Get your patient on Voranigo (Vorasidenib)
Voranigo prior authorization resources
Most recent state uniform prior authorization forms
Brand Resources
Voranigo patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
Recommended Evaluation Before Initiating VORANIGO
Before initiating VORANIGO, evaluate blood chemistry and liver laboratory tests [see Warnings and Precautions (5.1) and Adverse Reactions (6.1) ].
Patient Selection
Select patients with Grade 2 astrocytoma or oligodendroglioma for treatment with VORANIGO based on the presence of IDH1 or IDH2 mutations in tumor specimens [see Clinical Studies (14) ] .
Information on FDA-approved tests for detection of IDH1 or IDH2 mutations in Grade 2 astrocytoma or oligodendroglioma for selecting patients for treatment with VORANIGO is available at: https://www.fda.gov/CompanionDiagnostics.
Recommended Dosage and Administration
Recommended Dosage
Adult Patients
The recommended dosage of VORANIGO in adult patients is 40 mg orally once daily until disease progression or unacceptable toxicity.
Pediatric Patients 12 Years and Older
The recommended dosage of VORANIGO in pediatric patients 12 years and older is based on body weight:
- Patients weighing ≥40 kg: 40 mg orally once daily
- Patients weighing <40 kg: 20 mg orally once daily
Continue treatment with VORANIGO until disease progression or unacceptable toxicity.
Administration
Swallow VORANIGO tablets whole with water with or without food [see Clinical Pharmacology (12.3) ] . Do not split, crush or chew tablets.
Missed Dose
Take VORANIGO tablets at about the same time each day. If a dose is missed, take the missed dose as soon as possible within 6 hours. If a dose is missed by more than 6 hours, skip the missed dose and take the next dose at the scheduled time.
Vomiting
If vomiting occurs after taking a dose, do not take a replacement dose, and take the next dose at the scheduled time on the following day.
Dosage Modifications, Management and Monitoring for Adverse Reactions
The recommended VORANIGO dosage reductions for adverse reactions are provided in Table 1.
| Dosage Reduction | Recommended Dose and Schedule |
|---|---|
| Adult patients and Pediatric patients 12 years and older weighing at least 40 kg | |
| First | 20 mg once daily |
| Second | 10 mg once daily |
| Pediatric patients 12 years and older weighing less than 40 kg | |
| First | 10 mg once daily |
| Permanently discontinue VORANIGO in patients unable to tolerate 10 mg once daily. | |
The recommended management for adverse reactions and VORANIGO dosage modifications for adverse reactions are provided in Table 2.
| Adverse Reaction | Severity Adverse reactions graded by the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE) version 5.0. | Management and Dosage Modifications |
|---|---|---|
| Abbreviations: ALT = Alanine aminotransferase; AST = Aspartate aminotransferase; ULN = Upper limit of normal | ||
| Hepatotoxicity (Elevation of ALT or AST) [see Warnings and Precautions (5.1) ] | Grade 1 ALT or AST increase >ULN to 3 × ULN without concurrent total bilirubin >2 × ULN | Continue VORANIGO at current dose. Monitor liver laboratory tests weekly until recovery to |
| Grade 2 ALT or AST >3 to 5 × ULN without concurrent total bilirubin >2 × ULN | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
| |
| Grade 3 ALT or AST >5 to 20 × ULN without concurrent total bilirubin >2 × ULN | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
| |
| Grade 2 or 3 Any ALT or AST >3 to 20 × ULN with concurrent total bilirubin >2 × ULN | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
| |
| Grade 4 Any ALT or AST >20 × ULN | Permanently discontinue VORANIGO. | |
| Other Adverse Reactions [see Adverse Reactions (6.1) ] | Grade 3 | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
|
| Grade 4 | Permanently discontinue VORANIGO. | |

Voranigo prescribing information
INDICATIONS AND USAGE
VORANIGO is indicated for the treatment of adult and pediatric patients 12 years and older with Grade 2 astrocytoma or oligodendroglioma with a susceptible isocitrate dehydrogenase-1 (IDH1) or isocitrate dehydrogenase-2 (IDH2) mutation, as detected by an FDA-approved test, following surgery including biopsy, sub-total resection, or gross total resection [see Dosage and Administration (2.1) , Clinical Pharmacology (12.1) and Clinical Studies (14) ] .
DOSAGE AND ADMINISTRATION
Recommended Evaluation Before Initiating VORANIGO
Before initiating VORANIGO, evaluate blood chemistry and liver laboratory tests [see Warnings and Precautions (5.1) and Adverse Reactions (6.1) ].
Patient Selection
Select patients with Grade 2 astrocytoma or oligodendroglioma for treatment with VORANIGO based on the presence of IDH1 or IDH2 mutations in tumor specimens [see Clinical Studies (14) ] .
Information on FDA-approved tests for detection of IDH1 or IDH2 mutations in Grade 2 astrocytoma or oligodendroglioma for selecting patients for treatment with VORANIGO is available at: https://www.fda.gov/CompanionDiagnostics.
Recommended Dosage and Administration
Recommended Dosage
Adult Patients
The recommended dosage of VORANIGO in adult patients is 40 mg orally once daily until disease progression or unacceptable toxicity.
