Get your patient on Saphnelo (Anifrolumab-Fnia)
Saphnelo prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Saphnelo patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
Important Administration Information
SAPHNELO is intended for use under the guidance of a healthcare provider and may be administered as an intravenous infusion or as a subcutaneous injection.
SAPHNELO vials are for intravenous use only and must be diluted prior to intravenous administration [see Dosage and Administration (2.3) ] .
SAPHNELO prefilled syringe and autoinjector (SAPHNELO PEN) are for subcutaneous use only [see Dosage and Administration (2.4) ] .
Recommended Dosage
The recommended dosage of SAPHNELO is:
- 300 mg, administered as an intravenous infusion over a 30‑minute period every 4 weeks; or
- 120 mg, administered as a subcutaneous injection once every week.
Missed Dose
If a planned intravenous infusion is missed, administer SAPHNELO as soon as possible. Maintain a minimum interval of 14 days between infusions.
If a planned subcutaneous dose is missed, instruct the patient to administer SAPHNELO as soon as they remember. Thereafter, instruct the patient to start a new weekly schedule from the day the missed dose was administered or resume dosing on their usual day of administration, providing a minimum interval of 3 days between subcutaneous injections.
Intravenous Preparation and Administration Instructions
SAPHNELO is supplied as a single-dose vial. Prepare the diluted infusion solution using aseptic technique, by the following procedure:
- Visually inspect the vial for particulate matter and discoloration. SAPHNELO is a clear to opalescent, colorless to slightly yellow, solution. Discard the vial if the solution is cloudy, discolored or visible particles are observed. Do not shake the vial.
- Withdraw and discard 2 mL of solution from a 50 mL or 100 mL 0.9% Sodium Chloride Injection, USP infusion bag.
- Withdraw 2 mL of solution from the vial of SAPHNELO and add it to the infusion bag. Mix the solution by gentle inversion. Do not shake.
- Each vial is intended for one time use only. Discard any unused portion remaining in the vial.
- Administer the infusion solution immediately after preparation.
- If the infusion solution is not administered immediately, store the diluted solution of SAPHNELO at room temperature 59°F to 77°F (15°C to 25°C) for up to 4 hours, or refrigerated 36°F to 46°F (2°C to 8°C) for up to 24 hours. Do not freeze. Protect from light. If refrigerated, allow the diluted SAPHNELO solution to reach room temperature prior to administration.
- Administer the infusion solution intravenously over a 30-minute period through an infusion line containing a sterile, low-protein binding 0.2 to 15 micron in-line or add-on filter.
- To ensure the complete dose of SAPHNELO has been administered, flush the entire infusion line with 25 mL of 0.9% Sodium Chloride Injection, USP at the end of the infusion.
- Do not co-administer other medicinal products through the same infusion line.
- Dispose of any unused medicinal product or waste material in accordance with local requirements .
Subcutaneous Preparation and Administration Instructions
The SAPHNELO prefilled syringe and autoinjector (SAPHNELO PEN) are for subcutaneous use.
- Comprehensive instructions for subcutaneous administration of SAPHNELO using the prefilled syringe or autoinjector are provided in the ‘Instructions for Use’.
- Patients/caregivers may administer SAPHNELO prefilled syringe/SAPHNELO PEN after proper training in the subcutaneous injection technique and after the healthcare provider determines it is appropriate.
- Prior to administration, remove SAPHNELO from the refrigerator and allow it to come to room temperature for 60 minutes.
- Visually inspect SAPHNELO for particulate matter and discoloration prior to administration. SAPHNELO is a clear to opalescent, colorless to slightly yellow, solution. Discard the prefilled syringe or autoinjector if the solution is cloudy, discolored or visible particles are observed.
- Administer the subcutaneous injection into the thigh or abdomen, avoiding the 5 cm (approximately 2 inches) area around the navel. The upper arm can also be used if a healthcare professional or caregiver administers the injection.
- Do not inject into areas where the skin is tender, bruised, erythematous or hardened. When injecting in the same region, advise patients to use an injection site at least 3 cm (1‑inch) away from the last injection site.
- Dispose of any unused medicinal product or waste material in accordance with local requirements.
Switching Between Intravenous and Subcutaneous Administration
When transitioning patients from intravenous administration to subcutaneous administration of SAPHNELO, administer the first subcutaneous injection approximately 2 weeks after the last intravenous infusion.
When transitioning patients from subcutaneous administration to intravenous administration of SAPHNELO, administer the first intravenous infusion approximately 3 to 4 weeks after the last subcutaneous injection.

Saphnelo prescribing information
Dosage and Administration (2 ) 04/2026
INDICATIONS AND USAGE
SAPHNELO is indicated for the treatment of adult patients with moderate to severe systemic lupus erythematosus (SLE), who are receiving standard therapy.
Limitations of Use
The efficacy of SAPHNELO has not been evaluated in patients with severe active lupus nephritis or severe active central nervous system lupus. Use of SAPHNELO is not recommended in these situations.
DOSAGE AND ADMINISTRATION
Important Administration Information
SAPHNELO is intended for use under the guidance of a healthcare provider and may be administered as an intravenous infusion or as a subcutaneous injection.
SAPHNELO vials are for intravenous use only and must be diluted prior to intravenous administration [see Dosage and Administration (2.3) ] .
SAPHNELO prefilled syringe and autoinjector (SAPHNELO PEN) are for subcutaneous use only [see Dosage and Administration (2.4) ] .
Recommended Dosage
The recommended dosage of SAPHNELO is:
- 300 mg, administered as an intravenous infusion over a 30‑minute period every 4 weeks; or
- 120 mg, administered as a subcutaneous injection once every week.
Missed Dose
If a planned intravenous infusion is missed, administer SAPHNELO as soon as possible. Maintain a minimum interval of 14 days between infusions.
If a planned subcutaneous dose is missed, instruct the patient to administer SAPHNELO as soon as they remember. Thereafter, instruct the patient to start a new weekly schedule from the day the missed dose was administered or resume dosing on their usual day of administration, providing a minimum interval of 3 days between subcutaneous injections.
Intravenous Preparation and Administration Instructions
SAPHNELO is supplied as a single-dose vial. Prepare the diluted infusion solution using aseptic technique, by the following procedure:
- Visually inspect the vial for particulate matter and discoloration. SAPHNELO is a clear to opalescent, colorless to slightly yellow, solution. Discard the vial if the solution is cloudy, discolored or visible particles are observed. Do not shake the vial.
- Withdraw and discard 2 mL of solution from a 50 mL or 100 mL 0.9% Sodium Chloride Injection, USP infusion bag.
- Withdraw 2 mL of solution from the vial of SAPHNELO and add it to the infusion bag. Mix the solution by gentle inversion. Do not shake.
- Each vial is intended for one time use only. Discard any unused portion remaining in the vial.
- Administer the infusion solution immediately after preparation.
- If the infusion solution is not administered immediately, store the diluted solution of SAPHNELO at room temperature 59°F to 77°F (15°C to 25°C) for up to 4 hours, or refrigerated 36°F to 46°F (2°C to 8°C) for up to 24 hours. Do not freeze. Protect from light. If refrigerated, allow the diluted SAPHNELO solution to reach room temperature prior to administration.
- Administer the infusion solution intravenously over a 30-minute period through an infusion line containing a sterile, low-protein binding 0.2 to 15 micron in-line or add-on filter.
- To ensure the complete dose of SAPHNELO has been administered, flush the entire infusion line with 25 mL of 0.9% Sodium Chloride Injection, USP at the end of the infusion.
