Get your patient on Tyrvaya (Varenicline)
Tyrvaya prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Tyrvaya patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
Dosing Information
Spray TYRVAYA once in each nostril twice daily (approximately 12 hours apart). If a dose is missed, resume regular dosing at the next scheduled dose time.
Priming Instructions
Priming : Prime TYRVAYA before initial use by pumping seven (7) actuations into the air away from the face. When TYRVAYA has not been used for more than 5 days, re-prime with 1 spray into the air. Do not shake.

Tyrvaya prescribing information
INDICATIONS AND USAGE
TYRVAYA (varenicline solution) nasal spray is indicated for the treatment of the signs and symptoms of dry eye disease.
DOSAGE AND ADMINISTRATION
Dosing Information
Spray TYRVAYA once in each nostril twice daily (approximately 12 hours apart). If a dose is missed, resume regular dosing at the next scheduled dose time.
Priming Instructions
Priming : Prime TYRVAYA before initial use by pumping seven (7) actuations into the air away from the face. When TYRVAYA has not been used for more than 5 days, re-prime with 1 spray into the air. Do not shake.
DOSAGE FORMS AND STRENGTHS
Nasal spray delivering 0.03 mg of varenicline in each spray (0.05 mL).
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no available data on TYRVAYA use in pregnant women to inform any drug associated risks. In animal reproduction studies, varenicline did not produce malformations at clinically relevant doses.
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Pregnant rats and rabbits received varenicline succinate during organogenesis at oral doses up to 15 and 30 mg/kg/day, respectively. While no fetal structural abnormalities occurred in either species, maternal toxicity, characterized by reduced body weight gain, and reduced fetal weights occurred in rabbits at the highest dose (4864 times the MRHD on a mg/m 2 basis).
In a pre- and postnatal development study, pregnant rats received up to 15 mg/kg/day of oral varenicline succinate from organogenesis through lactation. Maternal toxicity, characterized by a decrease in body weight gain, was observed at 15 mg/kg/day (1216 times the MRHD on a mg/m 2 basis). Decreased fertility and increased auditory startle response occurred in offspring at the highest maternal dose of 15 mg/kg/day.
Lactation
Risk Summary
There are no data on the presence of varenicline in human milk, the effects on the breastfed infant, or the effects on milk production. In animal studies varenicline was present in milk of lactating rats. However, due to species-specific differences in lactation physiology, animal data may not reliably predict drug levels in human milk.
The lack of clinical data during lactation precludes a clear determination of the risk of TYRVAYA to an infant during lactation; however, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for TYRVAYA and any potential adverse effects on the breastfed child from TYRVAYA.
Pediatric Use
Safety and efficacy of TYRVAYA in pediatric patients have not been established.
Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and younger adult patients.
CONTRAINDICATIONS
None
ADVERSE REACTIONS
The most common adverse reaction reported in 82% of patients was sneezing. Events that were reported in 5-16% of patients were cough, throat irritation, and instillation-site (nose) irritation. (6 )
To report SUSPECTED ADVERSE REACTIONS, contact Oyster Point Pharma at 1-877-EYE-0123 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In three clinical studies of dry eye disease conducted with varenicline solution nasal spray, 349 patients received at least 1 dose of TYRVAYA. The majority of patients had 31 days of treatment exposure, with a maximum exposure of 105 days.
The most common adverse reactions reported in 82% of TYRVAYA treated patients was sneezing. Other common adverse reactions that were reported in >5% of patients include cough (16%), throat irritation (13%), and instillation-site (nose) irritation (8%).
DESCRIPTION
TYRVAYA nasal spray contains varenicline which is a partial nicotinic acetylcholine receptor agonist of α4β2, α4α6β2, α3β4, and α3α5β4 receptors and a full α7 receptor agonist.
Varenicline, as the tartrate salt, is a powder which is a white to off-white to slightly yellow solid whose chemical name is 7,8,9,10-tetrahydro-6,10-methano-6 H -pyrazino[2,3-h][3]benzazepine, (2 R ,3 R )-2,3-dihydroxybutanedioate (1:1). It is highly soluble in water. Varenicline tartrate has a molecular weight of 361.35 Daltons and a molecular formula of C 13 H 13 N 3 ⋅ C 4 H 6 O 6 . The chemical structure is:
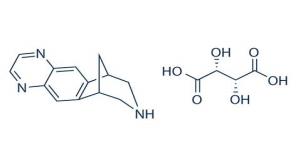
TYRVAYA (varenicline solution) nasal spray is formulated for intranasal use as a clear 0.6 mg/mL strength solution, at pH 6.4. After priming [see Dosage and Administration (2.2 )] , each actuation delivers a 0.05 mL spray containing 0.03 mg varenicline free base, equivalent to 0.05 mg of varenicline tartrate. The formulation also contains the following inactive ingredients: sodium phosphate dibasic heptahydrate, monobasic sodium phosphate anhydrous, sodium chloride, sodium hydroxide and/or hydrochloric acid (to adjust pH) and water for injection.
