Get your patient on Tzield (Teplizumab-Mzwv)
Tzield prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Tzield patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
- Confirm Stage 2 T1D by documenting at least two positive pancreatic islet autoantibodies in those who have dysglycemia without overt hyperglycemia using an oral glucose tolerance test (OGTT) or alternative method if appropriate and OGTT is not available (2.1 ).
- In patients who meet criteria for a diagnosis of Stage 2 type 1 diabetes, ensure the patient's diagnosis confirms an autoimmune origin and does not suggest type 2 diabetes (2.1 ).
- Prior to initiating TZIELD, obtain a complete blood count and liver enzyme tests. Evaluate patients for active EBV and CMV infection and confirm undetectable viral load (e.g., polymerase chain reaction testing). Use of TZIELD is not recommended in patients with certain laboratory abnormalities or patients with laboratory or clinical evidence of active infection with EBV or CMV (2.2 ).
- Premedicate with: (1) a nonsteroidal anti-inflammatory drug (NSAID) or acetaminophen, (2) an antihistamine, and (3) consider use of an antiemetic before each TZIELD dose for at least the first 5 days of the 14-day treatment course (2.3 ).
- Administer TZIELD by intravenous infusion once daily for 14 days. See full prescribing information for the dosing schedule, minimum infusion duration according to age, and recommendations regarding missed doses (2.4 , 2.5 ).
- See full prescribing information for recommendations on monitoring for changes in lymphocyte counts, liver enzymes, bilirubin, and symptoms of viral reactivation and discontinuing treatment (2.6 ).
- Must dilute TZIELD in 0.9% Sodium Chloride Injection, USP. See full prescribing information for detailed preparation and administration instructions (2.7 ).
Patient Selection
Select adult and pediatric patients 1 year of age and older with Stage 2 T1D for TZIELD treatment to delay the onset of Stage 3 T1D based on the confirmation of:
- At least two positive pancreatic islet cell autoantibodies, and
- Dysglycemia without overt hyperglycemia using an oral glucose tolerance test (if an oral glucose tolerance test is not available, an alternative method for diagnosing dysglycemia without overt hyperglycemia may be appropriate)
Ensure the patient's diagnosis confirms an autoimmune origin and does not suggest type 2 diabetes or other forms of diabetes. These may include, but are not limited to, genetic forms of diabetes, maturity-onset diabetes of the young (MODY), latent autoimmune diabetes in adults (LADA), or diabetes secondary to medications or surgery.
Laboratory and Infection Evaluation, and Vaccination Prior to Initiation
- Prior to initiating TZIELD, obtain a complete blood count and liver enzyme tests. Use of TZIELD is not recommended in patients with [see Warnings and Precautions (5.3) and Adverse Reactions (6.1) ] :
- Lymphocyte count less than 1,000 lymphocytes/mcL
- Hemoglobin less than 10 g/dL
- Platelet count less than 150,000 platelets/mcL
- Absolute neutrophil count less than 1,500 neutrophils/mcL
- Elevated alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 2 times the upper limit of normal (ULN) or bilirubin greater than 1.5 times ULN
- Active serious infection or chronic active infection other than localized skin infections [see Warnings and Precautions (5.3) ].
- Prior to initiating treatment, evaluate patients for active EBV and CMV infection and confirm undetectable viral load [e.g., polymerase chain reaction (PCR) testing]. Use of TZIELD is not recommended in patients with laboratory or clinical evidence of active infection with EBV or CMV [see Warnings and Precautions (5.1) ] .
- Administer all age-appropriate vaccinations prior to starting TZIELD [see Warnings and Precautions (5.6) ] :
- Administer live-attenuated (live) vaccines at least 8 weeks prior to TZIELD treatment.
- Administer inactivated (killed) vaccines or mRNA vaccines at least 2 weeks prior to treatment.
Important Premedication Instructions
Prior to each of the first 5 days of TZIELD infusion [see Warnings and Precautions (5.2) ] :
- Premedicate with a nonsteroidal anti-inflammatory drug (NSAID) or acetaminophen
- Premedicate with an antihistamine, and
- Consider premedication with an antiemetic
If needed, administer additional premedication doses.
Recommended Dosage and Administration
Calculate the dosage using a body surface area (BSA) and administer TZIELD once daily for 14 consecutive days as follows:
- Day 1: 65 mcg/m 2
- Day 2: 125 mcg/m 2
- Day 3: 250 mcg/m 2
- Day 4: 500 mcg/m 2
- Days 5 through 14: 1,030 mcg/m 2
Administer TZIELD by intravenous infusion over a minimum of:
- 30 minutes in adult and pediatric patients aged 8 years and older.
- 2 hours in pediatric patients aged 1 to less than 8 years [see Clinical Pharmacology (12.3) ] .
Recommendations Regarding Missed Doses
If a planned TZIELD infusion is missed, resume dosing by administering all remaining doses on consecutive days to complete the 14-day treatment course.
Do not administer two doses on the same day.
Recommended Monitoring During Treatment with TZIELD
- Monitor lymphocyte count regularly (every 2–3 days) during TZIELD infusion and monitor for lymphocyte recovery following completion of TZIELD.
- If prolonged severe lymphopenia (<500 cells per mcL lasting 1 week or longer) develops, permanently discontinue TZIELD.
- Monitor patients for signs and symptoms of viral reactivation during TZIELD treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD [see Warnings and Precautions (5.1) ] .
- Monitor liver enzymes and bilirubin during treatment. Discontinue TZIELD treatment in patients who develop elevated ALT or AST more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN [see Warnings and Precautions (5.2) ] .
Preparation Instructions
Using the dose calculated according to BSA [see Dosage and Administration (2.4) ], dilute TZIELD prior to preparing the infusion according to the instructions below:
TZIELD Dilution Preparation:
- Prior to dilution, inspect TZIELD visually before use (the supplied solution is clear and colorless). Do not use TZIELD if particulate matter or coloration is seen.
- Prepare TZIELD using aseptic technique.
- If the calculated dose is:
- 2,000 mcg or less, then prepare:
- One sterile glass vial with 18 mL of 0.9% Sodium Chloride Injection or
- One ≤50 mL polyvinylchloride (PVC) with di-(2-ethylhexyl)phthalate (DEHP) infusion bag with 18 mL of 0.9% Sodium Chloride Injection.