Pediatric Patients 12 Years and Older
The recommended dosage of VORANIGO in pediatric patients 12 years and older is based on body weight:
- Patients weighing ≥40 kg: 40 mg orally once daily
- Patients weighing <40 kg: 20 mg orally once daily
Continue treatment with VORANIGO until disease progression or unacceptable toxicity.
Administration
Swallow VORANIGO tablets whole with water with or without food [see Clinical Pharmacology (12.3) ] . Do not split, crush or chew tablets.
Missed Dose
Take VORANIGO tablets at about the same time each day. If a dose is missed, take the missed dose as soon as possible within 6 hours. If a dose is missed by more than 6 hours, skip the missed dose and take the next dose at the scheduled time.
Vomiting
If vomiting occurs after taking a dose, do not take a replacement dose, and take the next dose at the scheduled time on the following day.
Dosage Modifications, Management and Monitoring for Adverse Reactions
The recommended VORANIGO dosage reductions for adverse reactions are provided in Table 1.
| Dosage Reduction | Recommended Dose and Schedule |
|---|---|
| Adult patients and Pediatric patients 12 years and older weighing at least 40 kg | |
| First | 20 mg once daily |
| Second | 10 mg once daily |
| Pediatric patients 12 years and older weighing less than 40 kg | |
| First | 10 mg once daily |
| Permanently discontinue VORANIGO in patients unable to tolerate 10 mg once daily. | |
The recommended management for adverse reactions and VORANIGO dosage modifications for adverse reactions are provided in Table 2.
| Adverse Reaction | Severity Adverse reactions graded by the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI-CTCAE) version 5.0. | Management and Dosage Modifications |
|---|---|---|
| Abbreviations: ALT = Alanine aminotransferase; AST = Aspartate aminotransferase; ULN = Upper limit of normal | ||
| Hepatotoxicity (Elevation of ALT or AST) [see Warnings and Precautions (5.1) ] | Grade 1 ALT or AST increase >ULN to 3 × ULN without concurrent total bilirubin >2 × ULN | Continue VORANIGO at current dose. Monitor liver laboratory tests weekly until recovery to |
| Grade 2 ALT or AST >3 to 5 × ULN without concurrent total bilirubin >2 × ULN | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
| |
| Grade 3 ALT or AST >5 to 20 × ULN without concurrent total bilirubin >2 × ULN | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
| |
| Grade 2 or 3 Any ALT or AST >3 to 20 × ULN with concurrent total bilirubin >2 × ULN | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
| |
| Grade 4 Any ALT or AST >20 × ULN | Permanently discontinue VORANIGO. | |
| Other Adverse Reactions [see Adverse Reactions (6.1) ] | Grade 3 | First Occurrence: Withhold VORANIGO until recovery to ≤Grade 1 or baseline.
|
| Grade 4 | Permanently discontinue VORANIGO. | |
DOSAGE FORMS AND STRENGTHS
Tablets:
- 10 mg: White to off-white, round film-coated tablet imprinted with "10" in black ink on one side and plain on the other side. Each tablet contains 10 mg of vorasidenib.
- 40 mg: White to off-white, oblong film-coated tablet imprinted with "40" in black ink on one side and plain on the other side. Each tablet contains 40 mg of vorasidenib.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Based on findings from animal studies and its mechanism of action [see Clinical Pharmacology (12.1) ] , VORANIGO can cause fetal harm when administered to a pregnant woman. There are no available data on VORANIGO use in pregnant women to inform a drug-associated risk. In animal embryo-fetal development studies, oral administration of vorasidenib to pregnant rats and rabbits during the period of organogenesis caused embryo-fetal toxicity at ≥8 times the human exposure based on the AUC at the highest recommended dose (see Data ) . Advise pregnant women of the potential risk to the fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In an embryo-fetal development study, vorasidenib was administered to pregnant rats via oral gavage at dose levels of 10, 25, and 75 mg/kg/day during the period of organogenesis (gestation days 6 to 17). Embryo-fetal toxicity (higher incidence of early resorptions, and visceral malformations of kidney and testes) occurred in rats at the maternally toxic dose of 75 mg/kg/day (approximately 170 times the human exposure based on the AUC at the highest recommended dose). Malformation of heart occurred in a rat at a dose of 25 mg/kg (approximately 97 times the human exposure based on the AUC at the highest recommended dose). Dose-related delayed ossification of bones and short ribs associated with decreased fetal body weights was observed at 10 and 25 mg/kg/day in the absence of maternal toxicity and at 75 mg/kg/day. The dose of 10 mg/kg/day is ≥45 times the human exposure based on the AUC at the highest recommended dose.
In an embryo-fetal development study, oral administration of vorasidenib to pregnant rabbits at dose levels of 2, 6, and 18 mg/kg/day during the period of organogenesis (gestation days 6 to 19) resulted in maternal toxicity at all doses (≥1.5 times the human exposure based on the AUC at the highest recommended dose) and caused higher incidence of late resorptions at 18 mg/kg/day as well as decreased fetal weights and delayed ossification at doses ≥6 mg/kg/day (≥8 times the human exposure based on the AUC at the highest recommended dose).