- Do not co-administer other medicinal products through the same infusion line.
- Dispose of any unused medicinal product or waste material in accordance with local requirements .
Subcutaneous Preparation and Administration Instructions
The SAPHNELO prefilled syringe and autoinjector (SAPHNELO PEN) are for subcutaneous use.
- Comprehensive instructions for subcutaneous administration of SAPHNELO using the prefilled syringe or autoinjector are provided in the ‘Instructions for Use’.
- Patients/caregivers may administer SAPHNELO prefilled syringe/SAPHNELO PEN after proper training in the subcutaneous injection technique and after the healthcare provider determines it is appropriate.
- Prior to administration, remove SAPHNELO from the refrigerator and allow it to come to room temperature for 60 minutes.
- Visually inspect SAPHNELO for particulate matter and discoloration prior to administration. SAPHNELO is a clear to opalescent, colorless to slightly yellow, solution. Discard the prefilled syringe or autoinjector if the solution is cloudy, discolored or visible particles are observed.
- Administer the subcutaneous injection into the thigh or abdomen, avoiding the 5 cm (approximately 2 inches) area around the navel. The upper arm can also be used if a healthcare professional or caregiver administers the injection.
- Do not inject into areas where the skin is tender, bruised, erythematous or hardened. When injecting in the same region, advise patients to use an injection site at least 3 cm (1‑inch) away from the last injection site.
- Dispose of any unused medicinal product or waste material in accordance with local requirements.
Switching Between Intravenous and Subcutaneous Administration
When transitioning patients from intravenous administration to subcutaneous administration of SAPHNELO, administer the first subcutaneous injection approximately 2 weeks after the last intravenous infusion.
When transitioning patients from subcutaneous administration to intravenous administration of SAPHNELO, administer the first intravenous infusion approximately 3 to 4 weeks after the last subcutaneous injection.
DOSAGE FORMS AND STRENGTHS
Intravenous Infusion
- Injection: 300 mg/2 mL (150 mg/mL) as a clear to opalescent, colorless to slightly yellow solution in a single-dose vial.
Subcutaneous Injection
- Injection: 120 mg/0.8 mL as a clear to opalescent, colorless to slightly yellow solution in a single-dose prefilled syringe or single-dose autoinjector (SAPHNELO PEN).
USE IN SPECIFIC POPULATIONS
Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to SAPHNELO during pregnancy. For more information about the registry or to report a pregnancy while on SAPHNELO, healthcare providers should contact AstraZeneca at 1‑877‑693‑9268 or https://anifrolumabpregnancyandbreastfeedingstudy.us/ .
Risk Summary
The limited human data with SAPHNELO use in pregnant women are insufficient to inform on drug‑associated risk for major birth defects, miscarriage, or adverse maternal or fetal outcome. Monoclonal IgG antibodies are known to be actively transported across the placenta as pregnancy progresses; therefore, anifrolumab‑fnia exposure to the fetus may be greater during the third trimester of pregnancy.
In an enhanced pre- and post-natal development study with pregnant cynomolgus monkeys that received intravenous administration of anifrolumab-fnia, there was no evidence of embryotoxicity or fetal malformations with exposures up to approximately 28‑times the exposure at the maximum recommended human dose (MRHD) on an Area Under Curve (AUC) basis (see Data ) .
All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk : Pregnant women with SLE are at increased risk of adverse pregnancy outcomes, including worsening of the underlying disease, premature birth, miscarriage, and intrauterine growth restriction. Maternal lupus nephritis increases the risk of hypertension and preeclampsia/eclampsia. Passage of maternal autoantibodies across the placenta may result in adverse neonatal outcomes, including neonatal lupus and congenital heart block.
Data
Animal Data : In an enhanced pre- and post-natal development study, pregnant cynomolgus monkeys received anifrolumab-fnia at intravenous doses of 30 or 60 mg/kg once every 2 weeks from confirmation of pregnancy at Gestation Day 20, throughout the gestation period, and continuing until 1‑month post-partum (approximately Lactation Day 28). There was no evidence of anifrolumab-fnia related maternal toxicity, embryo-fetal toxicity, or post-natal developmental effects. No anifrolumab-fnia related effect on T-cell-dependent antibody response in the infants was noted up to Day 180 after birth. The no observed adverse effect level (NOAEL) for maternal and developmental toxicity was identified as 60 mg/kg (approximately 28‑times the MRHD on an AUC basis). In the infants, mean serum concentrations of anifrolumab‑fnia on Day 30 after birth increased with dose and were approximately 4.2% to 9.7% of the respective maternal concentrations. The anifrolumab-fnia concentrations in the infant serum were up to approximately 22‑times the concentrations in the maternal milk, suggesting that anifrolumab-fnia had transferred via the placenta.
Lactation
Risk Summary
No data are available regarding the presence of SAPHNELO in human milk, the effects on the breastfed child, or the effects on milk production. Anifrolumab-fnia was detected in the milk of female cynomolgus monkeys administered anifrolumab-fnia. Due to species-species differences in lactation physiology, animal data may not reliably predict drug levels in humans. Maternal IgG is known to be present in human milk. If anifrolumab-fnia is transferred into human milk, the effects of local gastrointestinal exposure and limited systemic exposure in the breastfed infant to anifrolumab-fnia are unknown.
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for SAPHNELO and any potential adverse effects on the breast-fed child from SAPHNELO or from the underlying maternal condition.
Pediatric Use
The safety and efficacy of SAPHNELO in pediatric patients have not been established.
Geriatric Use
Of the 952 patients with moderate to severe SLE exposed to SAPHNELO in clinical trials, 3% (n=33) were 65 and over. The number of patients aged 65 years of age and older was not sufficient to determine whether they respond differently from younger adult patients.
CONTRAINDICATIONS
SAPHNELO is contraindicated in patients with a history of anaphylaxis with anifrolumab-fnia [see Warnings and Precautions (5.2) ] .
WARNINGS AND PRECAUTIONS
- Serious Infections: Serious and sometimes fatal infections have occurred in patients receiving SAPHNELO. SAPHNELO increases the risk of respiratory infections and herpes zoster. Avoid initiating treatment during an active infection. Consider the individual benefit-risk if using in patients with severe or chronic infections. Consider interrupting therapy with SAPHNELO if patients develop a new infection during treatment. (5.1 )
- Hypersensitivity Reactions Including Anaphylaxis: Serious hypersensitivity reactions including anaphylaxis and angioedema have been reported. (5.2 )
- Malignancy: Consider the individual benefit-risk in patients with known risk factors for malignancy prior to prescribing SAPHNELO. (5.3 )
- Immunizations: Avoid use of live or live-attenuated vaccines in patients receiving SAPHNELO. (5.4 )
- Not Recommended for Use with Other Biologic Therapies. (5.5 )
Serious Infections
Serious and sometimes fatal infections (including COVID‑19) have occurred in patients receiving immunosuppressive agents, including SAPHNELO. In controlled trials, fatal infections occurred more frequently in patients receiving SAPHNELO [see Adverse Reactions (6.1) ] .
In controlled trials, SAPHNELO increased the risk of respiratory infections and herpes zoster (disseminated herpes zoster events have been reported) [see Adverse Reactions (6.1) ] .
Avoid initiating treatment with SAPHNELO in patients with any clinically significant active infection until the infection is resolved or adequately treated. Consider the benefit and risk of administering SAPHNELO in patients with a chronic infection, a history of recurrent infections, or known risk factors for infection. Instruct patients to seek medical advice if signs or symptoms of a clinically significant infection occur. If a patient develops an infection or is not responding to standard anti-infective therapy while on SAPHNELO, monitor the patient closely and consider interrupting SAPHNELO until the infection resolves.