- Greater than 2,000 mcg, then prepare:
- Two sterile glass vials with 18 mL of 0.9% Sodium Chloride Injection or
- Two ≤50 mL PVC with DEHP infusion bags with 18 mL of 0.9% Sodium Chloride Injection.
- 2,000 mcg or less, then prepare:
- Remove 2 mL of TZIELD from the single-dose vial and slowly add to the glass vial or PVC with DEHP infusion bag containing 18 mL of 0.9% Sodium Chloride Injection.
- Mix gently by slowly inverting the vial or rocking the infusion bag. The resulting 20 mL diluted TZIELD solution contains 100 mcg/mL of teplizumab-mzwv.
- If preparing a dose greater than 2,000 mcg, repeat the above process with second TZIELD vial and the glass vial, or PVC with DEHP infusion bag containing 18 mL of 0.9% Sodium Chloride Injection.
TZIELD Infusion Solution Preparation:
- Using an appropriately sized syringe, withdraw the volume of diluted TZIELD solution required for that day's calculated dose from the 100 mcg/mL dilution ( for a calculated dose 2,000 mcg or less ) or from both prepared 100 mcg/mL dilutions ( for a calculated dose more than 2,000 mcg ).
- Discard unused portion of remaining diluted TZIELD solution in the glass vial or infusion bag.
- Slowly add contents of the syringe containing the TZIELD dose to PVC with DEHP infusion bag containing 25 mL of 0.9% Sodium Chloride Injection ( for a calculated dose more than 2,000 mcg, add the cumulative volume for the calculated dose to a single infusion bag ). Gently rock the infusion bag to ensure that the solution mixes sufficiently. Do not shake.
- Prime the PVC with DEHP IV infusion set with the TZIELD infusion solution. Do not waste any infusion solution during the priming process.
- After infusion, flush the IV set with a volume of 0.9% Sodium Chloride Injection greater than or equal to the priming volume, to ensure full dose is administered. Same infusion rate should be used for flushing.
- If the TZIELD infusion solution is not used immediately, store the infusion at room temperature [15°C to 30°C (59°F to 86°F)]. Discard the TZIELD infusion solution if the infusion is unable to be completed within 4 hours of preparation.
Use of in-line filter is not recommended. If necessary, use a polyethylene sulfone (PES) filter.
Do not use light protected (colored) infusion sets.

Tzield prescribing information
WARNING: Viral Reactivation
- Serious, life-threatening cases of viral reactivation, including Epstein-Barr virus (EBV) and cytomegalovirus (CMV) reactivation have been reported with TZIELD. Patients who are immunocompromised are at increased risk. The majority of serious cases occurred in patients who continued TZIELD treatment despite persistent, severe lymphopenia. (5.1 , 5.4 )
- Test patients for active EBV and CMV infection prior to starting treatment. TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection. Adhere to lymphocyte count monitoring requirements and discontinuation recommendations. Monitor patients for signs and symptoms of viral reactivation following TZIELD treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD. (2.6 , 5.1 , 5.4 )
| Boxed Warning | 04/2026 |
| Indications and Usage (1 ) | 04/2026 |
| Dosage and Administration (2 ) | 04/2026 |
| Warnings and Precautions, Viral Reactivation (5.1 ) | 04/2026 |
INDICATIONS AND USAGE
TZIELD is indicated to delay the onset of Stage 3 type 1 diabetes (T1D) in adult and pediatric patients 1 year of age and older with Stage 2 type 1 diabetes [see Dosage and Administration (2.1) ] .
DOSAGE AND ADMINISTRATION
- Confirm Stage 2 T1D by documenting at least two positive pancreatic islet autoantibodies in those who have dysglycemia without overt hyperglycemia using an oral glucose tolerance test (OGTT) or alternative method if appropriate and OGTT is not available (2.1 ).
- In patients who meet criteria for a diagnosis of Stage 2 type 1 diabetes, ensure the patient's diagnosis confirms an autoimmune origin and does not suggest type 2 diabetes (2.1 ).
- Prior to initiating TZIELD, obtain a complete blood count and liver enzyme tests. Evaluate patients for active EBV and CMV infection and confirm undetectable viral load (e.g., polymerase chain reaction testing). Use of TZIELD is not recommended in patients with certain laboratory abnormalities or patients with laboratory or clinical evidence of active infection with EBV or CMV (2.2 ).
- Premedicate with: (1) a nonsteroidal anti-inflammatory drug (NSAID) or acetaminophen, (2) an antihistamine, and (3) consider use of an antiemetic before each TZIELD dose for at least the first 5 days of the 14-day treatment course (2.3 ).
- Administer TZIELD by intravenous infusion once daily for 14 days. See full prescribing information for the dosing schedule, minimum infusion duration according to age, and recommendations regarding missed doses (2.4 , 2.5 ).
- See full prescribing information for recommendations on monitoring for changes in lymphocyte counts, liver enzymes, bilirubin, and symptoms of viral reactivation and discontinuing treatment (2.6 ).
- Must dilute TZIELD in 0.9% Sodium Chloride Injection, USP. See full prescribing information for detailed preparation and administration instructions (2.7 ).
Patient Selection
Select adult and pediatric patients 1 year of age and older with Stage 2 T1D for TZIELD treatment to delay the onset of Stage 3 T1D based on the confirmation of:
- At least two positive pancreatic islet cell autoantibodies, and
- Dysglycemia without overt hyperglycemia using an oral glucose tolerance test (if an oral glucose tolerance test is not available, an alternative method for diagnosing dysglycemia without overt hyperglycemia may be appropriate)
Ensure the patient's diagnosis confirms an autoimmune origin and does not suggest type 2 diabetes or other forms of diabetes. These may include, but are not limited to, genetic forms of diabetes, maturity-onset diabetes of the young (MODY), latent autoimmune diabetes in adults (LADA), or diabetes secondary to medications or surgery.
