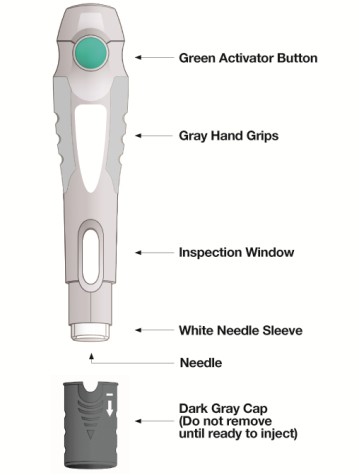
Get your patient on Skyrizi (Risankizumab-Rzaa)
Skyrizi prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Skyrizi patient education
Patient toolkit
Dosage & administration
2 DOSAGE AND ADMINISTRATION
- For the treatment of Crohn’s disease and ulcerative colitis: Obtain liver enzymes and bilirubin levels prior to initiating treatment with SKYRIZI. (2.1 , 5.4 )
- Complete all age-appropriate vaccinations as recommended by current immunization guidelines (2.1 , 5.5 )
Recommended Dosage
Plaque Psoriasis and Psoriatic Arthritis:
- 150 mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter. (2.3 , 2.4 )
- In patients with psoriatic arthritis SKYRIZI can be administered alone or in combination with non-biologic disease-modifying antirheumatic drugs (DMARDs). (2.4 )
Crohn’s Disease:
- The recommended induction dosage is 600 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8. The recommended maintenance dosage is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage to maintain therapeutic response. (2.6 )
Ulcerative Colitis:
- The recommended induction dosage is 1,200 mg administered by intravenous infusion over at least two hours at Week 0, Week 4, and Week 8. The recommended maintenance dosage is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage to maintain therapeutic response. (2.7 )
2.1 Procedures Prior to Treatment Initiation
- For the treatment of Crohn’s disease and ulcerative colitis, obtain liver enzymes and bilirubin levels prior to initiating treatment with SKYRIZI [see Warnings and Precautions (5.4 )]
- Evaluate patients for tuberculosis (TB) infection prior to initiating treatment with SKYRIZI [see Warnings and Precautions (5.3 )] .
- Complete all age-appropriate vaccinations as recommended by current immunization guidelines [see Warnings and Precautions (5.5 )].
2.2 General Considerations for Administration
• Visually inspect SKYRIZI for particulate matter and discoloration prior to administration. The solution may contain a few translucent to white particles.
○ SKYRIZI 150 mg/mL prefilled pen or prefilled syringe, 180 mg/1.2 mL prefilled syringe or prefilled cartridge, and 360 mg/2.4 mL prefilled cartridge: a colorless to yellow, and clear to slightly opalescent solution.
○ SKYRIZI 90 mg/mL prefilled syringe and 600 mg/10 mL vial: a colorless to slightly yellow, and clear to slightly opalescent solution.
○ Do not use if the solution contains large particles or is cloudy or discolored.
• Discard after use. Do not reuse.
2.000000000000000e+00 3 Recommended Dosage for Plaque Psoriasis
The recommended dosage is 150 mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter.
2.000000000000000e+00 4 Recommended Dosage for Psoriatic Arthritis
The recommended dosage is 150 mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter.
SKYRIZI may be administered alone or in combination with non-biologic disease-modifying antirheumatic drugs (DMARDs).
2.000000000000000e+00 5 Preparation and Administration Instruction s (Plaque Psoriasis and Psoriatic Arthritis)
- Administer SKYRIZI 150 mg/mL prefilled pen or prefilled syringe subcutaneously.
- Patients may self-inject SKYRIZI after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI.
- Before injecting, remove the carton with SKYRIZI from the refrigerator and without removing the prefilled pen or prefilled syringe from the carton, allow SKYRIZI to reach room temperature out of direct sunlight (30 to 90 minutes for the prefilled pen and 15 to 30 minutes for the prefilled syringe).
- Do not inject into areas where the skin is tender, bruised, erythematous, indurated or affected by psoriasis. Administration of SKYRIZI in the upper, outer arm may only be performed by a healthcare professional or caregiver.
- If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
The SKYRIZI “Instructions for Use” contains more detailed instructions on the preparation and administration of SKYRIZI [see Instructions for Use ] . Instruct the patient to read the Instructions for Use before administration.
2.6 Recommended Dosage for Crohn’s Disease
Adult Patients: Induction
The recommended induction dosage of SKYRIZI is 600 mg administered by intravenous infusion over a period of at least one hour at Week 0, Week 4, and Week 8.
Adult Patients: Maintenance
The recommended maintenance dosage of SKYRIZI is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage needed to maintain therapeutic response.
2.7 Recommended Dosage for Ulcerative Colitis
Adult Patients: Induction
The recommended induction dosage of SKYRIZI is 1,200 mg administered by intravenous infusion over a period of at least two hours at Week 0, Week 4, and Week 8.
Adult Patients: Maintenance
The recommended maintenance dosage of SKYRIZI is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage needed to maintain therapeutic response.
2.8 Preparation and Administration Instructions (Crohn’s Disease and Ulcerative Colitis)
Intravenous Induction Dosing Regimen:
1. SKYRIZI vial for intravenous administration is intended for administration by a healthcare provider using aseptic technique.
2. Prior to intravenous administration, determine the dose and number of SKYRIZI vials needed based on the patient’s indication (see table below). Withdraw 10 mL of SKYRIZI solution from a vial (600 mg/10 mL) and inject into an intravenous infusion bag or glass bottle containing 5% Dextrose Injection or 0.9% Sodium Chloride Injection (see Table 1 below) for a final concentration of approximately 1.2 mg/mL to 6 mg/mL. Discard any remaining solution in the vial.
Indication | I ntravenous Induction Dose | Number of SKYRIZI 600 mg/10 mL Vials | Total Volume of 5% Dextrose or 0.9% Sodium Chloride Injection |
| Crohn’s disease | 600 mg | 1 | 100 mL, or 250 mL, or 500 mL |
| Ulcerative colitis | 1,200 mg | 2 | 250 mL, or 500 mL |
3. Infuse the diluted solution intravenously over a period of at least one hour for the SKYRIZI 600 mg dose; at least two hours for the SKYRIZI 1,200 mg dose. If stored refrigerated, allow the diluted SKYRIZI solution in the infusion bag or glass bottle to warm to room temperature prior to the start of the intravenous infusion.
4. Do not administer SKYRIZI diluted solution concomitantly in the same intravenous line with other medicinal products.
Handling and Storage of the Vial and the Diluted Solution:
- Do not shake the vial or diluted solution in the infusion bag or glass bottle.
- Use the prepared infusion immediately. If not used immediately, store the diluted SKYRIZI solution refrigerated and protected from light for up to 20 hours between 36°F to 46°F (2°C to 8°C).
- Immediately after preparation or removal from refrigeration, the diluted SKYRIZI solution can be stored at room temperature at up to 77°F (25°C) (protected from sunlight) for up to 8 hours. Storage time at room temperature begins once the diluted solution has been prepared. The infusion should be completed within 8 hours after dilution in the infusion bag.
- Exposure to indoor light is acceptable during room temperature storage and administration.
- Do not freeze.
Subcutaneous Maintenance Dosing Regimen:
Using the single-dose 180 mg or 360 mg prefilled cartridge with On-Body Injector:
- SKYRIZI is intended for use under the guidance and supervision of a healthcare professional.
- Patients may self-inject SKYRIZI using the on-body injector with prefilled cartridge after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI.
- Before using the on-body injector with prefilled cartridge, remove the carton from the refrigerator and allow to reach room temperature out of direct sunlight (45 to 90 minutes) without removing the prefilled cartridge or on-body injector from the carton.
- Use the on-body injector to administer SKYRIZI 180 mg/1.2 mL or SKYRIZI 360 mg/2.4 mL prefilled cartridge subcutaneously on thigh or abdomen.
- Start the injection within 5 minutes after inserting the prefilled cartridge into the On-Body Injector.
- Do not inject into areas where the skin is tender, bruised, erythematous, indurated or affected by any lesions.
- If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
The SKYRIZI “Instructions for Use” contains more detailed instructions on the preparation and administration of SKYRIZI [see Instructions for Use ] . Instruct the patient to read the Instructions for Use before administration.
Using the 90 mg/mL or 180 mg/1.2 mL prefilled syringe:
• Administer each SKYRIZI 90 mg/mL or 180 mg/1.2 mL prefilled syringe subcutaneously.
• Patients may self-inject SKYRIZI after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI.
• Before injecting, remove the carton from the refrigerator and without removing the prefilled syringes from the carton, allow SKYRIZI to reach room temperature out of direct sunlight (15 to 30 minutes).
• Use the 90 mg/mL or 180 mg/1.2 mL prefilled syringe(s) to administer SKYRIZI 180 mg or SKYRIZI 360 mg subcutaneously as follows:
○ 180 mg maintenance dose:
▪ SKYRIZI 90 mg/mL prefilled syringes: Two 90 mg prefilled syringes are required. Inject one prefilled syringe after the other in different anatomic locations (such as thighs or abdomen).
▪ SKYRIZI 180 mg/1.2 mL prefilled syringe: One 180 mg prefilled syringe is required.
○ 360 mg maintenance dose:
▪ SKYRIZI 90 mg/mL prefilled syringes: Four 90 mg prefilled syringes are required. Inject one prefilled syringe after the other in different anatomic locations (such as thighs or abdomen).
▪ SKYRIZI 180 mg/1.2 mL prefilled syringes: Two 180 mg prefilled syringes are required. Inject one prefilled syringe after the other in different anatomic locations (such as thighs or abdomen).
• Do not inject into areas where the skin is tender, bruised, erythematous, indurated or affected by any lesions. Administration of SKYRIZI in the upper, outer arm may only be performed by a healthcare professional or caregiver.
• If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
The SKYRIZI “Instructions for Use” contains more detailed instructions on the preparation and administration of SKYRIZI [see Instructions for Use ] . Instruct the patient to read the Instructions for Use before administration.

Skyrizi prescribing information
1 INDICATIONS AND USAGE
SKYRIZI is an interleukin-23 antagonist indicated for the treatment of:
1.1 Plaque Psoriasis
SKYRIZI ® is indicated for the treatment of moderate-to-severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy.
1.2 Psoriatic Arthritis
SKYRIZI is indicated for the treatment of active psoriatic arthritis in adults.
1.3 Crohn’s Disease
SKYRIZI is indicated for the treatment of moderately to severely active Crohn's disease in adults.
1.4 Ulcerative Colitis
SKYRIZI is indicated for the treatment of moderately to severely active ulcerative colitis in adults.
2 DOSAGE AND ADMINISTRATION
- For the treatment of Crohn’s disease and ulcerative colitis: Obtain liver enzymes and bilirubin levels prior to initiating treatment with SKYRIZI. (2.1 , 5.4 )
- Complete all age-appropriate vaccinations as recommended by current immunization guidelines (2.1 , 5.5 )
Recommended Dosage
Plaque Psoriasis and Psoriatic Arthritis:
- 150 mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter. (2.3 , 2.4 )
- In patients with psoriatic arthritis SKYRIZI can be administered alone or in combination with non-biologic disease-modifying antirheumatic drugs (DMARDs). (2.4 )
Crohn’s Disease:
- The recommended induction dosage is 600 mg administered by intravenous infusion over at least one hour at Week 0, Week 4, and Week 8. The recommended maintenance dosage is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage to maintain therapeutic response. (2.6 )
Ulcerative Colitis:
- The recommended induction dosage is 1,200 mg administered by intravenous infusion over at least two hours at Week 0, Week 4, and Week 8. The recommended maintenance dosage is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage to maintain therapeutic response. (2.7 )
2.1 Procedures Prior to Treatment Initiation
- For the treatment of Crohn’s disease and ulcerative colitis, obtain liver enzymes and bilirubin levels prior to initiating treatment with SKYRIZI [see Warnings and Precautions (5.4 )]
- Evaluate patients for tuberculosis (TB) infection prior to initiating treatment with SKYRIZI [see Warnings and Precautions (5.3 )] .
- Complete all age-appropriate vaccinations as recommended by current immunization guidelines [see Warnings and Precautions (5.5 )].
2.2 General Considerations for Administration
• Visually inspect SKYRIZI for particulate matter and discoloration prior to administration. The solution may contain a few translucent to white particles.
○ SKYRIZI 150 mg/mL prefilled pen or prefilled syringe, 180 mg/1.2 mL prefilled syringe or prefilled cartridge, and 360 mg/2.4 mL prefilled cartridge: a colorless to yellow, and clear to slightly opalescent solution.