Hypersensitivity Reactions Including Anaphylaxis
Serious hypersensitivity reactions (including anaphylaxis) have been reported following SAPHNELO administration [see Contraindication (4) ] . Events of angioedema have also been reported [see Adverse Reactions (6.1) ] .
Other hypersensitivity reactions and infusion-related reactions have occurred following administration of SAPHNELO [see Adverse Reactions (6.1) ] . Consider pre-medication before infusion of SAPHNELO for patients with a history of these reactions.
SAPHNELO should be administered by healthcare providers prepared to manage hypersensitivity reactions, including anaphylaxis, and infusion-related reactions. If a serious infusion-related or hypersensitivity reaction (e.g., anaphylaxis) occurs, immediately interrupt the administration of SAPHNELO and initiate appropriate therapy.
Malignancy
There is an increased risk of malignancies with the use of immunosuppressants. The impact of SAPHNELO treatment on the potential development of malignancies is not known.
Consider the individual benefit-risk in patients with known risk factors for the development or reoccurrence of malignancy prior to prescribing SAPHNELO. In patients who develop malignancies, consider the benefit-risk of continued treatment with SAPHNELO.
Immunizations
Update immunizations, according to current immunization guidelines, prior to initiating SAPHNELO therapy. Avoid concurrent use of live or live-attenuated vaccines in patients treated with SAPHNELO.
Not Recommended for Concomitant Use with Other Biologic Therapies
SAPHNELO has not been studied in combination with other biologic therapies, including B-cell-targeted therapies. Therefore, use of SAPHNELO is not recommended for use in combination with biologic therapies.
ADVERSE REACTIONS
The following clinically significant adverse reactions are also discussed elsewhere in the labeling:
- Serious Infections [see Warnings and Precautions (5.1) ]
- Hypersensitivity Reactions Including Anaphylaxis [see Warnings and Precautions (5.2) ]
- Malignancy [see Warnings and Precautions (5.3) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse Reactions with Intravenous Administration
The safety of SAPHNELO was assessed in adult patients with moderate to severe SLE who received SAPHNELO 300 mg by intravenous infusion every 4 weeks (N=459) for 52 weeks, compared to placebo (N=466) in controlled clinical trials (Trials 1, 2 and 3) [see Clinical Studies (14.1) ]. The population studied had a mean age of 41 years (range: 18 to 69), of which 93% were female, 60% White, 13% Black/African American, and 10% Asian.
In the controlled-clinical trials, adverse reactions, irrespective of causality, were reported in 87% of patients receiving SAPHNELO and 79% of patients receiving placebo.
Adverse reactions that occurred at greater than or equal to 2% incidence are shown in Table 1.
Adverse Reaction | SAPHNELO (N=459) % | Placebo (N=466) % |
Upper respiratory tract infection Upper respiratory tract infections (including Upper respiratory tract infections, Nasopharyngitis, Pharyngitis) | 34 | 23 |
Bronchitis Bronchitis (including Bronchitis, Bronchitis viral, Tracheobronchitis) | 11 | 5.2 |
Infusion‑related reactions | 9.4 | 7.1 |
Herpes Zoster | 6.1 | 1.3 |
Cough | 5.0 | 3.2 |
Respiratory tract infection Respiratory tract infection (including Respiratory tract infection, Respiratory tract infection viral, Respiratory tract infection bacterial) | 3.3 | 1.5 |
Hypersensitivity | 2.8 | 0.6 |
All patients received standard therapy. | ||
Long-term Safety
Patients who completed Trials 2 and 3 (Phase III feeder trials) were eligible to continue on treatment in a randomized, double-blind, placebo-controlled long-term extension (LTE) trial, for an additional 3 years. The long-term safety of SAPHNELO was assessed in 257 patients who received SAPHNELO 300 mg every 4 weeks and 112 patients who received placebo in both a feeder trial and the LTE. Of these, 177 patients who received SAPHNELO (68.9%) and 52 patients who received placebo (46.4%) completed a total of 4 years on treatment. The overall long-term safety profile of SAPHNELO was consistent with Trials 1, 2 and 3.
Specific Adverse Reactions
Infections: In the 52‑week controlled-clinical trials, infections were reported in a greater proportion of patients while on treatment with SAPHNELO compared to placebo (69.7% versus 55.4%, respectively).
Herpes Zoster: In the 52‑week controlled-clinical trials, the incidence of herpes zoster in patients while on treatment with SAPHNELO was 6.1% and 1.3% in patients on placebo. Cases with multidermatomal involvement and disseminated presentation have been reported. Of the 28 SAPHNELO-treated patients with herpes zoster, 2 experienced disseminated disease requiring hospitalization compared to none among placebo-treated patients.
Hypersensitivity Reactions Including Anaphylaxis: During the SLE intravenous development program, there was one report of an anaphylactic reaction in a patient who received SAPHNELO 150 mg, and 4 reports of angioedema after 300 mg. In general, the hypersensitivity reactions were predominantly mild or moderate in intensity and did not lead to discontinuation of SAPHNELO.
In the 52‑week controlled‑clinical trials, hypersensitivity reactions occurred in 2.8% of patients while on treatment with SAPHNELO and 0.6% of patients on placebo. Serious hypersensitivity reactions were reported in 0.6% of patients receiving SAPHNELO, including angioedema (n=2).
Infusion‑related Reactions: Infusion‑related reactions were mild to moderate in intensity; the most common symptoms were headache, nausea, vomiting, fatigue, and dizziness.
In the 52‑week controlled‑clinical trials, the incidence of infusion‑related reactions while on treatment was 9.4% in patients while on treatment with SAPHNELO and 7.1% in patients on placebo.
Malignancies: In 52‑week controlled-clinical trials, malignancies (excluding non‑melanoma skin cancers) were observed in 0.7% and 0.6% of SAPHNELO-treated and placebo-treated patients. Malignant neoplasm (including non‑melanoma skin cancers) was reported in 1.3% of SAPHNELO-treated patients, compared to 0.6% placebo-treated patients. The malignancies that were reported in more than one patient treated with SAPHNELO included breast cancer and squamous cell carcinoma.
Adverse Reactions with Subcutaneous Administration
The safety profile of SAPHNELO administered subcutaneously in adult patients with moderate to severe SLE is derived from Trial 5 [see Clinical Studies (14.2) ] . The data reflect SAPHNELO exposure in 185 patients who received SAPHNELO 120 mg by subcutaneous injection once every week, with a median duration of 52 weeks, as compared with comparable exposure to placebo in 182 patients. All patients received standard therapy. The population studied had a mean age of 43 years (range: 18 to 70), of which 92% were female, 74% White, 9% Asian, 8% American Indian or Alaska Native, and 5% Black/African American.
The safety profile observed for SAPHNELO administered subcutaneously was consistent with the known safety profile of SAPHNELO administered intravenously .
Serious Infections in the Pool of Intravenous and Subcutaneous Trials
In the pool of four placebo-controlled clinical trials in adults with moderate to severe SLE where SAPHNELO was administered either intravenously or subcutaneously (Trials 1, 2, 3, and 5), the incidence of serious infections while on treatment was 5.4% in patients treated with SAPHNELO compared to 4.9% in patients receiving placebo. Fatal infections occurred while on treatment in 0.5% of SAPHNELO-treated patients and 0% in placebo-treated patients. The most frequent serious infection reported in patients treated with SAPHNELO was pneumonia. During the LTE of Trials 2 and 3, the most common serious infections were COVID‑19 and pneumonia.