Laboratory and Infection Evaluation, and Vaccination Prior to Initiation
- Prior to initiating TZIELD, obtain a complete blood count and liver enzyme tests. Use of TZIELD is not recommended in patients with [see Warnings and Precautions (5.3) and Adverse Reactions (6.1) ] :
- Lymphocyte count less than 1,000 lymphocytes/mcL
- Hemoglobin less than 10 g/dL
- Platelet count less than 150,000 platelets/mcL
- Absolute neutrophil count less than 1,500 neutrophils/mcL
- Elevated alanine aminotransferase (ALT) or aspartate aminotransferase (AST) greater than 2 times the upper limit of normal (ULN) or bilirubin greater than 1.5 times ULN
- Active serious infection or chronic active infection other than localized skin infections [see Warnings and Precautions (5.3) ].
- Prior to initiating treatment, evaluate patients for active EBV and CMV infection and confirm undetectable viral load [e.g., polymerase chain reaction (PCR) testing]. Use of TZIELD is not recommended in patients with laboratory or clinical evidence of active infection with EBV or CMV [see Warnings and Precautions (5.1) ] .
- Administer all age-appropriate vaccinations prior to starting TZIELD [see Warnings and Precautions (5.6) ] :
- Administer live-attenuated (live) vaccines at least 8 weeks prior to TZIELD treatment.
- Administer inactivated (killed) vaccines or mRNA vaccines at least 2 weeks prior to treatment.
Important Premedication Instructions
Prior to each of the first 5 days of TZIELD infusion [see Warnings and Precautions (5.2) ] :
- Premedicate with a nonsteroidal anti-inflammatory drug (NSAID) or acetaminophen
- Premedicate with an antihistamine, and
- Consider premedication with an antiemetic
If needed, administer additional premedication doses.
Recommended Dosage and Administration
Calculate the dosage using a body surface area (BSA) and administer TZIELD once daily for 14 consecutive days as follows:
- Day 1: 65 mcg/m 2
- Day 2: 125 mcg/m 2
- Day 3: 250 mcg/m 2
- Day 4: 500 mcg/m 2
- Days 5 through 14: 1,030 mcg/m 2
Administer TZIELD by intravenous infusion over a minimum of:
- 30 minutes in adult and pediatric patients aged 8 years and older.
- 2 hours in pediatric patients aged 1 to less than 8 years [see Clinical Pharmacology (12.3) ] .
Recommendations Regarding Missed Doses
If a planned TZIELD infusion is missed, resume dosing by administering all remaining doses on consecutive days to complete the 14-day treatment course.
Do not administer two doses on the same day.
Recommended Monitoring During Treatment with TZIELD
- Monitor lymphocyte count regularly (every 2–3 days) during TZIELD infusion and monitor for lymphocyte recovery following completion of TZIELD.
- If prolonged severe lymphopenia (<500 cells per mcL lasting 1 week or longer) develops, permanently discontinue TZIELD.
- Monitor patients for signs and symptoms of viral reactivation during TZIELD treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD [see Warnings and Precautions (5.1) ] .
- Monitor liver enzymes and bilirubin during treatment. Discontinue TZIELD treatment in patients who develop elevated ALT or AST more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN [see Warnings and Precautions (5.2) ] .
Preparation Instructions
Using the dose calculated according to BSA [see Dosage and Administration (2.4) ], dilute TZIELD prior to preparing the infusion according to the instructions below:
TZIELD Dilution Preparation:
- Prior to dilution, inspect TZIELD visually before use (the supplied solution is clear and colorless). Do not use TZIELD if particulate matter or coloration is seen.
- Prepare TZIELD using aseptic technique.
- If the calculated dose is:
- 2,000 mcg or less, then prepare:
- One sterile glass vial with 18 mL of 0.9% Sodium Chloride Injection or
- One ≤50 mL polyvinylchloride (PVC) with di-(2-ethylhexyl)phthalate (DEHP) infusion bag with 18 mL of 0.9% Sodium Chloride Injection.
- Greater than 2,000 mcg, then prepare:
- Two sterile glass vials with 18 mL of 0.9% Sodium Chloride Injection or
- Two ≤50 mL PVC with DEHP infusion bags with 18 mL of 0.9% Sodium Chloride Injection.
- 2,000 mcg or less, then prepare:
- Remove 2 mL of TZIELD from the single-dose vial and slowly add to the glass vial or PVC with DEHP infusion bag containing 18 mL of 0.9% Sodium Chloride Injection.
- Mix gently by slowly inverting the vial or rocking the infusion bag. The resulting 20 mL diluted TZIELD solution contains 100 mcg/mL of teplizumab-mzwv.
- If preparing a dose greater than 2,000 mcg, repeat the above process with second TZIELD vial and the glass vial, or PVC with DEHP infusion bag containing 18 mL of 0.9% Sodium Chloride Injection.
TZIELD Infusion Solution Preparation:
- Using an appropriately sized syringe, withdraw the volume of diluted TZIELD solution required for that day's calculated dose from the 100 mcg/mL dilution ( for a calculated dose 2,000 mcg or less ) or from both prepared 100 mcg/mL dilutions ( for a calculated dose more than 2,000 mcg ).
- Discard unused portion of remaining diluted TZIELD solution in the glass vial or infusion bag.
- Slowly add contents of the syringe containing the TZIELD dose to PVC with DEHP infusion bag containing 25 mL of 0.9% Sodium Chloride Injection ( for a calculated dose more than 2,000 mcg, add the cumulative volume for the calculated dose to a single infusion bag ). Gently rock the infusion bag to ensure that the solution mixes sufficiently. Do not shake.
- Prime the PVC with DEHP IV infusion set with the TZIELD infusion solution. Do not waste any infusion solution during the priming process.
- After infusion, flush the IV set with a volume of 0.9% Sodium Chloride Injection greater than or equal to the priming volume, to ensure full dose is administered. Same infusion rate should be used for flushing.
- If the TZIELD infusion solution is not used immediately, store the infusion at room temperature [15°C to 30°C (59°F to 86°F)]. Discard the TZIELD infusion solution if the infusion is unable to be completed within 4 hours of preparation.
Use of in-line filter is not recommended. If necessary, use a polyethylene sulfone (PES) filter.
Do not use light protected (colored) infusion sets.