○ SKYRIZI 90 mg/mL prefilled syringe and 600 mg/10 mL vial: a colorless to slightly yellow, and clear to slightly opalescent solution.
○ Do not use if the solution contains large particles or is cloudy or discolored.
• Discard after use. Do not reuse.
2.000000000000000e+00 3 Recommended Dosage for Plaque Psoriasis
The recommended dosage is 150 mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter.
2.000000000000000e+00 4 Recommended Dosage for Psoriatic Arthritis
The recommended dosage is 150 mg administered by subcutaneous injection at Week 0, Week 4, and every 12 weeks thereafter.
SKYRIZI may be administered alone or in combination with non-biologic disease-modifying antirheumatic drugs (DMARDs).
2.000000000000000e+00 5 Preparation and Administration Instruction s (Plaque Psoriasis and Psoriatic Arthritis)
- Administer SKYRIZI 150 mg/mL prefilled pen or prefilled syringe subcutaneously.
- Patients may self-inject SKYRIZI after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI.
- Before injecting, remove the carton with SKYRIZI from the refrigerator and without removing the prefilled pen or prefilled syringe from the carton, allow SKYRIZI to reach room temperature out of direct sunlight (30 to 90 minutes for the prefilled pen and 15 to 30 minutes for the prefilled syringe).
- Do not inject into areas where the skin is tender, bruised, erythematous, indurated or affected by psoriasis. Administration of SKYRIZI in the upper, outer arm may only be performed by a healthcare professional or caregiver.
- If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
The SKYRIZI “Instructions for Use” contains more detailed instructions on the preparation and administration of SKYRIZI [see Instructions for Use ] . Instruct the patient to read the Instructions for Use before administration.
2.6 Recommended Dosage for Crohn’s Disease
Adult Patients: Induction
The recommended induction dosage of SKYRIZI is 600 mg administered by intravenous infusion over a period of at least one hour at Week 0, Week 4, and Week 8.
Adult Patients: Maintenance
The recommended maintenance dosage of SKYRIZI is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage needed to maintain therapeutic response.
2.7 Recommended Dosage for Ulcerative Colitis
Adult Patients: Induction
The recommended induction dosage of SKYRIZI is 1,200 mg administered by intravenous infusion over a period of at least two hours at Week 0, Week 4, and Week 8.
Adult Patients: Maintenance
The recommended maintenance dosage of SKYRIZI is 180 mg or 360 mg administered by subcutaneous injection at Week 12, and every 8 weeks thereafter. Use the lowest effective dosage needed to maintain therapeutic response.
2.8 Preparation and Administration Instructions (Crohn’s Disease and Ulcerative Colitis)
Intravenous Induction Dosing Regimen:
1. SKYRIZI vial for intravenous administration is intended for administration by a healthcare provider using aseptic technique.
2. Prior to intravenous administration, determine the dose and number of SKYRIZI vials needed based on the patient’s indication (see table below). Withdraw 10 mL of SKYRIZI solution from a vial (600 mg/10 mL) and inject into an intravenous infusion bag or glass bottle containing 5% Dextrose Injection or 0.9% Sodium Chloride Injection (see Table 1 below) for a final concentration of approximately 1.2 mg/mL to 6 mg/mL. Discard any remaining solution in the vial.
| Indication | I ntravenous Induction Dose | Number of SKYRIZI 600 mg/10 mL Vials | Total Volume of 5% Dextrose or 0.9% Sodium Chloride Injection |
| Crohn’s disease | 600 mg | 1 | 100 mL, or 250 mL, or 500 mL |
| Ulcerative colitis | 1,200 mg | 2 | 250 mL, or 500 mL |
3. Infuse the diluted solution intravenously over a period of at least one hour for the SKYRIZI 600 mg dose; at least two hours for the SKYRIZI 1,200 mg dose. If stored refrigerated, allow the diluted SKYRIZI solution in the infusion bag or glass bottle to warm to room temperature prior to the start of the intravenous infusion.
4. Do not administer SKYRIZI diluted solution concomitantly in the same intravenous line with other medicinal products.
Handling and Storage of the Vial and the Diluted Solution:
- Do not shake the vial or diluted solution in the infusion bag or glass bottle.
- Use the prepared infusion immediately. If not used immediately, store the diluted SKYRIZI solution refrigerated and protected from light for up to 20 hours between 36°F to 46°F (2°C to 8°C).
- Immediately after preparation or removal from refrigeration, the diluted SKYRIZI solution can be stored at room temperature at up to 77°F (25°C) (protected from sunlight) for up to 8 hours. Storage time at room temperature begins once the diluted solution has been prepared. The infusion should be completed within 8 hours after dilution in the infusion bag.
- Exposure to indoor light is acceptable during room temperature storage and administration.
- Do not freeze.
Subcutaneous Maintenance Dosing Regimen:
Using the single-dose 180 mg or 360 mg prefilled cartridge with On-Body Injector:
- SKYRIZI is intended for use under the guidance and supervision of a healthcare professional.
- Patients may self-inject SKYRIZI using the on-body injector with prefilled cartridge after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI.
- Before using the on-body injector with prefilled cartridge, remove the carton from the refrigerator and allow to reach room temperature out of direct sunlight (45 to 90 minutes) without removing the prefilled cartridge or on-body injector from the carton.
- Use the on-body injector to administer SKYRIZI 180 mg/1.2 mL or SKYRIZI 360 mg/2.4 mL prefilled cartridge subcutaneously on thigh or abdomen.
- Start the injection within 5 minutes after inserting the prefilled cartridge into the On-Body Injector.
- Do not inject into areas where the skin is tender, bruised, erythematous, indurated or affected by any lesions.
- If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
The SKYRIZI “Instructions for Use” contains more detailed instructions on the preparation and administration of SKYRIZI [see Instructions for Use ] . Instruct the patient to read the Instructions for Use before administration.
Using the 90 mg/mL or 180 mg/1.2 mL prefilled syringe:
• Administer each SKYRIZI 90 mg/mL or 180 mg/1.2 mL prefilled syringe subcutaneously.
• Patients may self-inject SKYRIZI after training in subcutaneous injection technique. Provide proper training to patients and/or caregivers on the subcutaneous injection technique of SKYRIZI.
• Before injecting, remove the carton from the refrigerator and without removing the prefilled syringes from the carton, allow SKYRIZI to reach room temperature out of direct sunlight (15 to 30 minutes).
• Use the 90 mg/mL or 180 mg/1.2 mL prefilled syringe(s) to administer SKYRIZI 180 mg or SKYRIZI 360 mg subcutaneously as follows:
○ 180 mg maintenance dose:
▪ SKYRIZI 90 mg/mL prefilled syringes: Two 90 mg prefilled syringes are required. Inject one prefilled syringe after the other in different anatomic locations (such as thighs or abdomen).
▪ SKYRIZI 180 mg/1.2 mL prefilled syringe: One 180 mg prefilled syringe is required.
○ 360 mg maintenance dose:
▪ SKYRIZI 90 mg/mL prefilled syringes: Four 90 mg prefilled syringes are required. Inject one prefilled syringe after the other in different anatomic locations (such as thighs or abdomen).
▪ SKYRIZI 180 mg/1.2 mL prefilled syringes: Two 180 mg prefilled syringes are required. Inject one prefilled syringe after the other in different anatomic locations (such as thighs or abdomen).
• Do not inject into areas where the skin is tender, bruised, erythematous, indurated or affected by any lesions. Administration of SKYRIZI in the upper, outer arm may only be performed by a healthcare professional or caregiver.
• If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regular scheduled time.
The SKYRIZI “Instructions for Use” contains more detailed instructions on the preparation and administration of SKYRIZI [see Instructions for Use ] . Instruct the patient to read the Instructions for Use before administration.
3 DOSAGE FORMS AND STRENGTHS
Subcutaneous Injection
SKYRIZI Pen
- Injection: 150 mg/mL as a colorless to yellow and clear to slightly opalescent solution in each single-dose prefilled pen.
SKYRIZI Prefilled Syringe
- Injection: 90 mg/mL as a colorless to slightly yellow and clear to slightly opalescent solution in each single-dose prefilled syringe.
- Injection: 150 mg/mL as a colorless to yellow and clear to slightly opalescent solution in each single-dose prefilled syringe.
- Injection: 180 mg/1.2 mL (150 mg/mL) as a colorless to yellow and clear to slightly opalescent solution in each single-dose prefilled syringe.
SKYRIZI Prefilled Cartridge with Supplied On-Body Injector
- Injection: 180 mg/1.2 mL (150 mg/mL) as a colorless to yellow, and clear to slightly opalescent solution in each single-dose prefilled cartridge for use with the on-body injector.
- Injection: 360 mg/2.4 mL (150 mg/mL) as a colorless to yellow, and clear to slightly opalescent solution in each single-dose prefilled cartridge for use with the on-body injector.
Intravenous Infusion
SKYRIZI Vial
- Injection: 600 mg/10 mL (60 mg/mL) as a colorless to slightly yellow, and clear to slightly opalescent solution in each single-dose vial.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors outcomes in women who become pregnant while treated with SKYRIZI. Patients should be encouraged to enroll by calling 1-877-302-2161 or visiting http://glowpregnancyregistry.com.
Risk Summary
Available pharmacovigilance and clinical trial data with risankizumab use in pregnant women are insufficient to establish a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes. Although there are no data on risankizumab-rzaa, monoclonal antibodies can be actively transported across the placenta, and SKYRIZI may cause immunosuppression in the in utero - exposed infant . There are adverse pregnancy outcomes in women with inflammatory bowel disease (see Clinical Considerations) .
In an enhanced pre- and post-natal developmental toxicity study, pregnant cynomolgus monkeys were administered subcutaneous doses of 5 or 50 mg/kg risankizumab-rzaa once weekly during the period of organogenesis up to parturition. Increased fetal/infant loss was noted in pregnant monkeys at the 50 mg/kg dose (see Data) . The 50 mg/kg dose in pregnant monkeys resulted in approximately 5 times the exposure (AUC) in humans administered the maximum recommended induction dose (1,200 mg) and 32 times the exposure (AUC) to the maximum recommended maintenance dose (360 mg). No risankizumab-rzaa-related effects on functional or immunological development were observed in infant monkeys from birth through 6 months of age. The clinical significance of these findings for humans is unknown.
All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-associated maternal and embryo/fetal risk
Published data suggest that the risk of adverse pregnancy outcomes in women with inflammatory bowel disease is associated with increased disease activity. Adverse pregnancy outcomes include preterm delivery (before 37 weeks of gestation), low birth weight (less than 2,500 g) infants, and small for gestational age at birth.
Fetal/Neonatal adverse reactions
Transport of endogenous IgG antibodies across the placenta increases as pregnancy progresses, and peaks during the third trimester. Therefore, SKYRIZI may be present in infants exposed in utero . The potential clinical impact of risankizumab exposure in infants exposed in utero should be considered.
Data
Animal Data
An enhanced pre- and post-natal developmental toxicity study was conducted in cynomolgus monkeys. Pregnant cynomolgus monkeys were administered weekly subcutaneous doses of risankizumab-rzaa of 5 or 50 mg/kg from gestation day 20 to parturition, and the cynomolgus monkeys (mother and infants) were monitored for 6 months after delivery. No maternal toxicity was noted in this study. There were no treatment-related effects on growth and development, malformations, developmental immunotoxicology, or neurobehavioral development. However, a dose-dependent increase in fetal/infant loss was noted in the risankizumab-rzaa-treated groups (32% and 43% in the 5 mg/kg and 50 mg/kg groups, respectively) compared with the vehicle control group (19%). The increased fetal/infant loss in the 50 mg/kg group was considered to be related to risankizumab-rzaa treatment. The no-observed adverse effect level (NOAEL) for maternal toxicity was identified as 50 mg/kg, and the NOAEL for developmental toxicity was identified as 5 mg/kg. The 5 mg/kg dose in pregnant monkeys resulted in approximately 0.6 times the exposure (AUC) in humans administered the maximum recommended induction dose (1,200 mg) and 5 times the exposure (AUC) in humans administered the maximum recommended maintenance dose (360 mg). In the infants, mean serum concentrations increased in a dose-dependent manner and were approximately 17%-86% of the respective maternal concentrations. Following delivery, most adult female cynomolgus monkeys and all infants from the risankizumab-rzaa-treated groups had measurable serum concentrations of risankizumab-rzaa up to 91 days postpartum. Serum concentrations were below detectable levels at 180 days postpartum.
