadverse effects.Whenhigherdosage is indicated, the eveningdoseshouldbeincreasedbefore the daytimedoses.
Discontinuation or Dosage Reduction of Lorazepam Tablets
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue lorazepam tablets or reduce the dosage. If a patient develops withdrawal reactions, consider pausing the taper or increasing the dosage to the previous tapered dosage level. Subsequently decrease the dosage more slowly (see WARNINGS: Dependence and Withdrawal Reactions and DRUG ABUSE AND DEPENDENCE: Dependence ).
CONTRAINDICATIONS
Lorazepam is contraindicated in patientswith:
- hypersensitivity to benzodiazepines or to anycomponents of the formulation
- acutenarrow-angleglaucoma.
ADVERSE REACTIONS
Mostadversereactions to benzodiazepines,includingCNSeffectsandrespiratorydepression,aredose dependent,withmoresevereeffectsoccurringwithhighdoses.
In a sample of about3500patientstreated for anxiety, the mostfrequentadversereaction to lorazepam wassedation(15.9%),followedbydizziness(6.9%),weakness(4.2%),andunsteadiness(3.4%). The incidence of sedationandunsteadinessincreasedwithage.
Otheradversereactions to benzodiazepines,includinglorazepam arefatigue,drowsiness,amnesia,memoryimpairment,confusion,disorientation,depression,unmasking of depression,disinhibition,euphoria,suicidalideation/attempt,ataxia,asthenia,extrapyramidalsymptoms,convulsions/seizures,tremor,vertigo,eyefunction/visualdisturbance(includingdiplopiaandblurredvision),dysarthria/slurred speech,change in libido,impotence,decreasedorgasm;headache,coma;respiratorydepression,apnea,worsening of sleepapnea,worsening of obstructivepulmonarydisease;gastrointestinalsymptoms includingnausea,change in appetite,constipation,jaundice,increase in bilirubin,increase in liver transaminases,increase in alkalinephosphatase;hypersensitivityreactions,anaphylactoidreactions;dermatologicalsymptoms,allergicskinreactions,alopecia;syndrome of inappropriateantidiuretic hormone(SIADH),hyponatremia;thrombocytopenia,agranulocytosis,pancytopenia;hypothermia;andautonomicmanifestations.
Paradoxicalreactions,includinganxiety,excitation,agitation,hostility,aggression,rage,sleepdisturbances/insomnia,sexualarousal,andhallucinationsmayoccur.Small decreases in blood pressure andhypotensionmayoccur but areusually not clinicallysignificant,probablybeingrelated to therelief of anxietyproducedbylorazepam. To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
The concomitant use of benzodiazepines and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration. Benzodiazepines interact at GABA A sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of benzodiazepines and opioids, and monitor patients closely for respiratory depression and sedation.
The benzodiazepines, including lorazepam, produce increased CNS-depressant effects when administered with other CNS depressants such as alcohol, barbiturates, antipsychotics, sedative/hypnotics, anxiolytics, antidepressants, narcotic analgesics, sedative antihistamines, anticonvulsants, and anesthetics.
Concomitant use of clozapine and lorazepam may produce marked sedation, excessive salivation, hypotension, ataxia, delirium, and respiratory arrest.
Concurrent administration of lorazepam with valproate results in increased plasma concentrations and reduced clearance of lorazepam. Lorazepam dosage should be reduced to approximately 50% when coadministered with valproate.
Concurrent administration of lorazepam with probenecid may result in a more rapid onset or prolonged effect of lorazepam due to increased half-life and decreased total clearance. Lorazepam dosage needs to be reduced by approximately 50% when coadministered with probenecid.
The effects of probenecid and valproate on lorazepam may be due to inhibition of glucuronidation.
Administration of theophylline or aminophylline may reduce the sedative effects of benzodiazepines, including lorazepam.