DOSAGE FORMS AND STRENGTHS
Injection: 2 mg/2 mL (1 mg/mL) clear and colorless solution in a single-dose vial.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Available case reports from clinical trials with TZIELD are insufficient to identify a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes. Although there are no data on teplizumab-mzwv in nonclinical studies, monoclonal antibodies can be actively transported across the placenta, and TZIELD may cause immunosuppression in the utero - exposed infant (see Clinical Considerations ). To minimize exposure to a fetus, avoid use of TZIELD during pregnancy and at least 30 days prior to planned pregnancy.
TZIELD is not active in rodents. In animal reproduction studies, mice were given a surrogate anti-mouse CD3 antibody subcutaneously during organogenesis through lactation. Pups born to dams administered the murine surrogate antibody during pregnancy showed a reduction in the adaptive immune response consistent with the expected pharmacology (see Data ) .
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2%–4% and 15%–20%, respectively.
Report pregnancies to Provention Bio, Inc.'s Adverse Event reporting line at 1-800-633-1610 or visit https://ae.reporting.sanofi.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Transport of endogenous IgG antibodies across the placenta increases as pregnancy progresses, and peaks during the third trimester. Because teplizumab-mzwv may interfere with immune response to infections, risks and benefits should be considered prior to administering live vaccines to infants exposed to teplizumab-mzwv in utero. There are insufficient data regarding infant serum levels of teplizumab-mzwv at birth and the duration of persistence of teplizumab-mzwv in infant serum after birth to identify a specific timeframe to delay live virus immunizations in infants exposed in utero.
Data
Animal Data
In an embryo-fetal developmental toxicity study, pregnant mice were administered a murine surrogate anti-mouse CD3 antibody by subcutaneous injection at dose levels of 0, 0.03, 0.3, or 20 mg/kg on Gestation Days 6, 10, and 14. Increase in post-implantation loss occurred in the 20 mg/kg group, in the presence of maternal toxicity.
In a pre- and postnatal development toxicity study in pregnant mice, in which the murine surrogate antibody was administered every 3 days from gestation day 6 through lactation day 19 at doses of 0, 0.3, 3, or 20 mg/kg, no maternal toxicity or increased incidence of post-implantation loss was observed. Reductions in T cell populations and increases in B cells, and a reduction in the adaptive immune response to keyhole limpet hemocyanin (KLH) were observed in the offspring on postnatal days 35 and 84 at 20 mg/kg. The surrogate antibody was present in the offspring serum at level less than 1.5% that of maternal serum at the high dose. A trend towards reduction in fertility was observed in the offspring of dams administered the murine surrogate antibody at 20 mg/kg. The human relevance of this finding is unknown.
Lactation
Risk Summary
There are no data on the presence of teplizumab-mzwv in either human or animal milk, the effects on the breastfed child, or the effects on milk production. Endogenous maternal IgG and monoclonal antibodies are transferred into human milk. The effects of local gastrointestinal exposure and limited systemic exposure in the breastfed infant to teplizumab-mzwv are unknown.
Although the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for TZIELD and any potential adverse effects on the breastfed child from TZIELD or from the underlying maternal condition, a lactating woman may interrupt breastfeeding and pump and discard breast milk during treatment and for 20 days after TZIELD administration to minimize drug exposure to a breastfed child.
Pediatric Use
The safety and effectiveness of TZIELD have been established to delay the onset of Stage 3 T1D in pediatric patients 1 year of age and older with Stage 2 T1D.
Use of TZIELD for this indication is supported by evidence from an adequate and well-controlled study (Study TN-10) in adult and pediatric patients 8 years of age and older (including 29 pediatric patients) with Stage 2 T1D and from additional pharmacokinetic and safety data in 23 pediatric patients aged 1 to less than 8 years of age with Stage 2 T1D (PETITE T1D) [see Adverse Reactions (6.1) , Clinical Pharmacology (12.3) and Clinical Studies (14) ] .
Adverse reactions observed in pediatric patients 1 year of age and older who received TZIELD were consistent with those reported in adults [see Adverse Reactions (6.1) ] .
The safety and effectiveness of TZIELD have not been established in pediatric patients younger than 1 year of age.
Geriatric Use
Stage 2 type 1 diabetes is largely a condition that occurs in pediatric and younger adult patients. Clinical studies of TZIELD to delay the onset of Stage 3 T1D did not include patients 65 years of age and older.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Cytokine Release Syndrome (CRS): Premedicate, monitor liver enzymes, discontinue in those that develop elevated ALT or AST more than 5 times the upper limit of normal, and if severe CRS develops consider temporarily pausing dosing (5.2 ).
- Serious Infections: Use of TZIELD is not recommended in patients with active serious infection or chronic infection. Monitor for signs and symptoms of infection during and after TZIELD treatment. If a serious infection develops, discontinue TZIELD (5.3 ).
- Lymphopenia: Monitor white blood cell counts during the treatment period. If prolonged severe lymphopenia (<500 cells per mcL lasting 1 week or longer) develops, discontinue TZIELD (5.4 ).
- Hypersensitivity Reactions: If severe hypersensitivity reactions occur, discontinue TZIELD and treat promptly (5.5 ).
- Vaccinations: Administer all age-appropriate vaccinations prior to starting TZIELD. See recommendations regarding live-attenuated, inactivated, and mRNA vaccines (5.6 ).
Viral Reactivation
Serious, life-threatening cases of viral reactivation, including EBV and CMV have been reported with TZIELD. During and within 2 months of TZIELD treatment, if primary infection or reactivation of EBV or CMV occurs, it may present with increased severity, including EBV-associated lymphoproliferative disease and organ failure. Patients who are immunocompromised, including patients with Down syndrome, may be at increased risk. The majority of serious viral reactivation cases occurred in patients who continued TZIELD despite persistent, severe lymphopenia [see Warnings and Precautions 5.4 ].
Prior to initiating treatment with TZIELD, evaluate patients for active EBV and CMV infection and confirm undetectable viral load (e.g., PCR testing). TZIELD is not recommended in patients with laboratory or clinical evidence of active EBV or CMV infection [see Dosage and Administration (2.2) ].
