Get your patient on Radicava (Edaravone)
Radicava prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Radicava patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
- RADICAVA: The recommended dosage is 60 mg administered as an intravenous infusion over 60 minutes (2.1 )
- RADICAVA ORS: The recommended dosage is 105 mg (5 mL) taken orally or via feeding tube in the morning after overnight fasting. Food should not be consumed for 1 hour after administration except water (2.1 , 2.3 )
- For RADICAVA and RADICAVA ORS:
Dosage Information
The recommended dosage of RADICAVA and RADICAVA ORS is as follows:
- RADICAVA: an intravenous infusion of 60 mg administered over a 60-minute period
- RADICAVA ORS: 105 mg (5 mL) taken orally or via feeding tube in the morning after overnight fasting [see Dosage and Administration (2.3) ]
Administer RADICAVA or RADICAVA ORS according to the following schedule:
- An initial treatment cycle with daily dosing for 14 days, followed by a 14-day drug-free period
- Subsequent treatment cycles with daily dosing for 10 days out of 14-day periods, followed by 14-day drug-free periods
Preparation and Administration Information for RADICAVA Injection
RADICAVA is for intravenous infusion only.
Preparation
Do not use if the oxygen indicator has turned blue or purple before opening the package . Once the overwrap package is opened, use within 24 hours [see How Supplied/Storage and Handling (16.1 , 16.2) ].
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Administration
Administer each 60 mg dose of RADICAVA injection as two consecutive 30 mg intravenous infusion bags over a total of 60 minutes (infusion rate approximately 1 mg per minute [3.33 mL per minute]).
Promptly discontinue the infusion upon the first observation of any signs or symptoms consistent with a hypersensitivity reaction [see Warnings and Precautions (5.1 , 5.2) ] .
Other medications should not be injected into the infusion bag or mixed with RADICAVA.
Preparation and Administration Information for RADICAVA ORS Oral Suspension
See the Instruction for Use for further preparation and administration details.
Preparation
Prior to opening the bottle, turn it upside down (invert) and shake vigorously up and down for at least 30 seconds.
Administration
RADICAVA ORS can be administered by mouth or via feeding tube (see Feeding Tube Administration ) .
RADICAVA ORS should be taken in the morning on an empty stomach after overnight fasting. Food should not be consumed for 1 hour after administration except water [see Clinical Pharmacology (12.3) ] . See Table 1 for specific fasting conditions.
| Type of food/caloric supplement consumed | Fasting time before and after RADICAVA ORS dose administration with regards to meal type |
|---|---|
| High-fat meal (800-1,000 calories, 50% fat) | 8 hours before administration and one hour after administration |
| Low-fat meal (400-500 calories, 25% fat) | 4 hours before administration and one hour after administration |
| Caloric supplement (250 calories, e.g., protein drink) | 2 hours before administration and one hour after administration |
Administer RADICAVA ORS using a 5 mL oral syringe that comes with the product. A household teaspoon is not an adequate measuring device.
Dispose of any RADICAVA ORS that is not used within 15 days after opening the bottle or within the 30 days from the date of shipment indicated on the carton pharmacy label, which ever happens first.
Feeding Tube Administration
- Nasogastric (NG) tubes or percutaneous endoscopic gastrostomy (PEG) tubes made of silicone, polyvinyl chloride (PVC), or polyurethane can be used
- Before and after administration, use a catheter-tip syringe to flush the tube with at least 1 ounce (30 mL) of water
Switching from RADICAVA to RADICAVA ORS
Patients treated with 60 mg of RADICAVA intravenous infusion may be switched to 105 mg (5 mL) RADICAVA ORS using the same dosing frequency. Upon switching to RADICAVA ORS, patients should follow RADICAVA ORS dosing recommendations with regards to food consumption [see Dosage and Administration (2.3) ] .

Radicava prescribing information
INDICATIONS AND USAGE
RADICAVA and RADICAVA ORS are indicated for the treatment of amyotrophic lateral sclerosis (ALS).
DOSAGE AND ADMINISTRATION
- RADICAVA: The recommended dosage is 60 mg administered as an intravenous infusion over 60 minutes (2.1 )
- RADICAVA ORS: The recommended dosage is 105 mg (5 mL) taken orally or via feeding tube in the morning after overnight fasting. Food should not be consumed for 1 hour after administration except water (2.1 , 2.3 )
- For RADICAVA and RADICAVA ORS:
Dosage Information
The recommended dosage of RADICAVA and RADICAVA ORS is as follows:
- RADICAVA: an intravenous infusion of 60 mg administered over a 60-minute period
- RADICAVA ORS: 105 mg (5 mL) taken orally or via feeding tube in the morning after overnight fasting [see Dosage and Administration (2.3) ]
Administer RADICAVA or RADICAVA ORS according to the following schedule:
- An initial treatment cycle with daily dosing for 14 days, followed by a 14-day drug-free period
- Subsequent treatment cycles with daily dosing for 10 days out of 14-day periods, followed by 14-day drug-free periods
Preparation and Administration Information for RADICAVA Injection
RADICAVA is for intravenous infusion only.
