Get your patient on Miebo (Perfluorohexyloctane)
Miebo prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Patient education
Administration guides
Patient education materials
Dosing resources
Clinical information
Insurance resources
Prior authorization & coverage support
Financial assistance & copay programs
Specialty pharmacy coordination
Other resources
Dosage & administration
DOSAGE AND ADMINISTRATION
Instill one drop of MIEBO four times daily into each eye. (2.1 )
Recommended Dosage
Instill one drop of MIEBO four times daily into affected eye(s).
Contact lenses should be removed prior to and for at least 30 minutes after the administration of MIEBO.
Administration Instructions
Step 1. Remove the cap from eye drop bottle.
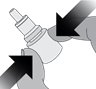 Step 2. Holding the bottle upright, gently squeeze the bottle.
Step 2. Holding the bottle upright, gently squeeze the bottle.
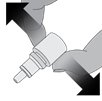 Step 3. While squeezing, turn the bottle upside down and release the pressure (drawing air into the bottle).
Step 3. While squeezing, turn the bottle upside down and release the pressure (drawing air into the bottle).
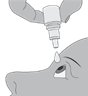 Step 4. Keeping the bottle upside down, place the bottle above your eye and squeeze it again to release a drop into your eye.
Step 4. Keeping the bottle upside down, place the bottle above your eye and squeeze it again to release a drop into your eye.
Repeat steps 1 - 4 for the second affected eye.

Miebo prescribing information
Contraindications, Hypersensitivity (4.1 ) 10/2025
INDICATIONS AND USAGE
MIEBO ® (perfluorohexyloctane ophthalmic solution) is indicated for the treatment of the signs and symptoms of dry eye disease (DED).
DOSAGE AND ADMINISTRATION
Instill one drop of MIEBO four times daily into each eye. (2.1 )
Recommended Dosage
Instill one drop of MIEBO four times daily into affected eye(s).
Contact lenses should be removed prior to and for at least 30 minutes after the administration of MIEBO.
Administration Instructions
Step 1. Remove the cap from eye drop bottle.
 Step 2. Holding the bottle upright, gently squeeze the bottle.
Step 2. Holding the bottle upright, gently squeeze the bottle.
 Step 3. While squeezing, turn the bottle upside down and release the pressure (drawing air into the bottle).
Step 3. While squeezing, turn the bottle upside down and release the pressure (drawing air into the bottle).
 Step 4. Keeping the bottle upside down, place the bottle above your eye and squeeze it again to release a drop into your eye.
Step 4. Keeping the bottle upside down, place the bottle above your eye and squeeze it again to release a drop into your eye.
Repeat steps 1 - 4 for the second affected eye.
DOSAGE FORMS AND STRENGTHS
MIEBO (perfluorohexyloctane ophthalmic solution) is a sterile, clear and colorless ophthalmic solution containing 100% perfluorohexyloctane.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no adequate and well controlled studies with MIEBO in pregnant women.
In animal reproduction studies with oral administration of perfluorohexyloctane during the period of organogenesis, no adverse maternal or developmental effects were observed in rats at doses up to 162 times the recommended human ophthalmic dose (RHOD) ( see Data ). Maternal toxicity, miscarriages and reduced fetal weights were observed in rabbits at all doses tested, with the lowest dose as 41 times the RHOD.
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects is 2 to 4%, and of miscarriage is 15 to 20%, of clinically recognized pregnancies.
Data
Animal Data
An embryofetal study was conducted in pregnant rabbits administered perfluorohexyloctane by oral gavage on gestation days 6 to 19, to target the period of organogenesis. Perfluorohexyloctane produced maternal toxicity, characterized by reduced body weight gain and food consumption, and miscarriages at all doses tested, with the lowest dose as ≥ 250 mg/kg/day (41 times the RHOD based on body surface area). Reduced fetal weights were also observed at ≥ 250 mg/kg/day but no fetal mortality or malformations. A no observed adverse effect level (NOAEL) for maternal toxicity was not established in rabbits.
An embryofetal study was conducted in pregnant rats administered perfluorohexyloctane by oral gavage on gestation days 6 to 17, to target the period of organogenesis. There was no evidence of embryofetal toxicity or teratogenicity at doses up to 2,000 mg/kg/day (162 times the RHOD).
Lactation
There are no data on the presence of perfluorohexyloctane in human milk, the effects on the breastfed infant, or the effects on milk production. The lack of clinical data during lactation precludes a clear determination of the risk of MIEBO to an infant during lactation; however, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for MIEBO.
Pediatric Use
The safety and effectiveness of MIEBO in pediatric patients below the age of 18 years have not been established.
Geriatric Use
No overall differences in safety and effectiveness have been observed between elderly and younger patients.
WARNINGS AND PRECAUTIONS
Use with Contact Lenses
MIEBO should not be administered while wearing contact lenses. Advise patients that contact lenses should be removed prior to and for at least 30 minutes after administration of MIEBO.