During treatment with TZIELD, regularly monitor lymphocyte counts [see Dosage and Administration (2.6) , Warnings and Precautions (5.4) ] and monitor patients for signs and symptoms of viral reactivation during treatment and for at least 2 months following the last infusion. If viral reactivation is suspected, discontinue TZIELD and obtain viral load (e.g., PCR) promptly. Consider appropriate expert consultation for diagnostic testing recommendations as some diagnostic tests may give inaccurate results in immunosuppressed patients. If viral reactivation is confirmed, permanently discontinue TZIELD [see Dosage and Administration (2.6) ]. Consider appropriate expert consultation for the management of severe viral reactivation.
Cytokine Release Syndrome
Cytokine release syndrome (CRS) has been observed in TZIELD-treated patients. In clinical trials, CRS was reported in 5% of TZIELD-treated patients compared to 0.8% of control-treated patients during the treatment period and through 28 days after the last study drug administration. CRS manifestations in TZIELD-treated patients included fever, nausea (with or without vomiting), fatigue, headache, myalgia, arthralgia, increased ALT, increased AST, and increased total bilirubin. These manifestations typically occurred during the first 5 days of TZIELD treatment [see Adverse Reactions (6.1) ] . To mitigate CRS:
- Premedicate with antipyretics, antihistamines and/or antiemetics prior to TZIELD treatment [see Dosage and Administration (2.3) ].
- Monitor liver enzymes and bilirubin during treatment. Discontinue TZIELD treatment in patients who develop elevated ALT or AST more than 5 times the upper limit of normal (ULN) or bilirubin more than 3 times ULN.
- Treat symptoms of CRS in TZIELD-treated patients with antipyretics, antihistamines and/or antiemetics. If severe CRS develops, consider:
- Temporarily pausing TZIELD dosing for 1–2 days and if symptoms have resolved or significantly improved, subsequently administering the remaining doses on consecutive days to complete the full 14-day course or
- Discontinuing TZIELD treatment .
Serious Infections
Bacterial and viral infections have occurred in TZIELD-treated patients. In clinical trials, TZIELD-treated patients had a higher rate of serious infections (3.5%) than control-treated patients (2%), including gastroenteritis, cellulitis, pneumonia, abscess, sepsis [see Adverse Reactions (6.1) ]. Use of TZIELD is not recommended in patients with active serious infection, or chronic infection other than localized skin infections. Monitor patients for signs and symptoms of infection during and after TZIELD treatment. If serious infection develops, treat appropriately, and discontinue TZIELD.
Lymphopenia
In clinical trials, 78% of TZIELD-treated patients developed lymphopenia compared to 11% of control-treated patients. For most TZIELD-treated patients who experienced lymphopenia, lymphocyte levels began to recover after the fifth day of treatment and returned to pre-treatment values within two weeks after treatment completion and without dose interruption. Severe lymphopenia (<500 cells per mcL) lasting 1 week or longer occurred in 0.9% of TZIELD-treated patients, and 0.5% of TZIELD-treated patients permanently discontinued TZIELD because of lymphopenia [see Adverse Reactions (6.1) ].
Obtain a CBC prior to starting TZIELD and monitor white blood cell counts during TZIELD treatment. If prolonged severe lymphopenia (<500 cells per mcL lasting 1 week or longer) develops, permanently discontinue TZIELD.
Hypersensitivity Reactions
Acute hypersensitivity reactions including serum sickness, angioedema, urticaria, rash, vomiting and bronchospasm occurred in TZIELD-treated patients [see Adverse Reactions (6.1) ]. If severe hypersensitivity reactions occur, discontinue use of TZIELD and treat promptly.
Vaccinations
The safety of immunization with live-attenuated vaccines in TZIELD-treated patients has not been studied. Additionally, TZIELD may interfere with the immune response to vaccination and decrease vaccine efficacy.
- Administer all age-appropriate vaccinations prior to starting TZIELD [see Dosage and Administration (2.2) ].
- Inactivated or mRNA vaccinations are not recommended within the 2 weeks prior to TZIELD treatment, during treatment, or up to 6 weeks after completion of treatment.
- Live-attenuated vaccinations are not recommended within the 8 weeks prior to starting TZIELD treatment, during treatment, or up to 52 weeks after treatment.
ADVERSE REACTIONS
The following serious adverse reactions are described elsewhere in the Prescribing Information:
- Viral Reactivation [see Warnings and Precautions (5.1) ]
- Cytokine Release Syndrome [see Warnings and Precautions (5.2) ]
- Serious Infections [see Warnings and Precautions (5.3) ]
- Lymphopenia [see Warnings and Precautions (5.4) ]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Placebo-Controlled Study in Adult and Pediatric Patients Aged 8 Years and Older with Stage 2 T1D
The data in Table 1 are derived from the placebo-controlled study (Study TN-10) in adult and pediatric patients aged 8 years and older with Stage 2 T1D [see Clinical Studies (14) ]. These data reflect exposure of 44 patients of whom 93% completed the full 14-day treatment course.
Pool of Five Controlled Clinical Studies in Stage 2 T1D and for an Unapproved Use
Adverse reactions in TZIELD-treated patients were also evaluated in a larger pool of adult and pediatric patients who participated in five controlled clinical studies (including Study TN-10 described above):
- One study in patients with Stage 2 T1D (Study TN-10) [see Clinical Studies (14) ],
- Three placebo-controlled studies in an unapproved population,
- One open-label standard-of-care controlled study of TZIELD in an unapproved population.
In this pool:
- 773 patients received TZIELD (44 patients with Stage 2 TID and 729 patients from an unapproved population), and
- 245 patients received either placebo or standard of care control (32 patients with Stage 2 T1D and 213 patients from an unapproved population).
In these studies, 436 patients received a 14-day dosing regimen of TZIELD with a total drug exposure that was comparable to the total drug exposure achieved with the recommended dosage [see Dosage and Administration (2.4) ] , 168 patients received a 14-day course of TZIELD with a lower total TZIELD drug exposure, and 169 patients received a 6-day course of TZIELD with a lower total TZIELD drug exposure. The mean age of TZIELD-treated patients was 17.6 years (median 15 years), 62% were less than18 years old (40% age 12 to 17; 21% age 8 to 11), and 64% were male. The population was 72% White, 26% Asian, 1% Black or African American, 1% were multiple or unknown race, and less than1% American Indian or Alaska Native; 5% were Hispanic or Latino ethnicity.