Postmarketing Experience
The following adverse reaction has been identified during post-approval use of SAPHNELO. Because the reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Arthralgia
DRUG INTERACTIONS
No formal drug interaction trials have been conducted.
DESCRIPTION
Anifrolumab-fnia is a type I interferon (IFN) receptor antagonist, immunoglobulin G1 kappa (IgG1κ) monoclonal antibody that is produced in mouse myeloma cells (NS0) by recombinant DNA technology. The molecular weight is approximately 148 kDa.
Intravenous Infusion
SAPHNELO (anifrolumab-fnia) injection is a sterile, preservative‑free, clear to opalescent, colorless to slightly yellow, solution in a single-dose vial for intravenous use.
Each 2 mL single-dose vial contains 300 mg (150 mg/mL) of anifrolumab-fnia, L-histidine (3 mg), L-histidine hydrochloride monohydrate (6 mg), L-lysine hydrochloride (18 mg), polysorbate 80 (1 mg), trehalose dihydrate (98 mg), and Water for Injection, USP. The pH is 5.9.
Subcutaneous Injection
SAPHNELO (anifrolumab-fnia) injection is a sterile, preservative-free, clear to opalescent, colorless to slightly yellow, solution in a prefilled syringe or autoinjector for subcutaneous injection.
Each 0.8 mL single-dose prefilled syringe or autoinjector delivers 120 mg of anifrolumab-fnia, histidine (1 mg), L-histidine hydrochloride monohydrate (3 mg), lysine hydrochloride (7 mg), polysorbate 80 (0.4 mg), trehalose (36 mg), and Water for Injection, USP. The pH is 5.9.
CLINICAL PHARMACOLOGY
Mechanism of Action
Anifrolumab-fnia is a human IgG1κ monoclonal antibody that binds to subunit 1 of the type I interferon receptor (IFNAR) with high specificity and affinity. This binding inhibits type I IFN signaling, thereby blocking the biologic activity of type I IFNs. Anifrolumab-fnia also induces the internalization of IFNAR1, thereby reducing the levels of cell surface IFNAR1 available for receptor assembly. Blockade of receptor mediated type I IFN signaling inhibits IFN responsive gene expression as well as downstream inflammatory and immunological processes. Inhibition of type I IFN blocks plasma cell differentiation and normalizes peripheral T-cell subsets.
Type I IFNs play a role in the pathogenesis of SLE. Approximately 60-80% of adult patients with active SLE express elevated levels of type I IFN inducible genes.
Pharmacodynamics
In patients with moderate to severe SLE, following the administration of SAPHNELO, neutralization (≥80%) of a type I IFN gene signature was observed from Week 4 to Week 52 in blood samples of patients with elevated levels of type I IFN inducible genes and returned to baseline levels within 8 to 12 weeks following withdrawal of SAPHNELO at the end of the 52‑week treatment period. However, the clinical relevance of the type I IFN gene signature neutralization is unclear.
In SLE patients with positive anti-dsDNA antibodies at baseline (Trials 2 and 3), treatment with SAPHNELO led to numerical reductions in anti-dsDNA antibodies over time through Week 52.
In patients with low complement levels (C3 and C4) at baseline (Trials 2 and 3), increases in complement levels were observed in patients receiving SAPHNELO through Week 52.
Pharmacokinetics
The pharmacokinetics (PK) of anifrolumab-fnia was studied in adult patients with SLE following intravenous doses ranging from one-third (100 mg) to 3.3 times (1000 mg) the approved intravenous dosage, 300 mg once every 4 weeks, and subcutaneous 120 mg weekly doses, as well as in healthy volunteers following a single intravenous dose at 300 mg and a single subcutaneous dose of 120 mg.
Anifrolumab-fnia exhibits non-linear PK in the dose range of 100 mg to 1000 mg with more than dose-proportional increases in the exposure as measured by AUC, following intravenous administration.
The estimated time to reach steady state is approximately 112 days for both intravenous and subcutaneous administration.
Following 300 mg intravenous administrations every 4 weeks, the accumulation ratio for C max was 1.11 and for C trough was 2.37. Following 120 mg subcutaneous administration weekly, the accumulation ratio for C max was 1.85 and C trough was 1.85.
Absorption
The bioavailability of anifrolumab-fnia was estimated to be 73% following subcutaneous injection. The estimated maximum serum concentration (C max ) of anifrolumab-fnia at steady state was 63.7 μg/mL.
Distribution
Based on population PK analysis, the estimated volume of distribution at steady state for a typical patient with SLE (68 kg) is 5.16 L.
Elimination
From population PK analysis, anifrolumab-fnia exhibited non-linear PK due to IFNAR1-mediated drug clearance.
The estimated systemic clearance (CL) for anifrolumab-fnia is 0.146 L/day.
Based on population PK analysis of patients who received SAPHNELO for one year, serum concentrations of anifrolumab-fnia were below detection in 95% of patients approximately 16 weeks after the last dose.
Specific Populations
There was no clinically meaningful difference in systemic clearance based on age, race, ethnicity, region, gender, IFN status or body weight, that requires dose adjustment.
Age: Based on population PK analyses, age (range 18 to 70 years) did not affect anifrolumab-fnia clearance. Limited PK data are available for geriatric patients; 3% (n=33) of the patients included in the PK analysis were 65 years or older [see Use in Specific Populations (8.5) ] .
Patients with Renal Impairment: No specific clinical trials have been conducted to investigate the effect of renal impairment on anifrolumab-fnia. Based on population PK analyses, anifrolumab-fnia clearance was comparable in SLE patients with mild (60-89 mL/min/1.73 m 2 ) and moderate (30-59 mL/min/1.73 m 2 ) decrease in eGFR values and patients with normal renal function (≥90 mL/min/1.73 m 2 ). There were no SLE patients with a severe decrease in eGFR or end stage renal disease (<30 mL/min/1.73 m 2 ); anifrolumab-fnia is not cleared renally.
Patients with urine protein/creatinine ratio (UPCR) >2 mg/mg were excluded from the clinical trials. Based on population PK analyses, increased UPCR did not significantly affect anifrolumab-fnia clearance.
Patients with Hepatic Impairment: No specific clinical trials have been conducted to investigate the effect of hepatic impairment on anifrolumab-fnia. IgG1 monoclonal antibodies are predominantly eliminated via catabolism and are not expected to undergo hepatic metabolism; changes in hepatic function are not expected to influence anifrolumab-fnia clearance. Based on population PK analyses, baseline hepatic function biomarkers (ALT and AST ≤2.0 × ULN, and total bilirubin) had no clinically relevant effect on anifrolumab-fnia clearance.
Drug Interactions
No formal drug-drug interaction trials have been conducted.
Based on population PK analysis, concomitant use of oral corticosteroids, anti-malarials, immunosuppressants (azathioprine, methotrexate, mycophenolate mofetil, mycophenolic acid, and mizoribine), NSAIDs, ACE inhibitors, and HMG-CoA reductase inhibitors did not significantly affect the PK of anifrolumab-fnia.
Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies (ADA) in the trials described below with the incidence of anti-drug antibodies in other trials, including those of anifrolumab-fnia.
In Trials 2, 3, and the long-term extension trial, treatment emergent anti-anifrolumab-fnia antibodies were detected in 9 of 350 patients (ADA incidence 2.6%) who received SAPHNELO at the recommended intravenous dosing regimen for up to 4 years.