8.2 Lactation
Risk Summary
There are no data on the presence of risankizumab-rzaa in human milk, the effects on the breastfed infant, or the effects on milk production. Endogenous maternal IgG and monoclonal antibodies are transferred in human milk. The effects of local gastrointestinal exposure and limited systemic exposure in the breastfed infant to risankizumab-rzaa are unknown. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for SKYRIZI and any potential adverse effects on the breastfed infant from SKYRIZI or from the underlying maternal condition.
8.4 Pediatric Use
The safety and effectiveness of SKYRIZI have not been established in pediatric patients.
8.5 Geriatric Use
Of the 6,862 subjects exposed to SKYRIZI, a total of 664 were 65 years and older (243 subjects with plaque psoriasis, 246 subjects with psoriatic arthritis, 72 subjects with Crohn’s disease and 103 subjects with ulcerative colitis), and 71 subjects were 75 years and older.
Clinical studies of SKYRIZI, within each indication, did not include sufficient numbers of subjects 65 years of age and older to determine whether they respond differently from younger adult subjects.
No clinically meaningful differences in the pharmacokinetics of risankizumab-rzaa were observed based on age [see Clinical Pharmacology (12.3 )] .
4 CONTRAINDICATIONS
SKYRIZI is contraindicated in patients with a history of serious hypersensitivity reaction to risankizumab-rzaa or any of the excipients [see Warnings and Precautions (5.1 )] .
5 WARNINGS AND PRECAUTIONS
- Hypersensitivity Reactions: Serious hypersensitivity reactions, including anaphylaxis, may occur. (5.1 )
- Infections: SKYRIZI may increase the risk of infection. Instruct patients to seek medical advice if signs or symptoms of clinically important infection occur. If such an infection develops, do not administer SKYRIZI until the infection resolves. (5.2 )
- Tuberculosis (TB): Evaluate for TB prior to initiating treatment with SKYRIZI. (5.3 )
- Hepatotoxicity in Treatment of Inflammatory Bowel Disease: Drug-induced liver injury during induction has been reported. Monitor liver enzymes and bilirubin levels at baseline and, during induction, up to at least 12 weeks of treatment. Monitor thereafter according to routine patient management. (5.4 )
- Administration of Vaccines: Avoid use of live vaccines. (5.5 )
5.1 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, have been reported with use of SKYRIZI. If a serious hypersensitivity reaction occurs, discontinue SKYRIZI and initiate appropriate therapy immediately [see Adverse Reactions (6.1 )].
5.000000000000000e+00 2 Infections
SKYRIZI may increase the risk of infections [see Adverse Reactions (6.1 )] .
Treatment with SKYRIZI should not be initiated in patients with any clinically important active infection until the infection resolves or is adequately treated.
In patients with a chronic infection or a history of recurrent infection, consider the risks and benefits prior to prescribing SKYRIZI. Instruct patients to seek medical advice if signs or symptoms of clinically important infection occur. If a patient develops such an infection or is not responding to standard therapy, monitor the patient closely and do not administer SKYRIZI until the infection resolves.
5.000000000000000e+00 3 Tuberculosis
Evaluate patients for tuberculosis (TB) infection prior to initiating treatment with SKYRIZI. Across the Phase 3 psoriasis clinical studies, of the 72 subjects with latent TB who were concurrently treated with SKYRIZI and appropriate TB prophylaxis during the studies, none developed active TB during the mean follow-up of 61 weeks on SKYRIZI. Two subjects taking isoniazid for treatment of latent TB discontinued treatment due to liver injury. Of the 31 subjects from the PsO-3 study with latent TB who did not receive prophylaxis during the study, none developed active TB during the mean follow-up of 55 weeks on SKYRIZI. Consider anti-TB therapy prior to initiating SKYRIZI in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed. Monitor patients for signs and symptoms of active TB during and after SKYRIZI treatment. Do not administer SKYRIZI to patients with active TB.
5.000000000000000e+00 4 Hepatotoxicity in Treatment of Inflammatory Bowel Disease
A serious adverse reaction of drug-induced liver injury in conjunction with a rash that required hospitalization was reported in a patient with Crohn’s disease (ALT 54x ULN, AST 30x ULN, and total bilirubin 2.2x ULN) following two 600 mg intravenous doses of SKYRIZI. The liver test abnormalities resolved following administration of steroids. SKYRIZI was subsequently discontinued.
For the treatment of Crohn’s disease and ulcerative colitis, evaluate liver enzymes and bilirubin at baseline, and during induction at least up to 12 weeks of treatment. Monitor thereafter according to routine patient management.
Consider other treatment options in patients with evidence of liver cirrhosis. Prompt investigation of the cause of liver enzyme elevation is recommended to identify potential cases of drug-induced liver injury. Interrupt treatment if drug-induced liver injury is suspected, until this diagnosis is excluded. Instruct patients to seek immediate medical attention if they experience symptoms suggestive of hepatic dysfunction.
5.5 Administration of Vaccines
Avoid use of live vaccines in patients treated with SKYRIZI. Medications that interact with the immune system may increase the risk of infection following administration of live vaccines. Prior to initiating therapy with SKYRIZI, complete all age-appropriate vaccinations according to current immunization guidelines. No data are available on the response to live or inactive vaccines.
6 ADVERSE REACTIONS
The following adverse reactions are discussed in other sections of labeling:
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse drug reaction rates observed in the clinical trials of a drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Plaque Psoriasis
A total of 2234 subjects were treated with SKYRIZI in clinical development trials in plaque psoriasis. Of these, 1208 subjects with psoriasis were exposed to SKYRIZI for at least one year.
Data from placebo- and active-controlled trials were pooled to evaluate the safety of SKYRIZI for up to 16 weeks. In total, 1306 subjects were evaluated in the SKYRIZI 150 mg group.
Table 2 summarizes the adverse drug reactions that occurred at a rate of at least 1% and at a higher rate in the SKYRIZI group than the placebo group during the 16-week controlled period of pooled clinical trials.
| Adverse Drug Reactions | SKYRIZI N = 1306 n (%) | Placebo N = 300 n (%) |
| Upper respiratory infections a | 170 (13.0) | 29 (9.7) |
| Headache b | 46 (3.5) | 6 (2.0) |
| Fatigue c | 33 (2.5) | 3 (1.0) |
| Injection site reactions d | 19 (1.5) | 3 (1.0) |
| Tinea infections e | 15 (1.1) | 1 (0.3) |
| a Includes: respiratory tract infection (viral, bacterial or unspecified), sinusitis (including acute), rhinitis, nasopharyngitis, pharyngitis (including viral), tonsillitis b Includes: headache, tension headache, sinus headache, cervicogenic headache c Includes: fatigue, asthenia d Includes: injection site bruising, erythema, extravasation, hematoma, hemorrhage, infection, inflammation, irritation, pain, pruritus, reaction, swelling, warmth e Includes: tinea pedis, tinea cruris, body tinea, tinea versicolor, tinea manuum, tinea infection, onychomycosis | ||
Adverse drug reactions that occurred in < 1% but > 0.1% of subjects in the SKYRIZI group and at a higher rate than in the placebo group through Week 16 were folliculitis and urticaria.
Specific Adverse Drug Reactions
Infections
In the first 16 weeks, infections occurred in 22.1% of the SKYRIZI group (90.8 events per 100 patient-years) compared with 14.7% of the placebo group (56.5 events per 100 patient-years) and did not lead to discontinuation of SKYRIZI. The rates of serious infections for the SKYRIZI group and the placebo group were ≤0.4%. Serious infections in the SKYRIZI group included cellulitis, osteomyelitis, sepsis, and herpes zoster. In Trials PsO-1 and PsO-2, through Week 52, the rate of infections (73.9 events per 100 patient-years) was similar to the rate observed during the first 16 weeks of treatment.
SafetyThrough Week 52
Through Week 52, no new adverse reactions were identified, and the rates of the adverse reactions were similar to those observed during the first 16 weeks of treatment. During this period, serious infections that led to trial discontinuation included pneumonia.
Plaque Psoriasisofthe Scalp or Genital Area
The overall safety profile observed in clinical trials of subjects with moderate to severe plaque psoriasis of the scalp or genital area treated with SKYRIZI is generally consistent with the safety profile observed in previous clinical trials of subjects with moderate to severe plaque psoriasis [see Clinical Studies (14.1 )] .
Psoriatic Arthritis
The overall safety profile observed in subjects with psoriatic arthritis treated with SKYRIZI is generally consistent with the safety profile in subjects with plaque psoriasis. Additionally, in the Phase 3 placebo-controlled trials the incidence of hepatic events was higher in the SKYRIZI group (5.4%, 16.7 events per 100 patient-years) compared to the placebo group (3.9%, 12.6 events per 100 patient-years). Of these, the most common events that were reported more frequently in both the placebo group and the SKYRIZI group were ALT increased (placebo: n=12 (1.7%); SKYRIZI: n=16 (2.3%)), AST increased (placebo: n=9 (1.3%); SKYRIZI: n=13 (1.8%)), and GGT increased (placebo: n=5 (0.7%); SKYRIZI: n=8 (1.1%)). There were no serious hepatic events reported. The incidence of hypersensitivity reactions was higher in the SKYRIZI group (n=16, 2.3%) compared to the placebo group (n=9, 1.3%). In the Phase 3 placebo-controlled trials, hypersensitivity reactions reported at a higher rate in the SKYRIZI group included rash (placebo: n=4 (0.6%); SKYRIZI: n=5 (0.7%), allergic rhinitis (placebo: n=1 (0.1%); SKYRIZI: n=2 (0.3%), and facial swelling (placebo: n=0 (0.0%); SKYRIZI n=1 (0.1%). One case of anaphylaxis was reported in a subject who received SKYRIZI in the Phase 2 clinical trial.
Crohn’s Disease
SKYRIZI was studied up to 12 weeks in subjects with moderately to severely active Crohn’s disease in two randomized, double-blind, placebo-controlled induction trials (CD-1, CD-2) and a randomized, double-blind, placebo-controlled, dose-finding trial (CD-4; NCT02031276). Long-term safety up to 52 weeks was evaluated in subjects who responded to induction therapy in a randomized, double-blind, placebo-controlled maintenance trial (CD-3) [see Clinical Studies (14.3 )] .
In the two induction trials (CD-1, CD-2) and the dose finding trial (CD-4), 620 subjects received the SKYRIZI intravenous induction regimen at Weeks 0, 4 and 8. In the maintenance trial (CD-3), 297 subjects who achieved clinical response, defined as a reduction in CDAI of at least 100 points from baseline after 12 weeks of induction treatment with intravenous SKYRIZI in trials CD-1 and CD-2, received a maintenance regimen of SKYRIZI either 180 mg or 360 mg subcutaneously at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks.
Adverse reactions reported in > 3% of subjects in induction trials and at a higher rate than placebo are shown in Table 3.
| Adverse Drug Reactions | SKYRIZI 600 mg Intravenous Infusion a N = 620 n (%) | Placebo N = 432 n (%) |
| Upper respiratory infections b | 66 (10.6) | 40 (9.3) |
| Headache c | 41 (6.6) | 24 (5.6) |
| Arthralgia | 31 (5.0) | 19 (4.4) |
| a SKYRIZI 600 mg as an intravenous infusion at Week 0, Week 4, and Week 8. b Includes: influenza like illness, nasopharyngitis, influenza, pharyngitis, upper respiratory tract infection, viral upper respiratory tract infection, COVID-19, nasal congestion, respiratory tract infection viral, viral pharyngitis, tonsillitis, upper respiratory tract inflammation c Includes: headache, tension headache | ||
Adverse reactions reported in >3% of subjects in the maintenance trial and at a higher rate than placebo are shown in Table 4.
| Adverse Drug Reactions | SKYRIZI 180 mg Subcutaneous Injection N = 155 n (%) | SKYRIZI 360 mg Subcutaneous Injection N = 142 n (%) | Placebo N = 143 n (%) |
| Arthralgia | 13 (8.4) | 13 (9.2) | 12 (8.4) |
| Abdominal pain b | 9 (5.8) | 12 (8.5) | 6 (4.2) |
| Injection site reactions c,d | 7 (4.5) | 8 (5.6) | 4 (2.8) |
| Anemia | 7 (4.5) | 7 (4.9) | 6 (4.2) |
| Pyrexia | 4 (2.6) | 7 (4.9) | 4 (2.8) |
| Back pain | 3 (1.9) | 6 (4.2) | 3 (2.1) |
| Arthropathy | 1 (0.6) | 5 (3.5) | 2 (1.4) |
| Urinary tract infection | 1 (0.6) | 5 (3.5) | 4 (2.8) |
| a SKYRIZI 180 mg or 360 mg at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks b Includes: abdominal pain, abdominal pain upper, abdominal pain lower c Includes: injection site rash, injection site erythema, injection site swelling, injection site urticaria, injection site warmth, injection site pain, injection site hypersensitivity, injection site reaction d Some subjects had multiple occurrences of injection site reactions. In this table, injection site reactions are counted only once per subject for the rate calculations. | |||
Specific Adverse Drug Reactions
Infections
In the maintenance trial (CD-3) through Week 52, the rate of infections was 32.3% (50.2 events per 100 patient-years) in subjects who received SKYRIZI 180 mg and 36.6% (60.8 events per 100 patient-years) in subjects who received SKYRIZI 360 mg compared to 36.4% (60.3 events per 100 patient-years) in subjects who received placebo after SKYRIZI induction. The rate of serious infections was 2.6% (2.7 events per 100 patient-years) in subjects who received SKYRIZI 180 mg and 5.6% (7.4 events per 100 patient-years) in subjects who received SKYRIZI 360 mg compared to 2.1% (2.4 events per 100 patient-years) in subjects who received placebo after SKYRIZI induction.
