Get your patient on Zegalogue (Dasiglucagon)
Zegalogue prior authorization resources
Most recent state uniform prior authorization forms
Zegalogue patient education
Patient toolkit
Dosage & administration
DOSAGE AND ADMINISTRATION
- ZEGALOGUE autoinjector and prefilled syringe are for subcutaneous injection only. (2.1 )
- The dose in adults and pediatric patients aged 6 years and older is 0.6 mg. (2.2 )
- Administer ZEGALOGUE according to the printed instructions on the protective case label and the Instructions For Use. (2.1 )
- Visually inspect ZEGALOGUE prior to administration. The solution should appear clear, colorless, and free from particles. If the solution is discolored or contains particulate matter, do not use. (2.1 )
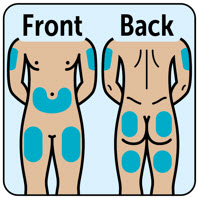
- Administer the injection into the lower abdomen, buttocks, thigh, or outer upper arm. (2.1 )
- Call for emergency assistance immediately after administering the dose. (2.1 )
- If there has been no response after 15 minutes, an additional dose of ZEGALOGUE from a new device may be administered while waiting for emergency assistance. (2.1 )
- When the patient has responded to treatment, give oral carbohydrates. (2.1 )
- Do not attempt to reuse ZEGALOGUE. Each device contains a single dose of dasiglucagon and cannot be reused. (2.1 )
Administration Instructions
ZEGALOGUE autoinjector and prefilled syringe are for subcutaneous injection only.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires the help of others to recover, instruct the patient to inform those around them about ZEGALOGUE and its Instructions For Use. Administer ZEGALOGUE as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions For Use at the time they receive a prescription for ZEGALOGUE. Emphasize the following instructions to the patient or caregiver:
- Administer ZEGALOGUE according to the printed instructions on the protective case label and the Instructions For Use.
- Visually inspect ZEGALOGUE prior to administration. The solution should appear clear, colorless, and free from particles. If the solution is discolored or contains particulate matter, do not use.
- Administer the injection in the lower abdomen, buttocks, thigh, or outer upper arm.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, an additional dose of ZEGALOGUE may be administered while waiting for emergency assistance.
- When the patient has responded to treatment, give oral carbohydrates to restore liver glycogen and prevent recurrence of hypoglycemia.
- Do not attempt to reuse ZEGALOGUE. Each ZEGALOGUE device contains a single dose of dasiglucagon and cannot be reused.
Recommended Dosage
The recommended dose of ZEGALOGUE in adults and pediatric patients aged 6 years and older is 0.6 mg administered by subcutaneous injection into the lower abdomen, buttocks, thigh, or outer upper arm.
If there has been no response after 15 minutes, an additional 0.6 mg dose of ZEGALOGUE from a new device may be administered.

Zegalogue prescribing information
INDICATIONS AND USAGE
ZEGALOGUE ® is indicated for the treatment of severe hypoglycemia in pediatric and adult patients with diabetes aged 6 years and above.
DOSAGE AND ADMINISTRATION
- ZEGALOGUE autoinjector and prefilled syringe are for subcutaneous injection only. (2.1 )
- The dose in adults and pediatric patients aged 6 years and older is 0.6 mg. (2.2 )
- Administer ZEGALOGUE according to the printed instructions on the protective case label and the Instructions For Use. (2.1 )
- Visually inspect ZEGALOGUE prior to administration. The solution should appear clear, colorless, and free from particles. If the solution is discolored or contains particulate matter, do not use. (2.1 )
- Administer the injection into the lower abdomen, buttocks, thigh, or outer upper arm. (2.1 )
- Call for emergency assistance immediately after administering the dose. (2.1 )
- If there has been no response after 15 minutes, an additional dose of ZEGALOGUE from a new device may be administered while waiting for emergency assistance. (2.1 )
- When the patient has responded to treatment, give oral carbohydrates. (2.1 )
- Do not attempt to reuse ZEGALOGUE. Each device contains a single dose of dasiglucagon and cannot be reused. (2.1 )
Administration Instructions
ZEGALOGUE autoinjector and prefilled syringe are for subcutaneous injection only.
