Get your patient on Thallous Chloride Tl 201- Thallous Chloride, Tl 201 injection, Solution (Thallous Chloride, Tl 201)
Thallous Chloride Tl 201- Thallous Chloride, Tl 201 injection, Solution prescribing information
INDICATIONS AND USAGE
Thallous Chloride Tl 201 Injection is indicated for use with planar scintigraphy or single-photon emission computed tomography (SPECT) for the following applications:
- Myocardial perfusion imaging in adults for the diagnosis of coronary artery disease by localization of:
- Non-reversible defects (myocardial infarction)
- Reversible defects (myocardial ischemia) when used in conjunction with exercise or pharmacologic stress
- Localization of sites of parathyroid hyperactivity pre- and post-operatively in adults with elevated serum calcium and parathyroid hormone levels
DOSAGE AND ADMINISTRATION
- • For myocardial perfusion imaging:
- For localization of parathyroid hyperactivity, planar or SPECT: 75 MBq to 130 MBq (2 mCi to 3.5 mCi)
- Administer by intravenous injection. (2.2 )
- See full prescribing information for administration and imaging instructions and radiation dosimetry information. (2.3 , 2.4 )
2.1 Radiation Safety – Drug Handling
Handle Thallous Chloride Tl 201 Injection with appropriate safety measures to minimize radiation exposure [see Warnings and Precautions (5.3 )] . Use waterproof gloves, effective radiation shielding, and other appropriate safety measures when preparing and handling Thallous Chloride Tl 201 Injection.
Radiopharmaceuticals should be used by or under the control of healthcare providers who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
Recommended Dose
Myocardial Perfusion Imaging in Adults
- Planar scintigraphy: 37 MBq to 74 MBq (1 mCi to 2 mCi) administered intravenously
- SPECT: 74 MBq to 111 MBq (2 mCi to 3 mCi) administered intravenously
Parathyroid Hyperactivity Localization in Adults
Planar or SPECT: 75 MBq to 130 MBq (2 mCi to 3.5 mCi) administered intravenously
2.3 Administration and Imaging Instructions
Patient Preparation Instruct patients to hydrate before and after Thallous Chloride Tl 201 Injection administration and to void before imaging and frequently thereafter following Thallous Chloride Tl 201 Injection administration [see Warnings and Precautions (5.3 )] .
Administration
- Use aseptic technique and radiation shielding when withdrawing and administering Thallous Chloride Tl 201 Injection.
- Visually inspect the drug for particulate matter and discoloration prior to administration, whenever the solution and container permit. Do not use if contents are turbid or discolored.
- Measure the patient dose with a dose calibrator immediately prior to administration.
- Use within 6 days from the manufacturer’s calibration date or 9 days from the date of manufacture, whichever comes first.
- Dispose of unused products in a safe manner in compliance with applicable regulations.
Myocardial Perfusion Imaging For resting myocardial studies, begin imaging 10 minutes to 20 minutes after administration of Thallous Chloride Tl 201 Injection. Target-to-background ratios are improved when patients are injected upright and in the fasting state; the upright position reduces the hepatic and gastric thallium-201 concentration.
For exercise stress testing, administer Thallous Chloride Tl 201 Injection at the start of a period of maximum stress, which is sustained for approximately 30 seconds after injection. Begin imaging within 10 minutes after administration to obtain maximum target-to-background ratios. Within 2 hours after the completion of the stress testing, the target-to-background ratios may decrease in lesions that are attributable to transient ischemia.
Parathyroid Hyperactivity Localization For localization of parathyroid hyperactivity, administer Thallous Chloride Tl 201 Injection before, with, or after a minimal dose of a thyroid imaging agent such as sodium pertechnetate Tc 99m injection or sodium iodide I 123 capsules to enable thyroid subtraction imaging.