Lipid Elevations
Elevations in lipid parameters (total cholesterol and low-density lipoprotein cholesterol [LDL-C]) were first assessed at 4 weeks following initiation of SKYRIZI in the induction trials (CD-1, CD-2). Increases from baseline and increases relative to placebo were observed at Week 4 and remained stable to Week 12. Following SKYRIZI induction, mean total cholesterol increased by 9.4 mg/dL from baseline to a mean absolute value of 175.1 mg/dL at Week 12. Similarly, mean LDL-C increased by 6.6 mg/dL from baseline to a mean absolute value of 92.6 mg/dL at Week 12. Mean LDL-C increased by 3.1 mg/dL from baseline to a mean absolute value of 99.0 mg/dL at Week 52 with SKYRIZI 180 mg maintenance treatment and by 2.3 mg/dL from baseline to a mean absolute value of 102.2 mg/dL at Week 52 with SKYRIZI 360 mg maintenance treatment (CD-3).
Ulcerative Colitis
SKYRIZI was studied up to 12 weeks in subjects with moderately to severely active ulcerative colitis in a randomized, double-blind, placebo-controlled induction trial (UC-1) and a randomized, double-blind, placebo-controlled, dose-finding trial (UC-3). Long-term safety up to 52 weeks was evaluated in subjects who responded to induction therapy in a randomized, double-blind, placebo-controlled maintenance trial (UC-2) [see Clinical Studies (14.4 )].
In the induction trials (UC-1 and UC-3), 712 subjects received the SKYRIZI 1,200 mg intravenous induction regimen at Weeks 0, 4 and 8. In the maintenance trial (UC-2), 347 subjects who achieved clinical response, defined as a decrease in mMS of ≥2 points and ≥30% from baseline and a decrease in RBS ≥1 from baseline or an absolute RBS ≤1, received a maintenance regimen of SKYRIZI either 180 mg or 360 mg subcutaneously at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks.
The adverse reaction reported in ≥3% subjects treated with SKYRIZI in the ulcerative colitis induction trials (UC-1 and UC-3) and at a higher rate than placebo was arthralgia (3% SKYRIZI vs 1% placebo).
Adverse reactions reported in ≥3% of subjects treated with SKYRIZI in the maintenance trial (UC-2) and at a higher rate than placebo are shown in Table 5.
| Adverse Drug Reactions | SKYRIZI 180 mg Subcutaneous Injection N = 170 n (%) | SKYRIZI 360 mg Subcutaneous Injection N = 177 n (%) | Placebo N = 173 n (%) |
| Arthralgia | 9 (5.3) | 17 (9.6) | 8 (4.6) |
| Pyrexia | 8 (4.7) | 7 (4.0) | 6 (3.5) |
| Injection site reactions b,c | 5 (2.9) | 5 (2.8) | 2 (1.2) |
| Rash d | 7 (4.1) | 1 (0.6) | 3 (1.7) |
| a SKYRIZI 180 mg or 360 mg at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks b Includes: application site pain, injection site erythema, injection site pain, injection site pruritus, injection site reaction c Some subjects had multiple occurrences of injection site reactions. In this table, injection site reactions are counted only once per subject for the rate calculations. d Includes: rash and rash macular | |||
Specific Adverse Drug Reactions
The rates of infections, serious infections, and lipid elevations in subjects with UC who received SKYRIZI compared to subjects who received placebo in the induction trials (UC-1 and UC-3) and maintenance trial (UC-2) were similar to the rates in subjects with CD who received SKYRIZI compared to subjects who received placebo in the induction trials (CD-1, CD-2, and CD-4) and maintenance trial (CD-3).
6.2 Immunogenicity
The observed incidence of anti-drug antibodies is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies (ADA) in the trials described below with the incidence of ADA in other trials, including those of SKYRIZI (risankizumab).
Plaque Psoriasis
By Week 52, approximately 24% (263/1079) of subjects treated with SKYRIZI at the recommended dose developed antibodies to risankizumab-rzaa. Of the subjects who developed antibodies to risankizumab-rzaa, approximately 57% (14% of all subjects treated with SKYRIZI) had antibodies that were classified as neutralizing. Higher antibody titers in approximately 1% of subjects treated with SKYRIZI were associated with lower risankizumab-rzaa concentrations and reduced clinical response.
Psoriatic Arthritis
By Week 28, approximately 12.1% (79/652) of subjects treated with SKYRIZI at the recommended dose developed antibodies to risankizumab-rzaa. None of the subjects who developed antibodies to risankizumab-rzaa had antibodies that were classified as neutralizing. Antibodies to risankizumab-rzaa were not associated with changes in clinical response for psoriatic arthritis. A higher proportion of subjects with anti-drug antibodies experienced hypersensitivity reactions (6.3% (5/79)) and injection site reactions (2.5% (2/79)) compared to subjects without anti-drug antibodies (3.8% (22/574) with hypersensitivity reactions and 0.7% (4/574) with injection site reactions). None of these hypersensitivity and injection site reactions led to discontinuation of risankizumab-rzaa.
Crohn’s D isease
By Week 64, antibodies to risankizumab-rzaa developed in approximately 3.4% (2/58) of subjects treated with SKYRIZI induction followed by 360 mg maintenance regimen. No subjects (0/57) treated with SKYRIZI induction followed by 180 mg maintenance regimen developed antibodies to risankizumab-rzaa. None of the subjects who developed antibodies to risankizumab-rzaa had antibodies that were classified as neutralizing.
Ulcerative Colitis
By Week 64, antibodies to risankizumab-rzaa developed in approximately 8.9% (8/90) or 4.4% (4/91) of subjects treated with SKYRIZI induction followed by the 180 mg or 360 mg maintenance regimen, respectively. Of the subjects who developed antibodies to risankizumab-rzaa, 75% (6.7% of all subjects treated with SKYRIZI induction followed by the 180 mg maintenance regimen) or 50% (2.2% of all subjects treated with SKYRIZI induction followed by the 360 mg maintenance regimen), respectively, had antibodies that were classified as neutralizing.
6.3 Postmarketing Experience
The following adverse reactions have been reported during post-approval of SKYRIZI. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to SKYRIZI exposure:
- Skin and subcutaneous tissue disorders : eczema and rash
11 DESCRIPTION
Risankizumab-rzaa, an interleukin-23 (IL-23) antagonist, is a humanized immunoglobulin G1 (IgG1) monoclonal antibody. Risankizumab-rzaa is produced by recombinant DNA technology in Chinese hamster ovary cells and has an approximate molecular weight of 149 kDa.
SKYRIZI (risankizumab-rzaa) injection 90 mg/mL prefilled syringe for subcutaneous use
Each SKYRIZI prefilled syringe contains a sterile, preservative-free, colorless to slightly yellow, and clear to slightly opalescent solution. Each syringe delivers 90 mg of risankizumab-rzaa, and inactive ingredients polysorbate 20 (0.2 mg), sodium succinate (0.63 mg), sorbitol (41 mg), succinic acid (0.059 mg), and Water for Injection, USP. The pH is 6.2.
SKYRIZI (risankizumab-rzaa) injection 150 mg/mL prefilled syringe or prefilled pen for subcutaneous use
Each SKYRIZI prefilled pen or prefilled syringe contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution. Each syringe and pen delivers 150 mg of risankizumab-rzaa and the inactive ingredients glacial acetic acid (0.054 mg), polysorbate 20 (0.2 mg), sodium acetate (0.75 mg), trehalose (63.33 mg), and Water for Injection, USP. The pH is 5.7.
SKYRIZI (risankizumab-rzaa) injection 180 mg/1.2 mL prefilled syringe for subcutaneous use
Each SKYRIZI prefilled syringe contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution. Each syringe delivers 180 mg of risankizumab-rzaa, and inactive ingredients glacial acetic acid (0.065 mg), polysorbate 20 (0.24 mg), sodium acetate (0.898 mg), trehalose (76.0 mg), and Water for Injection, USP. The pH is 5.7.
SKYRIZI (risankizumab-rzaa) injection 180 mg/ 1.2 mL (150 mg/ mL ) prefilled cartridge for use with supplied on-body-injector for subcutaneous use
Each SKYRIZI prefilled cartridge contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution. Each cartridge delivers 180 mg of risankizumab-rzaa, and the inactive ingredients glacial acetic acid (0.065 mg), polysorbate 20 (0.24 mg), sodium acetate (0.9 mg), trehalose (76 mg), and Water for Injection, USP. The pH is 5.7.
SKYRIZI (risankizumab-rzaa) injection 360 mg/2.4 mL (150 mg/mL) prefilled cartridge for use with the supplied o n- b ody i njector for subcutaneous use
Each SKYRIZI prefilled cartridge contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution. Each cartridge delivers 360 mg of risankizumab-rzaa, and the inactive ingredients glacial acetic acid (0.13 mg), polysorbate 20 (0.48 mg), sodium acetate (1.8 mg), trehalose (152 mg), and Water for Injection, USP. The pH is 5.7.
SKYRIZI 600 mg/10 mL (60 mg/mL) in a vial for intravenous infusion
SKYRIZI (risankizumab-rzaa) injection 600 mg/10 mL (60 mg/mL) is a sterile, preservative-free, colorless to slightly yellow, and clear to slightly opalescent solution in a 10 mL single-dose vial.
Each 10 mL single-dose vial contains 600 mg of risankizumab-rzaa, and the inactive ingredients glacial acetic acid (0.54 mg), polysorbate 20 (2 mg), sodium acetate (7.5 mg), trehalose (633.3 mg), and Water for Injection, USP. The pH is 5.7.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Risankizumab-rzaa is a humanized IgG1 monoclonal antibody that selectively binds to the p19 subunit of human IL-23 cytokine and inhibits its interaction with the IL-23 receptor. IL-23 is a naturally occurring cytokine that is involved in inflammatory and immune responses.
Risankizumab-rzaa inhibits the release of pro-inflammatory cytokines and chemokines.
12.2 Pharmacodynamics
No formal pharmacodynamics studies have been conducted with risankizumab-rzaa.
12.3 Pharmacokinetics
Risankizumab-rzaa plasma concentrations, after single dose administration increased dose proportionally from 18 mg to 360 mg when administered subcutaneously (0.12 to 2.4 times the lowest recommended dose and 0.05 to 1 times the highest recommended dose) and from 200 mg to 1,800 mg when administered as an up to 3-hour intravenous infusion (0.2 to 3 times the recommended dose) in healthy subjects.
In subjects with plaque psoriasis treated with 150 mg subcutaneously at Weeks 0, 4, and every 12 weeks thereafter, steady-state peak concentration (C max ) and trough concentration (C trough ) are estimated to be 12 mcg/mL and 2 mcg/mL, respectively.
With the same subcutaneous dosing regimen, the pharmacokinetics of risankizumab-rzaa in subjects with psoriatic arthritis were similar to that in subjects with plaque psoriasis.
In subjects with Crohn’s disease treated with 600 mg intravenous induction dose at Weeks 0, 4, and 8, followed by 180 mg or 360 mg subcutaneous maintenance dose at Week 12 and every 8 weeks thereafter, the median C max and C trough are estimated to be 156 mcg/mL and 38.8 mcg/mL, respectively, during Weeks 8-12; and the steady state median C max and C trough are estimated to be 14.0 mcg/mL and 4.1 mcg/mL, respectively for 180 mg or 28.0 mcg/mL and 8.1 mcg/mL, respectively, for 360 mg, during Weeks 40-48.