Instruct patients and their caregivers on the signs and symptoms of severe hypoglycemia. Because severe hypoglycemia requires the help of others to recover, instruct the patient to inform those around them about ZEGALOGUE and its Instructions For Use. Administer ZEGALOGUE as soon as possible when severe hypoglycemia is recognized.
Instruct the patient or caregiver to read the Instructions For Use at the time they receive a prescription for ZEGALOGUE. Emphasize the following instructions to the patient or caregiver:
- Administer ZEGALOGUE according to the printed instructions on the protective case label and the Instructions For Use.
- Visually inspect ZEGALOGUE prior to administration. The solution should appear clear, colorless, and free from particles. If the solution is discolored or contains particulate matter, do not use.
- Administer the injection in the lower abdomen, buttocks, thigh, or outer upper arm.
- Call for emergency assistance immediately after administering the dose.
- If there has been no response after 15 minutes, an additional dose of ZEGALOGUE may be administered while waiting for emergency assistance.
- When the patient has responded to treatment, give oral carbohydrates to restore liver glycogen and prevent recurrence of hypoglycemia.
- Do not attempt to reuse ZEGALOGUE. Each ZEGALOGUE device contains a single dose of dasiglucagon and cannot be reused.
Recommended Dosage
The recommended dose of ZEGALOGUE in adults and pediatric patients aged 6 years and older is 0.6 mg administered by subcutaneous injection into the lower abdomen, buttocks, thigh, or outer upper arm.
If there has been no response after 15 minutes, an additional 0.6 mg dose of ZEGALOGUE from a new device may be administered.
DOSAGE FORMS AND STRENGTHS
ZEGALOGUE injection is a clear, colorless solution available as:
- 0.6 mg/0.6 mL single-dose autoinjector
- 0.6 mg/0.6 mL single-dose prefilled syringe
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no available data on dasiglucagon use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes.
Untreated hypoglycemia in pregnancy can cause complications and may be fatal.
In animal reproduction studies, daily subcutaneous administration of dasiglucagon to pregnant rabbits and rats during the period of organogenesis did not cause adverse developmental effects at exposures 7 and 709 times the human dose of 0.6 mg based on AUC, respectively (see Data ) .
Data
Animal Data
In an embryo-fetal development study, pregnant rats were treated daily with subcutaneous doses of 2, 10, and 24 mg/kg/day during the period of organogenesis (gestation day 6 to 17). Maternal toxicity, in terms of decreased body weight gain, lower fetal body weight, and delayed bone ossification, was observed at ≥10 mg/kg/day (≥475 times the human dose, based on AUC).
In an embryo-fetal development study, pregnant rabbits were treated daily with subcutaneous doses of 0.1, 0.3, and 1 mg/kg/day during the period of organogenesis (gestation day 6 to 19). Lower fetal body weight and delayed bone ossification were observed at 1 mg/kg/day (100 times the human dose, based on AUC), a dose that also induced maternal toxicity in terms of decreased body weight gain. At ≥0.3 mg/kg/day (≥20 times the human dose), dasiglucagon caused fetal skeletal and visceral malformations. No adverse fetal developmental effects were observed at 0.1 mg/kg/day, corresponding to exposure 7 times the human dose.
Lactation
Risk Summary
There is no information on the presence of dasiglucagon in either human or animal milk, or the effects of the drug on the breastfed infant or milk production. Dasiglucagon is a peptide and would be expected to be broken down to its constituent amino acids in the infant's digestive tract and is therefore unlikely to cause harm to an exposed infant.
Pediatric Use
The safety and effectiveness of ZEGALOGUE for the treatment of severe hypoglycemia in patients with diabetes have been established in pediatric patients aged 6 years and above. Use of ZEGALOGUE for this indication is supported by evidence from a study in 42 pediatric patients with type 1 diabetes [see Clinical Studies (14.2) ] .
The safety and effectiveness of ZEGALOGUE have not been established in pediatric patients younger than 6 years of age.
Geriatric Use
Clinical studies of ZEGALOGUE included too few patients 65 years of age and older to determine whether these patients respond differently from younger adult patients.