In Trial 5, treatment emergent anti-anifrolumab-fnia antibodies were detected in 6 of 107 patients (ADA incidence 5.6%) treated with SAPHNELO at the recommended subcutaneous dosing regimen during the 52‑week treatment period. No neutralizing antibodies were detected.
Because of the low occurrence of anti-drug antibodies, the effect of these antibodies on the pharmacokinetics, pharmacodynamics, safety, and/or effectiveness of anifrolumab-fnia products is unknown.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic and genotoxic potential of anifrolumab-fnia have not been evaluated. In rodent models of IFNAR1 blockade, increased carcinogenic potential has been observed. The clinical relevance of these findings is unknown.
Effects on male and female fertility have not been directly evaluated in animal studies. No anifrolumab-fnia-related adverse effects on indirect measures of male or female fertility, based on semen analysis, spermatogenesis staging, menses cycle, organ weights and histopathological findings in the reproductive organs were observed in 9-month repeat-dose toxicity studies in cynomolgus monkeys at doses up to 50 mg/kg intravenous once weekly (approximately 58‑times the MRHD on an AUC basis) and 60 mg/kg subcutaneous once weekly (approximately 52-times the subcutaneous MRHD on an AUC basis).
CLINICAL STUDIES
Intravenous Administration in Adults with Moderate to Severe SLE
Trial Design and Population
The safety and efficacy of SAPHNELO were evaluated in three 52-week treatment period, multicenter, randomized, double-blind, placebo-controlled trials (Trial 1 [NCT01438489], Trial 2 [NCT02446912] and Trial 3 [NCT02446899]). Patients were diagnosed with SLE according to the American College of Rheumatology (1982 revised) classification criteria. All patients were ≥18 years of age and had moderate to severe disease, with a SLE Disease Activity Index 2000 (SLEDAI-2K) score ≥6 points, organ level involvement based on the British Isles Lupus Assessment Group (BILAG) assessment, and a Physician’s Global Assessment [PGA] score ≥1, despite receiving standard SLE therapy consisting of either one or any combination of oral corticosteroids (OCS), antimalarials and/or immunosuppressants at baseline. Patients continued to receive their existing SLE therapy at stable doses during the clinical trials, with the exception of OCS (prednisone or equivalent) where tapering was a component of the protocol. Patients who had severe active lupus nephritis and patients who had severe active central nervous system lupus were excluded. The use of other biologic agents and cyclophosphamide were not permitted during the trials; patients receiving other biologic therapies were required to complete a wash-out period of at least 5 half‑lives prior to enrollment. All three trials were conducted in North America, Europe, South America and Asia. Patients received SAPHNELO or placebo, administered by intravenous infusion, every 4 weeks.
Efficacy of SAPHNELO was established based on assessment of clinical response using the composite endpoints, the British Isles Lupus Assessment Group based Composite Lupus Assessment (BICLA) and the SLE Responder Index (SRI‑4).
BICLA response at Week 52, was defined as improvement in all organ domains with moderate or severe activity at baseline:
- Reduction of all baseline BILAG A to B/C/D and baseline BILAG B to C/D, and no BILAG worsening in other organ systems, as defined by ≥1 new BILAG A or ≥2 new BILAG B;
- No worsening from baseline in SLEDAI-2K, where worsening is defined as an increase from baseline of >0 points in SLEDAI-2K;
- No worsening from baseline in patients’ lupus disease activity, where worsening is defined by an increase ≥0.30 points on a 3-point PGA visual analogue scale (VAS);
- No discontinuation of treatment;
- No use of restricted medication beyond the protocol-allowed threshold.
SRI‑4 response, was defined as meeting each of the following criteria at Week 52 compared with baseline:
- Reduction from baseline of ≥4 points in the SLEDAI-2K;
- No new organ system affected as defined by 1 or more BILAG A or 2 or more BILAG B items compared to baseline;
- No worsening from baseline in the patients’ lupus disease activity defined by an increase ≥0.30 points on a 3‑point PGA VAS;
- No discontinuation of treatment;
- No use of restricted medication beyond the protocol-allowed threshold.
Trial 1 randomized 305 patients (1:1:1) who received SAPHNELO, 300 mg or 1000 mg, or placebo once every 4 weeks for up to 52 weeks. The primary endpoint was a combined assessment of the SRI-4 and the sustained reduction in OCS (<10 mg/day and ≤OCS dose at Week 1, sustained for 12 weeks) measured at Week 24.
Trial 2 and 3 were similar in design. Trial 2 randomized 457 patients (1:2:2) who received SAPHNELO 150 mg, 300 mg once every 4 weeks or placebo. Trial 3 randomized 362 patients (1:1) who received SAPHNELO 300 mg once every 4 weeks or placebo. The primary endpoints were improvement in disease activity evaluated at 52 weeks, measured by SRI‑4 in Trial 2 and BICLA in Trial 3 (defined above). The common secondary efficacy endpoints included in both trials were the maintenance of OCS reduction, improvement in cutaneous SLE activity, and flare rate. During Weeks 8-40, patients with a baseline OCS ≥10 mg/day were required to taper their OCS dose to ≤7.5 mg/day, unless there was worsening of disease activity. Both trials evaluated the efficacy of SAPHNELO 300 mg once every 4 weeks versus placebo; a dose of 150 mg was also evaluated for dose-response in Trial 2.
Patient demographics and disease characteristics were generally similar and balanced across treatment arms (Table 2).
Total Population | |||
Trial 1 (N=305) | Trial 2 (N=457) | Trial 3 (N=362) | |
Mean Age (years) | 40 | 41 | 42 |
Female (%) | 93 | 92 | 93 |
White (%) | 42 | 71 | 60 |
Black/African American (%) | 13 | 14 | 12 |
Asian (%) | 7 | 5 | 17 |
Hispanic or Latino (%) | 42 | 19 | 30 |
Baseline SLEDAI-2K score | |||
Mean (SD) | 10.9 (4.1) | 11.3 (3.72) | 11.5 (3.76) |
≥10 points, n (%) | 182 (60) | 328 (72) | 260 (72) |
BILAG organ system scoring (Overall) | |||
At least one A, n (%) | 152 (50) | 217 (48) | 176 (49) |
No A and at least 2 Bs, n (%) | 134 (44) | 211 (46) | 169 (47) |
Positive Anti-dsDNA levels, n (%) | 185 (77) | 207 (45) | 159 (44) |
Abnormal ANA, n (%) | 299 (98) | 412 (90) | 325 (90) |
Abnormal Complement C3 level, n (%) | 119 (39) | 157 (34) | 144 (40) |
Abnormal Complement C4 level, n (%) | 74 (24) | 95 (21) | 95 (26) |
Baseline SLE treatment | |||
OCS, n (%) | 258 (85) | 381 (83) | 292 (81) |
Antimalarials, n (%) | 219 (72) | 334 (73) | 252 (70) |
Immunosuppressants, n (%) | 150 (49) | 214 (47) | 174 (48) |
Randomization was stratified by disease severity (SLEDAI-2K score at baseline, <10 vs ≥10 points), OCS dose on Day 1 (<10 mg/day vs ≥10 mg/day prednisone or equivalent) and interferon gene signature test results (high vs low).
Trials 1, 2, and 3 Results
Although other intravenous SAPHNELO dosages were studied in Trials 1 and 2, only data for the approved intravenous SAPHNELO dosage of 300 mg every 4 weeks are presented below.