In subjects with ulcerative colitis treated with 1,200 mg intravenous induction dose at Weeks 0, 4, and 8, followed by 180 mg or 360 mg subcutaneous maintenance dose at Week 12 and every 8 weeks thereafter, the median C max and C trough are estimated to be 350 and 87.7 mcg/mL, respectively, during the induction period (Weeks 8-12); and the steady state median C max and C trough are estimated to be 19.6 and 4.64 µg/mL, respectively, for 180 mg or 39.2 mcg/mL and 9.29 mcg/mL, respectively, for 360 mg, during the maintenance period (Weeks 40-48).
Based on population pharmacokinetic analyses, the pharmacokinetics of risankizumab-rzaa in subjects with ulcerative colitis was generally similar to that in subjects with Crohn’s disease.
Absorption
The absolute bioavailability of risankizumab-rzaa was estimated to be 74 to 89% following subcutaneous injection. In healthy subjects, following administration of a single subcutaneous dose, C max was reached by 3 to 14 days.
Distribution
The estimated steady-state volume of distribution (inter-subject CV%) was 11.2 L (34%) in subjects with plaque psoriasis, and 7.68 L (64%) in subjects with Crohn’s disease.
Elimination
The estimated systemic clearance (inter-subject CV%) was 0.31 L/day (24%) and 0.30 L/day (34%) and terminal elimination half-life was approximately 28 days and 21 days in subjects with plaque psoriasis and Crohn’s disease, respectively.
Metabolism
The metabolic pathway of risankizumab-rzaa has not been characterized. As a humanized IgG1 monoclonal antibody, risankizumab-rzaa is expected to be degraded into small peptides and amino acids via catabolic pathways in a manner similar to endogenous IgG.
Specific Populations
Risankizumab-rzaa exposures (C trough ) in geriatric patients (≥65 years) are comparable to those in younger adult patients within each indication. No studies have been conducted to determine the effect of renal or hepatic impairment on the pharmacokinetics of risankizumab-rzaa.
Body Weight
Risankizumab-rzaa clearance and volume of distribution increase and plasma concentrations decrease as body weight increases; however, no dose adjustment is recommended based on body weight.
Drug Interaction Studies
Cytochrome P450 Substrates
No clinically significant changes in exposure of caffeine (CYP1A2 substrate), warfarin (CYP2C9 substrate), omeprazole (CYP2C19 substrate), metoprolol (CYP2D6 substrate), or midazolam (CYP3A substrate) were observed when used concomitantly with risankizumab-rzaa in subjects with plaque psoriasis (risankizumab-rzaa 150 mg administered subcutaneously at Weeks 0, 4, 8, and 12) and subjects with Crohn’s disease or ulcerative colitis (risankizumab-rzaa 1,800 mg administered intravenously at Weeks 0, 4, and 8, i.e., 3 times and 1.5 times the recommended dose for Crohn’s disease and ulcerative colitis, respectively).
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity and mutagenicity studies have not been conducted with SKYRIZI.
No effects on male fertility parameters were observed in sexually mature male cynomolgus monkeys dosed weekly for 26 weeks with 50 mg/kg risankizumab-rzaa at 4 times the exposure (AUC) in humans administered the maximum recommended induction dose (1,200 mg) and 39 times the exposure in humans administered the maximum recommended maintenance dose (360 mg).
14 CLINICAL STUDIES
14.1 Plaque Psoriasis
Four multicenter, randomized, double-blind trials [PsO-1 (NCT02684370), PsO-2 (NCT02684357), PsO-3 (NCT02672852), and PsO-4 (NCT02694523)] enrolled 2,109 subjects 18 years of age and older with moderate to severe plaque psoriasis who had a body surface area (BSA) involvement of ≥10%, a static Physician’s Global Assessment (sPGA) score of ≥3 (“moderate”) in the overall assessment (plaque thickness/induration, erythema, and scaling) of psoriasis on a severity scale of 0 to 4, and a Psoriasis Area and Severity Index (PASI) score ≥12.
Overall, subjects had a median baseline PASI score of 17.8 and a median BSA of 20%. Baseline sPGA score was 4 (“severe”) in 19% of subjects. A total of 10% of trial subjects had a history of diagnosed psoriatic arthritis.
Across all trials, 38% of subjects had received prior phototherapy, 48% had received prior non-biologic systemic therapy, and 42% had received prior biologic therapy for the treatment of psoriasis.
Trials PsO-1 and PsO-2
In trials PsO-1 and PsO-2, 997 subjects were enrolled (including 598 subjects randomized to the SKYRIZI 150 mg group, 200 subjects randomized to the placebo group, and 199 to the biologic active control group). Subjects received treatment at Weeks 0, 4, and every 12 weeks thereafter.
Both trials assessed the responses at Week 16 compared with placebo for the two co-primary endpoints:
- the proportion of subjects who achieved an sPGA score of 0 (“clear”) or 1 (“almost clear”)
- the proportion of subjects who achieved at least a 90% reduction from baseline PASI (PASI 90)
Secondary endpoints included the proportion of subjects who achieved PASI 100, sPGA 0, and Psoriasis Symptom Scale (PSS) 0 at Week 16.
The results are presented in Table 6.
| PsO-1 | PsO-2 | |||
| SKYRIZI (N=304) n (%) | Placebo (N=102) n (%) | SKYRIZI (N=294) n (%) | Placebo (N=98) n (%) | |
| sPGA 0 or 1 (“clear or almost clear”) a | 267 (88) | 8 (8) | 246 (84) | 5 (5) |
| PASI 90 a | 229 (75) | 5 (5) | 220 (75) | 2 (2) |
| sPGA 0 (“clear”) | 112 (37) | 2 (2) | 150 (51) | 3 (3) |
| PASI 100 | 109 (36) | 0 (0) | 149 (51) | 2 (2) |
| a Co-primary endpoints | ||||
Examination of age, gender, race, body weight, baseline PASI score and previous treatment with systemic or biologic agents did not identify differences in response to SKYRIZI among these subgroups at Week 16.
In PsO-1 and PsO-2 at Week 52, subjects receiving SKYRIZI achieved sPGA 0 (58% and 60%, respectively), PASI 90 (82% and 81%, respectively), and PASI 100 (56% and 60%, respectively).
Patient Reported Outcomes
Improvements in signs and symptoms related to pain, redness, itching and burning at Week 16 compared to placebo were observed in both trials as assessed by the PSS. In PsO-1 and PsO-2, about 30% of the subjects who received SKYRIZI achieved PSS 0 (“none”) at Week 16 compared to 1% of the subjects who received placebo.
Trial PsO-3
Trial PsO-3 enrolled 507 subjects (407 randomized to SKYRIZI 150 mg and 100 to placebo). Subjects received treatment at Weeks 0, 4, and every 12 weeks thereafter.
At Week 16, SKYRIZI was superior to placebo on the co-primary endpoints of sPGA 0 or 1 (84% SKYRIZI and 7% placebo) and PASI 90 (73% SKYRIZI and 2% placebo). The respective response rates for SKYRIZI and placebo at Week 16 were: sPGA 0 (46% SKYRIZI and 1% placebo); PASI 100 (47% SKYRIZI and 1% placebo); and PASI 75 (89% SKYRIZI and 8% placebo).
Maintenance and Durability of Response
In PsO-1 and PsO-2, among the subjects who received SKYRIZI and had PASI 100 at Week 16, 80% (206/258) of the subjects who continued on SKYRIZI had PASI 100 at Week 52. For PASI 90 responders at Week 16, 88% (398/450) of the subjects had PASI 90 at Week 52.
In PsO-3, subjects who were originally on SKYRIZI and had sPGA 0 or 1 at Week 28 were re-randomized to continue SKYRIZI every 12 weeks or withdrawal of therapy. At Week 52, 87% (97/111) of the subjects re-randomized to continue treatment with SKYRIZI had sPGA 0 or 1 compared to 61% (138/225) who were re-randomized to withdrawal of SKYRIZI.
Plaque Psoriasis of the Scalp or Genital Area (Trial PsO-5)
The efficacy of SKYRIZI was assessed in a multicenter, randomized, double-blind, placebo-controlled trial [PsO-5 (NCT05969223)] that enrolled subjects 18 years of age and older with moderate to severe plaque psoriasis of the scalp (Trial S), defined as Psoriasis Scalp Severity Index (PSSI) ≥12, scalp Investigator Global Assessment (scalp IGA) ≥3 (“moderate”), and ≥30% of the scalp affected, or moderate to severe plaque psoriasis of the genital area (Trial G), defined as static Physician’s Global Assessment of Genitalia (sPGA-G) ≥3 (“moderate”) at baseline. All subjects had BSA ≥1% and sPGA ≥3 (“moderate”) at baseline.
In trial PsO-5, subjects were randomized to receive either SKYRIZI 150 mg or placebo subcutaneously at Weeks 0 and 4. Starting at Week 16, all subjects received SKYRIZI 150 mg every 12 weeks until the last dose at Week 40.
Plaque Psoriasis of the Scalp
PsO-5 Trial S enrolled 105 subjects with moderate to severe plaque psoriasis of the scalp. The median age of enrolled subjects at baseline was 44 years (range 20 to 83 years) and 10% of the subjects were 65 years of age and older. Fifty-six (56)% of the subjects were male, 83% were White, 8% were Black or African American, and 5% were Asian; for ethnicity, 36% of the subjects identified as Hispanic or Latino. At baseline, 74% of the subjects had moderate plaque psoriasis of the scalp (scalp IGA of 3) and 26% had severe plaque psoriasis of the scalp (scalp IGA of 4). Median baseline PSSI was 32. Baseline BSA involvement was ≥10% for 62% of the subjects and <10% for the remaining subjects. Median baseline BSA involvement was 11%. Baseline sPGA score was 4 (“severe”) in 24% of the subjects.
At baseline, 54% of subjects were naïve to both non-biologic systemic and biologic therapy, 0% of subjects had received prior phototherapy, 15% had received prior non-biologic systemic therapy, and 37% had received prior biologic therapy.
The results are presented in Table 7.
| Endpoint | SKYRIZI (N=51) n (%) | Placebo (N=54) n (%) | Treatment Difference (95% CI) |
| scalp IGA of 0 or 1 (“clear or almost clear”) a | 31 (61) | 7 (13) | 47 (31, 63) |
| PSSI 90 b | 27 (53) | 7 (13) | 40 (24, 55) |
| PSSI 100 c | 23 (45) | 7 (13) | 31 (15, 47) |
| a Primary endpoint b Achievement of ≥90% improvement from baseline in PSSI c Achievement of 100% improvement from baseline in PSSI | |||
Plaque Psoriasis of the Genital Area
PsO-5 Trial G enrolled 109 subjects with moderate to severe plaque psoriasis of the genital area. The median age of enrolled subjects at baseline was 45 years (range 19 to 77 years) and 15% of the subjects were 65 years of age and older. Sixty-five (65)% of the subjects were male, 90% were White, 4% were Asian, and 2% were Black or African American; for ethnicity, 29% of the subjects identified as Hispanic or Latino. At baseline, 62% of the subjects had moderate plaque psoriasis of the genital area (sPGA-G of 3) and 38% had severe or very severe plaque psoriasis of the genital area (sPGA-G of 4 or 5). Baseline BSA involvement was ≥10% for 63% of the subjects and <10% for the remaining subjects. Median baseline BSA involvement was 11%. Baseline sPGA score was 4 (“severe”) in 19% of the subjects.
At baseline, 61% of subjects were naïve to both non-biologic systemic and biologic therapy, 3% of subjects had received prior phototherapy, 17% had received prior non-biologic systemic therapy, and 26% had received prior biologic therapy.