CONTRAINDICATIONS
ZEGALOGUE is contraindicated in patients with:
- Pheochromocytoma because of the risk of substantial increase in blood pressure [see Warnings and Precautions (5.1) ]
- Insulinoma because of the risk of hypoglycemia [see Warnings and Precautions (5.2) ]
WARNINGS AND PRECAUTIONS
- Substantial Increase in Blood Pressure in Patients with Pheochromocytoma: Contraindicated in patients with pheochromocytoma because ZEGALOGUE may stimulate the release of catecholamines from the tumor. (4 , 5.1 )
- Hypoglycemia in Patients with Insulinoma: In patients with insulinoma, administration may produce an initial increase in blood glucose, but ZEGALOGUE may stimulate exaggerated insulin release from an insulinoma and cause subsequent hypoglycemia. If a patient develops symptoms of hypoglycemia after a dose of ZEGALOGUE, give glucose orally or intravenously. (4 , 5.2 )
- Hypersensitivity and Allergic Reactions: Allergic reactions have been reported with glucagon products and may include generalized rash, and in some cases anaphylactic shock with breathing difficulties and hypotension. (5.3 )
- Lack of Efficacy in Patients with Decreased Hepatic Glycogen: ZEGALOGUE is effective in treating hypoglycemia only if sufficient hepatic glycogen is present. Patients in states of starvation, with adrenal insufficiency or chronic hypoglycemia may not have adequate levels of hepatic glycogen for ZEGALOGUE to be effective. Patients with these conditions should be treated with glucose. (5.4 )
Substantial Increase in Blood Pressure in Patients with Pheochromocytoma
ZEGALOGUE is contraindicated in patients with pheochromocytoma because glucagon products may stimulate the release of catecholamines from the tumor [see Contraindications (4) ] . If the patient develops a substantial increase in blood pressure and a previously undiagnosed pheochromocytoma is suspected, 5 to 10 mg of phentolamine mesylate, administered intravenously, has been shown to be effective in lowering blood pressure.
Hypoglycemia in Patients with Insulinoma
In patients with insulinoma, administration of glucagon products may produce an initial increase in blood glucose; however, ZEGALOGUE administration may directly or indirectly (through an initial rise in blood glucose) stimulate exaggerated insulin release from an insulinoma and cause hypoglycemia. ZEGALOGUE is contraindicated in patients with insulinoma [see Contraindications (4) ] . If a patient develops symptoms of hypoglycemia after a dose of ZEGALOGUE, give glucose orally or intravenously.
Hypersensitivity and Allergic Reactions
Allergic reactions have been reported with glucagon products; these include generalized rash, and in some cases anaphylactic shock with breathing difficulties and hypotension. Advise patients to seek immediate medical attention if they experience any symptoms of serious hypersensitivity reactions.
Lack of Efficacy in Patients with Decreased Hepatic Glycogen
ZEGALOGUE is effective in treating hypoglycemia only if sufficient hepatic glycogen is present. Patients in states of starvation, with adrenal insufficiency or chronic hypoglycemia may not have adequate levels of hepatic glycogen for ZEGALOGUE administration to be effective. Patients with these conditions should be treated with glucose.
ADVERSE REACTIONS
The following important adverse reactions are described elsewhere in the labeling:
- Hypersensitivity and Allergic Reactions [see Warnings and Precautions (5.3) ]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of ZEGALOGUE cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In clinical trials, 316 adult patients with type 1 diabetes and 20 pediatric patients with type 1 diabetes were exposed to ZEGALOGUE.