Radiation Dosimetry
Estimated absorbed radiation doses from an intravenous injection of Thallous Chloride Tl 201 Injection are shown in Table 1.
| Organ/ Tissue | Absorbed Dose per Unit Activity Administered (mGy/MBq) |
| Adrenals | 0.063 |
| Brain | 0.057 |
| Breasts | 0.034 |
| GB Wall | 0.083 |
| GI Tract | |
| LLI Wall | 0.300 |
| Small Intestine | 0.379 |
| Stomach | 0.171 |
| ULI Wall | 0.297 |
| Heart Wall | 0.247 |
| Kidneys | 0.410 |
| Liver | 0.094 |
| Lungs | 0.047 |
| Muscle | 0.046 |
| Ovaries | 0.102 |
| Pancreas | 0.075 |
| Red Marrow | 0.044 |
| Bone Surfaces | 0.094 |
| Skin | 0.032 |
| Spleen | 0.166 |
| Testes | 0.209 |
| Thymus | 0.046 |
| Thyroid | 0.542 |
| Urinary Bladder Wall | 0.063 |
| Uterus | 0.086 |
| Total Body | 0.058 |
| Effective Dose (mSv/MBq) | 0.145 |
1 Assumed percentage of 98.3% thallium-201, 0.3% thallium-200, 1.2% thallium-202, and 0.2% lead-203
DOSAGE FORMS AND STRENGTHS
Injection: a clear, colorless solution available in the following strength at calibration time:
- 207.2 MBq (5.6 mCi) per 5.6 mL (37 MBq (1 mCi) per mL) in a multiple-dose vial
USE IN SPECIFIC POPULATIONS
Lactation: Temporarily discontinue breastfeeding. A lactating woman should pump and discard breast milk for at least 96 hours after Thallous Chloride Tl 201 Injection administration. (8.2 )
Pregnancy
Risk Summary
There are no data on Thallous Chloride Tl 201 Injection use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Studies using human placentas demonstrate that thallous chloride Tl 201 crosses the placenta. No animal reproductive studies have been conducted.
All radiopharmaceuticals, including Thallous Chloride Tl 201 Injection have the potential to cause fetal harm depending on the fetal stage of development and the magnitude of the radiation dose. If considering administration of Thallous Chloride Tl 201 Injection to a pregnant woman, inform the patient about the potential for adverse pregnancy outcomes based on the radiation dose from Thallous Chloride Tl 201 Injection and gestational timing of exposure.
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 3% and 10% to 20%, respectively.
Lactation
Risk Summary
Thallous chloride Tl 201 has been detected in the milk of lactating women. There are no data on the effects of thallous chloride Tl 201 on milk production or the effects of thallous chloride Tl 201 on the breastfed infant.
Exposure of the breastfed infant to thallous chloride Tl 201 can be minimized by advising a lactating woman to temporarily discontinue breastfeeding and pump and discard milk for a minimum of 96 hours after administration of Thallous Chloride Tl 201 Injection. Minimize close contact with infants if the administered dose would result in an effective dose greater than 1 mSv to the infant. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Thallous Chloride Tl 201 Injection and any potential adverse effects on the breastfed infant from Thallous Chloride Tl 201 Injection or from the underlying maternal condition.
Pediatric Use
The safety and effectiveness of Thallous Chloride Tl 201 Injection in pediatric patients have not been established.
Geriatric Use
Clinical studies of Thallous Chloride Tl 201 Injection for myocardial perfusion imaging did not include sufficient numbers of subjects aged younger than 65 years to determine whether they respond differently from younger subjects. Other reported experience has not revealed clinically relevant differences in the response of elderly in comparison to younger patients.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Hypersensitivity Reactions: Anaphylactic reactions characterized by cardiovascular, respiratory, and cutaneous symptoms may occur. Have resuscitation equipment and trained staff readily available. (5.1 )
- Risk Associated with Stress Testing: Induction of cardiovascular stress may be associated with serious adverse reactions. Perform stress testing in a setting where cardiac resuscitation equipment and trained staff are readily available. (5.2 )
- Radiation Risk: Ensure safe handling to minimize radiation exposure to patients and health care providers. (2.1 , 5.3 )
5.1 Hypersensitivity Reactions
Thallous Chloride Tl 201 Injection may cause anaphylactic reactions characterized by cardiovascular, respiratory, and cutaneous symptoms [see Adverse Reactions (6 )] . Have resuscitation equipment and trained staff readily available.
Risks Associated with Stress Testing
Induction of cardiovascular stress might be associated with serious adverse reactions such as myocardial infarction, arrhythmia, hypotension or hypertension, ECG abnormalities, chest pain, bronchoconstriction, and cerebrovascular events [see Adverse Reactions (6 )] . Perform stress testing in the setting where cardiac resuscitation equipment and trained staff are readily available. Perform pharmacologic stress in accordance with the pharmacologic stress agent’s prescribing information.