Preparation
Do not use if the oxygen indicator has turned blue or purple before opening the package . Once the overwrap package is opened, use within 24 hours [see How Supplied/Storage and Handling (16.1 , 16.2) ].
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Administration
Administer each 60 mg dose of RADICAVA injection as two consecutive 30 mg intravenous infusion bags over a total of 60 minutes (infusion rate approximately 1 mg per minute [3.33 mL per minute]).
Promptly discontinue the infusion upon the first observation of any signs or symptoms consistent with a hypersensitivity reaction [see Warnings and Precautions (5.1 , 5.2) ] .
Other medications should not be injected into the infusion bag or mixed with RADICAVA.
Preparation and Administration Information for RADICAVA ORS Oral Suspension
See the Instruction for Use for further preparation and administration details.
Preparation
Prior to opening the bottle, turn it upside down (invert) and shake vigorously up and down for at least 30 seconds.
Administration
RADICAVA ORS can be administered by mouth or via feeding tube (see Feeding Tube Administration ) .
RADICAVA ORS should be taken in the morning on an empty stomach after overnight fasting. Food should not be consumed for 1 hour after administration except water [see Clinical Pharmacology (12.3) ] . See Table 1 for specific fasting conditions.
| Type of food/caloric supplement consumed | Fasting time before and after RADICAVA ORS dose administration with regards to meal type |
|---|---|
| High-fat meal (800-1,000 calories, 50% fat) | 8 hours before administration and one hour after administration |
| Low-fat meal (400-500 calories, 25% fat) | 4 hours before administration and one hour after administration |
| Caloric supplement (250 calories, e.g., protein drink) | 2 hours before administration and one hour after administration |
Administer RADICAVA ORS using a 5 mL oral syringe that comes with the product. A household teaspoon is not an adequate measuring device.
Dispose of any RADICAVA ORS that is not used within 15 days after opening the bottle or within the 30 days from the date of shipment indicated on the carton pharmacy label, which ever happens first.
Feeding Tube Administration
- Nasogastric (NG) tubes or percutaneous endoscopic gastrostomy (PEG) tubes made of silicone, polyvinyl chloride (PVC), or polyurethane can be used
- Before and after administration, use a catheter-tip syringe to flush the tube with at least 1 ounce (30 mL) of water
Switching from RADICAVA to RADICAVA ORS
Patients treated with 60 mg of RADICAVA intravenous infusion may be switched to 105 mg (5 mL) RADICAVA ORS using the same dosing frequency. Upon switching to RADICAVA ORS, patients should follow RADICAVA ORS dosing recommendations with regards to food consumption [see Dosage and Administration (2.3) ] .
DOSAGE FORMS AND STRENGTHS
RADICAVA is supplied for intravenous infusion in a single-dose polypropylene bag containing 30 mg of edaravone in 100 mL of clear, colorless aqueous solution.
RADICAVA ORS is supplied as an oral suspension in a multi-dose amber glass bottle 105 mg/5 mL of white to off-white color.
USE IN SPECIFIC POPULATIONS
Pregnancy: Based on animal data, may cause fetal harm. (8.1 )
Pregnancy
Risk Summary
There are no adequate data on the developmental risk associated with the use of RADICAVA or RADICAVA ORS in pregnant women. In animal studies, administration of edaravone to pregnant rats and rabbits resulted in adverse developmental effects (increased mortality, decreased growth, delayed sexual development, and altered behavior) at clinically relevant doses. Most of these effects occurred at doses that were also associated with maternal toxicity (see Animal Data ).
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively. The background risk for major birth defects and miscarriage in patients with ALS is unknown.
Data
Animal Data
In rats, intravenous administration of edaravone (0, 3, 30, or 300 mg/kg/day) throughout the period of organogenesis resulted in reduced fetal weight at all doses. In dams allowed to deliver naturally, offspring weight was reduced at the highest dose tested. Maternal toxicity was also observed at the highest dose tested. There were no adverse effects on reproductive function in the offspring. A no-effect dose for embryofetal developmental toxicity was not identified; the low dose is less than the recommended human dose of 60 mg for RADICAVA on a body surface area (mg/m 2 ) basis.
In rabbits, intravenous administration of edaravone (0, 3, 20, or 100 mg/kg/day) throughout the period of organogenesis resulted in embryofetal death at the highest dose tested, which was associated with maternal toxicity. The higher no-effect dose for embryofetal developmental toxicity is approximately 6 times the recommended human dose (RHD) for RADICAVA on a body surface area (mg/m 2 ) basis.