The data in Table 1 reflect exposure of 116 adult patients to ZEGALOGUE in 2 placebo-controlled trials (mean age 40 years). Table 2 reflects exposure of 20 pediatric patients exposed to ZEGALOGUE in a placebo-controlled trial. Eight patients were 7 to 11 years old, and 12 were 12 to 17 years old [see Clinical Studies (14) ] .
| Adverse reaction type | Placebo (N=53) | Dasiglucagon (N=116) |
|---|---|---|
| % of Patients | % of Patients | |
| Nausea | 4% | 57% |
| Vomiting | 2% | 25% |
| Headache | 4% | 11% |
| Diarrhea | 0% | 5% |
| Injection site pain | 0% | 2% |
| Adverse reaction type | Placebo (N=11) | Dasiglucagon Age 6-11 years (N=8) | Dasiglucagon Age 12-17 years (N=12) | Dasiglucagon All (N=20) |
|---|---|---|---|---|
| % of Patients | % of Patients | % of Patients | % of Patients | |
| Nausea | 0% | 25% | 92% | 65% |
| Vomiting | 0% | 25% | 67% | 50% |
| Headache | 0% | 0% | 17% | 10% |
| Injection site pain | 0% | 0% | 8% | 5% |
Other Adverse Reactions
Other adverse reactions in patients treated with dasiglucagon occurring within 12 hours of treatment include: hypertension, hypotension, bradycardia, presyncope, palpitations, and orthostatic intolerance.
Immunogenicity
As with all therapeutic peptides, there is a potential for immunogenicity with ZEGALOGUE. The detection of antibody formation is highly dependent on the sensitivity and specificity of the assay. Additionally, the observed incidence of antibody (including neutralizing antibody) positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies to ZEGALOGUE with the incidence of antibodies to other products may be misleading.
In clinical trials, 4/498 (<1%) of ZEGALOGUE-treated patients developed treatment-emergent anti-drug antibodies (ADAs). Two patients receiving a single dose of ZEGALOGUE had detectable ADAs to dasiglucagon for at least 14 months after dosing. One ADA-positive patient receiving multiple doses of ZEGALOGUE had ADAs with transient neutralizing activity and with cross-reactivity against native glucagon. Although no safety or efficacy concerns were noted for these ADA-positive subjects, it is unknown whether ADAs may affect pharmacokinetics, pharmacodynamics, safety, and/or effectiveness of the drug [see Clinical Studies (14) ] .
DRUG INTERACTIONS
| Beta-Blockers | |
| Clinical Impact: | Patients taking beta-blockers may have a transient increase in pulse and blood pressure when given ZEGALOGUE. |
| Indomethacin | |
| Clinical Impact: | In patients taking indomethacin, ZEGALOGUE may lose its ability to raise blood glucose or may even produce hypoglycemia. |
| Warfarin | |
| Clinical Impact: | ZEGALOGUE may increase the anticoagulant effect of warfarin. |
DESCRIPTION
ZEGALOGUE contains dasiglucagon hydrochloride, which is a glucagon analog and an antihypoglycemic agent. Dasiglucagon is comprised of 29 amino acids. The molecular formula of dasiglucagon (anhydrous, free-base) is C 152 H 222 N 38 O 50 , and its molecular mass is 3382 g/mol (anhydrous, free-base). Dasiglucagon hydrochloride has the following chemical structure:

ZEGALOGUE injection is a preservative free, sterile, aqueous, clear, and colorless solution for subcutaneous use in a single-dose prefilled syringe and an autoinjector. Each prefilled syringe and autoinjector contains 0.63 mg of dasiglucagon provided as dasiglucagon hydrochloride, which is a salt with 3 - 5 equivalents of hydrochloride, and contains the following inactive ingredients: 3.82 mg tromethamine, 6.44 mg sodium chloride, and water for injection. Hydrochloric acid and/or sodium hydroxide may have been added to adjust pH to 6.5.
CLINICAL PHARMACOLOGY
Mechanism of Action
Dasiglucagon is a glucagon receptor agonist, which increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for dasiglucagon to produce an antihypoglycemic effect.
Pharmacodynamics
After administration of ZEGALOGUE in adult patients with type 1 diabetes, the mean glucose increase from baseline at 90 minutes was 168 mg/dL (Figure 1).

Figure 1 Mean plasma glucose over time in adults with type 1 diabetes administered 0.6 mg dasiglucagon
In pediatric patients (7 to 17 years) with type 1 diabetes, the mean glucose increase at 60 minutes after administration of ZEGALOGUE was 162 mg/dL (Figure 2).