CLINICAL PHARMACOLOGY
Mechanism of Action
The efficacy of TYRVAYA in dry eye disease is believed to be the result of varenicline's activity at heteromeric sub-type(s) of the nicotinic acetylcholine (nACh) receptor where its binding produces agonist activity and activates the trigeminal parasympathetic pathway resulting in increased production of basal tear film as a treatment for dry eye disease. Varenicline binds with high affinity and selectivity at human α4β2, α4α6β2, α3β4, α3α5β4 and α7 neuronal nicotinic acetylcholine receptors. The exact mechanism of action is unknown at this time.
Pharmacokinetics
Absorption/Distribution
Following administration of 0.12 mg (0.06 mg per 50-µL spray in each nostril), a strength of varenicline that is higher than the labeled concentration, varenicline can be detected in plasma by 5 minutes, generally achieves peak concentration within 2 hours, with a mean C max of 0.34 ng/mL, and has an AUC 0-inf of 7.46 h•ng/mL. The systemic exposure (AUC 0-inf ) following this intranasal dose was approximately 7.5% of the exposure observed following a 1 mg oral dose of varenicline.
Metabolism/Elimination
The mean ± SD elimination half-life of varenicline after intranasal administration is approximately 19 ± 10 hours. Varenicline undergoes minimal metabolism with 92% excreted as unchanged drug in the urine.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Lifetime carcinogenicity studies were performed in CD-1 mice and Sprague-Dawley rats. There was no evidence of a carcinogenic effect in mice administered varenicline by oral gavage for 2 years at doses up to 20 mg/kg/day (810 times the maximum recommended human dose [MRHD], on a mg/m 2 basis). Rats were administered varenicline (1, 5, and 15 mg/kg/day) by oral gavage for 2 years. In male rats (n = 65 per sex per dose group), incidences of hibernoma (tumor of the brown fat) were increased at the mid dose (1 tumor, 5 mg/kg/day, 405 times the MRHD on a mg/m 2 basis) and maximum dose (2 tumors, 15 mg/kg/day, 1216 times the MRHD on a mg/m 2 basis). The clinical relevance of this finding to humans has not been established. There was no evidence of carcinogenicity in female rats.
Mutagenesis
Varenicline was not genotoxic, with or without metabolic activation, in the following assays: Ames bacterial mutation assay; mammalian CHO/HGPRT assay; and tests for cytogenetic aberrations in vivo in rat bone marrow and in vitro in human lymphocytes.
Impairment of Fertility
There was no evidence of impairment of fertility in either male or female Sprague-Dawley rats administered varenicline succinate up to 15 mg/kg/day (1216 times the MRHD on a mg/m 2 basis). Maternal toxicity, characterized by a decrease in body weight gain, was observed at 15 mg/kg/day. A decrease in fertility was noted in the offspring of pregnant rats administered varenicline succinate at an oral dose of 15 mg/kg/day. The decrease in fertility in the offspring of treated female rats was not evident at an oral dose of 3 mg/kg/day (243 times the MRHD, on a mg/m 2 basis).
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In three clinical studies of dry eye disease conducted with varenicline solution nasal spray, 349 patients received at least 1 dose of TYRVAYA. The majority of patients had 31 days of treatment exposure, with a maximum exposure of 105 days.
The most common adverse reactions reported in 82% of TYRVAYA treated patients was sneezing. Other common adverse reactions that were reported in >5% of patients include cough (16%), throat irritation (13%), and instillation-site (nose) irritation (8%).
HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
TYRVAYA (varenicline solution) nasal spray is available in a carton containing two (2) nasal spray amber glass Type I bottles. Each bottle consists of a white nasal pump and blue dust cover, delivering 0.03 mg varenicline per spray (0.05 mL). Each bottle delivers one spray in each nostril twice daily for 15 days.
Two nasal spray bottles in each carton, containing 60 sprays per bottle, equivalent to 30-days’ supply with one spray in each nostril twice daily (NDC 73521-030-02).
16.2 Storage and Handling
- Store TYRVAYA nasal spray at 20°C to 25°C (68°F to 77°F). Do not freeze.
- Discard TYRVAYA nasal spray bottle 30 days after opening bottle.