Radiation Risk
Thallous Chloride Tl 201 Injection contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe handling to minimize radiation exposure to patients and health care providers. Advise patients to hydrate before and after administration and to void frequently after administration [see Dosage and Administration (2.1 , 2.3 )].
5.4 Injection Site Reactions and Tissue Damage
Injection site reactions and extravasation have been reported after administration of Thallous Chloride Tl 201 Injection [see Adverse Reactions (6 )] . Administer Thallous Chloride Tl 201 Injection strictly into the vein to avoid local tissue accumulation and irradiation. Confirm intravenous patency before injection.
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1 )]
- Risk Associated with Stress Testing [see Warnings and Precautions (5.2 )]
- Injection Site Reactions and Tissue Damage [see Warnings and Precautions (5.4 )]
The following adverse reactions associated with the use of Thallous Chloride Tl 201 Injection were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure:
Cardiovascular, respiratory, and cerebrovascular disorders: myocardial infarction, arrhythmia, hypotension, bronchoconstriction, and cerebrovascular events in patients who have undergone stress testing
Gastrointestinal disorders: nausea, vomiting, and diarrhea
General disorders and administration site conditions: injection site reactions (burning, pain, redness, swelling, warmth, and tissue damage with chronic ulcer formation), chills, fever, and sweating
Immune system disorders: hypersensitivity (anaphylaxis, hypotension, shortness of breath, pruritus, flushing, and diffuse rash)
DESCRIPTION
11.1 Drug Characteristics
Thallous Chloride Tl 201 Injection is a sterile, radioactive diagnostic drug for intravenous use.
Each mL contains 37 MBq (1 mCi) thallous chloride Tl 201 at calibration time and the following inactive ingredients: 9 mg sodium chloride and 0.9% (v/v) benzyl alcohol. The pH is adjusted to between 4.5 to 7.0 with hydrochloric acid and/or sodium hydroxide.
Thallium-201 is cyclotron produced. At the time of calibration it contains no more than 1% thallium-200, no more than 1% thallium-202, no more than 0.25% lead-203, and no less than 98% thallium-201 as a percentage of total activity. No carrier has been added.
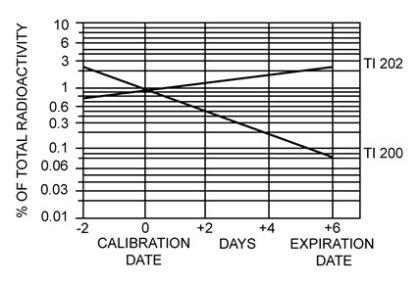
The concentration of each radionuclidic contaminant changes with time. Figure 1 shows maximum concentration of thallium-200 (Tl 200) and thallium-202 (Tl 202) radionuclidic contaminants as a function of time.
Figure 1. Radionuclidic Contaminants
Nuclear Physical Characteristics
Thallium-201, with a physical half-life of 72.9 hours, decays by electron capture to mercury-201. Photons that are useful for detection and imaging are listed in Table 2. The lower energy x-rays obtained from the mercury-201 daughter of thallium-201 are recommended for myocardial imaging, because the mean percent disintegration at 68.9 to 80.3 keV is much greater than the combination of gamma-4 and gamma-6 mean percent disintegration.
Radiation | Mean Percent/ Disintegration | Energy (keV) |
Gamma-4 | 2.7 | 135.3 |
Gamma-6 | 10.0 | 167.4 |
Mercury x-rays | 94.4 | 68.9-80.3 |
The specific gamma ray constant for thallium-201 is 0.45 R/mCi-hr at 1 cm. The first half-value thickness of lead (Pb) is 0.026 cm. A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 3. For example, the use of 0.194 cm of lead will decrease the external radiation exposure by a factor of about 1,000.