The effects on offspring of edaravone (0, 3, 20, or 200 mg/kg/day), administered by intravenous injection to rats from GD 17 throughout lactation, were assessed in two studies. In the first study, offspring mortality was observed at the high dose and increased activity was observed at the mid and high doses. In the second study, there was an increase in stillbirths, offspring mortality, and delayed physical development (vaginal opening) at the highest dose tested. Reproduction function in offspring was not affected in either study. Maternal toxicity was evident in both studies at all but the lowest dose tested. The no-effect dose for developmental toxicity (3 mg/kg/day) is less than the RHD on a mg/m 2 basis.
Reproductive and developmental toxicology studies of edaravone using the oral route have not been conducted.
Lactation
Risk Summary
There are no data on the presence of edaravone in human milk, the effects on the breastfed infant, or the effects of the drug on milk production. Edaravone and its metabolites are excreted in the milk of lactating rats. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for RADICAVA and RADICAVA ORS and any potential adverse effects on the breastfed infant from RADICAVA and RADICAVA ORS or from the underlying maternal condition.
Pediatric Use
Safety and effectiveness of RADICAVA or RADICAVA ORS in pediatric patients have not been established.
Geriatric Use
Of the 184 patients with ALS who received RADICAVA in 3 placebo-controlled clinical trials, a total of 53 patients were 65 years of age and older, including 2 patients 75 years of age and older. No overall differences in safety or effectiveness were observed between these patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
CONTRAINDICATIONS
RADICAVA and RADICAVA ORS are contraindicated in patients with a history of hypersensitivity to edaravone or any of the inactive ingredients in this product. Hypersensitivity reactions and anaphylactic reactions have occurred [see Warnings and Precautions (5.1 , 5.2) ].
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions
Hypersensitivity reactions (redness, wheals, and erythema multiforme) and cases of anaphylaxis (urticaria, decreased blood pressure, and dyspnea) have been reported in spontaneous postmarketing reports with RADICAVA.
Patients should be monitored carefully for hypersensitivity reactions. If hypersensitivity reactions occur, discontinue RADICAVA and/or RADICAVA ORS, treat per standard of care, and monitor until the condition resolves [see Contraindications (4) ].
Sulfite Allergic Reactions
RADICAVA and RADICAVA ORS contain sodium bisulfite, a sulfite that may cause allergic type reactions, including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown. Sulfite sensitivity occurs more frequently in asthmatic than non-asthmatic people.
ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1) ]
- Sulfite Allergic Reactions [see Warnings and Precautions (5.2) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In randomized, placebo-controlled trials, 184 patients with ALS were administered RADICAVA 60 mg in treatment cycles for 6 months. The population consisted of Japanese patients who had a median age of 60 years (range 29-75) and were 59% male. Most (93%) of these patients were living independently at the time of screening.
Most Common Adverse Reactions Observed During Clinical Studies
Table 2 lists the adverse reactions that occurred in ≥2% of patients in the RADICAVA-treated group and that occurred at least 2% more frequently than in the placebo-treated group in randomized placebo-controlled ALS trials. The most common adverse reactions that occurred in ≥10% of RADICAVA-treated patients were contusion, gait disturbance, and headache.
| Adverse Reaction | RADICAVA (N=184) % | Placebo (N=184) % |
|---|---|---|
| Contusion | 15 | 9 |
| Gait disturbance | 13 | 9 |
| Headache | 10 | 6 |
| Dermatitis | 8 | 5 |
| Eczema | 7 | 4 |
| Respiratory failure, respiratory disorder, hypoxia | 6 | 4 |
| Glycosuria | 4 | 2 |
| Tinea infection | 4 | 2 |
Additional Adverse Reactions with RADICAVA ORS
In an open-label study in patients with ALS (n=185) treated with RADICAVA ORS for 6 months, fatigue was observed in 7.6% of patients.
Postmarketing Experience
The following adverse reactions have been identified during postapproval use of RADICAVA. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders: Hypersensitivity reactions and anaphylaxis [see Warnings and Precautions (5.1 , 5.2) ].
DESCRIPTION
The active ingredient in RADICAVA and RADICAVA ORS is edaravone, which is a member of the substituted 2-pyrazolin-5-one class. The chemical name of edaravone is [3-methyl-1-phenyl-2-pyrazolin-5-one]. The molecular formula is C 10 H 10 N 2 O and the molecular weight is 174.20.
The chemical structure is:

Edaravone is a white crystalline powder with a melting point of 129.7°C. It is freely soluble in acetic acid, methanol, or ethanol and slightly soluble in water or diethyl ether.
RADICAVA injection is a clear, colorless liquid provided as a sterile solution.
