Get your patient on Methimazole - Methimazole tablet (Methimazole)
Methimazole - Methimazole tablet prescribing information
INDICATIONS AND USAGE
Methimazole tablets are indicated:
- In patients with Graves' disease with hyperthyroidism or toxic multinodular goiter for whom surgery or radioactive iodine therapy is not an appropriate treatment option.
- To ameliorate symptoms of hyperthyroidism in preparation for thyroidectomy or radioactive iodine therapy.
DOSAGE AND ADMINISTRATION
Methimazole tablets are administered orally. The total daily dosage is usually given in 3 divided doses at approximately 8-hour intervals.
Adult - The initial daily dosage is 15 mg for mild hyperthyroidism, 30 to 40 mg for moderately severe hyperthyroidism, and 60 mg for severe hyperthyroidism, divided into 3 doses at 8-hour intervals. The maintenance dosage is 5 to 15 mg daily.
Pediatric - Initially, the daily dosage is 0.4 mg/kg of body weight divided into 3 doses and given at 8-hour intervals. The maintenance dosage is approximately 1/2 of the initial dose.
CONTRAINDICATIONS
Methimazole tablets are contraindicated in the presence of hypersensitivity to the drug or any of the other product components.
ADVERSE REACTIONS
Major adverse reactions (which occur with much less frequency than the minor adverse reactions) include inhibition of myelopoiesis (agranulocytosis, granulocytopenia, thrombocytopenia, and aplastic anemia), drug fever, a lupus-like syndrome, insulin autoimmune syndrome (which can result in hypoglycemic coma), hepatitis (jaundice may persist for several weeks after discontinuation of the drug), periarteritis, and hypoprothrombinemia. Nephritis occurs very rarely. There have been postmarketing case reports of acute pancreatitis.
There are reports of a vasculitis, often associated with the presence of anti-neutrophilic cytoplasmic antibodies (ANCA), resulting in severe complications (see WARNINGS ).
Minor adverse reactions include skin rash, urticaria, nausea, vomiting, epigastric distress, arthralgia, paresthesia, loss of taste, abnormal loss of hair, myalgia, headache, pruritus, drowsiness, neuritis, edema, vertigo, skin pigmentation, jaundice, sialadenopathy, and lymphadenopathy.
Anticoagulants (oral):
Due to potential inhibition of vitamin K activity by methimazole, the activity of oral anticoagulants (e.g., warfarin) may be increased; additional monitoring of PT/INR should be considered, especially before surgical procedures.
β-adrenergic blocking agents:
Hyperthyroidism may cause an increased clearance of beta blockers with a high extraction ratio. A dose reduction of beta-adrenergic blockers may be needed when a hyperthyroid patient becomes euthyroid.
Digitalis glycosides:
Serum digitalis levels may be increased when hyperthyroid patients on a stable digitalis glycoside regimen become euthyroid; a reduced dosage of digitalis glycosides may be needed.
Theophylline:
Theophylline clearance may decrease when hyperthyroid patients on a stable theophylline regimen become euthyroid; a reduced dose of theophylline may be needed.
DESCRIPTION
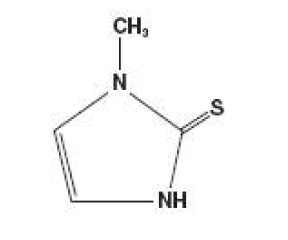
Methimazole (1-methylimidazole-2-thiol) is a white, crystalline substance that is freely soluble in water. It differs chemically from the drugs of the thiouracil series primarily because it has a 5- instead of a 6-membered ring.
Methimazole tablet, USP contains 5 or 10 mg (43.8 or 87.6 μmol) methimazole, an orally administered antithyroid drug.
Each tablet also contains lactose monohydrate, magnesium stearate, pregelatinized starch and talc.
The molecular weight is 114.16, and the molecular formula is C 4 H 6 N 2 S. The structural formula is as follows:
CLINICAL PHARMACOLOGY
Methimazole inhibits the synthesis of thyroid hormones and thus is effective in the treatment of hyperthyroidism. The drug does not inactivate existing thyroxine and tri-iodothyronine that are stored in the thyroid or circulating in the blood nor does it interfere with the effectiveness of thyroid hormones given by mouth or by injection.
Methimazole is readily absorbed in the gastrointestinal tract, metabolized in the liver, and excreted in the urine.
HOW SUPPLIED
Methimazole tablets, USP 5 mg - round, white to off white bi-convex tablet. Debossed HP bisect 70 on one side and plain on the reverse side.
They are available in:
Bottles of 100 NDC 72603-315-01
Methimazole tablets, USP 10 mg - round, white to off white bi-convex tablet. Debossed HP bisect 71 on one side and plain on the reverse side.
They are available in:
Bottles of 100 NDC 72603-316-01
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
Dispense in tight, light-resistant container.
Manufactured by:
Avet Pharmaceuticals Labs Inc.
East Brunswick, NJ 08816
Manufactured for:
Northstar Rx LLC.
Memphis, TN 38141
Toll-Free 1-800-206-7821
51U190000003US01
Revised: 05/2024