Lactation
Risk Summary
There are no data on the presence of vorasidenib or its metabolites in human milk, their effects on the breastfed child, or on milk production. Because of the potential for adverse reactions in breastfed children from VORANIGO, advise women not to breastfeed during treatment with VORANIGO and for 2 months after the last dose.
Females and Males of Reproductive Potential
Based on animal embryo-fetal toxicity studies, VORANIGO can cause fetal harm when administered to pregnant women [see Use in Specific Populations (8.1) ] .
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to starting VORANIGO [see Use in Specific Populations (8.1) ] .
Contraception
Females
Advise females of reproductive potential to use effective nonhormonal contraception during treatment with VORANIGO and for 3 months after the last dose. VORANIGO can render some hormonal contraceptives ineffective [see Drug Interactions (7.2) ] .
Males
Advise male patients with female partners of reproductive potential to use effective contraception during treatment with VORANIGO and for 3 months after the last dose.
Infertility
Based on findings in animals, VORANIGO may impair fertility in females and males of reproductive potential. The effects on female and male fertility were not reversible in rats [see Nonclinical Toxicology (13.1) ] .
Pediatric Use
The safety and effectiveness of VORANIGO have been established in pediatric patients aged 12 years and older for the treatment of Grade 2 IDH1- or IDH2-mutant astrocytoma or oligodendroglioma. Use of VORANIGO for this indication in this age group is supported by evidence from an adequate and well-controlled study of VORANIGO in adult and pediatric patients with additional population pharmacokinetic data demonstrating that age had no clinically meaningful effect on the pharmacokinetics of vorasidenib. In addition, the course of IDH1- or IDH2-mutant astrocytoma or oligodendroglioma is sufficiently similar between adults and pediatric patients to allow extrapolation of pharmacokinetic data in adults to pediatric patients [see Adverse Reactions (6.1) , Clinical Pharmacology (12.3) and Clinical Studies (14) ] .
The exposure of vorasidenib in pediatric patients 12 years and older is predicted to be within range of exposure observed in adults at the recommended dosages [see Clinical Pharmacology (12.3) ] .
The safety and effectiveness of VORANIGO have not been established in pediatric patients younger than 12 years of age for any indication.
Geriatric Use
Of the 167 patients who were randomized and received VORANIGO 40 mg once daily in the INDIGO trial, 1.2% (2 patients) were 65 years or older. Clinical studies of VORANIGO did not include sufficient numbers of patients aged ≥65 to determine whether they respond differently from younger subjects.
Renal Impairment
No dosage adjustment is recommended for patients with creatinine clearance (CL cr ) >40 mL/min.
The pharmacokinetics and safety of vorasidenib in patients with CL cr ≤40 mL/min or renal impairment requiring dialysis have not been studied [see Clinical Pharmacology (12.3) ] . For patients with CL cr ≤40 mL/min or who require dialysis, monitor for increased adverse reactions and modify the dosage for adverse reactions as recommended [see Dosage and Administration (2.4) , Clinical Pharmacology (12.3) ] .
Hepatic Impairment
No dosage adjustment is recommended for patients with mild or moderate (Child-Pugh Class A or B) hepatic impairment [see Clinical Pharmacology (12.3) ] .
The pharmacokinetics and safety of vorasidenib in patients with severe hepatic impairment (Child-Pugh Class C) have not been studied. For patients with severe hepatic impairment, monitor for increased adverse reactions and modify the dosage for adverse reactions as recommended [see Dosage and Administration (2.4) , Clinical Pharmacology (12.3) ] .
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Hepatotoxicity : Monitor liver function tests every 2 weeks during the first 2 months of treatment, then monthly for the first 2 years of treatment, and as clinically indicated. Withhold, reduce the dose or discontinue VORANIGO based on severity. (2.3 , 5.1 )
- Embryo-Fetal Toxicity : VORANIGO can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective nonhormonal contraception. (5.2 , 8.1 , 8.3 )
Hepatotoxicity
VORANIGO can cause hepatic transaminase elevations, which can lead to hepatic failure, hepatic necrosis, and autoimmune hepatitis.
In the pooled safety population [see Adverse Reactions (6.1) ] , 58% of patients treated with VORANIGO experienced increased ALT and 44% of patients experienced increased AST. Grade 3 or 4 increased ALT or AST occurred in 9% and 4.8% of patients respectively. Among these patients, 4.1% (10/244) had concurrent Grade 3 to 4 ALT or AST elevations. A total of 34% of patients treated with VORANIGO had increased gamma-glutamyl transferase (GGT), of these 2.2% were Grade 3 or 4. Bilirubin increases occurred in 4.8% of patients treated with VORANIGO, with 0.4% Grade 3 or 4. Nine percent of patients treated with VORANIGO had increased alkaline phosphatase, with 0.9% Grade 3 or 4.
Two patients met the laboratory criteria for Hy's Law and had concurrent elevations in ALT or AST >3 times the upper limit of normal and total bilirubin >2 times the upper limit of normal; these events were associated with cases of autoimmune hepatitis and hepatic failure. The median time to first onset of increased ALT or AST was 57 days (range: 1 to 1049).