Figure 2 Mean plasma glucose over time in pediatric patients with type 1 diabetes administered 0.6 mg dasiglucagon
Cardiac Electrophysiology
At a dose resulting in more than 5 times the concentration achieved with the recommended therapeutic dose, ZEGALOGUE does not prolong the QT interval to any clinically relevant extent.
Pharmacokinetics
Absorption
ZEGALOGUE absorption following subcutaneous injection of 0.6 mg resulted in a mean peak plasma concentration of 5110 pg/mL (1510 pmol/L) at around 35 minutes.
Distribution
The mean apparent volume of distribution was 47 L to 57 L following subcutaneous administration.
Elimination
The half-life was approximately 30 minutes.
Metabolism
Metabolism data indicated that dasiglucagon is cleared like native glucagon through proteolytic degradation pathways in blood, liver, and kidney.
Specific Populations
After administration of ZEGALOGUE in pediatric patients with type 1 diabetes, the mean peak plasma concentration of 3920 pg/mL occurred at around 21 minutes.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term studies in animals to evaluate carcinogenicity of dasiglucagon have not been performed.
Mutagenesis
Dasiglucagon was not mutagenic or clastogenic in a standard battery of genotoxicity tests: bacterial mutagenicity (Ames), human lymphocyte chromosome aberration, and rat bone marrow micronucleus.
Impairment of Fertility
In a fertility and early embryofetal development study in rats, dasiglucagon administered by subcutaneous injection (0.5, 2, and 8 mg/kg/day) did not impair fertility in male and female rats at exposures 179 and 269 times the human dose of 0.6 mg (based on AUC), respectively.
CLINICAL STUDIES
Three randomized, double-blind, placebo-controlled, multicenter trials were conducted in patients with type 1 diabetes. Two trials (Trial A and Trial B) were conducted in adult patients, and one trial (Trial C) was conducted in pediatric patients aged 6 to 17 years. In all 3 trials, patients were randomized to ZEGALOGUE 0.6 mg, placebo, or (in Trials A and C) glucagon for injection 1.0 mg. ZEGALOGUE and the comparators were administered as single subcutaneous injections following a controlled induction of hypoglycemia using intravenous administration of insulin. During this procedure, a plasma glucose concentration of <60 mg/dL was targeted in Trials A and B, whereas the target was <80 mg/dL in Trial C.
The primary efficacy endpoint for all 3 trials was time to plasma glucose recovery (treatment success), defined as an increase in blood glucose of ≥20 mg/dL from time of administration, without additional intervention within 45 minutes. In Trials A and B, plasma glucose values were collected and assessed at pre-dose, and at 4, 6, 8, 10, 12, 15, 17, 20, 25, 30, 40, 45, 50, 60, 75, 90 minutes after treatment. Trial C assessed plasma glucose at the same timepoints as did Trials A and B, with the exception of the 25, 40, 50, 75 and 90-minute post-treatment timepoints. The primary hypothesis test was superiority of ZEGALOGUE versus placebo. There was no formal hypothesis test of ZEGALOGUE versus glucagon for injection.
Adult Patients
Trial A, NCT03378635: A total of 170 patients were randomized 2:1:1 to ZEGALOGUE, placebo, and glucagon for injection, stratified by injection sites (abdominal region, buttocks, thigh). The mean age of the patients was 39.1 years (96% were < 65 years), and the mean duration of diabetes was 20.0 years; 63% were male; 92% were White. The mean baseline plasma glucose was 58.8 mg/dL. The median time to plasma glucose recovery was statistically significantly shorter for ZEGALOGUE (10 minutes) versus placebo (40 minutes) (Table 4). Figure 3 shows the cumulative proportions of patients achieving plasma glucose recovery over time. The median time to plasma glucose recovery was numerically similar between ZEGALOGUE (10 minutes) and glucagon for injection (12 minutes).