The reduction in disease activity seen in the BICLA and SRI-4 was related primarily to improvement in the mucocutaneous and musculoskeletal organ systems. Flare rate was reduced in SAPHNELO patients compared to placebo patients although the difference was not statistically significant.
BICLA responder analysis
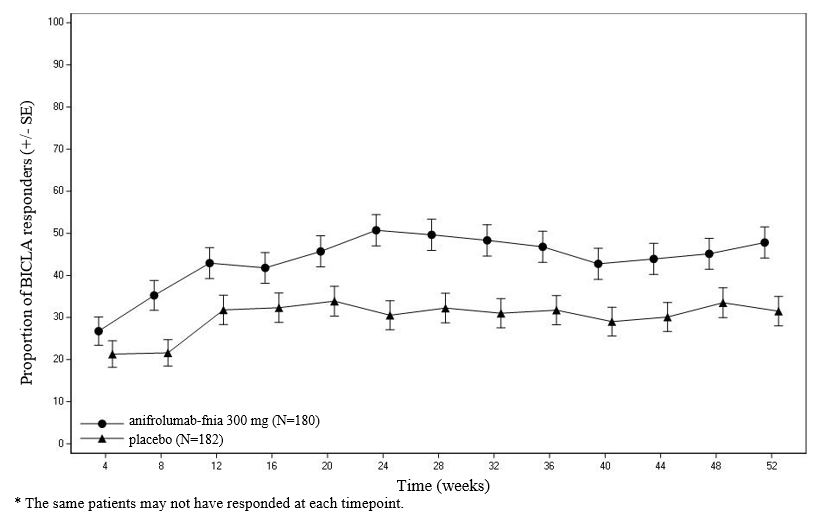
BICLA was the primary endpoint in Trial 3, SAPHNELO 300 mg once every 4 weeks demonstrated statistically significant and clinically meaningful efficacy in overall disease activity compared with placebo, with greater improvements in all components of the composite endpoint. In Trial 1 and 2, BICLA was a pre-specified analysis. The BICLA results are presented in Table 3.
Trial 1 Not formally tested in a pre-specified testing scheme and findings should be interpreted with caution. Based on post hoc analysis. | Trial 2 | Trial 3 Primary endpoint. | ||||
SAPHNELO 300 mg once every 4 weeks (N=99) | Placebo (N=102) | SAPHNELO 300 mg once every 4 weeks (N=180) | Placebo (N=184) | SAPHNELO 300 mg once every 4 weeks (N=180) | Placebo (N=182) | |
BICLA Response Rate In all 3 trials, patients who discontinued investigational product or initiated restricted medications beyond the protocol-specified thresholds are considered non-responders. For consistency, the results presented for Trial 2 represent the post-hoc analysis using the restricted medication thresholds as defined in Trial 3. | ||||||
Responder, n (%) | 54 (54.6) | 27 (25.8) | 85 (47.1) | 55 (30.2) | 86 (47.8) | 57 (31.5) |
Difference in Response Rates (95% CI) | 28.8 (15.7, 41.9) | 17.0 (7.2, 26.8) | 16.3 (6.3, 26.3) p-value=0.001 | |||
Components of BICLA Response | ||||||
BILAG Improvement, n (%) | 54 (54.5) | 28 (27.5) | 85 (47.2) | 58 (31.5) | 88 (48.9) | 59 (32.4) |
No Worsening of SLEDAI-2K, n (%) | 73 (73.7) | 61 (59.8) | 121 (67.2) | 104 (56.5) | 122 (67.8) | 94 (51.6) |
No Worsening of PGA, n (%) | 76 (76.8) | 62 (60.8) | 117 (65.0) | 105 (57.1) | 122 (67.8) | 95 (52.2) |
The response rates and associated difference and 95% CI are calculated using a Cochran-Mantel-Haenszel approach adjusted for stratification factors. The reported percentages for the components are unadjusted. | ||||||
In Trial 3, examination of subgroups by age, race, gender, ethnicity, disease severity [SLEDAI-2K at baseline], and baseline OCS use did not identify differences in response to SAPHNELO.
Figure 1 shows the proportion of BICLA responders through the 52-week treatment period in Trial 3.
Figure 1 Trial 3: Proportion (%) of BICLA Responders in Adults with Moderate to Severe SLE by Visit•
SRI-4 responder analysis
SRI-4 was the primary endpoint in Trial 2, treatment with SAPHNELO did not result in statistically significant improvements over placebo. In Trials 1 and 3, SRI-4 was a pre-specified analysis. The SRI-4 results are presented in Table 4.
Trial 1 Not formally tested in a pre-specified testing scheme and findings should be interpreted with caution. | Trial 2 Primary endpoint. | Trial 3 | ||||
SAPHNELO 300 mg once every 4 weeks (N=99) | Placebo (N=102) | SAPHNELO 300 mg once every 4 weeks (N=180) | Placebo (N=184) | SAPHNELO 300 mg once every 4 weeks (N=180) | Placebo (N=182) | |
SRI-4 Response Rate In all 3 trials, patients who discontinued investigational product or initiated restricted medications beyond the protocol-specified thresholds are considered non-responders. For consistency, the results presented for Trial 2 represent the post-hoc analysis using the restricted medication thresholds as defined in Trial 3. The most commonly involved SLEDAI-2K organ domains were mucocutaneous, musculoskeletal and immune. | ||||||
Responder, n (%) | 62 (62.8) | 41 (38.8) | 88 (49.0) | 79 (43.0) | 100 (55.5) | 68 (37.3) |
Difference in Response Rates (95% CI) | 24.0 (10.9, 37.2) | 6.0 (-4.2, 16.2) | 18.2 (8.1, 28.3) | |||
Components of SRI-4 Response | ||||||
SLEDAI-2K improvement, n (%) | 62 (62.6) | 41 (40.2) | 89 (49.4) | 80 (43.5) | 101 (56.1) | 71 (39.0) |
No worsening of BILAG, n (%) | 75 (75.8) | 61 (59.8) | 119 (66.1) | 105 (57.1) | 125 (69.4) | 94 (51.6) |
No worsening of PGA, n (%) | 76 (76.8) | 62 (60.8) | 117 (65.0) | 105 (57.1) | 122 (67.8) | 95 (52.2) |
The response rates and associated difference and 95% CI are calculated using a Cochran-Mantel-Haenszel approach adjusted for stratification factors. The reported percentages for the components are unadjusted. | ||||||
Effect on Concomitant Steroid Treatment
In Trial 3, among the 47% of patients with a baseline OCS use ≥10 mg/day, SAPHNELO demonstrated a statistically significant difference in the proportion of patients able to reduce OCS use by at least 25% to ≤7.5 mg/day at Week 40 and maintain the reduction through Week 52 (p-value = 0.004); 52% (45/87) of patients in the SAPHNELO group versus 30% (25/83) in the placebo group achieved this level of steroid reduction (difference 21% [95% CI 6.8, 35.7]). Consistent trends in favor of SAPHNELO compared to placebo, on effect of reduction of OCS use, were observed in Trial 1 and 2, but the difference was not statistically significant.