The results are presented in Table 8.
| Endpoint | SKYRIZI (N=55) n (%) | Placebo (N=54) n (%) | Treatment Difference (95% CI) |
| sPGA-G of 0 or 1 (“clear or minimal ”) a | 38 (69) | 7 (13) | 57 (42, 72) |
| sPGA-G of 0 (“clear”) | 28 (51) | 3 (6) | 47 (33, 61) |
| GPI- NRS reduction of ≥4-point from baseline b | N=41 20 (49) | N=45 3 (7) | 43 (27, 59) |
| GenPs-SFQ item 2 score of 0 (never) or 1 (rarely) c,d | N=31 22 (71) | N=32 7 (22) | 46 (27, 66) |
| a Primary endpoint b Improvement of genital itch severity as measured by a reduction of at least 4 points in the 11-point Genital Psoriasis Itch (GPI) Numeric Rating Scale (NRS) from the Genital Psoriasis Symptom Scale (GPSS) among subjects with baseline score ≥4 c Genital Psoriasis Sexual Frequency Questionnaire (GenPs-SFQ) Item 2 (In the past week, how often did your genital psoriasis limit the frequency of your sexual activity?) score ranges from 0 to 4 (0 = never, 1 = rarely, 2 = sometimes, 3 = often, 4 = always); where higher scores indicate greater limitations on the frequency of sexual activity in the past week d Among subjects with baseline score ≥2 | |||
14.2 Psoriatic Arthritis
The safety and efficacy of SKYRIZI were assessed in 1407 subjects in 2 randomized, double-blind, placebo-controlled trials (964 in PsA-1 [NCT03675308] and 443 in PsA-2 [NCT03671148]) in subjects 18 years and older with active psoriatic arthritis (PsA).
Subjects in these trials had a diagnosis of PsA for at least 6 months based on the Classification Criteria for Psoriatic Arthritis (CASPAR), a median duration of PsA of 4.9 years at baseline, ≥ 5 tender joints and ≥ 5 swollen joints, and active plaque psoriasis or psoriatic nail disease at baseline. Regarding baseline clinical presentation, 55.9% of subjects had ≥3% BSA with active plaque psoriasis; 63.4% and 27.9% of subjects had enthesitis and dactylitis, respectively. In PsA-1 where psoriatic nail disease was further assessed, 67.3% had psoriatic nail disease.
In PsA-1, all subjects had a previous inadequate response or intolerance to non-biologic DMARD therapy and were biologic naïve. In PsA-2, 53.5% of subjects had a previous inadequate response or intolerance to non-biologic DMARD therapy, and 46.5% of subjects had a previous inadequate response or intolerance to biologic therapy.
In both trials, subjects were randomized to receive SKYRIZI 150 mg or placebo at Weeks 0, 4, and 16. Starting from Week 28, all subjects received SKYRIZI every 12 weeks. Both trials included a long-term extension for up to an additional 204 weeks. Regarding use of concomitant medications, 59.6% of subjects were receiving concomitant methotrexate (MTX), 11.6% were receiving concomitant non-biologic DMARDs other than MTX, and 28.9% were receiving SKYRIZI monotherapy.
For both trials, the primary endpoint was the proportion of subjects who achieved an American College of Rheumatology (ACR) 20 response at Week 24.
Clinical Response
In both trials, treatment with SKYRIZI resulted in significant improvement in measures of disease activity compared with placebo at Week 24. See Tables 9 and 10 for key efficacy results.
In both trials, similar responses were seen regardless of concomitant non-biologic DMARD use, number of prior non-biologic DMARDs, age, gender, race, and BMI. In PsA-2, responses were seen regardless of prior biologic therapy.
| Endpoint | Placebo N=481 Response Rate | SKYRIZI N=483 Response Rate | Difference from Placebo (95% CI) |
| ACR20 Response• | |||
| Week 16 | 33.4% | 56.3% a | 23.1% (16.8, 29.4) |
| Week 24 | 33.5% | 57.3% a | 24.0% (18.0, 30.0) |
| ACR50 Response• | |||
| Week 16 | 11.1% | 26.4% | 15.4% (10.6, 20.2) |
| Week 24 | 11.3% | 33.4 % | 22.2% (17.3, 27.2) |
| ACR70 Response• | |||
| Week 16 | 2.7% | 11.8% | 9.2% (6.1, 12.4) |
| Week 24 | 4.7% | 15.3% | 10.5% (6.9, 14.2) |
| a. multiplicity-controlled p≤0.001, SKYRIZI vs. placebo comparison. •A Subject was considered as a non-responder after initiation of rescue medication or concomitant medications for PsA that could meaningfully impact efficacy assessment. | |||
| Endpoint | Placebo N=219 Response Rate | SKYRIZI N=224 Response Rate | Difference from Placebo (95% CI) |
| ACR20 Response• | |||
| Week 16 | 25.3% | 48.3% a | 22.6% (13.9, 31.2) |
| Week 24 | 26.5% | 51.3% a | 24.5% (15.9, 33.0) |
| ACR50 Response• | |||
| Week 16 | 6.8% | 20.3% | 13.5% (7.3, 19.7) |
| Week 24 | 9.3% | 26.3% | 16.6% (9.7, 23.6) |
| ACR70 Response• | |||
| Week 16 | 3.4% | 11.2% | 7.8% (3.0, 12.6) |
| Week 24 | 5.9% | 12.0% | 6.0% (0.8, 11.3) |
| a. multiplicity-controlled p≤0.001, SKYRIZI vs. placebo comparison. •A Subject was considered as a non-responder after initiation of rescue medication or concomitant medications for PsA that could meaningfully impact efficacy assessment. | |||
The percent of subjects achieving ACR20 responses in trial PsA-1 through Week 24 is shown in Figure 1.
Figure 1. Percent of Subjects Achieving ACR20 Responses in Trial PsA-1 through Week 24
The results of the components of the ACR response criteria for both trials are shown in Table 11.
| PsA-1 | PsA-2 | |||
| Placebo (N=481) Mean (SD) | SKYRIZI (N=483) Mean (SD) | Placebo (N=219) Mean (SD) | SKYRIZI (N=224) Mean (SD) | |
| Number of Swollen Joints (0-66) | ||||
| Baseline | 12.2 (8.0) | 12.1 (7.8) | 13.6 (9.0) | 13.0 (8.7) |
| Mean change at Week 16 | -5.5 (7.0) | -7.7 (7.2) | -5.4 (8.5) | -8.0 (7.4) |
| Mean change at Week 24 | -6.7 (7.2) | -8.7 (7.2) | -6.5 (7.8) | -9.1 (7.6) |
| Number of Tender Joints (0-68) | ||||
| Baseline | 20.5 (12.8) | 20.8 (14.0) | 22.3 (13.8) | 22.8 (14.9) |
| Mean change at Week 16 | -6.3 (11.1) | -10.7 (11.4) | -6.0 (13.1) | -11.3 (13.0) |
| Mean change at Week 24 | -7.9 (10.7) | -12.0 (12.3) | -8.3 (11.3) | -13.0 (12.5) |
| Patient’s Assessment of Pain a | ||||
| Baseline | 57.1 (22.6) | 57.1 (22.6) | 57.0 (23.1) | 55.0 (23.5) |
| Mean change at Week 16 | -8.6 (23.7) | -18.4 (26.3) | -5.7 (22.7) | -14.4 (26.4) |
| Mean change at Week 24 | -10.9 (25.4) | -21.4 (26.5) | -8.7 (25.3) | -15.3 (26.5) |
| Patient’s Global Assessment a | ||||
| Baseline | 57.4 (22.1) | 57.9 (21.7) | 56.2 (23.0) | 56.2 (21.8) |
| Mean change at Week 16 | -10.2 (23.9) | -19.4 (25.7) | -4.9 (23.6) | -17.0 (27.1) |
| Mean change at Week 24 | -11.1 (25.1) | -22.6 (26.9) | -8.7 (25.4) | -17.7 (27.7) |
| Physician Global Assessment a | ||||
| Baseline | 62.4 (17.0) | 61.3 (17.6) | 60.7 (16.4) | 63.0 (17.0) |
| Mean change at Week 16 | -18.3 (22.5) | -31.1 (23.4) | -19.0 (23.3) | -32.7 (24.7) |
| Mean change at Week 24 | -22.2 (22.8) | -34.8 (23.2) | -21.3 (25.2) | -35.5 (25.6) |
| Health Assessment Questionnaire - Disability Index (HAQ-DI) b | ||||
| Baseline | 1.2 (0.7) | 1.2 (0.7) | 1.1 (0.6) | 1.1 (0.6) |
| Mean change at Week 16 | -0.1 (0.5) | -0.3 (0.5) | -0.1 (0.5) | -0.2 (0.5) |
| Mean change at Week 24 | -0.1 (0.5) | -0.3 (0.5) | -0.1 (0.4) | -0.2 (0.5) |
| High sensitivity C-reactive protein (hs-CRP) mg/L | ||||
| Baseline | 11.3 (14.1) | 11.9 (15.9) | 8.2 (17.1) | 7.4 (10.9) |
| Mean change at Week 16 | -0.3 (14.7) | -4.8 (14.2) | -0.1 (6.8) | -2.1 (7.5) |
| Mean change at Week 24 | -0.2 (11.7) | -4.3 (12.8) | -0.5 (14.5) | -1.8 (13.4) |
| SD= Standard Deviation. a. Assessment based on Visual Analog Scale (100 mm) with the left end indicating “no pain” (for patient’s assessment of pain), “very well” (for patient global assessment), or “no arthritis activity” (for physician global assessment) and the right end indicating “the worst possible pain” (for patient assessment of pain), “poor” (for patient global assessment), or “extremely active arthritis” (for physician global assessment). b. Disability Index of the Health Assessment Questionnaire; 0 = no difficulty to 3 = inability to perform, measures the patient’s ability to perform the following: dressing, arising, eating, walking, hygiene, reaching, gripping, and activities of daily living. | ||||
Treatment with SKYRIZI resulted in improvement in dactylitis and enthesitis in subjects with pre-existing dactylitis or enthesitis.
In patients with coexistent plaque psoriasis receiving SKYRIZI, the skin lesions of psoriasis improved with treatment, relative to placebo, as measured by the Psoriasis Area Severity Index (PASI 90) at Week 24.
Physical Function
In both trials, patients treated with SKYRIZI showed statistically significant improvement from baseline in physical function compared with placebo as assessed by HAQ-DI at Week 24 (Table 9 ). The mean difference (95% CI) from placebo in HAQ-DI change from baseline at Week 24 was -0.20 (-0.26, -0.14) in trial PsA-1 and -0.16 (-0.26, -0.07) in trial PsA-2.
In both trials, a greater proportion of subjects achieved a reduction of at least 0.35 in HAQ-DI score from baseline in the SKYRIZI group compared with placebo at Week 24.
Other Health Related Outcomes
In both trials, general health status was assessed by the 36-Item Short Form Health Survey (SF-36 V2). Fatigue was assessed by Functional Assessment of Chronic Illness Therapy Fatigue Scale (FACIT-Fatigue).
In both trials at Week 24, subjects treated with SKYRIZI showed improvements in the SF-36 physical component summary scores compared with subjects who received placebo. There were also numerical improvements in subjects treated with SKYRIZI in physical functioning, role physical, bodily pain, general health, vitality, social functioning, mental health, role emotional domain scores and mental component summary scores in both trials at week 24 compared to placebo. In both trials at Week 24, subjects treated with SKYRIZI showed improvements in FACIT-Fatigue scores compared with subjects who received placebo.
14.3 Crohn’s Disease
Induction Trials ( Trials CD-1 and CD-2)
In two 12-week induction trials (CD-1; NCT03105128 and CD-2; NCT03104413), subjects with moderately to severely active Crohn’s disease were randomized to receive SKYRIZI 600 mg, SKYRIZI 1,200 mg, or placebo as an intravenous infusion at Week 0, Week 4, and Week 8. Moderately to severely active CD was defined as a Crohn’s Disease Activity Index (CDAI) of 220 to 450 and Simple Endoscopic Score for Crohn’s disease (SES-CD) ≥6 (or ≥4 for isolated ileal disease). Subjects with inadequate response, loss of response, or intolerance to oral aminosalicylates, corticosteroids, immunosuppressants, and/or biologic therapy were enrolled.
At baseline, the median CDAI was 307 (range: 76 – 634) and 307 (range: 72 – 651), and the median SES-CD was 12 (range: 4 – 45) and 13 (range 4 – 40), in CD-1 and CD-2, respectively. In CD-1, 58% (491/850) of subjects had failed or were intolerant to treatment with one or more biologic therapies (prior biologic failure). All subjects in CD-2 had prior biologic failure. At baseline, 30% and 34% of patients were receiving corticosteroids, 24% and 23% of patients were receiving immunomodulators (azathioprine, 6-mercaptopurine, methotrexate), and 31% and 19% of patients were receiving aminosalicylates in CD-1 and CD-2, respectively. In CD-1 and CD-2 combined, the median age was 36 years (ranging from 16 to 80 years), 81% (1145/1419) of subjects were white, and 53% (753/1419) were male.