Permanent discontinuation of VORANIGO was required for 2.9% of patients with ALT elevations, 1.6% of AST elevations, and 0.4% of GGT elevations. Dosage reductions of VORANIGO were required for 7% of patients with ALT elevations, 1.2% of AST elevations, and 0.4% of GGT elevations. Dosage interruptions were required in 14% of patients with ALT elevations, 6% of AST elevations, and 1.6% of GGT elevations.
Monitor liver laboratory tests (AST, ALT, GGT, total bilirubin and alkaline phosphatase) prior to the start of VORANIGO, every 2 weeks during the first 2 months of treatment, then monthly for the first 2 years of treatment, and as clinically indicated, with more frequent testing in patients who develop transaminase elevations.
Reduce the dose, withhold, or permanently discontinue VORANIGO based on severity [see Dosage and Administration (2.3) and Adverse Reactions (6.1) ].
Embryo-Fetal Toxicity
Based on findings from animal studies, VORANIGO can cause fetal harm when administered to a pregnant woman. In animal embryo-fetal development studies, oral administration of vorasidenib to pregnant rats during the period of organogenesis caused embryo-fetal toxicities at doses ≥45 times the human exposure based on the area under the concentration-time curve (AUC) at the highest recommended dose. Oral administration of vorasidenib to pregnant rabbits during the period of organogenesis resulted in embryo-fetal toxicity at doses ≥8 times the human exposure based on the AUC at the highest recommended dose.
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective nonhormonal contraception during treatment with VORANIGO and for 3 months after the last dose, since VORANIGO can render some hormonal contraceptives ineffective [see Drug Interactions (7.2) ] . Advise male patients with female partners of reproductive potential to use effective contraception during treatment with VORANIGO and for 3 months after the last dose [see Use in Specific Populations (8.1 , 8.3) ] .
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hepatotoxicity [see Warnings and Precautions (5.1) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse reactions described in the WARNINGS AND PRECAUTIONS reflect exposure to VORANIGO 40 mg orally once daily until disease progression or unacceptable toxicity in the 244 patients with astrocytoma or oligodendroglioma with susceptible IDH1 or IDH2 mutation in trials AG881-C-002 (NCT02481154, n=11), AG120-881-001 (NCT03343197, n=14) and INDIGO (NCT04164901, n=167 randomized patients and n=52 crossover patients). Among the 244 patients who received VORANIGO, 78% were exposed for 6 months or longer and 44% were exposed for greater than one year. In this pooled safety population, the most common (≥15%) adverse reactions were fatigue (33%), headache (28%), COVID-19 (28%), musculoskeletal pain (24%), diarrhea (21%), nausea (20%), and seizure (16%). In this pooled safety population, the most common (≥2%) Grade 3 or 4 laboratory abnormalities were increased ALT (9%), increased AST (4.8%), increased GGT (2.2%), and decreased neutrophils (2.2%).
INDIGO
The safety of VORANIGO was evaluated in 330 patients with Grade 2 astrocytoma or oligodendroglioma with an IDH1 or IDH2 mutation who received at least one dose of either VORANIGO 40 mg daily (N=167) or placebo (N=163) in the INDIGO trial [see Clinical Studies (14) ] . Patients received VORANIGO 40 mg orally once daily or placebo orally once daily until disease progression or unacceptable toxicity. Among the 167 patients who were randomized and received VORANIGO, the median duration of exposure to VORANIGO was 12.7 months (range: 1 to 30 months) with 153 patients (92%) exposed to VORANIGO for at least 6 months and 89 (53%) exposed for at least 1 year.
The demographics of patients randomized to VORANIGO were: median age 41 years (range: 21 to 71 years); 60% male, 74% White, 20% race not reported, 3% Asian, and 1.2% Black or African American; and 5% were Hispanic or Latino.
Serious adverse reactions occurred in 7% of patients who received VORANIGO. The most common serious adverse reactions occurring in ≥2% of patients who received VORANIGO includes seizure (3%).
Permanent discontinuation of VORANIGO due to an adverse reaction occurred in 3.6% of patients. Adverse reactions which resulted in permanent discontinuation of VORANIGO in ≥2% of patients included ALT increased (3%).
Dosage interruptions of VORANIGO due to an adverse reaction occurred in 30% of patients. Adverse reactions which required dose interruption in ≥5% of patients included ALT increased (14%), COVID-19 (9%), and AST increased (6%).
Dose reductions of VORANIGO due to an adverse reaction occurred in 11% of patients. Adverse reactions which required dose reduction in ≥5% of patients included ALT increased (8%).
The most common (≥15%) adverse reactions were fatigue (37%), COVID-19 (33%), musculoskeletal pain (26%), diarrhea (25%), and seizure (16%).
Grade 3 or 4 (≥2%) laboratory abnormalities were ALT increased (10%), AST increased (4.8%), GGT increased (3%) and neutrophil decreased (2.4%).