Common Adverse Reactions
Table 1 presents common (≥ 5%) adverse reactions that occurred during treatment and through 28 days after the last study drug administration in Study TN-10. Adverse reactions observed in pediatric patients 8 years and older who received TZIELD were consistent with those reported in adult patients in this study.
| Adverse Reaction | Placebo N=32 | TZIELD N=44 |
|---|---|---|
| Lymphopenia | 6% | 73% |
| Rash Composite of rash-related terms including rash erythematous, rash macular, rash papular, rash maculo-papular, rash pruritic | 0% | 36% |
| Leukopenia | 0% | 21% |
| Headache | 6% | 11% |
| Neutropenia | 3% | 5% |
| Increased alanine aminotransferase | 3% | 5% |
| Nausea | 3% | 5% |
| Diarrhea | 0% | 5% |
| Nasopharyngitis | 0% | 5% |
Cytokine Release Syndrome (CRS)
In Study TN-10, CRS was reported in 2% of TZIELD-treated patients compared to 0% of placebo-treated patients.
Of the 39 TZIELD-treated patients that developed CRS (5% of all TZIELD-treated patients) in the pool of 5 clinical trials, 13% of the CRS cases were serious adverse reactions [see Warnings and Precautions (5.2) ] . Liver transaminase elevations were observed in 56% of TZIELD-treated patients who experienced CRS: 64% were up to 2.5 times ULN, 32% were more than 2.5 to 5 times ULN, and 4.5% were 5–10 times ULN.
Serious Infections
In Study TN-10, serious infections (cellulitis, gastroenteritis, pneumonia, wound infection) were reported in 9% (4/44) of TZIELD-treated patients compared to 0% (0/32) of placebo-treated patients any time during or after the first dose of study treatment.
Rash and Hypersensitivity Reactions
Hypersensitivity reactions were reported with TZIELD in Study TN-10. Serum sickness was observed in 2% (1/44) of TZIELD-treated patients compared to 0% (0/32) of placebo-treated patients. The patient who developed serum sickness had a prior history of positive anti-nuclear antibody and presented with arthralgias and elevated c-reactive protein and low C4 complement five days after completing their course of TZIELD; illness resolved in 2.5 months.
In the pool of 5 clinical trials of patients:
- Anaphylaxis (with hypoxia and bronchospasm) was observed in one TZIELD-treated patient who was hospitalized.
- Angioedema (periorbital and facial) was observed in 0.3% TZIELD-treated patients, compared to 0% in control-treated patients. Peripheral and generalized edema was reported in 1.6% of TZIELD-treated patients and 0% of control-treated patients.
- Rash was observed in 48% of TZIELD-treated patients compared to 15% in control-treated patients, with 33 excess cases of rash per 100 patients. The majority of rashes observed with TZIELD treatment were not serious and resolved without intervention; although 0.3% (2/773) of TZIELD-treated patients had a serious rash compared to 0% (0/245) of placebo- treated patients.
- Urticaria was reported in 1.9% of TZIELD-treated patients and in 1.2% of control-treated patients.
Immunogenicity: Anti-Drug Antibody-Associated Adverse Reactions
In Study TN-10, rash occurred in 39% of TZIELD-treated patients who developed anti- teplizumab-mzwv antibodies and in 33% of TZIELD-treated patients who did not develop anti- teplizumab-mzwv antibodies [see Clinical Pharmacology (12.6) ].
Open Label Study in Pediatric Patients Age 1 to Less Than 8 Years with Stage 2 T1D
The safety of TZIELD was evaluated in a non-randomized, single arm, open-label, multicenter study in 23 pediatric patients age 1 to less than 8 years with Stage 2 T1D ( PETITE-T1D Study; NCT05757713)[see Clinical Pharmacology (12.3) ]. In this trial, 87% completed the full 14-day treatment course. The median age was 4.9 years (1 patient was less than 2 years old; 52% were 2 to less than 5 years old; 44% were 5 to less than 8 years old; range 1.7 to 6.8 years) and 52% were female. The majority of the population (96%) was White and one patient (4%) was Asian; 3 patients (13%) were Hispanic or Latino ethnicity. At baseline, 87% of participants had a first-degree relative with T1D. The majority (87%) were positive for 3 or more diabetes-related autoantibodies. The most common autoantibodies were anti-insulin (87%), anti-ICA (85%), anti-GAD65 (83%), anti-ZnT8 (74%), and anti-IA2 (68%). The median HbA1c was 5.5%.
Overall, the safety profile of TZIELD observed in pediatric patients less than 8 years of age with Stage 2 T1D was consistent with that observed in patients 8 years of age and older with Stage 2 T1D. The most common adverse reactions that occurred in patients less than 8 years of age were vomiting (52%) and diarrhea (30%).
Other Adverse Reactions in Adult and Pediatric Patients Age 1 Year and Older with Stage 2 T1D
Lymphopenia
In Study TN-10, lymphopenia was reported in 73% of TZIELD-treated patients compared to 6% of placebo-treated patients. The average lymphocyte count nadir occurred at Day 5 of treatment, with recovery and return to baseline by Week 6 [see Warnings and Precautions (5.4) ].
Neutropenia
In Study TN-10, neutropenia was observed in 7% of TZIELD-treated patients compared to 3% of placebo-treated patients.
Anemia and Thrombocytopenia
In the pool of 5 clinical trials of patients, anemia was reported in 27% of TZIELD-treated patients compared to 21% of placebo-treated patients, and thrombocytopenia was reported in 13% of TZIELD-treated patients compared to 5% of placebo-treated patients during the 14-day treatment course; recovery occurred within 2 to 4 weeks of treatment. In these clinical trials, 1.8% of TZIELD-treated patients discontinued treatment due to hemoglobin less than 8.5 g/dL (or a decrease of more than 2 g/dL to a value less than 10 g/dL), and 1% discontinued TZIELD due to platelet count less than 50,000 platelets/mcL.
Liver Enzyme Elevations
Liver enzyme elevations were observed in TZIELD-treated patients, both in the context of CRS and in patients without CRS. In the pool of 5 clinical trials, elevated aminotransferases were reported in 25% of TZIELD-treated patients compared to 11% of placebo-treated patients during the 14-day treatment course. On laboratory analysis, 5.1% of TZIELD-treated patients experienced a peak ALT more than 3 times the ULN compared to 0.8% of control-treated patients. Most liver enzyme elevations were transient and resolved 1–2 weeks after treatment; 98% resolved by follow-up week 14.