In CD-1 and CD-2, the co-primary endpoints were clinical remission and endoscopic response at Week 12. Secondary endpoints included clinical response and endoscopic remission (see Table 12 and Table 13 ). The SKYRIZI 1,200 mg dosage did not demonstrate additional treatment benefit over the 600 mg dosage and is not a recommended regimen [see Dosage and Administration (2.6 )] .
| Endpoint | Placebo | SKYRIZI 600 mg Intravenous Infusion a | Treatment Difference b (95% CI) |
| Clinical Remission c,d | |||
| Total Population | N=175 25% | N=336 45% | 21% e (12%, 29%) |
| Prior biologic failure f | N=97 26% | N=195 42% | |
| Without prior biologic failure | N=78 23% | N=141 49% | |
| Endoscopic Response c, g | |||
| Total Population | N=175 12% | N=336 40% | 28% e (21%, 35%) |
| Prior biologic failure f | N=97 11% | N=195 33% | |
| Without prior biologic failure | N=78 13% | N=141 50% | |
| Clinical Response h | |||
| Total Population | N=175 37% | N=336 60% | 23% e (14%, 32%) |
| Prior biologic failure f | N=97 34% | N=195 58% | |
| Without prior biologic failure | N=78 40% | N=141 62% | |
| Endoscopic Remission i | |||
| Total Population | N=175 9% | N=336 24% | 15% e (9%, 21%) |
| Prior biologic failure f | N=97 5% | N=195 18% | |
| Without prior biologic failure | N=78 14% | N=141 32% | |
| a. SKYRIZI 600 mg as an intravenous infusion at Week 0, Week 4, and Week 8 b. Adjusted treatment difference (95% CI) based on Cochran-Mantel-Haenszel method adjusted for randomization stratification factors c. Co-primary endpoints d. CDAI <150 e. p <0.001 f. Prior biologic failure includes inadequate response, loss of response, or intolerance to one or more biologic treatments for CD g. A decrease in SES-CD > 50% from baseline, or a decrease of at least 2 points for subjects with a baseline score of 4 and isolated ileal disease, based on central reading h. A reduction of CDAI ≥ 100 points from baseline i. SES-CD ≤ 4 and at least a 2-point reduction from baseline, with no individual subscore greater than 1, based on central reading | |||
| Endpoint | Placebo N=187 | SKYRIZI 600 mg Intravenous Infusion b N=191 | Treatment Difference c (95% CI) |
| Clinical Remission d, e | 20% | 42% | 22% f (13%, 31%) |
| Endoscopic Response d,g | 11% | 29% | 18% f (10%, 25%) |
| Clinical Response h | 30% | 60% | 29% f (20%, 39%) |
| Endoscopic Remission i | 4% | 19% | 15% f (9%, 21%) |
| a. All subjects enrolled in CD-2 had prior biologic failure. Prior biologic failure includes inadequate response, loss of response, or intolerance to one or more biologic treatments for CD b. SKYRIZI 600 mg as an intravenous infusion at Week 0, Week 4, and Week 8 c. Adjusted treatment difference (95% CI) based on Cochran-Mantel-Haenszel method adjusted for randomization stratification factors d. Co-primary endpoints e. CDAI score <150 f. p < 0.001 g. A decrease in SES-CD > 50% from baseline, or a decrease of at least 2 points for subjects with a baseline score of 4 and isolated ileal disease, based on central reading h. A reduction of CDAI ≥ 100 points from baseline i. SES-CD ≤ 4 and at least a 2-point reduction versus from baseline, with and no individual subscore greater than 1, based on central reading | |||
Onset of clinical response and clinical remission based on CDAI occurred as early as Week 4 in a greater proportion of subjects treated with the SKYRIZI 600 mg induction regimen compared to placebo.
Reductions in stool frequency and abdominal pain were observed in a greater proportion of subjects treated with the SKYRIZI 600 mg induction regimen compared to placebo at Week 12.
Trial CD-3
The maintenance trial CD-3 evaluated 382 subjects who achieved clinical response defined as a reduction in CDAI of at least 100 points from baseline after 12 weeks of induction treatment with intravenous SKYRIZI in trials CD-1 and CD-2. Subjects were randomized to receive a maintenance regimen of SKYRIZI 180 mg or SKYRIZI 360 mg or placebo at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks.
The co-primary endpoints in CD-3 were clinical remission and endoscopic response at Week 52 (see Table 14 ).
| Endpoint | Placebo a | SKYRIZI 180 mg Subcutaneous Injection b | SKYRIZI 360 mg Subcutaneous Injection c | Treatment Difference vs Placebo d (95% CI ) | ||
| SKYRIZI 180 mg | SKYRIZI 360 mg | |||||
| Clinical Remission e,f | ||||||
| Total Population | N=130 46% | N=135 61% | N=117 57% | 17% g (6%, 28%) | 14% g (3%, 26%) | |
| Prior biologic failure h | N=99 40% | N=95 56% | N=83 51% | |||
| Without prior biologic failure | N=31 65% | N=40 75% | N=34 71% | |||
| Endoscopic Response e,i | ||||||
| Total Population | N=130 22% | N=135 50% | N=117 48% | 30% g (20%, 39%) | 31% g (21%, 41%) | |
| Prior biologic failure h | N=99 21% | N=95 44% | N=83 44% | |||
| Without prior biologic failure | N=31 23% | N=40 65% | N=34 59% | |||
| a. The placebo group consisted of patients who were in response to SKYRIZI and were randomized to receive placebo at the start of maintenance therapy. b. SKYRIZI 180 mg at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks c. SKYRIZI 360 mg at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks d. Adjusted treatment difference and 95% CI computed using Cochran-Mantel-Haenszel method adjusted for randomization stratification factors e. Co-primary endpoints f. CDAI <150 g. p <0.05 h. Prior biologic failure includes inadequate response, loss of response, or intolerance to one or more biologic treatments for CD i. A decrease in SES-CD > 50% from baseline, or a decrease of at least 2 points for subjects with a baseline score of 4 and isolated ileal disease, based on central reading | ||||||
Endoscopic remission was observed at Week 52 in 33% (44/135) of subjects treated with the SKYRIZI 180 mg maintenance regimen and 41% (48/117) of subjects treated with the SKYRIZI 360 mg maintenance regimen, compared to 13% (17/130) of subjects treated with placebo. This endpoint was not statistically significant under the prespecified multiple testing procedure.
14.4 Ulcerative Colitis
Induction Trial ( Trial UC-1)
In the 12-week induction trial (UC-1; NCT03398148), 966 subjects with moderately to severely active ulcerative colitis were randomized and received SKYRIZI 1,200 mg or placebo as an intravenous infusion at Week 0, Week 4, and Week 8. Disease activity was assessed by the modified Mayo score (mMS), a 3-component Mayo score (0-9) which consists of the following subscores (0 to 3 for each subscore): stool frequency (SFS), rectal bleeding (RBS), and findings on centrally read endoscopy score (ES). An ES of 2 was defined by marked erythema, lack of vascular pattern, any friability, and/or erosions; an ES of 3 was defined by spontaneous bleeding and ulceration. Enrolled subjects had a mMS between 5 and 9, with an ES of 2 or 3. Subjects with inadequate response, or intolerance to oral aminosalicylates, corticosteroids, immunomodulators, biologics, Janus Kinase inhibitors (JAKi), and/or sphingosine-1-phosphate receptor modulators (S1PRM) were enrolled.
At baseline in UC-1, the median mMS was 7; 37% had severely active disease (mMS >7); 69% had an ES of 3. In UC-1, 52% (499/966) of subjects had failed (inadequate response or intolerance) treatment with one or more biologics, JAKi or S1PRM. Of these 499 subjects, 484 (97%) failed biologics and 90 (18%) failed JAK inhibitors. Enrolled subjects were permitted to use a stable dose of oral corticosteroids (up to 20 mg/day prednisone or equivalent), immunomodulators, and aminosalicylates. At baseline, 36% of subjects were receiving corticosteroids, 16% of subjects were receiving immunomodulators (including azathioprine, 6-mercaptopurine, methotrexate), and 73% of subjects were receiving aminosalicylates in UC-1.
In UC-1, the primary endpoint was clinical remission defined using the mMS at Week 12 (see Table 15 ). Key secondary endpoints included clinical response, endoscopic improvement, and histologic endoscopic mucosal improvement (see Table 15 ).
| Endpoint | Placebo | SKYRIZI 1,200 mg Intravenous Infusion a | Treatment Difference (95% CI) b |
| Clinical Remission c | |||
| Total Population | N=320 8% | N=646 24% | 16% h (12%, 20%) |
| Prior biologic, JAKi, or S1PRM failure d | N=168 6% | N=331 14% | |
| Without prior biologic, JAKi, or S1PRM failure | N=152 9% | N=315 33% | |
| Clinical Response e | |||
| Total Population | N=320 36% | N=646 65% | 29% h (23%, 35%) |
| Prior biologic, JAKi, or S1PRM failure d | N=168 32% | N=331 56% | |
| Without prior biologic, JAKi, or S1PRM failure | N=152 41% | N=315 75% | |
| Endoscopic Improvement f | |||
| Total Population | N=320 12% | N=646 36% | 25% h (20%, 30%) |
| Prior biologic, JAKi, or S1PRM failure d | N=168 10% | N=331 26% | |
| Without prior biologic, JAKi, or S1PRM failure | N=152 14% | N=315 47% | |
| Histologic Endoscopic Mucosal Improvement (HEMI) g | |||
| Total Population | N=320 7% | N=646 24% | 17% h (13%, 21%) |
| Prior biologic, JAKi, or S1PRM failure d | N=168 7% | N=331 16% | |
| Without prior biologic, JAKi, or S1PRM failure | N=152 8% | N=315 33% | |
| a SKYRIZI 1,200 mg as an intravenous infusion at Week 0, Week 4, and Week 8 b Adjusted treatment difference (95% CI) based on Cochran-Mantel-Haenszel method adjusted for stratification factors c Per mMS: SFS ≤ 1 and not greater than baseline, RBS = 0, and ES ≤ 1 without friability d Prior failure includes inadequate response or intolerance to treatment with one or more of the following: biologic therapies, Janus Kinase inhibitors (JAKi), and/or sphingosine-1-phosphate receptor modulators (S1PRM) e Per mMS: decrease ≥ 2 points and ≥ 30% from baseline, and a decrease in RBS ≥ 1 from baseline or an absolute RBS ≤ 1 f ES ≤ 1 without the evidence of friability g ES ≤ 1 without the evidence of friability and Geboes score ≤ 3.1 (indicating neutrophil infiltration in <5% of crypts, no crypt destruction and no erosions, ulcerations or granulation tissue) h p < 0.001 | |||
UC-1 was not designed to evaluate the relationship of histologic endoscopic mucosal improvement at week 12 to disease progression and long-term outcomes.
Rectal Bleeding and Stool Frequency Subscores
Decreases in rectal bleeding and stool frequency subscores in subjects treated with SKYRIZI compared to placebo were observed as early as 4 weeks.
Endoscopic Assessment
Endoscopic remission was defined as ES of 0. At Week 12, a greater proportion of subjects treated with SKYRIZI compared to placebo achieved endoscopic remission (11% vs 3%).
Bowel Urgency
A greater proportion of subjects treated with the SKYRIZI 1,200 mg induction regimen compared to placebo had no bowel urgency (44% vs 27%) at Week 12.
Fatigue
In UC-1, subjects treated with SKYRIZI experienced a clinically meaningful improvement in fatigue, assessed by change from baseline in FACIT-F score, at Week 12, compared to placebo-treated subjects. The effect of SKYRIZI to improve fatigue after 12 weeks of induction has not been established.
Other UC Symptoms
The proportion of subjects who had no nocturnal bowel movements was greater in subjects treated with SKYRIZI compared to placebo at Week 12 (67% vs 43%).
Maintenance Trial UC-2
The maintenance trial (UC-2; NCT03398135) evaluated 547 subjects who received one of three SKYRIZI induction regimens, including the 1,200 mg regimen, for 12 weeks in Trials UC-1 or UC-3 and demonstrated clinical response per mMS after 12 weeks. Subjects were randomized to receive a maintenance regimen of subcutaneous (SC) SKYRIZI 180 mg or SKYRIZI 360 mg or placebo at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks.
In UC-2, 75% (411/547) of subjects had failed (inadequate response or intolerance) treatment with one or more biologics, JAKi, or S1PRM. Of these 411 subjects, 407 (99%) failed biologics and 78 (19%) failed JAK inhibitors.