Subcutaneous Administration in Adults with Moderate to Severe SLE
The safety and efficacy of SAPHNELO administered subcutaneously were evaluated in a 52-week treatment period, multicenter, randomized, double-blind, placebo-controlled trial (Trial 5 [NCT04877691]). All patients were ≥18 years of age, diagnosed with SLE according to the American College of Rheumatology (1997 revised) classification criteria, and had moderate to severe disease, with a SLEDAI-2K score ≥6 points, organ level involvement based on BILAG assessment, and a PGA score ≥1, despite receiving standard SLE therapy consisting of either one or any combination of OCS, antimalarials and/or immunosuppressants at baseline. Patients continued to receive their existing SLE therapy at stable doses during the trial, with the exception of OCS (prednisone or equivalent) where tapering was a component of the protocol. Patients who had severe active lupus nephritis or severe active central nervous system lupus were excluded. Patients were randomized (1:1) to receive SAPHNELO 120 mg plus standard therapy or placebo plus standard therapy by subcutaneous injection once every week. Randomization was stratified by SLEDAI‑2K score at baseline (<10 vs ≥10 points), OCS dose on Day 1 (<10 mg/day vs ≥10 mg/day prednisone or equivalent) and interferon gene signature test results (high vs low).
A pre-specified interim analysis was conducted when the first 220 randomized patients completed Week 52 or had withdrawn from the trial. Of these, 89% were female, 45% Hispanic or Latino, 78% White, 7% American Indian or Alaska Native, 7% Asian, and 4% Black/African American. The mean age was 43 years. At baseline, the mean SLEDAI‑2K score was 10.9 (SD: 3.4) and 67% had high disease activity (SLEDAI‑2K score ≥10), 45% had severe disease (BILAG A) in at least 1 organ system and 50% had moderate disease (BILAG B) in at least 2 organ systems. The most commonly affected organ systems (BILAG A or B at baseline) were the musculoskeletal (95%) and mucocutaneous (92%) systems; 2% cardiorespiratory and 2% renal organ domain involvement. At baseline, 95% had abnormal ANA, 40% were positive for anti-dsDNA antibodies; 33% of patients had abnormal C3, and 24% abnormal C4. Background SLE standard therapy included OCS (82%; mean daily dose, prednisone or equivalent, 9.8 mg), immunosuppressants (56%), and anti-malarials (80%). During Weeks 8-40, patients with a baseline OCS ≥10 mg/day were required to taper their OCS dose to ≤7.5 mg/day, unless there was worsening of disease activity.
The primary endpoint was BICLA response rate measured at Week 52. At the interim analysis, SAPHNELO by subcutaneous administration demonstrated a statistically significant and clinically meaningful reduction of overall disease activity compared with placebo (Table 5).
SAPHNELO 120 mg once weekly (N=109) | Placebo (N=111) | |
BICLA Response | ||
| 64 (58.5) | 48 (43.2) |
| 15.3 (2.1, 28.5) p-value=0.0231 Based on an interim analysis using Pocock alpha spending function with an information fraction of 0.6. The primary endpoint was tested at the alpha level of 0.0354 at the interim analysis. | |
Components of BICLA Response | ||
| 64 (58.5) | 48 (43.3) |
| 80 (73.4) | 77 (68.9) |
| 80 (73.5) | 78 (70.0) |
The response rates, associated difference, and 95% CI are calculated using a Cochran-Mantel-Haenszel approach adjusted for stratification factors. | ||
Subgroup analysis by disease severity (based on baseline SLEDAI-2K, <10 points, ≥10 points) and baseline OCS use (<10 mg/day, ≥10 mg/day) did not identify differences in response to SAPHNELO.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
SAPHNELO (anifrolumab-fnia) injection is a sterile, preservative-free, clear to opalescent, colorless to slightly yellow solution in a single-dose vial for intravenous infusion or a prefilled syringe or autoinjector for subcutaneous injection.
SAPHNELO is available as follows:
Packaging Unit and Strength | Pack Size and NDC |
300 mg/2 mL (150 mg/mL) single-dose vial (NDC 0310-3040-00) | One vial in a carton: NDC 0310-3040-00 |
120 mg/0.8 mL single-dose prefilled syringe (NDC 0310-3080-75) | One syringe in a carton: NDC 0310-3080-02 |
120 mg/0.8 mL single-dose autoinjector (SAPHNELO PEN) (NDC 0310-3080-25) | One autoinjector in a carton: NDC 0310-3080-01 |
The prefilled syringe (including needle cover) and autoinjector (including cap) are not made with natural rubber latex.
Storage and Handling
Store SAPHNELO in a refrigerator at 36°F to 46°F (2°C to 8°C) in the original carton to protect from light.
- Do not freeze. Do not shake. Do not expose to heat. Do not use SAPHNELO past the expiration date.
If needed, SAPHNELO prefilled syringe or autoinjector can be stored at room temperature 68°F to 77°F (20°C to 25°C) for up to 7 days in the original carton to protect from light. After the SAPHNELO prefilled syringe or autoinjector has reached room temperature, do not return to the refrigerator. Discard SAPHNELO prefilled syringe or autoinjector if not used within 7 days at room temperature storage.
INSTRUCTIONS FOR USE SAPHNELO® (saf-NEL-low) (anifrolumab-fnia) injection, for subcutaneous use Single-dose prefilled syringe 120 mg/0.8 mL | ||
This Instructions for Use contains information on how to inject using SAPHNELO prefilled syringe. Read this Instructions for Use before you start using SAPHNELO prefilled syringe and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment. Your healthcare provider should show you or your caregiver how to use SAPHNELO prefilled syringe the right way. If you or your caregiver have any questions, talk to your healthcare provider. SAPHNELO prefilled syringe is for use under the skin (subcutaneous) only. | ||
Important storage information and warnings | ||
| ||
Do not use SAPHNELO prefilled syringe if it has:
Do not shake SAPHNELO prefilled syringe. | ||
If any of the above happens, throw away the prefilled syringe in an FDA-cleared sharps disposal container and use a new prefilled syringe. Keep SAPHNELO prefilled syringe and all medicines out of the sight and reach of children. | ||
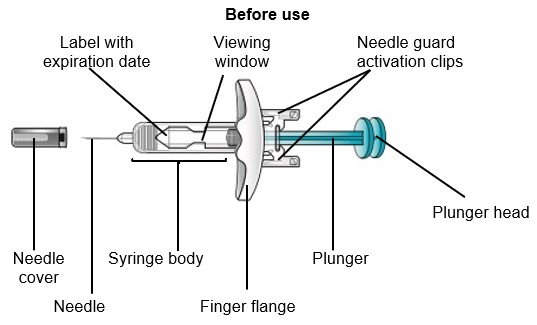
SAPHNELO prefilled syringe parts | ||
Do not remove the needle cover until right before injecting SAPHNELO. Do not touch the needle guard activation clips. This will keep you from activating the needle guard too soon. | ||
 |  | |
Preparing to inject using SAPHNELO prefilled syringe | ||
Step 1 – Gather supplies for your injection | ||
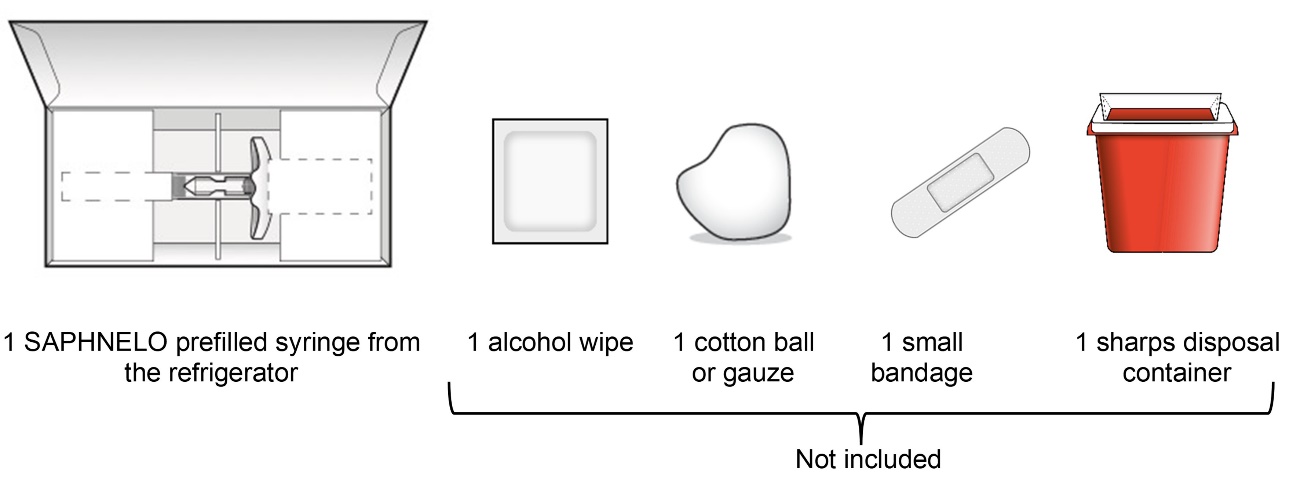 | ||
See Step 9 for instructions on how to throw away (dispose of) the used SAPHNELO prefilled syringe. | ||
Step 2 – Inspect the carton and wait 60 minutes | ||
Select a clean, well-lit, flat work surface, such as a table. Check the expiration date (EXP) on the carton.