Procedure-Related Venous Thrombosis
Venous thrombus and thrombophlebitis have been reported in patients receiving teplizumab intravenously administered via peripherally inserted central catheter (PICC). In the pool of 5 clinical trials of patients, deep vein thrombus was reported in 0.4% of TZIELD-treated patients compared to 0 placebo-treated patients. One teplizumab-treated patient (4.3%) in the PETITE-T1D study experienced a deep vein thrombosis.
Other Laboratory Abnormalities
In the pool of 5 clinical trials, other laboratory abnormalities including decreased bicarbonate (15% in TZIELD-treated patients, compared to 7% in placebo-treated patients) and decreased blood calcium (19% in TZIELD-treated patients, compared to 13% in placebo-treated patients) were noted.
DESCRIPTION
Teplizumab-mzwv is a CD3-directed monoclonal antibody (humanized IgG1 kappa) that has a molecular weight of approximately 150 kilodalton (kDa) and is expressed from a recombinant Chinese hamster ovary (CHO) cell line.
TZIELD (teplizumab-mzwv) injection is supplied as a sterile, preservative-free, clear and colorless solution in a 2 mg/2 mL (1 mg/mL) single-dose vial for intravenous use. Each mL contains 1 mg of teplizumab-mzwv, dibasic sodium phosphate (0.26 mg), monobasic sodium phosphate (0.98 mg), polysorbate 80 (0.05 mg), sodium chloride (8.78 mg), and water for injection. The pH is 6.1.
CLINICAL PHARMACOLOGY
Mechanism of Action
Teplizumab-mzwv binds to CD3 (a cell surface antigen present on T lymphocytes) and delays the onset of Stage 3 T1D in adult and pediatric patients aged 1 year and older with Stage 2 T1D. The mechanism may involve partial agonistic signaling and deactivation of pancreatic beta cell autoreactive T lymphocytes. Teplizumab-mzwv leads to an increase in the proportion of regulatory T cells and of exhausted CD8+ T cells in peripheral.
Pharmacodynamics
Clinical studies have shown that teplizumab-mzwv binds to CD3 molecules on the surface of both CD4+ and CD8+ T cells during TZIELD treatment, with internalization of the teplizumab-mzwv/CD3 complex from the surface of T cells. Pharmacodynamic effects include lymphopenia in the absence of depletion of T cells with a nadir approximately on the 5 th day of dosing, during a 14-day course of TZIELD treatment [see Warnings and Precautions (5.4) ]. Teplizumab-mzwv exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of teplizumab-mzwv have not been fully characterized.
Pharmacokinetics
Distribution
The central volume of distribution (Vd) of teplizumab-mzwv was 2.27 L in a 60 kg subject.
Elimination
Teplizumab-mzwv showed saturable binding and elimination. The clearance of teplizumab-mzwv is 2.66 L/day in a 60 kg subject.
Metabolism
Teplizumab-mzwv is expected to be metabolized into small peptides by catabolic pathways.
Specific Populations
No clinically significant differences in the pharmacokinetics of teplizumab-mzwv were observed based on age (1 to 35 years old), sex, or racial groups (White, Asians).
Pediatric Patients 1 to Less Than 8 Years Old
No clinically significant difference in the AUC of teplizumab-mzwv was observed in pediatric patients aged 1 to less than 8 years compared to that in adult and pediatric patients aged 8 years and older. By extending the infusion duration from 30 minutes to 2 hours in pediatric patients aged 1 to less than 8 years, the C max of teplizumab-mzwv was comparable to that in adult and pediatric patients aged 8 years and older.
Body weight
BSA-based dosing normalizes the exposure to teplizumab-mzwv across body weight.
Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies in other studies, including those of teplizumab-mzwv or of other teplizumab products.
In the placebo-controlled study in patients aged 8 years of age and older with Stage 2 T1D (Study TN-10) [see Clinical Studies (14) ] , approximately 57% (24/42) of TZIELD-treated patients developed anti-teplizumab-mzwv antibodies, 46% (11/24) of whom developed neutralizing antibodies. There was a higher incidence of rash in TZIELD-treated patients who developed anti-teplizumab-mzwv antibodies (39%) compared to those who did not develop anti-teplizumab-mzwv antibodies (33%) [see Adverse Reactions (6.1) ] .
Results from the analysis up to Week 52 from the PETITE-T1D study in patients 1 to less than 8 years of age with Stage 2 T1D , approximately 87% (20/23) of TZIELD-treated patients developed anti-teplizumab-mzwv antibodies, 70% (14/20) of whom developed neutralizing antibodies [see Adverse Reactions (6.1) ] . There was a higher incidence of skin and subcutaneous tissue disorders (most of which were mild or moderate) in TZIELD-treated patients who developed anti-teplizumab-mzwv antibodies (70%) compared to those who did not develop anti-teplizumab-mzwv antibodies (33%) [see Adverse Reactions (6.1) ] .
There is insufficient information to characterize the effects of ADA on pharmacokinetics, pharmacodynamics, or effectiveness of TZIELD in patients aged 1 year and older with Stage 2 T1D.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
No long-term studies have been performed to assess the carcinogenic potential of teplizumab-mzwv.
No studies have been performed to assess the mutagenic potential of teplizumab-mzwv. As an antibody, teplizumab-mzwv is not expected to interact directly with DNA.
Fertility and reproductive performance were unaffected in female and male mice that received a murine surrogate anti-mouse CD3 antibody administered by the subcutaneous route at doses up to 20 mg/kg.