Adverse reactions and select laboratory abnormalities reported in the INDIGO trial are shown in Tables 3 and 4.
| VORANIGO 40 mg daily (n=167) | Placebo (n=163) | |||
|---|---|---|---|---|
| Adverse Reaction Adverse reactions are based on NCI CTCAE v5.0. | All Grades (%) | Grades 3 or 4 (%) | All Grades (%) | Grades 3 or 4 (%) |
| General Disorders | ||||
| Fatigue Grouped term includes asthenia. | 37 | 0.6 | 36 | 1.2 |
| Infections and Infestations | ||||
| COVID-19 | 33 | 0 | 29 | 0 |
| Nervous System Disorders | ||||
| Seizure Grouped term includes partial seizures, generalized tonic-clonic seizure, epilepsy, clonic convulsion, and simple partial seizures. | 16 | 4.2 | 15 | 3.7 |
| Musculoskeletal and Connective Tissue Disorders | ||||
| Musculoskeletal pain Grouped term includes arthralgia, back pain, non-cardiac chest pain, pain in extremity, myalgia, neck pain, musculoskeletal chest pain, arthritis, and musculoskeletal stiffness. | 26 | 0 | 25 | 1.8 |
| Gastrointestinal Disorders | ||||
| Diarrhea Grouped term includes feces soft and frequent bowel movements. | 25 | 0.6 | 17 | 0.6 |
| Constipation | 13 | 0 | 12 | 0 |
| Abdominal pain Grouped term includes abdominal pain upper, abdominal discomfort, abdominal pain lower, abdominal tenderness, and epigastric discomfort. | 13 | 0 | 12 | 0 |
| Decreased appetite | 9 | 0 | 3.7 | 0 |
| VORANIGO 40 mg daily N=167 | Placebo N=163 | |||
|---|---|---|---|---|
| Parameter | All Grades Based on NCI CTCAE v5.0. (% The denominator used to calculate percentages is N, the number of subjects in the Safety Analysis Set within each treatment group. ) | Grades3 or 4 (%) | All Grades(%) | Grades3 or 4 (%) |
| Abbreviations: AST = Aspartate Aminotransferase; ALT = Alanine Aminotransferase; GGT = Gamma-Glutamyl Transferase; ALP = Alkaline Phosphatase | ||||
| Chemistry | ||||
| Increased ALT | 59 | 10 | 25 | 0 |
| Increased AST | 46 | 4.8 | 20 | 0 |
| Increased Creatinine | 11 | 0.6 | 7 | 0 |
| Decreased Calcium | 10 | 0 | 7 | 0 |
| Increased Glucose Includes adverse reaction term hyperglycemia. | 10 | 0 | 4.3 | 0 |
| Increased GGT | 38 | 3 | 10 | 1.8 |
| Decreased Phosphate Includes adverse reaction terms hypophosphatemia and blood phosphorus decreased. | 8 | 0.6 | 4.9 | 0 |
| Increased Potassium | 23 | 0.6 | 20 | 0 |
| Increased ALP | 10 | 1.2 | 7 | 0.6 |
| Hematology | ||||
| Increased Hemoglobin | 13 | 0 | 3.1 | 0 |
| Decreased Lymphocytes | 11 | 1.8 | 8 | 0.6 |
| Decreased Leukocytes | 13 | 0.6 | 12 | 0.6 |
| Decreased Neutrophils | 14 | 2.4 | 12 | 1.8 |
| Decreased Platelets | 12 | 0 | 4.3 | 0 |
DRUG INTERACTIONS
- CYP1A2 Inhibitors : Avoid concomitant use of strong and moderate CYP1A2 inhibitors. (7.1 )
- CYP1A2 Inducers : Avoid concomitant use of moderate CYP1A2 inducers and smoking tobacco. (7.1 )
- Certain CYP3A Substrates : Avoid concomitant use with CYP3A substrates, where a minimal concentration change can reduce efficacy. (7.2 )
- Hormonal Contraception : If concomitant use cannot be avoided, use with nonhormonal contraception methods. (7.2 )
Effect of Other Drugs on VORANIGO
| Strong and Moderate CYP1A2 Inhibitors | |
| Clinical Impact |
|
| Prevention or Management |
|
| Moderate CYP1A2 Inducers | |
| Clinical Impact |
|
| Prevention or Management |
|
Effect of VORANIGO on Other Drugs
| Certain CYP3A Substrates | |
| Clinical Impact |
|
| Prevention or Management |
|
| Hormonal Contraception | |
| Clinical Impact |
|
| Prevention or Management |
|
DESCRIPTION
VORANIGO tablets contain vorasidenib, an isocitrate dehydrogenase-1 (IDH1) and isocitrate dehydrogenase-2 (IDH2) inhibitor. Vorasidenib is present as the hemicitric acid hemihydrate co-crystal. The chemical name of the co-crystal is 6-(6-chloropyridin-2-yl)- N 2 , N 4 -bis[(2 R )-1,1,1-trifluoropropan-2-yl]-1,3,5-triazine-2,4-diamine, 2-hydroxypropane-1,2,3-tricarboxylic acid, hydrate (2:1:1). It has the following chemical structure:
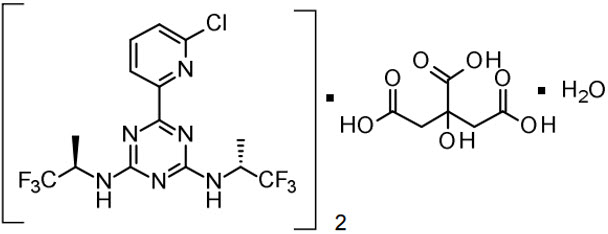
The molecular formula is C 14 H 13 ClF 6 N 6 • ½ C 6 H 8 O 7• ½ H 2 O and the molecular weight is 519.8 g/mol. Vorasidenib hemicitric acid hemihydrate is a white to off-white solid practically insoluble in aqueous solutions between pH 1.2 to 6.8.