Trial B, NCT03688711: A total of 45 patients were randomized 3:1 to ZEGALOGUE and placebo stratified by injection sites (buttocks, deltoid). The mean age of the patients was 41.0 years (95% were < 65 years), and the mean duration of diabetes was 22.5 years; 57% were male; 93% were White. The mean baseline plasma glucose was 55.0 mg/dL. The median time to plasma glucose recovery was statistically significantly shorter for ZEGALOGUE (10 minutes) versus placebo (35 minutes) (Table 4).
| Trial A | Trial B | |||
|---|---|---|---|---|
| ZEGALOGUE N=82 | Placebo N=43 | ZEGALOGUE N=34 | Placebo N=10 | |
| N is the number of patients who were randomized and treated. | ||||
| Median time to recovery [95% CI log-log confidence interval ] | 10 min [10; 10] p < 0.001 versus placebo (log-rank test stratified by injection sites) | 40 min [30; 40] | 10 min [8; 12] | 35 min [20; -) |

Figure 3 Time to plasma glucose recovery in Trial A
Pediatric Patients
Trial C, NCT03667053: Pediatric patients aged 6 to 17 years and weighing ≥20 kg, were randomized 2:1:1 to ZEGALOGUE, placebo, and glucagon for injection, stratified by injection sites (abdominal region, thigh) and age groups (6-11 years and 12-17 years). A total of 42 patients were randomized. The mean age was 12.5 years (range 7 to 17 years), and the mean duration of diabetes was 5.9 years; 56% were male; 95% were White. The mean baseline plasma glucose was 72.0 mg/dL. The median time to plasma glucose recovery was statistically significantly shorter for ZEGALOGUE (10 minutes) versus placebo (30 minutes) (Table 5). Figure 4 shows the cumulative proportions of pediatric patients achieving plasma glucose recovery over time. The median time to plasma glucose recovery was numerically similar between ZEGALOGUE (10 minutes) and glucagon for injection (10 minutes).
| Trial C | ||
|---|---|---|
| ZEGALOGUE N=20 | Placebo N=11 | |
| N is the number of patients who were randomized and treated. | ||
| Median time to recovery [95% CI log-log confidence interval ] | 10 min [8; 12] p < 0.001 versus placebo (log-rank test stratified by injection site and age group) | 30 min [20; -) |

Figure 4 Time to plasma glucose recovery in Trial C
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
ZEGALOGUE injection is a clear, colorless solution supplied as follows:
| Presentation | Strength | Package Size | NDC |
|---|---|---|---|
| Single-dose autoinjector | 0.6 mg/0.6 mL | 1 | 80644-0012-1 |
| Single-dose autoinjector | 0.6 mg/0.6 mL | 2 | 80644-0012-2 |
| Single-dose prefilled syringe | 0.6 mg/0.6 mL | 1 | 80644-0013-1 |
| Single-dose prefilled syringe | 0.6 mg/0.6 mL | 2 | 80644-0013-2 |
Recommended Storage
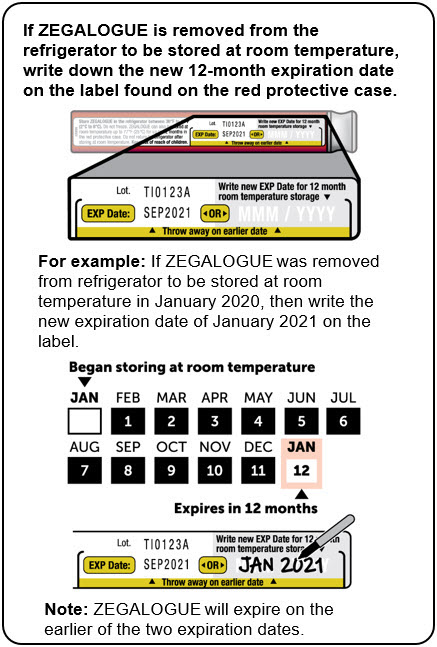
Store ZEGALOGUE in a refrigerator, 2°C to 8°C (36°F to 46°F). Keep away from the cooling element. Do not freeze.
ZEGALOGUE can be kept at room temperature between 20°C to 25°C (68°F to 77°F) for up to 12 months. Record the date when the product was removed from the refrigerator in the space provided on the protective case. Do not return the product to the refrigerator after storing at room temperature. Store in the provided protective case and protect from light.