DESCRIPTION
Lorazepam USP,anantianxietyagent,has the chemicalformula,7-chloro-5-( o -chlorophenyl)-1,3-dihydro-3-hydroxy-2 H -1,4-benzodiazepin-2-one: 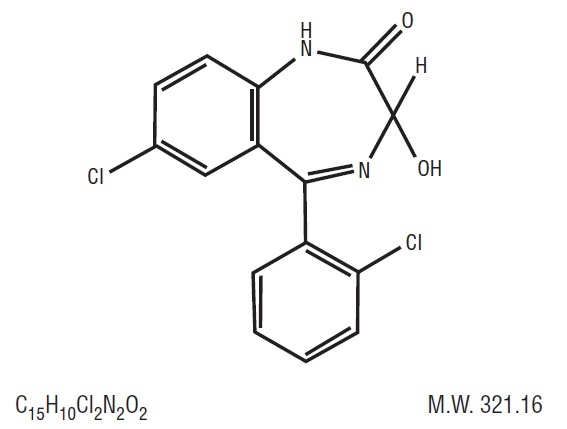 It is a nearlywhitepowderalmostinsoluble in water.Eachlorazepam tablet, USP to be takenorally,contains 0.5 mg, 1 mg, or 2 mg of lorazepam, USP. The inactiveingredientspresent are anhydrous lactose, magnesium stearate, microcrystalline cellulose, and polacrilin potassium.
It is a nearlywhitepowderalmostinsoluble in water.Eachlorazepam tablet, USP to be takenorally,contains 0.5 mg, 1 mg, or 2 mg of lorazepam, USP. The inactiveingredientspresent are anhydrous lactose, magnesium stearate, microcrystalline cellulose, and polacrilin potassium.
CLINICAL PHARMACOLOGY
Studies in healthyvolunteersshowthat in singlehighdoseslorazepam has a tranquilizingaction on the centralnervoussystemwith no appreciableeffect on the respiratory or cardiovascularsystems.
Lorazepam is readilyabsorbedwithanabsolutebioavailability of 90%.Peakconcentrations in plasmaoccurapproximately 2 hoursfollowingadministration. The peakplasmalevel of lorazepam from a 2 mgdose is approximately 20 ng/mL.
The meanhalf-life of unconjugatedlorazepam inhumanplasma is about 12 hoursand for its majormetabolite,lorazepam glucuronide,about 18 hours.Atclinicallyrelevantconcentrations,lorazepam isapproximately 85% bound to plasmaproteins.Lorazepam is rapidlyconjugatedatits3-hydroxy group into lorazepam glucuronidewhich is thenexcreted in the urine. Lorazepam glucuronidehas no demonstrablecentralnervoussystem(CNS)activityinanimals.
The plasmalevels of lorazepam areproportionaltothedosegiven. There is no evidence of accumulation of lorazepam onadministrationup to 6 months.
Studiescomparing young andelderlysubjectshaveshownthatadvancingagedoes not have a significanteffect on the pharmacokinetics of lorazepam.However, in one studyinvolvingsingleintravenousdoses of 1.5 to 3 mg of lorazepam Injection,meantotalbodyclearance of lorazepam decreasedby20% in 15 elderlysubjects of 60 to 84 years of agecompared to that in 15youngersubjects of 19 to 38 years of age.
HOW SUPPLIED
Lorazepam Tablets, USP are available in the following dosage strengths:
0.5 mg, white to off-white, round, flat-faced beveled edge tablets debossed with ‘U32’ on one side and plain on the other side.
Bottles of 10 tablets NDC 13107-083-11
Bottles of 100 tablets NDC 13107-083-01
Bottles of 500 tablets NDC 13107-083-05
Bottles of 1000 tablets NDC 13107-083-99
1 mg, white to off-white, round, flat-faced beveled edge tablets debossed with ‘U33’ on one side and bisect on the other side.
Bottles of 10 tablets NDC 13107-084-11
Bottles of 100 tablets NDC 13107-084-01
Bottles of 500 tablets NDC 13107-084-05
Bottles of 1000 tablets NDC 13107-084-99
2 mg, white to off-white, round, flat-faced beveled edge tablets debossed with a bisect separating “U” and “34” on one side and “2” on other side.
Bottles of 10 tablets NDC 13107-085-11
Bottles of 100 tablets NDC 13107-085-01
Bottles of 500 tablets NDC 13107-085-05
Bottles of 1000 tablets NDC 13107-085-99
Keep bottles tightly closed.
Keep out of reach of children. Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as described in the USP. Dispense with Medication Guide available at: www.aurobindousa.com/medication-guides Distributed by: Aurobindo Pharma USA, Inc. 279 Princeton-Hightstown Road East Windsor, NJ 08520 Manufactured by: Aurobindo Pharma Limited Hyderabad-500 032, India Revised: 06/2023 Dispense with Medication Guide available at: www.aurobindousa.com/medication-guides