cm of Lead (Pb) | Coefficient of Attenuation |
| 0.026 | 0.5 |
| 0.052 | 10 -1 |
| 0.089 | 10 -2 |
| 0.194 | 10 -3 |
| 0.310 | 10 -4 |
To correct for physical decay of the radionuclide, the fractions that remain at selected intervals after calibration time are shown in Table 4.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
| 0• | 1 | 66 | 0.53 |
| 6 | 0.95 | 72 | 0.50 |
| 12 | 0.89 | 78 | 0.48 |
| 18 | 0.84 | 84 | 0.45 |
| 24 | 0.80 | 90 | 0.43 |
| 30 | 0.75 | 96 | 0.40 |
| 36 | 0.74 | 108 | 0.36 |
| 42 | 0.67 | 120 | 0.32 |
| 48 | 0.63 | 132 | 0.29 |
| 54 | 0.60 | 144 | 0.25 |
| 60 | 0.57 |
• Calibration Time
CLINICAL PHARMACOLOGY
Mechanism of Action
Thallous chloride Tl 201 with no carrier added accumulates in viable myocardium in a manner analogous to that of potassium. Experiments in healthy subjects using labeled microspheres have shown that the myocardial distribution of thallous chloride Tl 201 correlates with regional perfusion.
In clinical studies, Thallous Chloride Tl 201 images have been found to visualize areas of infarction as “cold” or nonlabeled regions that were confirmed by electrocardiographic and enzyme changes. Regions of transient myocardial ischemia corresponding to areas perfused by coronary arteries with partial stenoses have been visualized when Thallous Chloride Tl 201 Injection was administered in conjunction with an exercise stress test. Anatomic configurations may interfere with visualization of the right coronary artery territory.
Pharmacodynamics
The pharmacodynamics of thallous chloride Tl 201 have not been established.
Pharmacokinetics
Distribution After intravenous administration, thallous chloride Tl 201 clears from the blood with maximal concentration in normal myocardium occurring at about 10 minutes. It will, in addition, localize in parathyroid adenomas; it is not specific since it will localize to a lesser extent in sites of parathyroid hyperplasia and other abnormal tissues such as thyroid adenoma, neoplasia (e.g., parathyroid carcinoma), and sarcoid. Biodistribution is generally proportional to organ blood flow at the time of injection. Blood clearance of thallous chloride Tl 201 is primarily by the myocardium, thyroid, liver, kidneys, and stomach with the remainder distributing fairly uniformly throughout the body. The dosimetry data in Table 1 reflect this distribution pattern and are based on a biological half-life of 2.4 days.
Elimination Five minutes after intravenous administration only 5% to 8% of injected activity remained in the blood. A biexponential disappearance curve was obtained, with 91.5 % of the blood radioactivity disappearing with a half-time of about 5 minutes. The remainder had a half-time of about 40 hours.
Excretion Approximately 4% to 8% of the injected dose was excreted in the urine in the first 24 hours. The whole body disappearance half-time was 9.8 ± 2.5 days. Kidney concentration was found to be about 3% of the injected activity and the testicular content was 0.15%. Net thyroid activity was determined to be only 0.2% of the injected dose, and the activity disappeared in 24 hours. From anterior and posterior whole-body scans, it was determined that about 45% of the injected dose was in the large intestines and contiguous structures (liver, kidneys, abdominal musculature).
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term animal studies have been performed to evaluate carcinogenic potential, mutagenic potential, or whether this drug affects fertility in males or females.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied Thallous Chloride Tl 201 Injection is supplied as a clear, colorless solution in the following strength at calibration time:
207.2 MBq (5.6 mCi) per 5.6 mL (37 MBq (1 mCi) per mL) in a multiple-dose vial (NDC 69945-120-56)
Storage and Handling
Store Thallous Chloride Tl 201 Injection in the original container with radiation shielding at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature].
Dispose of any unused product in accordance with all federal, state, and local laws and institutional requirements.
This preparation is for use by persons under license by the Nuclear Regulatory Commission or the relevant regulatory authority of an Agreement State.
Mechanism of Action
Thallous chloride Tl 201 with no carrier added accumulates in viable myocardium in a manner analogous to that of potassium. Experiments in healthy subjects using labeled microspheres have shown that the myocardial distribution of thallous chloride Tl 201 correlates with regional perfusion.
In clinical studies, Thallous Chloride Tl 201 images have been found to visualize areas of infarction as “cold” or nonlabeled regions that were confirmed by electrocardiographic and enzyme changes. Regions of transient myocardial ischemia corresponding to areas perfused by coronary arteries with partial stenoses have been visualized when Thallous Chloride Tl 201 Injection was administered in conjunction with an exercise stress test. Anatomic configurations may interfere with visualization of the right coronary artery territory.