Discard ZEGALOGUE after the end of the 12-month period at room temperature storage, or after the expiration date stated on the product, whichever occurs first.
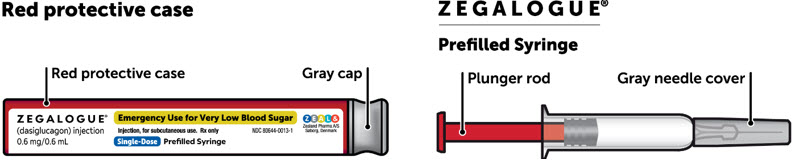
The inside of the gray cap on ZEGALOGUE autoinjector contains dry natural rubber (a derivative of latex).
The inside of the gray needle cover on ZEGALOGUE prefilled syringe contains dry natural rubber (a derivative of latex).
INSTRUCTIONS FOR USE
ZEGALOGUE ® (ze' gah log) (dasiglucagon) injection
0.6 mg/0.6 mL
Emergency Use for Very Low Blood Sugar Single-Dose Prefilled Syringe Injection, for subcutaneous use
Important: ZEGALOGUE is used to treat very low blood sugar (severe hypoglycemia) where you need help from others.
ZEGALOGUE contains 1 dose of dasiglucagon in a prefilled syringe and cannot be reused.
Read and understand this Instructions For Use before an emergency happens.
Show your family and friends where you keep ZEGALOGUE and explain how to use it by sharing these instructions, so they know how to use ZEGALOGUE before an emergency happens.
The inside of the gray needle cover contains dry natural rubber, which may cause allergic reactions in people with latex allergy.
| Read before Injecting ZEGALOGUE |
|---|
|
| Storage information | |
|---|---|
|  |
| Description of parts |
|
| Before injection | ||||
| Choose injection site and expose skin
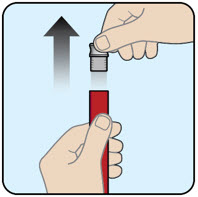
| Hold the red protective case upright and remove gray cap
| |||
| How to inject |
|---|
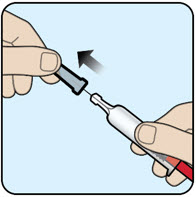
| Step 1 Remove the gray needle cover
|
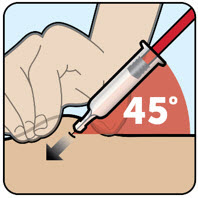
| Step 2 Pinch the skin and insert the needle
|
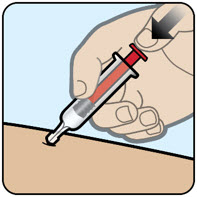
| Step 3 Give the injection
|
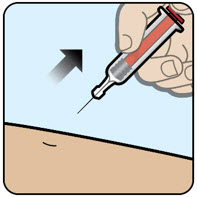
| Step 4 Remove the needle
|
| After injection |
|---|
|
Hypoglycemia may happen again after receiving ZEGALOGUE treatment.
Early symptoms of hypoglycemia may include:
|
|
| If not treated early, hypoglycemia may worsen and the person may have severe hypoglycemia. Signs of severe hypoglycemia include confusion, seizures, unconsciousness, and death. |
| How to throw away (dispose of) ZEGALOGUE | |
|---|---|
 Dispose of your ZEGALOGUE and the gray needle cover in an FDA-cleared sharps disposal container. Dispose of your ZEGALOGUE and the gray needle cover in an FDA-cleared sharps disposal container. | Put your expired or used needles and syringes in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash. If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
|
This Instructions For Use has been approved by the U.S. Food and Drug Administration.
Manufactured for: Zealand Pharma A/S Sydmarken 11 2860 Søborg, Denmark
Manufactured by: Rechon Lifescience AB Malmö, Sweden
REP-5121-C22 Revised 04/2021
Mechanism of Action
Dasiglucagon is a glucagon receptor agonist, which increases blood glucose concentration by activating hepatic glucagon receptors, thereby stimulating glycogen breakdown and release of glucose from the liver. Hepatic stores of glycogen are necessary for dasiglucagon to produce an antihypoglycemic effect.