CLINICAL STUDIES
The effectiveness of TZIELD was investigated in the following study:
Study TN-10; NCT01030861
A randomized, double-blind, event-driven, placebo-controlled study in 76 patients, 8 to 49 years of age with Stage 2 type 1 diabetes. Stage 2 T1D was defined as having both of the following:
- Two or more of the following pancreatic islet autoantibodies:
- Glutamic acid decarboxylase 65 (GAD) autoantibodies
- Insulin autoantibody (IAA)
- Insulinoma-associated antigen 2 autoantibody (IA-2A)
- Zinc transporter 8 autoantibody (ZnT8A)
- Islet cell autoantibody (ICA)
- Dysglycemia on oral glucose tolerance testing
In this study, patients were randomized to receive TZIELD or placebo once daily by intravenous infusion for 14 days. Patients in the TZIELD group had a total drug exposure that was comparable to the total drug exposure achieved with the recommended total TZIELD dosage [see Dosage and Administration (2.4) ]. The primary efficacy endpoint in this study was the time from randomization to development of Stage 3 type 1 diabetes diagnosis.
Baseline Patient Characteristics
In this study, 45% were female; 97% White, 1% Asian, and 1% reported multiracial background; 3% were Hispanic or Latino ethnicity; and 95% were from the United States. The median age was 14 years. The age range was 8.5 to 49.5 years old (72% were <18 years old; 18% were ≥30 years old) (Table 2).
| TZIELD N=44 | Placebo N=32 | |
|---|---|---|
| Age Group | ||
| ≥ 18 Years | 34% | 19% |
| < 18 years | 66% | 81% |
| Pediatric Age Group Quartiles | ||
| 8 to <11 years | 21% | 25% |
| 11 to <14 years | 27% | 31% |
| 14 to <18 years | 18% | 25% |
Baseline Disease Characteristics
Table 3 displays the baseline disease characteristics in Study TN-10.
| TZIELD N=44 | Placebo N=32 | |
|---|---|---|
| Abbreviations: HbA1c=hemoglobin A1c, SD=standard deviation, HLA = human leukocyte antigen, GAD65=Glutamic acid decarboxylase 65 (GAD) autoantibodies, IAA=Insulin autoantibody, IA-2A=Insulinoma-associated antigen 2 autoantibody, ZnT8A=Zinc transporter 8 autoantibody, ICA=Islet cell autoantibody | ||
| Glucose, mg/dL The glucose data are area under the time-concentration curve (AUC) values from the oral glucose tolerance test | ||
| median (min, max) | 165 (115, 207) | 154 (103, 200) |
| HbA1c, % | ||
| median (min, max) | 5.2 (4.6, 6.1) | 5.3 (4.3, 5.6) |
| HLA-DR4 | ||
| Missing | 5% | 0 |
| Absent | 34% | 34% |
| Present | 61% | 66% |
| HLA-DR3 | ||
| Missing | 5% | 0 |
| Absent | 48% | 53% |
| Present | 48% | 47% |
| HLA-DR3/DR4 | ||
| Both DR3 and DR4 | 25% | 22% |
| DR3 only | 23% | 25% |
| DR4 only | 36% | 44% |
| Missing | 5% | 0 |
| Neither DR3 nor DR4 | 11% | 9% |
| Autoantibodies Positive (N) | ||
| 1 | 2% | 0 |
| 2 | 27% | 22% |
| 3 | 25% | 16% |
| 4 | 27% | 44% |
| 5 | 18% | 19% |
| Autoantibody Type Positive | ||
| GAD65 | 91% | 88% |
| IAA | 43% | 34% |
| IA-2A | 59% | 75% |
| ICA | 66% | 88% |
| ZnT8 | 73% | 75% |
Efficacy Results
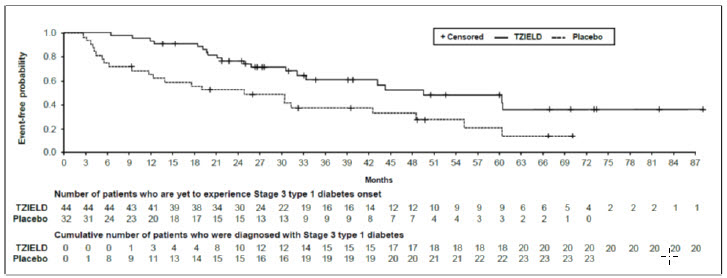
In Study TN-10, Stage 3 type 1 diabetes was diagnosed in 20 (45%) of the TZIELD-treated patients and in 23 (72%) of the placebo-treated patients. A Cox proportional hazards model, stratified by age and oral glucose tolerance test status at randomization, demonstrated that the median time from randomization to Stage 3 type 1 diabetes diagnosis was 50 months in the TZIELD group and 25 months in the placebo group, for a difference of 25 months. With a median follow-up time of 51 months, therapy with TZIELD resulted in a statistically significant delay in the development of Stage 3 type 1 diabetes, hazard ratio 0.41 (95% CI: 0.22 to 0.78; p=0.0066) (Figure 1).
Study TN-10 was not designed to assess whether there were differences in the effectiveness between subgroups based on demographic characteristics or baseline disease characteristics.
| Figure 1: Kaplan-Meier Curve of Time to Diagnosis of Stage 3 Type 1 Diabetes in Adult and Pediatric Patients Aged 8 Years and Older with Stage 2 Type 1 Diabetes by Treatment Group (Study TN-10) ITT population |
|
HOW SUPPLIED/STORAGE AND HANDLING
TZIELD (teplizumab-mzwv) injection is a clear and colorless solution (2 mg/2 mL (1 mg/mL)) supplied in a single-dose vial as follows:
| Carton Contents | NDC |
|---|---|
| 1 single dose vial | NDC 73650-316-01 |
| 10 single dose vials | NDC 73650-316-10 |
| 14 single dose vials | NDC 73650-316-14 |
Refrigerate TZIELD vials at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. Store upright. Do not freeze or shake the vials.
If not used immediately, store the infusion at room temperature [15°C to 30°C (59°F to 86°F)] and complete infusion within 4 hours of the start of preparation. Discard the diluted solution if not administered within 4 hours of preparation [see Dosage and Administration (2.7) ] .
Mechanism of Action
Teplizumab-mzwv binds to CD3 (a cell surface antigen present on T lymphocytes) and delays the onset of Stage 3 T1D in adult and pediatric patients aged 1 year and older with Stage 2 T1D. The mechanism may involve partial agonistic signaling and deactivation of pancreatic beta cell autoreactive T lymphocytes. Teplizumab-mzwv leads to an increase in the proportion of regulatory T cells and of exhausted CD8+ T cells in peripheral.