RADICAVA injection is supplied for intravenous infusion in a polypropylene bag containing 30 mg edaravone in 100 mL isotonic, sterile, aqueous solution, which is further overwrapped with polyvinyl alcohol (PVA) secondary packaging. The overwrapped package also contains an oxygen absorber and oxygen indicator to minimize oxidation. Each bag contains the following inactive ingredients: L-cysteine hydrochloride hydrate (10 mg), sodium bisulfite (20 mg). Sodium chloride is added for isotonicity and phosphoric acid and sodium hydroxide are added to adjust to pH 4.
RADICAVA ORS (edaravone) oral suspension is a white to off-white color, opaque, homogenous suspension containing 105 mg of edaravone per 5 mL of suspension.
RADICAVA ORS contains the following inactive ingredients: L-cysteine hydrochloride hydrate, polyvinyl alcohol, simethicone emulsion, sodium bisulfite, sorbitol, and xanthan gum. Phosphoric acid and sodium hydroxide are added to adjust to pH 4.
CLINICAL PHARMACOLOGY
Mechanism of Action
The mechanism by which RADICAVA and RADICAVA ORS exert their therapeutic effect in patients with ALS is unknown.
Pharmacodynamics
Cardiac Electrophysiology
At exposures at least 5 times higher than that of the recommended doses of RADICAVA and RADICAVA ORS, edaravone does not prolong the QT interval to any clinically relevant extent.
Pharmacokinetics
RADICAVA is administered by IV infusion. The maximum plasma concentration (C max ) of edaravone was reached by the end of infusion. There was a trend of more than dose-proportional increase in area under the concentration-time curve (AUC) and C max of edaravone. With multiple-dose administration, edaravone does not accumulate in plasma.
RADICAVA ORS administered orally at the dose of 105 mg, under fasted conditions, was shown to demonstrate an equivalent AUC with RADICAVA (60 mg administered intravenously for 60 min) and C max not less than that of RADICAVA. RADICAVA ORS demonstrated similar pharmacokinetics following administration via feeding tubes and oral administration.
In healthy subjects, RADICAVA ORS does not accumulate with once-daily administration, and the maximum plasma concentration (C max ) and area under the concentration-time curve (AUC) of edaravone is more than dose-proportional over the dose range of 30 to 300 mg (approximately 0.3 and 3 times the recommended dosage, respectively).
Absorption
RADICAVA ORS has a median time to maximum concentration (T max ) occurring at approximately 0.5 hour (range, 0.25 to 0.75 hours) after fasted oral administration. The absolute oral bioavailability is about 57% because of first-pass effect when comparing RADICAVA ORS (105 mg oral suspension) and RADICAVA (60 mg intravenous formulation).
Effect of Food
Following oral administration of RADICAVA ORS to healthy subjects 1 hour before or 8 hours after high-fat meals (800-1000 calories, 50% fat), 4 hours after low-fat meals (400-500 calories, 25% fat), or 2 hours after caloric supplement (250 calories, e.g., protein drink), the C max and AUC did not decrease significantly (less than 20% and 10% changes in C max and AUC, respectively) [see Dosage and Administration (2.3) ] .
Following oral administration of RADICAVA ORS to healthy subjects, the C max decreased by 82%, and the AUC decreased by 61% with a high-fat meal compared to fasted conditions. Following administration of RADICAVA ORS 4 hours after high-fat meals, the C max and AUC decreased by 44% and 24%, respectively.
Following administration of RADICAVA ORS 2 hours after low-fat meals, the C max and AUC decreased by 45% and 21%, respectively.
Distribution
Edaravone is bound to human serum proteins (92%), mainly to albumin, with no concentration dependence in the range of 0.1 to 50 micromol/L. Edaravone has a mean volume of distribution after intravenous administration of 63.1 L.
Elimination
The mean terminal elimination half-life of edaravone is approximately 4.5 to 9 hours. The half-lives of its metabolites are 3 to 6 hours. Following intravenous administration, the total clearance of edaravone is estimated to be 35.9 L/h.
Metabolism
Edaravone is metabolized to a sulfate conjugate and a glucuronide conjugate, which are not pharmacologically active. The glucuronide conjugation of edaravone involves multiple uridine diphosphate glucuronosyltransferase (UGT) isoforms (UGT1A1, UGT1A6, UGT1A7, UGT1A8, UGT1A9, UGT1A10, UGT2B7, and UGT2B17). In human plasma, edaravone is mainly detected as the sulfate conjugate, which is presumed to be formed by sulfotransferases. RADICAVA ORS results in 1.3- and 1.7-fold higher exposures for both sulfate and glucuronide metabolites, respectively, when compared to RADICAVA because of first-pass metabolism.
Excretion
In Japanese and Caucasian healthy volunteer studies, edaravone was excreted mainly in the urine as its glucuronide conjugate (60-80% of the dose up to 48 hours). Approximately 6-8% of the dose was recovered in the urine as the sulfate conjugate, and < 1% of the dose was recovered in the urine as the unchanged drug. In vitro studies suggest that the sulfate conjugate of edaravone is hydrolyzed back to edaravone, which is then converted to the glucuronide conjugate in the kidney before excretion into the urine.