VORANIGO is available as a 10 mg and 40 mg strength film-coated tablet for oral administration. The strengths reflect the amount of active ingredient vorasidenib in each tablet. Each tablet core contains the following inactive ingredients: croscarmellose sodium, magnesium stearate, microcrystalline cellulose, silicified microcrystalline cellulose and sodium lauryl sulfate. The tablet coating includes hypromellose, lactose monohydrate, macrogol and titanium dioxide. Each tablet is printed with black ink that contains black iron oxide, hypromellose and propylene glycol.
CLINICAL PHARMACOLOGY
Mechanism of Action
Vorasidenib is a small molecule inhibitor that targets isocitrate dehydrogenase-1 and 2 (IDH1 and IDH2) enzymes. In vitro, vorasidenib inhibited the IDH1 wild type and mutant variants, including R132H and the IDH2 wild type and mutant variants. In cell-based and in vivo tumor models expressing IDH1 or IDH2 mutated proteins, vorasidenib decreased production of 2-hydroxyglutarate (2-HG) and partially restored cellular differentiation.
Pharmacodynamics
Exposure-Response Relationships
Vorasidenib decreases 2-HG tumor concentrations in patients with IDH1 or IDH2 mutated glioma. Relative to tumors from patients in the untreated group, the posterior median percentage reduction (95% credible interval) in tumor 2-HG was 64% (22%, 88%) to 93% (76%, 98%) in tumors from patients who received vorasidenib at exposures that were 0.3 to 0.8 times the exposure observed with the highest recommended dosage.
The exposure-response relationship and time course of pharmacodynamic response for the safety and effectiveness of vorasidenib have not been fully characterized.
Cardiac Electrophysiology
When the recommended dose of VORANIGO is administered 1 hour prior to a meal, a mean increase in the QTc interval >20 msec is unlikely. There is insufficient information to characterize the QTc effects of VORANIGO at higher vorasidenib concentrations, e.g., when administered with a meal or when co-administered with a moderate CYP1A2 inhibitor [see Clinical Pharmacology (12.3) ] .
Pharmacokinetics
Vorasidenib maximum plasma concentration (C max ) and AUC increased approximately proportionally over the dose range of 10 to 200 mg (0.2 to 4 times the exposure of the highest approved recommended dosage) following once daily administration of single and multiple doses. At the highest approved recommended dosage, steady state mean (CV%) C max is 133 ng/mL (73%) and AUC is 1,988 h•ng/mL (95%). Steady state is achieved within 28 days of once daily dosing and the mean accumulation ratio of AUC is 4.4.
Absorption
The median (minimum, maximum) time to maximum plasma concentrations (t max ) at steady state is 2 hours (0.5 to 4 hours).
The mean absolute bioavailability of vorasidenib is 34%.
Food Effect
A high-fat and high-calorie (total 800-1,000 calories, of which 500-600 from fat) meal increased vorasidenib C max 3.1-fold and AUC 1.4-fold, compared to the fasting conditions.
A low-fat and low-calorie (total 400-500 calories, of which 100-125 from fat) meal increased vorasidenib C max 2.3-fold and AUC 1.4-fold, compared to the fasting conditions.
Distribution
The mean (CV%) volume of distribution at steady state of vorasidenib is 3,930 L (40%).
The protein binding is 97% in human plasma independent of vorasidenib concentrations in vitro.
The brain tumor-to-plasma concentration ratio is 1.6.
Elimination
The mean (CV%) steady state terminal half-life is 10 days (57%) and oral clearance is 14 L/h (56%).
Metabolism
Vorasidenib is primarily metabolized by CYP1A2 with minor contributions from CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6 and CYP3A. Non-CYP pathways may contribute up to 30% of its metabolism.
Excretion
Following a single oral radiolabeled dose of vorasidenib, 85% of the dose was recovered in feces (56% unchanged) and 4.5% was recovered in urine.
Specific Populations
No clinically significant effects on the pharmacokinetics of vorasidenib were observed based on age (16 to 75 years), sex, race (White, Black or African American, Asian, American Indian/Alaskan Native, Native Hawaiian or Other Pacific Islander), ethnicity (Hispanic and non-Hispanic), body weight (43.5 to 168 kg), mild or moderate hepatic impairment (Child-Pugh Class A or B) or CL cr >40 mL/min (as Cockcroft-Gault). The pharmacokinetics of vorasidenib has not been studied in patients with severe hepatic impairment (Child-Pugh Class C), in patients with CL cr ≤40 mL/min or in patients with renal impairment who require dialysis.
Pediatric Patients
The exposure of vorasidenib in pediatric patients ≥12 years of age is predicted to be within range of that observed in adults at the approved recommended dosage.