The primary endpoint in UC-2 was clinical remission using mMS at Week 52 (see Table 16 ). Key secondary endpoints included corticosteroid-free clinical remission, endoscopic improvement, and histologic endoscopic mucosal improvement (see Table 16 ).
| Endpoint | Placebo a | SKYRIZI 180 mg SC Injection b | SKYRIZI 360 mg SC Injection c |
| Clinical remission d | |||
| Total Population | N=182 26% | N=179 45% | N=186 41% |
| Treatment Difference vs Placebo e (95% CI) | 20% j [11%, 29%] | 16% j [7%, 25%] | |
| Prior biologic, JAKi, or S1PRM failure f | N=138 24% | N=134 41% | N=139 32% |
| Without prior biologic, JAKi, or S1PRM failure | N=44 32% | N=45 58% | N=47 67% |
| Corticosteroid-free clinical remission g | |||
| Total Population | N=182 26% | N=179 45% | N=186 40% |
| Treatment Difference vs Placebo e (95% CI) | 20% j [11%, 29%] | 16% j [7%, 25%] | |
| Prior biologic, JAKi, or S1PRM failure f | N=138 24% | N=134 40% | N=139 32% |
| Without prior biologic, JAKi, or S1PRM failure | N=44 32% | N=45 58% | N=47 64% |
| Endoscopic improvement h | |||
| Total Population | N=182 31% | N=179 51% | N=186 48% |
| Treatment Difference vs Placebo e (95% CI) | 20% j [11%, 30%] | 18% j [8%, 27%] | |
| Prior biologic, JAKi, or S1PRM failure f | N=138 30% | N=134 48% | N=139 39% |
| Without prior biologic, JAKi, or S1PRM failure | N=44 34% | N=45 60% | N=47 76% |
| Histologic Endoscopic Mucosal Improvement i | |||
| Total Population | N=182 24% | N=179 43% | N=186 42% |
| Treatment Difference vs Placebo e (95% CI) | 20% j [11%, 29%] | 20% j [11%, 29%] | |
| Prior biologic, JAKi, or S1PRM failure f | N=138 22% | N=134 39% | N=139 33% |
| Without prior biologic, JAKi, or S1PRM failure | N=44 30% | N=45 55% | N=47 69% |
| a The placebo group consisted of subjects who were in response to 12 weeks of SKYRIZI induction and were randomized to receive placebo at the start of maintenance therapy. b SKYRIZI 180 mg at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks c SKYRIZI 360 mg at Week 12 and every 8 weeks thereafter for up to an additional 52 weeks d Per mMS: SFS ≤ 1 and not greater than baseline, RBS = 0, and ES ≤ 1 without friability e Adjusted treatment difference (95% CI) based on Cochran-Mantel-Haenszel method adjusted for stratification factors f Prior failure includes inadequate response or intolerance to treatment with one or more of the following: biologic therapies, Janus Kinase inhibitors (JAKi), and/or sphingosine-1-phosphate receptor modulators (S1PRM) g Clinical remission per mMS at Week 52 and corticosteroid-free for ≥90 days h ES ≤ 1 without the evidence of friability i ES ≤ 1without the evidence of friability and Geboes score ≤ 3.1 (indicating neutrophil infiltration in <5% of crypts, no crypt destruction and no erosions, ulcerations or granulation tissue) j p < 0.001 | |||
Endoscopic Assessment
Endoscopic remission was defined as ES of 0. In UC-2, a greater proportion of subjects treated with SKYRIZI 180 mg and SKYRIZI 360 mg compared to placebo achieved endoscopic remission at Week 52 (23% and 24% vs 15%).
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
SKYRIZI (risankizumab-rzaa) injection is supplied in the following strengths:
| Strength | Pack Size | NDC |
| Subcutaneous Injection | ||
| 150 mg/mL single-dose pen | Carton of 1 | 0074-2100-01 |
| 90 mg/mL single-dose prefilled syringe | Carton of 2 | 0074-7040-02 |
| Carton of 4 | 0074-7042-04 | |
| 180 mg/1.2 mL (150 mg/mL) single-dose prefilled syringe | Carton of 1 | 0074-8300-01 |
| Carton of 2 | 0074-8350-01 | |
| 150 mg/mL single-dose prefilled syringe | Carton of 1 | 0074-1050-01 |
| 180 mg/1.2 mL (150 mg/mL) single-dose prefilled cartridge with on-body injector | Kit | 0074-1065-01 |
| 360 mg/2.4 mL (150 mg/mL) single-dose prefilled cartridge with on-body injector | Kit | 0074-1070-01 |
| Intravenous Infusion | ||
| 600 mg/10 mL (60 mg/mL) single-dose vial | Carton of 1 | 0074-5015-01 |
Subcutaneous Injection
SKYRIZI 150 mg/mL prefilled syringe or prefilled pen contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution. Each prefilled syringe or prefilled pen consists of a 1 mL glass syringe with a fixed 27-gauge ½ inch needle with needle guard.
SKYRIZI 90 mg/mL prefilled syringe contains a sterile, preservative-free, colorless to slightly yellow and clear to slightly opalescent solution. Each prefilled syringe consists of a 1 mL glass syringe with a fixed 29-gauge ½ inch needle with needle guard.
SKYRIZI 180 mg/1.2 mL (150 mg/mL) prefilled syringe contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution. Each prefilled syringe consists of a 2.25 mL glass syringe with a fixed 27-gauge ½ inch needle with needle guard.
SKYRIZI 180 mg/1.2 mL (150 mg/mL) cyclic olefin polymer prefilled cartridge with a septum and cap contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution for use with supplied on-body injector administration device.
SKYRIZI 360 mg/2.4 mL (150 mg/mL) cyclic olefin polymer prefilled cartridge with a septum and cap contains a sterile, preservative-free, colorless to yellow, and clear to slightly opalescent solution for use with supplied on-body injector administration device.
Intravenous Infusion
SKYRIZI 600 mg/10 mL (60 mg/mL) vial contains a sterile and preservative-free, colorless to slightly yellow, and clear to slightly opalescent solution. Each glass vial is closed with a stopper and blue flip cap.
Storage and Handling
- Store in a refrigerator at 36°F to 46° F (2°C to 8°C).
- Do not freeze.
- Do not shake.
- Keep in the original cartons to protect from light.
- Not made with natural rubber latex.
I NSTRUCTIONS FOR USE SKYRIZI ® (sky-RIZZ-ee) Pen (risankizumab-rzaa) injection, for subcutaneous use
Read Before First Use
Refer to the Medication Guide for product information.
Read this Instructions for Use before using SKYRIZI Pen (risankizumab-rzaa) injection.
Before using SKYRIZI, you should receive training from your healthcare provider on how to inject SKYRIZI.
SKYRIZI Single-Dose Pen
Important Information
- Store SKYRIZI in the refrigerator at 36°F to 46°F (2°C to 8°C).
- Keep SKYRIZI in the original carton to protect from light until you are ready to use.
- Before injecting, take the SKYRIZI carton out of the refrigerator. Leave the carton at room temperature and out of direct sunlight for 30 to 90 minutes.
- The liquid in the inspection window should look clear to yellow and may contain tiny white or clear particles.
- Do not use SKYRIZI if the liquid is discolored , cloudy or contains flakes or large particles .
- Do not use SKYRIZI if the expiration date (EXP) has passed.
- Do not use SKYRIZI if the liquid has been frozen, even if it has been thawed.
- Do not shake SKYRIZI.
- Do not use if the SKYRIZI Pen has been dropped or damaged.
- Do not use SKYRIZI if carton perforations are broken. Return product to pharmacy.
- Do not remove the dark gray cap until right before injection.
- SKYRIZI is not made with natural rubber latex.
Prepare SKYRIZI injection  | Take the SKYRIZI carton out of the refrigerator. Leave the carton at room temperature and out of direct sunlight for 30 to 90 minutes before injecting.
|
 | Check expiration date (EXP). Do not use the Pen if expiration date has passed. Place the following on a clean, flat surface:
|
 | Choose an injection site:
|
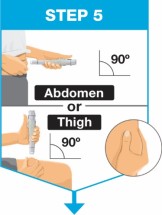 | Hold the Pen with the dark gray cap pointing up.
|
Give SKYRIZI injection 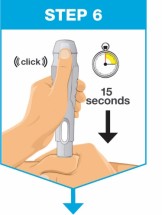 | Hold the Pen with your fingers on the gray hand grips. Turn the Pen so that the white needle sleeve points toward the injection site and you can see the green activator button. Pinch the skin at your injection site to make a raised area and hold it firmly. Place the white needle sleeve straight (90-degree angle) against the raised injection site. |
 | Hold the Pen so that you can see the green activator button and inspection window. Push and keep pressing the Pen down against the raised injection site.
|
 | Keep pressing the Pen down against the injection site. The injection is complete when:
|
After SKYRIZI injection  | When the injection is complete, slowly pull the Pen straight out from the skin. The white needle sleeve will cover the needle tip and make another “click.” After completing the injection, place a cotton ball or gauze pad on the skin at the injection site.
|
 | Throw away (dispose of) the used Pen in a FDA-cleared sharps disposal container right away after use. ● Do not dispose of used Pens in your household trash unless your community guidelines permit this. ● Do not recycle your sharps disposal container. The dark gray cap, alcohol swab, cotton ball or gauze pad, and packaging may be placed in your household trash. For more information, see “ Used SKYRIZI Prefilled Pen Disposal ” . |
Important Information
- Store SKYRIZI in the refrigerator at 36°F to 46°F (2°C to 8°C).
- Keep SKYRIZI in the original carton to protect from light until you are ready to use.
- Before injecting , take the SKYRIZI carton out of the refrigerator. Leave the carton at room temperature and out of direct sunlight for 30 to 90 minutes .
- The liquid in the inspection window should look clear to yellow and may contain tiny white or clear particles.
- Do not use SKYRIZI if the liquid is discolored , cloudy or contains flakes or large particles .
- Do not use SKYRIZI if the expiration date (EXP) has passed.
- Do not use SKYRIZI if the liquid has been frozen , even if it has been thawed.
- Do not shake SKYRIZI.
- Do not use if the SKYRIZI Pen has been dropped or damaged .
- Do not use SKYRIZI if carton perforations are broken. Return product to pharmacy .
- Do not remove the dark gray cap until right before injection.
- SKYRIZI is not made with natural rubber latex.
Keep the SKYRIZI Pen and sharps disposal container out of the reach of children.
Call your healthcare provider or (866) SKYRIZI or (866) 759-7494 if you need help or do not know how to proceed.
Questions About Using the SKYRIZI Pen
Q. What if I need help on how to inject SKYRIZI?
A. Call your healthcare provider or (866) SKYRIZI or (866) 759-7494 if you need help.
Q. I have removed the dark gray cap and pressed the green activator button. Why isn’t my injection starting?
A. The green activator button will not start the injection unless the white needle sleeve is pressed firmly against the injection site.
Q. How do I know when the injection is complete?
A. The injection is complete if the Pen makes a second “click” or the yellow indicator fills the inspection window. This takes up to 15 seconds.
Q. What should I do if there are more than a few drops of liquid on the injection site?
A. Call (866) SKYRIZI or (866) 759-7494 for help.
Q. What should I do with the used Pen after my injection?
A. Dispose of the used Pen in a sharps disposal container right after use. Do not dispose of the used Pen in your household trash.
You can sign up to receive sharps containers for SKYRIZI Pen disposal at no additional cost by going to www.SKYRIZI.comor calling (866) SKYRIZI or (866) 759-7494 .
Call (866) SKYRIZI or (866) 759-7494 or go to www.SKYRIZI.com for help with your injection.
 | To help remember when to inject, mark your calendar with the date you give your SKYRIZI injection. |
Keep the SKYRIZI Pen and sharps disposal container out of the reach of children.
Call your healthcare provider or (866) SKYRIZI or (866) 759-7494 if you need help or have questions about the use of SKYRIZI.
Used SKYRIZI Prefilled Pen Disposal
If you do not have a FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used Pens.
For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA’s website at: www.fda.gov/safesharpsdisposal.
Do not recycle your used sharps disposal container.
Manufactured by: AbbVie Inc., North Chicago, IL 60064, U.S.A. US License Number 1889 SKYRIZI ® is a registered trademark of AbbVie Biotechnology Ltd. © 2019-2024 AbbVie Inc. 20086179
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 6/2024
12.1 Mechanism of Action
Risankizumab-rzaa is a humanized IgG1 monoclonal antibody that selectively binds to the p19 subunit of human IL-23 cytokine and inhibits its interaction with the IL-23 receptor. IL-23 is a naturally occurring cytokine that is involved in inflammatory and immune responses.
Risankizumab-rzaa inhibits the release of pro-inflammatory cytokines and chemokines.