Specific Populations
Geriatric Patients
No age effect on edaravone pharmacokinetics has been found [see Use in Specific Populations (8.5) ].
Patients with Renal Impairment
Following single IV infusion of 30 mg edaravone (half the recommended dosage of RADICAVA) over 60 minutes, mean C max and AUC 0-∞ of unchanged edaravone were 1.15- and 1.20-fold greater in the subjects with mild renal impairment (eGFR 60-89 mL/min/1.73m 2 ), and were 1.25- and 1.29-fold greater in the subjects with moderate renal impairment (eGFR 30-59 mL/min/1.73m 2 ) when compared to subjects with normal renal function, respectively. These changes in exposures are not considered to be clinically significant and therefore no dosage adjustments are necessary in patients with mild to moderate renal impairment. The effects of severe renal impairment on the pharmacokinetics of edaravone have not been studied.
Patients with Hepatic Impairment
Following single IV infusion of 30 mg edaravone (half of the recommended dose of RADICAVA) over 60 minutes, mean C max and AUC 0-∞ of unchanged edaravone were 1.20- and 1.07-fold greater in the subjects with mild hepatic impairment (Child-Pugh score 5 or 6), were 1.24- and 1.14-fold greater in the subjects with moderate hepatic impairment (Child-Pugh score 7 to 9), and were 1.20- and 1.19-fold greater in the subjects with severe hepatic impairment (Child-Pugh score 10 to 14) when compared to subjects with normal hepatic function, respectively. These changes in exposures are not considered to be clinically significant and therefore no dosage adjustments are necessary in patients with hepatic impairment.
Male and Female Patients
No gender effect on edaravone pharmacokinetics has been found.
Racial or Ethnic Groups
There were no significant racial differences in C max and AUC of edaravone between Japanese and Caucasian subjects.
Drug Interaction Studies
The pharmacokinetics of edaravone is not expected to be significantly affected by inhibitors of cytochrome P450 (CYP) enzymes, UGTs, or major transporters. Concomitant oral administration of edaravone 120 mg (higher than the recommended dose of 105 mg for ORS) with sildenafil (CYP3A4 substrate), rosuvastatin (BCRP substrate), and furosemide (OAT3 substrate) did not produce any changes in C max and AUC of these drugs.
In vitro studies demonstrated that, at the recommended dosage, edaravone and its metabolites are not expected to significantly inhibit CYP enzymes (CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A), conjugating enzymes UGT1A1 and UGT2B7, or other transporters (P-gp, OATP1B1, OATP1B3, OAT1, OCT2, MATE1, and MATE2-K) in humans. Edaravone and its metabolites are not expected to induce CYP1A2, or CYP2B6, at the recommended dosage of edaravone.
In vitro data indicated that edaravone was not a substrate of OATP1B1 or OATP1B3.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In a 26-week carcinogenicity study in male and female transgenic (Tg rasH2) mice, oral administration of edaravone (0 [water control], 0 [vehicle control], 100, 150, or 350 mg/kg) resulted in no increase in tumors.
In a 2-year carcinogenicity study in rats, oral administration of edaravone (0, 50, 100, or 200 mg/kg in males; 0, 50, 100, or 250 mg/kg in females) resulted in no increase in tumors.
Carcinogenicity studies of edaravone using the intravenous route have not been conducted.
Mutagenesis
Edaravone was negative in in vitro (bacterial reverse mutation and Chinese hamster lung chromosomal aberration) and in vivo (mouse micronucleus) assays.
Impairment of Fertility
Intravenous administration of edaravone (0, 3, 20, or 200 mg/kg) prior to and throughout mating in male and female rats and continuing in females to gestation day 7 had no effect on fertility; however, disruption of the estrus cycle and mating behavior was observed at the highest dose tested. No effects on reproductive function were observed at the lower doses, which are up to approximately 3 times the RHD for RADICAVA (60 mg) on a body surface area (mg/m 2 ) basis.
Fertility studies of edaravone using the oral route have not been conducted.
Animal Toxicology and/or Pharmacology
Oral administration of edaravone (0, 10, 30, 100, or 300 mg/kg/day) in dogs for 39 weeks resulted in neurotoxicity, characterized by white matter vacuolation in the spinal cord and vacuolation and nerve fiber atrophy in the sciatic nerve, at the two highest doses tested. The microscopic findings were accompanied by gait abnormalities, loss of patellar reflex, and inability to rise. Plasma edaravone exposures (C max and AUC) at the higher no-effect dose for neurotoxicity (30 mg/kg/day) were approximately 2 times those in humans at the recommended human dose of RADICAVA ORS (105 mg).