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
Effect of Other Drugs on Vorasidenib
Strong and Moderate CYP1A2 Inhibitors: Concomitant use of ciprofloxacin (moderate CYP1A2 inhibitor) increased vorasidenib plasma C max 1.3-fold and AUC 2.5-fold.
Concomitant use with fluvoxamine (strong CYP1A2 inhibitor) is predicted to increase vorasidenib C max and AUC by ≥5-fold.
Moderate CYP1A2 Inducers: Concomitant use with phenytoin or rifampicin (moderate CYP1A2 inducers) is predicted to decrease vorasidenib steady-state C max by 30% and AUC by 40%.
Gastric Acid Reducing Agents: No clinically significant difference in vorasidenib pharmacokinetics was observed following concomitant use with omeprazole (an acid-reducing agent).
Effect of Vorasidenib on Other Drugs
CYP3A Substrates: Concomitant use of multiple doses of vorasidenib with CYP3A substrates is predicted to decrease the concentrations of these substrates.
UGT1A4 Substrate: No clinically significant difference in lamotrigine pharmacokinetics was observed following the administration of lamotrigine with multiple doses of vorasidenib.
P-gp and BCRP Substrates: No clinically significant difference in the pharmacokinetics of digoxin (P-gp substrate) or rosuvastatin (BCRP substrate) is predicted when used concomitantly with vorasidenib.
In vitro Studies
Vorasidenib is an inducer of CYP2B6, CYP2C8, CYP2C9, CYP2C19 and CYP3A and UGT1A4.
Vorasidenib is not a substrate of P-gp, BCRP, or OATP1B1 and OATP1B3.
Vorasidenib is an inhibitor of BCRP. Vorasidenib does not inhibit P-gp and OATP1B1.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with vorasidenib. Vorasidenib and its major circulating metabolite, AGI-69460, were not mutagenic in an in vitro bacterial reverse mutation (Ames) assay. Vorasidenib was not clastogenic in an in vitro human lymphocyte micronucleus assay, or an in vivo rat bone marrow micronucleus assay.
Fertility studies in animals have not been conducted with vorasidenib. In repeat-dose toxicity studies up to 13 weeks in duration with oral administration of vorasidenib in rats, adverse effects in female reproductive organs included atrophy, decreased/absent corpora lutea, increased atretic follicles, and interstitial cell vacuolation of the ovaries, atrophy, hypertrophy, and metaplasia of the uterus, hyperplasia of the cervix, atrophy, hyperplasia, and mucification of the vagina, and estrous cycle variations at doses ≥3 mg/kg/day (≥12 times the exposure based on AUC in humans at the highest recommended dose). Adverse effects in male reproductive organs in rats included tubular degeneration and atrophy of the testes, degeneration of seminiferous tubules, cellular debris in the epididymides, epithelial atrophy and inflammation in the prostate, and atrophy in the seminal vesicles at doses ≥3 mg/kg/day (≥12 times the exposure based on AUC in humans at the highest recommended dose). Findings in the male rats were not reversible. In the 4-week repeat-dose toxicity studies in monkeys, oral administration of vorasidenib led to adverse effects in male and female reproductive organs including fibrotic hypoplasia of the testes in males at doses ≥10 mg/kg/day (approximately 17 times the exposure based on AUC in humans at the highest recommended dose) and decreased uterine weights in females at doses ≥10 mg/kg/day (approximately 22 times the exposure based on AUC in humans at the highest recommended dose). Findings in male and female monkeys were reversible.
Animal Toxicology and Pharmacology
In repeat-dose toxicity studies, oral administration of vorasidenib to rats for 28 days led to ototoxicity findings of reversible neutrophil infiltration of the epithelial lining of the middle ear and Eustachian tube at doses >3 mg/kg/day (>12-times the human exposure based on the AUC at the highest recommended dose). Additional findings in a 28-day ototoxicity study included edema in the tympanic cavity at doses >30 mg/kg/day. In addition, oral administration of vorasidenib to monkeys for 13 weeks resulted in dilated cardiomyopathy and secondary congestive heart failure at a dose of 20 mg/kg/day (105 times the human exposure based on the AUC at the highest recommended dose). Skeletal muscle was a target organ in the repeat dose toxicology studies in rats and monkeys at doses ≥13 times the AUC in patients at the highest recommended dose. Findings included decreased hind limb muscle tone, abnormal gait with limited hind limb usage, and low carriage, associated with small size and atrophy of the muscle in rats and necrosis and mononuclear/mixed cell infiltrates in the muscle in monkeys.
CLINICAL STUDIES
The efficacy of VORANIGO was evaluated in the INDIGO trial (Study AG881-C-004), a randomized, multicenter, double-blind, placebo-controlled study of 331 patients (NCT04164901). Eligible patients were required to have IDH1- or IDH2-mutant Grade 2 astrocytoma or oligodendroglioma with prior surgery including biopsy, sub-total resection, or gross total resection. Patients were required to have measurable, non-enhancing disease; patients with centrally confirmed minimal, non-nodular, non-measurable enhancement were eligible. Patients who received prior anti-cancer treatment, including chemotherapy or radiation therapy were excluded. Patients were randomized to receive either VORANIGO 40 mg orally once daily or placebo orally once daily until disease progression or unacceptable toxicity. IDH1 or IDH2 mutation status was prospectively determined by the Life Technologies Corporation Oncomine Dx Target Test.
