Get your patient on Primidone - Primidone tablet (Primidone)
Primidone - Primidone tablet prescribing information
INDICATIONS AND USAGE
Primidone tablets, used alone or concomitantly with other anticonvulsants, is indicated in the control of grand mal, psychomotor, and focal epileptic seizures. It may control grand mal seizures refractory to other anticonvulsant therapy.
DOSAGE AND ADMINISTRATION
Usual Dosage
Patients 8 years of age and older who have received no previous treatment may be started on primidone tablets according to the following regimen using either 50 mg or scored 250 mg primidone tablets:
Days 1 to 3: 100 to 125 mg at bedtime.
Days 4 to 6: 100 to 125 mg twice a day.
Days 7 to 9: 100 to 125 mg three times a day.
Day 10 to maintenance: 250 mg three times a day.
For most adults and children 8 years of age and over, the usual maintenance dosage is three to four 250 mg primidone tablets in divided doses (250 mg three times a day or four times a day). If required, an increase to five or six 250 mg tablets daily may be made, but daily doses should not exceed 500 mg four times a day.
Dosage should be individualized to provide maximum benefit. In some cases, serum blood level determinations of primidone may be necessary for optimal dosage adjustment. The clinically effective serum level for primidone is between 5 to 12 mcg/mL.
INITIAL: ADULTS AND CHILDREN OVER 8 | ||||||
KEY: •=50 mg tablet; ●=250 mg tablet | ||||||
DAY | 1 | 2 | 3 | 4 | 5 | 6 |
AM NOON PM | •• | •• | •• | •• •• | •• •• | •• •• |
DAY | 7 | 8 | 9 | 10 | 11 | 12 |
AM NOON PM | •• •• •• | •• •• •• | •• •• •• | Adjust to Maintenance | ||
Patients Already Receiving Other Anticonvulsants
Primidone tablets should be started at 100 to 125 mg at bedtime and gradually increased to maintenance level as the other drug is gradually decreased. This regimen should be continued until satisfactory dosage level is achieved for the combination, or the other medication is completely withdrawn. When therapy with primidone tablets alone is the objective, the transition from concomitant therapy should not be completed in less than 2 weeks.
Pediatric Dosage
For children under 8 years of age, the following regimen may be used:
Days 1 to 3: 50 mg at bedtime.
Days 4 to 6: 50 mg twice a day.
Days 7 to 9: 100 mg twice a day.
Day 10 to maintenance: 125 mg three times a day to 250 mg three times a day.
For children under 8 years of age, the usual maintenance dosage is 125 to 250 mg three times daily or, 10 to 25 mg/kg/day in divided doses.
CONTRAINDICATIONS
Primidone is contraindicated in: 1) patients with porphyria and 2) patients who are hypersensitive to phenobarbital (see ACTIONS ).
ADVERSE REACTIONS
The most frequently occurring early side effects are ataxia and vertigo. These tend to disappear with continued therapy, or with reduction of initial dosage. Occasionally, the following have been reported: nausea, anorexia, vomiting, fatigue, hyperirritability, emotional disturbances, sexual impotency, diplopia, nystagmus, drowsiness, and morbilliform skin eruptions. Granulocytopenia, agranulocytosis, and red-cell hypoplasia and aplasia, have been reported rarely. These and, occasionally, other persistent or severe side effects may necessitate withdrawal of the drug. Megaloblastic anemia may occur as a rare idiosyncrasy to primidone and to other anticonvulsants. The anemia responds to folic acid without necessity of discontinuing medication.
To report SUSPECTED ADVERSE REACTIONS, contact Carnegie Pharmaceuticals LLC at 1-732-783-7010 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Chemical name: 5-ethyldihydro-5-phenyl-4,6 (1H, 5H)-pyrimidinedione. Structural formula:
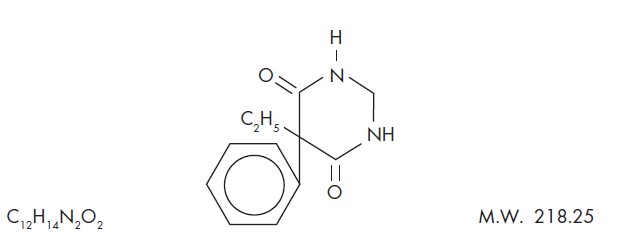
Primidone is a white, crystalline, highly stable substance, M.P. 279-284°C. It is poorly soluble in water (60 mg per 100 mL at 37°C) and in most organic solvents. It possesses no acidic properties, in contrast to its barbiturate analog.
Primidone Tablets, USP 50 mg and 250 mg contain the following inactive ingredients: lactose monohydrate, NF; magnesium stearate, NF; methyl cellulose, USP; microcrystalline cellulose, NF; purified water, USP; sodium lauryl sulfate, NF; sodium starch glycolate, NF; talc, USP
HOW SUPPLIED
Primidone Tablets, USP
50 mg: White, round, flat-faced, bevel-edged tablets, debossed “CP” above “11” on one side and bisected on the other side, in bottles of 100 (NDC 80005-117-11), 500 (NDC 80005-117-14) and 1000 (NDC 80005-117-15).
250 mg: White round, flat-faced, bevel-edged tablets, debossed “CP” bisect “118” on one side and plain on the other side, in bottles of 100 (NDC 80005-118-11), 500 ( NDC 80005-118-14) and 1000 ( NDC 80005-118-15).
Storage
Store at 20° to 25°C (68° to 77°F)
[see USP Controlled Room Temperature].
Dispense in a tight, light-resistant container with a child-resistant closure.
Manufactured and Distributed by:
Carnegie Pharmaceuticals LLC
Delran, NJ 08075, USA
Rev. 06/2024