CLINICAL STUDIES
The efficacy of RADICAVA ORS is based on a bioavailability study comparing RADICAVA and RADICAVA ORS [see Clinical Pharmacology (12.3) ] .
The efficacy of RADICAVA for the treatment of ALS was established in a 6-month, randomized, placebo- controlled, double-blind study conducted in Japanese patients with ALS who were living independently and met the following criteria at screening:
- Functionality retained most activities of daily living (defined as scores of 2 points or better on each individual item of the ALS Functional Rating Scale-Revised [ALSFRS-R; described below])
- Normal respiratory function (defined as percent-predicted forced vital capacity values of [%FVC] ≥80%)
- Definite or Probable ALS based on El Escorial revised criteria
- Disease duration of 2 years or less
The study enrolled 69 patients in the RADICAVA arm and 68 in the placebo arm. Baseline characteristics were similar between these groups, with over 90% of patients in each group being treated with riluzole.
RADICAVA was administered as an intravenous infusion of 60 mg given over a 60-minute period according to the following schedule:
- An initial treatment cycle with daily dosing for 14 days, followed by a 14-day drug-free period (Cycle 1).
- Subsequent treatment cycles with daily dosing for 10 days out of 14-day periods, followed by 14-day drug-free periods (Cycles 2-6).
The primary efficacy endpoint was a comparison of the change between treatment arms in the ALSFRS-R total scores from baseline to Week 24. The ALSFRS-R scale consists of 12 questions that evaluate the fine motor, gross motor, bulbar, and respiratory function of patients with ALS (speech, salivation, swallowing, handwriting, cutting food, dressing/hygiene, turning in bed, walking, climbing stairs, dyspnea, orthopnea, and respiratory insufficiency). Each item is scored from 0-4, with higher scores representing greater functional ability. The decline in ALSFRS-R scores from baseline was significantly less in the RADICAVA-treated patients as compared to placebo (see Table 3 ). The distribution of change in ALSFRS-R scores from baseline to Week 24 by percent of patients is shown in Figure 1.
| Treatment | Change from Baseline LS Mean ± SE (95% CI) | Treatment Difference (RADICAVA – placebo [95% CI]) | p -value |
|---|---|---|---|
| RADICAVA | −5.01±0.64 | 2.49 (0.99, 3.98) | 0.0013 |
| Placebo | −7.50±0.66 |
Figure 1 Distribution of Change from Baseline to Week 24 in ALSFRS-R Scores

HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
RADICAVA
RADICAVA is supplied as a 30 mg/100 mL (0.3 mg/mL) clear, colorless, sterile solution for intravenous infusion in single-dose polypropylene bags, each overwrapped with polyvinyl alcohol (PVA) secondary packaging containing an oxygen absorber and oxygen indicator, which should be pink to reflect appropriate oxygen levels [see Dosage and Administration (2.2) and How Supplied/Storage and Handling (16.2) ] . These are supplied in cartons as listed below.
NDC 70510-2171-1 30 mg/100 mL (0.3 mg/mL) single-dose bag NDC 70510-2171-2 2 bags per carton
RADICAVA ORS
RADICAVA ORS is supplied in a white to off-white suspension in multi-dose child resistant 60 mL amber glass bottle which is supplied as two configurations:
- RADICAVA ORS Starter Kit (14-day treatment cycle), including two (2) inner cartons, each containing one (1) bottle of 735 mg/35mL (105 mg/5 mL dose), two oral dosing syringes and one bottle adapter.
- RADICAVA ORS Kit (10-day treatment cycle), including one (1) bottle of 1050 mg/50 mL (105 mg/5 mL dose) with two oral dosing syringes and one bottle adapter.
These are supplied in cartons as listed below:
| Unit of sale | NDC number | Package configuration |
|---|---|---|
| RADICAVA ORS Starter Kit | NDC 70510-2321-1 | Carton of One (1) bottle of 35mL (105 mg/5 mL dose) |
| NDC 70510-2321-2 | Carton of two (2) NDC 70510-2321-1 | |
| RADICAVA ORS Kit | NDC 70510-2322-1 | Carton of One (1) bottle of 50 mL (105 mg/5 mL dose) |
Storage and Handling
RADICAVA
Store RADICAVA at up to 25°C (77°F). Excursions permitted from 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from light. Store in overwrapped package to protect from oxygen degradation until time of use. The oxygen indicator will turn blue or purple if the oxygen has exceeded acceptable levels. Once the overwrap package is opened, use within 24 hours.
RADICAVA ORS
Pharmacy
Store RADICAVA ORS refrigerated between 2°C to 8°C (36°F to 46°F) and protect from light. Do not freeze. Store upright.
Patient
Store RADICAVA ORS upright at room temperature between 20°C to 25°C (68°F to 77°F). Protect from light.
Discard 15 days after opening bottle or if unopened 30 days from date of shipment indicated on the carton pharmacy label.