Randomization was stratified by local 1p19q status (co-deleted or not co-deleted) and baseline tumor size (diameter ≥2 cm or <2 cm). Patients who were randomized to placebo were allowed to cross over to receive VORANIGO after documented radiographic disease progression. Tumor assessments were performed every 12 weeks.
A total of 331 patients were randomized, 168 to the VORANIGO arm and 163 to the placebo arm. The median age was 40 years (range: 16 to 71); 57% were male; 78% were White, 4% were Asian, 1% were Black or African American and 16% had race not reported; 78% were not Hispanic or Latino; 52% oligodendroglioma and 48% astrocytoma; 79% had one prior surgery and 21% had ≥2 prior surgeries. In the VORANIGO arm, 14% of patients had biopsy, 48% had sub-total resection and 51% had gross-total resection. The majority of IDH1 mutations consisted of R132H (87%). The other alleles were reported as follows: R132C (5%), R132G (3%), R132L (1%), and R132S (1%). IDH2 mutations consisted of R172K (2%) and R172G (1%).
The major efficacy outcome was progression-free survival (PFS) as evaluated by a blinded independent review committee (BIRC) per modified Response Assessment in Neuro-Oncology for Low Grade Glioma (RANO-LGG) criteria.
Efficacy results are summarized in Table 7 and Figure 1.
| Efficacy Parameter | VORANIGO 40 mg daily (n=168) | Placebo (n=163) |
|---|---|---|
| CI = Confidence Interval | ||
| Progression-Free Survival (PFS) | ||
| Number of Events, n (%) | ||
| Progressive disease | 47 (28) | 88 (54) |
| Death | 0 | 0 |
| Hazard ratio (95% CI) Stratified Cox proportional hazard model, stratified by 1p19q status and baseline tumor size. | 0.39 (0.27, 0.56) | |
| p-value Based on one-sided stratified log-rank test compared to the pre-specified α of 0.000359 (one-sided). | <0.0001 | |
Figure 1: Kaplan-Meier Curve for Progression-Free Survival per BIRC for the INDIGO Trial
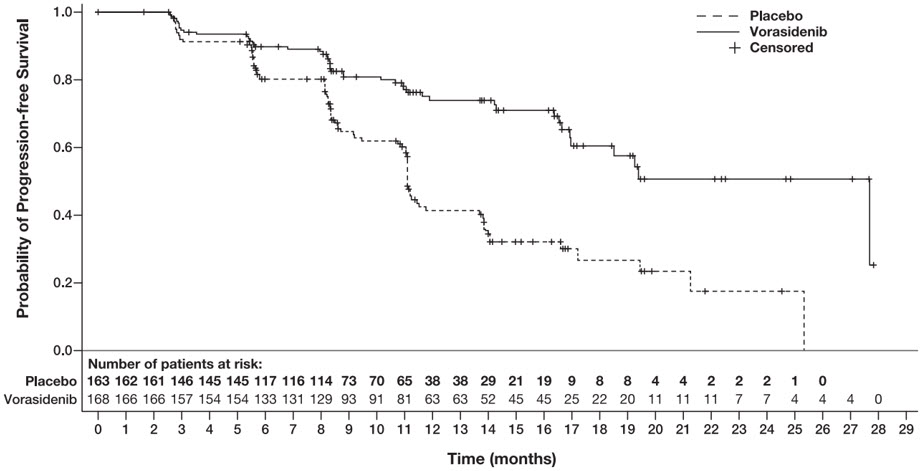
The major efficacy analyses are supported by a prospectively defined key secondary outcome measure time to next intervention (defined as the time from randomization to the initiation of first subsequent anticancer therapy or death due to any cause). The median time to next intervention was not reached for patients in the VORANIGO arm and 17.8 months for patients in the placebo arm (HR=0.26; 95% CI: [0.15, 0.43], p <0.0001).
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
VORANIGO ® (vorasidenib) tablets are supplied in two strengths:
10 mg tablets: White to off-white, round film-coated tablet imprinted with "10" in black ink on one side and plain on the other side.
- Each carton contains one 30-count bottle of 10 mg tablets with desiccant canister(s) and child-resistant cap (NDC 72694-879-10)
40 mg tablets: White to off-white, oblong film-coated tablet imprinted with "40" in black ink on one side and plain on the other side.
- Each carton contains one 30-count bottle of 40 mg tablets with desiccant canister(s) and child-resistant cap (NDC 72694-728-40)
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Mechanism of Action
Vorasidenib is a small molecule inhibitor that targets isocitrate dehydrogenase-1 and 2 (IDH1 and IDH2) enzymes. In vitro, vorasidenib inhibited the IDH1 wild type and mutant variants, including R132H and the IDH2 wild type and mutant variants. In cell-based and in vivo tumor models expressing IDH1 or IDH2 mutated proteins, vorasidenib decreased production of 2-hydroxyglutarate (2-HG) and partially restored cellular differentiation.