Check the carton for damage.
| ||
Let SAPHNELO prefilled syringe come to room temperature for 60 minutes before injecting.
|  | |
Step 3 – Remove the prefilled syringe from the carton and inspect | ||
Open the carton and remove SAPHNELO prefilled syringe by holding the middle of the syringe body.
| 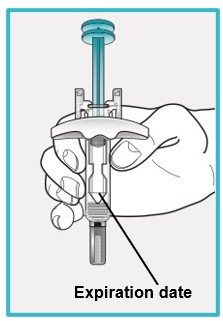 | |
Check the expiration date on the prefilled syringe.
| ||
Check the prefilled syringe for damage.
| ||
Check the liquid through the viewing window.
| ||
Injecting using your SAPHNELO prefilled syringe | ||
Step 4 – Choose an injection site | ||
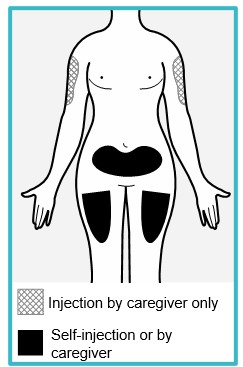
You or your caregiver can inject in the front of your thigh or the lower part of your stomach (abdomen). A caregiver may also inject your upper arm. Do not try to inject yourself in the upper arm. Choose an injection site that is at least 1-inch (3 cm) away from where you last injected. Do not inject:
|  | |
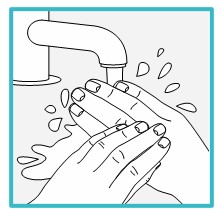
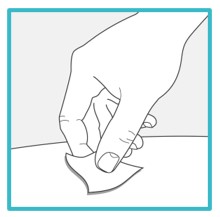
Step 5 – Wash your hands and clean the injection site | ||
Wash your hands well with soap and water. Clean the injection site with an alcohol wipe or with soap and water. Let the site air dry. | ||
|  |  |
Step 6 – Pull off the needle cover | ||
Hold the SAPHNELO prefilled syringe body with 1 hand and carefully pull the needle cover straight off with your other hand.
Go to Step 7 right away after removing the needle cover. | 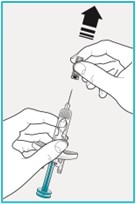 | |
Step 7 – Injecting SAPHNELO | ||
Hold SAPHNELO prefilled syringe in 1 hand as shown. Use your other hand to gently pinch and hold the cleaned injection site.
Inject using the SAPHNELO prefilled syringe by following the steps in figures a, b, and c. | 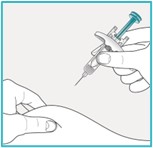 | |
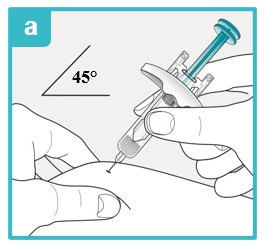 | 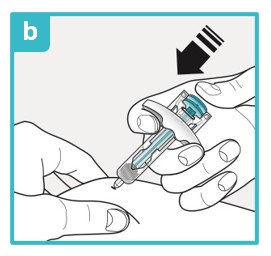 | 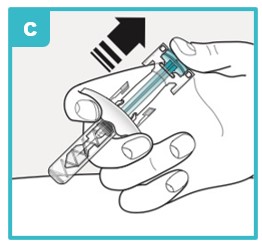 |
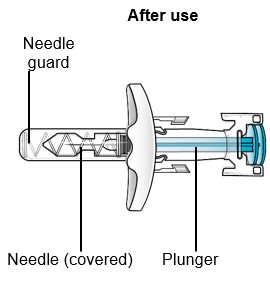
Using a 45 degree angle, fully insert the needle into the pinched skin. Do not reposition the SAPHNELO prefilled syringe after you insert the needle into the skin. | Use your thumb to push down the plunger head. To make sure you inject all the medicine and activate the needle guard, keep pushing firmly on the plunger head until it is fully down as far as it will go. | Slowly let go of the plunger head until the needle guard covers the needle. If the needle is not covered, carefully throw away (dispose of) the SAPHNELO prefilled syringe right away (see Step 9). |
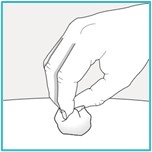
Step 8 – Check the injection site | ||
There may be a small amount of blood or liquid at the injection site. This is normal. If needed, press a cotton ball or gauze on the area and apply a small bandage.
|  | |
Step 9 – Throw away (dispose of) the used SAPHNELO prefilled syringe | ||
Each prefilled syringe contains 1 single-dose of SAPHNELO and cannot be used again. Do not recap the needle. Put your used SAPHNELO prefilled syringe in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) SAPHNELO prefilled syringe in your household trash. | 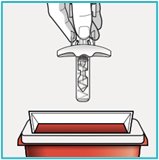 | |
If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
When your sharp disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA’s website at: http://www.fda.gov/safesharpsdisposal . Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container. Manufactured by: AstraZeneca AB, Södertälje, Sweden SE-15185 US License No. 2059 Distributed by: AstraZeneca Pharmaceuticals LP, Wilmington, DE 19850 ©AstraZeneca 2026 SAPHNELO is a registered trademark of AstraZeneca. For more information, go to https://www.SAPHNELO.com or call 1-800-236-9933. | ||
This Instructions for Use has been approved by the U.S. Food and Drug Administration Approved: April 2026 | ||
Mechanism of Action
Anifrolumab-fnia is a human IgG1κ monoclonal antibody that binds to subunit 1 of the type I interferon receptor (IFNAR) with high specificity and affinity. This binding inhibits type I IFN signaling, thereby blocking the biologic activity of type I IFNs. Anifrolumab-fnia also induces the internalization of IFNAR1, thereby reducing the levels of cell surface IFNAR1 available for receptor assembly. Blockade of receptor mediated type I IFN signaling inhibits IFN responsive gene expression as well as downstream inflammatory and immunological processes. Inhibition of type I IFN blocks plasma cell differentiation and normalizes peripheral T-cell subsets.
Type I IFNs play a role in the pathogenesis of SLE. Approximately 60-80% of adult patients with active SLE express elevated levels of type I IFN inducible genes.