INSTRUCTIONS FOR USE RADICAVA (ra di ká vah) ORS (edaravone) oral suspension
Read this Instructions for Use before you take RADICAVA ORS ® for the first time and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
Important information about measuring RADICAVA ORS: Always measure your prescribed dose of RADICAVA ORS using the oral syringe provided. Ask your healthcare provider or pharmacist who provided the medicine any questions you have about how to measure your prescribed dose. If you miss a dose, do not take 2 doses of RADICAVA ORS the next day. Do not take a dose of RADICAVA ORS on days 15 through 28.
How to prepare RADICAVA ORS : Keep this Instructions for Use handy when preparing the treatment.
- If your healthcare provider prescribes a Starter Kit, you will receive 2 bottles of RADICAVA ORS. Each bottle will contain 35 mL of RADICAVA ORS for a total of 70 mL to be used for your first treatment cycle of 14 days. Only open the second bottle when you have finished the first bottle.
- If you were not prescribed the Starter Kit, for each treatment cycle you will receive 1 bottle of RADICAVA ORS that contains a total of 50 mL of RADICAVA ORS. After the first treatment cycle, RADICAVA ORS is to be used for 10 days out of 14-day periods.
- Only use the bottle adapter and the 2 reusable 5 mL oral syringes provided with the bottle. Do not use a household teaspoon to measure your medicine.
How to store RADICAVA ORS:
- Store RADICAVA ORS upright at room temperature between 68°F to 77°F (20°C to 25°C). Protect from light.
- Opening the bottle:
- When you open the bottle of RADICAVA ORS for the first use, write the date you open the bottle on the bottle label.
- After opening the bottle of RADICAVA ORS, use within 15 days.
- After a bottle of Radicava ORS has been opened and used, a white crust may form on the neck or on the side of the bottle. This is due to normal use and RADICAVA ORS can continue to be used as prescribed.
- Keep bottle tightly closed between each use.
- Throw away (discard) any RADICAVA ORS that is not used within 15 days after opening the bottle or within 30 days from the date of shipment shown on the carton pharmacy label, whichever happens first. Ask your pharmacist how to properly throw away (discard) RADICAVA ORS you are no longer able to use.
- Keep RADICAVA ORS and all medicines out of the reach of children.
Each RADICAVA ORS carton contains:
|  |
| Figure A |
Important information:
- Keep these instructions for future use.
- Do not share RADICAVA ORS with anyone else.
- Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
- People who have problems using their hands may need assistance to draw up and give the correct dose of RADICAVA ORS.
How to take RADICAVA ORS:
Take RADICAVA ORS as prescribed by your healthcare provider.
Dosing information:
RADICAVA ORS has 2 different dosing schedules:
- For the first treatment cycle, you will take RADICAVA ORS every day for 14 days, followed by 14 days without the medicine.
- For the cycles after the first treatment cycle, you will take RADICAVA ORS daily for 10 out of 14 days, followed by 14 days without the medicine.
How RADICAVA ORS will be provided:
- If your healthcare provider prescribes the Starter Kit, you will receive 2 bottles of RADICAVA ORS. Each bottle contains 35 mL of RADICAVA ORS for a total of 70 mL to deliver 14 doses.
- There may be some medicine that remains in each bottle after the dose on day 7 and day 14 of the first treatment cycle. Throw away (discard) any medicine that remains.
- If you were not prescribed the Starter Kit, for each treatment cycle, you will receive 1 bottle of RADICAVA ORS that contains a total of 50 mL of RADICAVA ORS to deliver 10 doses.
- There may be some medicine that remains in each bottle after 10 doses. Throw away (discard) any medicine that remains.
Fasting information:
- Do not eat or drink anything 8 hours before each dose of RADICAVA ORS if you eat a high-fat meal.
- Do not eat or drink anything 4 hours before each dose of RADICAVA ORS if you eat a low-fat meal.
- Do not eat or drink anything 2 hours before each dose of RADICAVA ORS if you take a calorie supplement.
- You should wait at least 1 hour after taking your medicine before eating or drinking anything except water.
Step 1: Before each use of RADICAVA ORS: Before opening the bottle, turn it upside down (invert) and shake vigorously up and down for at least 30 seconds (see Figure B ). Look at the liquid medicine to make sure it is mixed well. If you see any small pieces at the bottom of the bottle, invert the bottle and shake it up and down for another 30 seconds or until you can no longer see the pieces at the bottom of the bottle (see Figure B ). If the liquid medicine all looks the same, you can proceed to Step 2.
| Shake up and down for at least 30 seconds. |  |
| Figure B |
Step 2: Open the bottle by firmly pressing down on the bottle cap and turning it counterclockwise (to the left). Place the open bottle upright on a flat surface. Do not throw away the bottle cap; you will need to replace it after taking each dose.