Instructions for Use
TYRVAYA ® (Teer-vye-ah) (varenicline solution) nasal spray, for intranasal use
Read this Instructions for Use before you start using TYRVAYA and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Important information you need to know before using TYRVAYA: | Parts of your TYRVAYA nasal spray: |
| 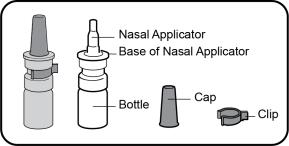 |
Steps for priming TYRVAYA before first Use | |
Step 1. Remove the cap and the clip. Do not throw away the cap or the clip. The cap and the clip will be placed back on to the nasal applicator after each use. | 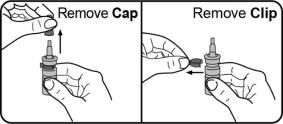 |
Step 2. Hold the nasal spray bottle upright and away from your face. Place 1 finger on each side of the base of the nasal applicator and place your thumb underneath the bottle. |  |
Step 3. Prime the nasal spray bottle by fully pressing and releasing the nasal applicator 7 times with your thumb and fingers. You may feel some resistance but be sure to press all the way down. You may not see a spray released each time you press and release the nasal applicator. You should see a spray by the 7 th time you press and release. Spray away from yourself and others. TYRVAYA is now primed for use. | 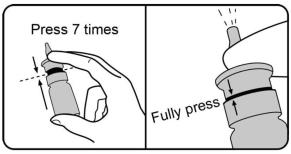 |
- Reprime : If you do not use TYRVAYA for more than 5 days , you will need to reprime the nasal spray bottle with 1 spray before you start using it. To reprime, hold the nasal spray bottle upright and away from your face and fully press and release the nasal spray applicator 1 time .
- Avoid priming the nasal spray bottle more than needed : Priming the nasal spray bottle more than needed will reduce the amount of medicine in the nasal spray bottle.
Steps for using TYRVAYA nasal spray after priming | |
Step 1. Blow your nose to clear your nostrils, if needed. | 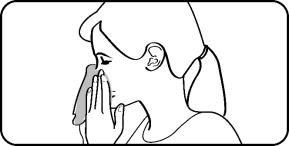 |
Step 2. Remove the cap and clip. Do not throw away the cap or the clip. The cap and the clip will be placed back on to the nasal applicator after each use. | 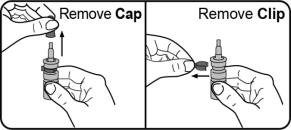 |
Step 3. Hold the nasal spray bottle upright. Place 1 finger on each side of the base of the nasal applicator and your thumb underneath the bottle. | 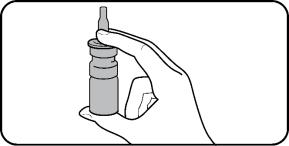 |
Step 4. Tilt your head back slightly without lying down. | 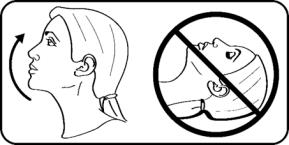 |
Step 5. Insert the nasal applicator into the left or right nostril. Tilt the nasal applicator and point the tip of the nasal applicator towards the top of the ear on the same side as your nostril. Do not press the tip of the nasal applicator against the wall of the inside of your nose. Leave a space between the tip of the nasal applicator and the wall of the inside of your nose. | 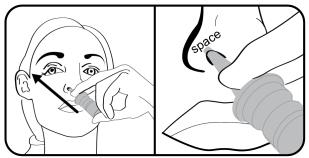 |
Step 6. Place your tongue to the roof of your mouth and breathe gently while fully pressing and releasing the nasal applicator 1-time to release a spray into your nostril. Repeat Steps 5 and 6 to deliver a second spray in the other nostril. | 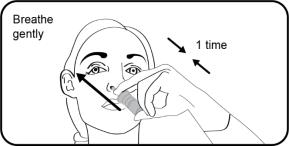 |
Step 7. Wipe the nasal applicator with a clean tissue. | 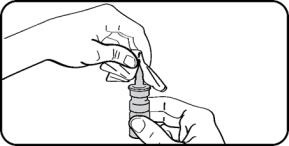 |
Step 8. Replace the clip and the cap. Repeat Steps 1 to 8 each time you use TYRVAYA. | 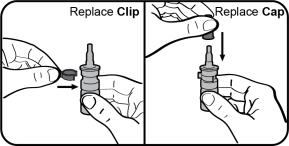 |
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
TYRVAYA ® is a registered trademark of Oyster Point Pharma, Inc., a Viatris company.
TYRVAYA ® and/or the use of TYRVAYA ® in a method may be covered by one or more patents or patent applications, available at www.oysterpointrx.com/patent-notices.
Manufactured for: Oyster Point Pharma, Inc., 202 Carnegie Center, Suite 106, Princeton, NJ 08540
©2023 Oyster Point Pharma, Inc.
Revised: 2/2024
OYP:IFU:TYRVA:R1
Mechanism of Action
The efficacy of TYRVAYA in dry eye disease is believed to be the result of varenicline's activity at heteromeric sub-type(s) of the nicotinic acetylcholine (nACh) receptor where its binding produces agonist activity and activates the trigeminal parasympathetic pathway resulting in increased production of basal tear film as a treatment for dry eye disease. Varenicline binds with high affinity and selectivity at human α4β2, α4α6β2, α3β4, α3α5β4 and α7 neuronal nicotinic acetylcholine receptors. The exact mechanism of action is unknown at this time.