ADVERSE REACTIONS
Most common ocular adverse reaction was blurred vision. Blurred vision was reported in less than 4% of individuals. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In patients with DED, 614 patients received at least one dose of MIEBO in two randomized controlled clinical trials across 68 sites in the United States. The most common ocular adverse reaction was blurred vision. Blurred vision and conjunctival redness were reported in 1-3% of individuals.
In four premarketing studies (three open-label [n=127], one randomized [n=24 treated with at least one dose of perfluorohexyloctane]) the most common adverse reaction was hypersensitivity.
DESCRIPTION
MIEBO ® (perfluorohexyloctane ophthalmic solution) is a sterile, clear and colorless liquid containing 100% perfluorohexyloctane, for topical ophthalmic use.
The active ingredient is 1,1,1,2,2,3,3,4,4,5,5,6,6-tridecafluorotetradecane and is a semifluorinated alkane. It has a molecular formula of C 14 H 17 F 13 and a molecular weight of 432.26 g/mol. The chemical structure is:
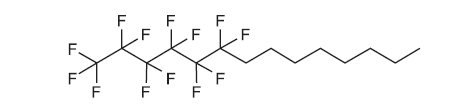
Perfluorohexyloctane is practically immiscible with water. It is miscible with ethanol and most
organic solvents. Each multiple-dose bottle contains 3 mL of perfluorohexyloctane, 1.338 g/mL as a clear and colorless liquid.
CLINICAL PHARMACOLOGY
Mechanism of Action
Perfluorohexyloctane, a semifluorinated alkane, contains 6 perfluorinated carbon atoms and 8 hydrogenated carbon atoms. Perfluorohexyloctane forms a monolayer at the air-liquid interface of the tear film which can be expected to reduce evaporation. The exact mechanism of action for MIEBO in DED is not known.
Pharmacokinetics
The pharmacokinetics of perfluorohexyloctane following topical ocular administration of MIEBO has not been quantitatively characterized in humans. A single pharmacokinetic (PK) study was conducted that showed low systemic perfluorohexyloctane blood levels after topical ocular administration. Perfluorohexyloctane was not metabolized by human liver microsomes in vitro.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been conducted to evaluate the carcinogenic potential of perfluorohexyloctane.
Perfluorohexyloctane was not mutagenic or clastogenic in a standard battery of genotoxicity tests, including a bacterial mutagenicity assay (Ames assay), an in vitro chromosome aberration assay using human peripheral lymphocytes, and an in vivo bone marrow micronucleus assay in rats.
CLINICAL STUDIES
In two randomized, multicenter, double-masked, saline-controlled trials (GOBI and MOJAVE), a total of 1,217 patients with a history of DED and clinical signs of meibomian gland dysfunction were randomized to MIEBO or saline 0.6% (1:1 ratio) to evaluate safety and efficacy after receiving MIEBO four times daily (QID) for 57 days.The mean age of the 614 patients who received MIEBO was 57 years (range, 19-87 years). The majority of patients were female (76%).
Effects on Signs of Dry Eye Disease
Total corneal fluorescein staining (tCFS) was recorded at each study visit using a standardized grading system of 0-3 for each of the five areas on the cornea (inferior, superior, central, nasal, and temporal), totaling a maximum tCFS score for each eye of 15. The average baseline tCFS was approximately 6.7 in GOBI and 7.0 in MOJAVE. At Days 15 and 57, a statistically significant reduction in tCFS favoring MIEBO was observed in both studies (Figure 1).