First-time use of a bottle only. This must only be done 1 time for each bottle. Insert the ribbed end of the bottle adapter into the bottle by firmly pressing it in as far as it will go (see Figure C ). Do not remove the bottle adapter.
 |
| Figure C |
Step 3: Remove the oral syringe from plastic wrap and make sure the plunger is inserted all the way into the barrel. Push the oral syringe plunger toward its tip to remove excess air (see Figure D ). Insert the oral syringe into the opening of the bottle adapter until it is firmly in place (see Figure E ). Turn the bottle upside down and slowly pull the plunger to remove a small amount of liquid (see Figure F ).
 |  |  |
| Figure D | Figure E | Figure F |
Step 4: Keep the bottle upside down and pull the plunger until it goes up to the last line (5 mL) (see Figure G ). While keeping the plunger in the same position, turn the bottle upright, and place it carefully on a flat surface. Remove the oral syringe by gently twisting or pulling it out from the bottle (see Figure H ). Check the amount of medicine again before moving on to the next step (see Figure I ).
Note: Do not remove the oral syringe while the bottle is upside down (medicine may leak out through the adapter).
Note: If the dose is not correct, insert the oral syringe tip firmly into the bottle adapter. Push the plunger all the way in so that the medicine flows back into the bottle. Turn the bottle upside down. Repeat Step 4.
 |  |  |
| Figure G | Figure H | Figure I |
Step 5: Place the tip of the oral syringe in the mouth and aim towards the inside of the cheek. Slowly press down on the plunger until the oral syringe is empty. Swallow all of the medicine (see Figure J ). If needed, you can use up to 8 ounces or 1 cup of water to help swallow the medicine.
Note: It is normal for a small amount of medicine to remain in the tip of the syringe after taking.
 |
| Figure J |
Step 6: Leave the bottle adapter inside the bottle. Place the bottle cap on the bottle and turn the cap clockwise (to the right). Keep bottle tightly closed between each use (see Figure K ).
 |
| Figure K |
Step 7: Remove the plunger from the oral syringe barrel by pulling the plunger and the barrel away from each other. Rinse the oral syringe (plunger and barrel) with water only . Allow it to air dry.
 |
| Figure L |
Step 8: When the oral syringe (plunger and barrel) is dry, put the plunger back into the oral syringe barrel. Do not throw away the oral syringe . Store the syringe in a clean, dry place.
You must complete Steps 1 through 4 under "How to take RADICAVA ORS" before starting Step 9 under "How to take a dose of RADICAVA ORS oral suspension through a feeding tube."
How to take a dose of RADICAVA ORS oral suspension through a feeding tube:
Step 9: Using a catheter-tip syringe, flush the feeding tube with 1 ounce (30 mL) of water (see Figure M ).
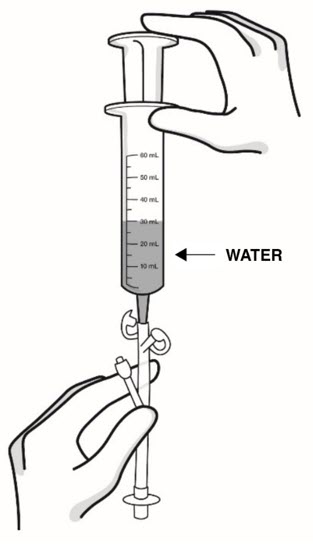 |
| Figure M |
Step 10: Place the oral syringe provided (containing the 5 mL of RADICAVA ORS) into the feeding tube. Slowly push down the plunger until the oral syringe is empty (see Figure N ).
 |
| Figure N |
Step 11: Using a catheter-tip syringe, flush the feeding tube with 1 ounce (30 mL) of water after taking the dose of RADICAVA ORS (see Figure O ).
 |
| Figure O |
Step 12: Leave the adapter in the bottle. Place the bottle cap on the bottle and turn it clockwise (to the right) to close the bottle. Keep the bottle tightly closed between each use (see Figure P ).
 |
| Figure P |
Step 13: Remove the plunger from the oral syringe barrel by pulling the plunger and barrel away from each other. Rinse the oral syringe (plunger and barrel) with water only (see Figure Q ). Allow it to air dry.
 |
| Figure Q |
Step 14: When the oral syringe (plunger and barrel) are dry, put the plunger back into the oral syringe barrel. Do not throw away the oral syringe. Store the oral syringe in a clean, dry place.
Manufactured for: Shionogi Inc. Florham Park, NJ 07932
For more information, go to www.Radicava.com or call 1-888-292-0058.
RADICAVA ORS is a registered trademark of Shionogi Inc. © 2026 Shionogi Inc. All Rights Reserved.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
22022-4
Issued: 05/2026
Mechanism of Action
The mechanism by which RADICAVA and RADICAVA ORS exert their therapeutic effect in patients with ALS is unknown.