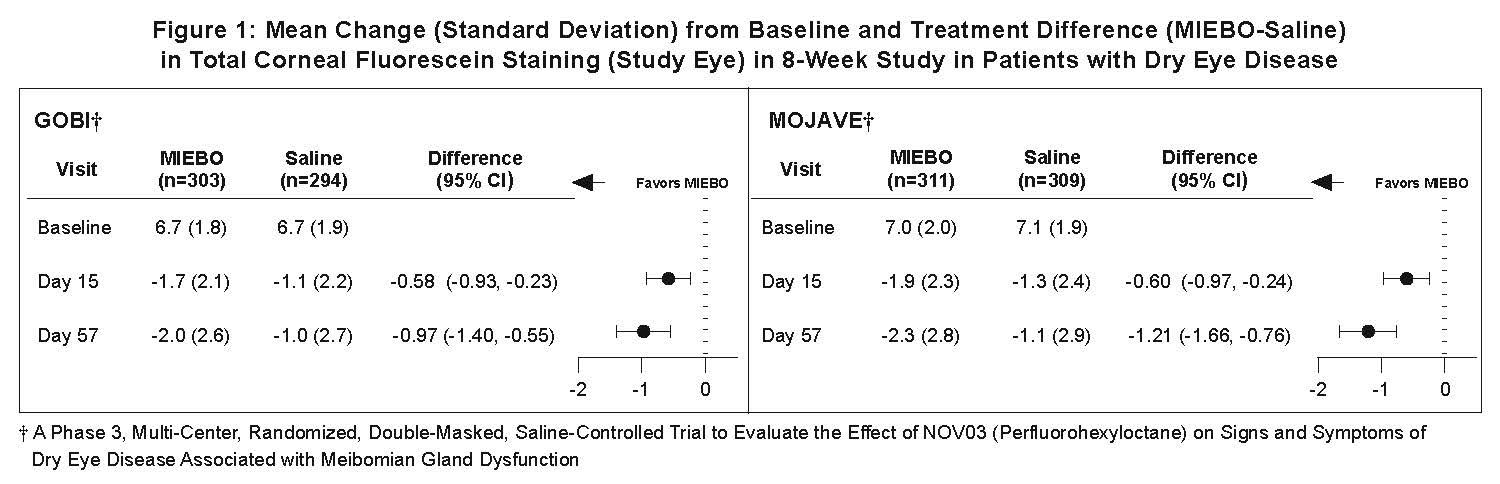
Effects on Symptoms of Dry Eye Disease
Eye dryness score was rated by patients using a visual analogue scale (VAS) (0=no discomfort, 100=maximal discomfort) at each study visit. The baseline VAS eye dryness average score was approximately 67 in GOBI and 65 in MOJAVE. At Days 15 and 57, a statistically significant reduction in VAS eye dryness score favoring MIEBO was observed in both studies (Figure 2).
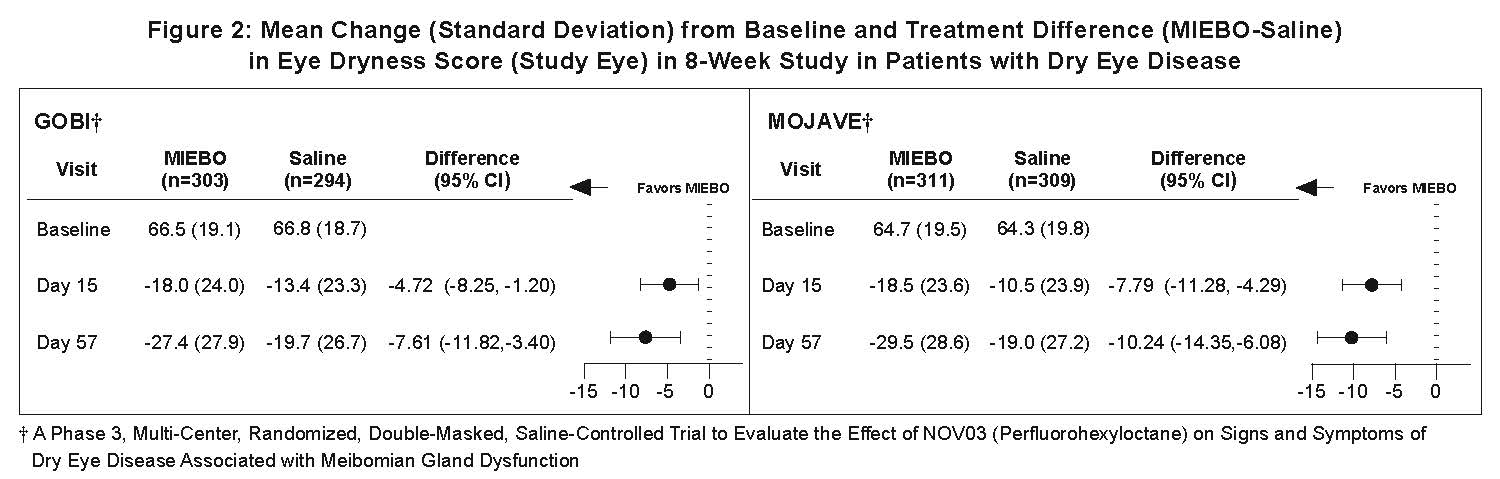
HOW SUPPLIED/STORAGE AND HANDLING
MIEBO ® (perfluorohexyloctane ophthalmic solution) is supplied as a sterile, clear and colorless liquid in multiple-dose 5 mL polypropylene bottles with dropper tips and screw caps, packaged in a carton - NDC 24208-377-05.
Storage
Store MIEBO at 15ºC to 25ºC (59ºF to 77ºF). After opening, MIEBO can be used until the expiration date on the bottle.
Mechanism of Action
Perfluorohexyloctane, a semifluorinated alkane, contains 6 perfluorinated carbon atoms and 8 hydrogenated carbon atoms. Perfluorohexyloctane forms a monolayer at the air-liquid interface of the tear film which can be expected to reduce evaporation. The exact mechanism of action for MIEBO in DED is not known.